Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Mycoplasmas are prokaryotes of the class Mollicutes and represent the smallest known free-living organisms. Their small size of 150 to 350 nm is more on the order of viruses than of bacteria. They lack a cell wall and are bound by a cell membrane. Many of the biologic properties of mycoplasmas are due to the absence of a rigid cell wall, including resistance to β-lactam antibiotics and marked pleomorphism among individual cells. The mycoplasmal cell membrane contains phospholipids, glycolipids, sterols, and various proteins. Mycoplasmas are able to grow in cell-free media and possess RNA and DNA. The entire genomes of many of the Mycoplasma species have been sequenced and have been found to be among the smallest of prokaryotic genomes; the Mycoplasma genitalium genome consists of only 580,070 DNA base pairs and only 470 predicted coding regions. The elimination of genes related to synthesis of amino acids, fatty acid metabolism, and cholesterol necessitates a parasitic dependence on their host for exogenous nutrients, such as nucleic acid precursors, amino acids, fatty acids, and sterols. In mammals, Mycoplasma species most commonly colonize mucosal surfaces, such as the respiratory and genital tracts. At least 16 different species of Mollicutes colonize the mucosa of humans.
Ureaplasma species and Mycoplasma hominis are the mycoplasmas most commonly isolated from the genital tract of women and are associated with maternal and fetal infection. This chapter focuses on these two species in the maternal and fetal, neonatal, and very young infant populations. Mycoplasmal illnesses in other populations, such as immunocompromised older children and nonpregnant adults, are not discussed. Mycoplasma pneumoniae, M. genitalium, and Mycoplasma fermentans are mentioned briefly.
The originally identified species Ureaplasma urealyticum has been divided into two species, Ureaplasma parvum and Ureaplasma urealyticum, based on 16S ribosomal RNA sequences; the 14 described serovars were reassigned, with U. parvum containing 4 serovars (1, 3, 6, and 14) and U. urealyticum containing the remaining 10 serovars (2, 4, 5, 7, 8, 9, 10, 11, 12, and 13). Most of the available literature simply refers to U. urealyticum without differentiation into U. parvum or U. urealyticum . A few more recent investigations distinguish between U. parvum and U. urealyticum in their results, although not enough data are available to fully determine if the two species or 14 serovars differ in pathogenicity; initial data suggest that Ureaplasma virulence is species and serovar independent. In this chapter, Ureaplasma is used to refer to U. parvum and U. urealyticum without differentiation.
Ureaplasma and M. hominis are commensal organisms in the lower female genital tract. Colonization of the female lower urogenital tract by Ureaplasma and M. hominis generally occurs as a result of sexual activity. Sexual contact is the major mode of transmission of these organisms, and colonization increases dramatically with increasing numbers of sexual partners.
In an asymptomatic woman, these mycoplasmas may be found throughout the lower urogenital tract, including the external cervical os, vagina, labia, and urethra. The vagina yields the largest number of organisms, followed by the periurethral area and the cervix. Ureaplasma is isolated less often from urine than from the cervix, but M. hominis is present in the urine and in the cervix with approximately the same frequency. In asymptomatic men, mycoplasmas also have been isolated from urine, semen, and the distal urethra.
Ureaplasma can be isolated from the vagina of 40% to 80% of sexually active, asymptomatic women; M. hominis is found in 21% to 70%. Both microorganisms can be found concurrently in 31% to 60% of women. In men, colonization with each is less prevalent. In women, colonization has been linked to younger age, lower socioeconomic status, multiple sexual partners, black ethnicity, oral contraceptive use, and recent antimicrobial therapy. In addition, mycoplasmas are prevalent in the lower genital tract of pregnant women. When genital mycoplasmas are present at the first prenatal visit, usually they persist throughout the pregnancy. Studies suggest that postmenopausal women are infrequently colonized with genital mycoplasmas.
Three disease associations have been established for Ureaplasma and M. hominis in the urinary tract: urethritis in men caused by Ureaplasma, urinary calculi caused by Ureaplasma, and pyelonephritis caused by M. hominis . Intraurethral inoculation of human volunteers and nonhuman primates with Ureaplasma produces urethritis. Serologic studies and clinical responsiveness in antimicrobial treatment trials also support a causative role of this organism in urethritis. The common presence of ureaplasmas in the urethra of asymptomatic men suggests either that only certain serovars of ureaplasmas are pathogenic or that predisposing factors, such as lack of mucosal immunity, must exist in individuals in whom symptomatic infection develops. Alternatively, disease may develop only on initial exposure to ureaplasmas. Ureaplasma also has been implicated in urethroprostatitis and epididymitis.
Ureaplasma has been shown to have a limited role in the production of urinary calculi. Ureaplasma produces urease, which splits urea into ammonia and carbon dioxide, and has been shown to induce crystallization of struvite and calcium phosphates in artificial urine in vitro, indicating a capacity of the pathogen to induce stone formation. Renal calculi have been induced experimentally by inoculation of pure cultures of Ureaplasma directly into the bladder and renal pelvis of rats. Ureaplasma has been isolated from stones recovered by surgery in 6 of 15 patients. In 4 of the 6 patients, no other urease-producing organisms were isolated either in the stone or in urine sampled from the renal pelvis. Proteus mirabilis is the most common infectious cause of similar stones in humans. The frequency with which Ureaplasma reaches the kidney, the predisposing factors that allow this to occur, and the relative frequency of renal calculi induced by this organism compared with that of calculi induced by other organisms are unknown.
Even with the high incidence of M. hominis in the lower urogenital tract, this organism has been isolated from the upper urinary tract only in patients with symptoms of acute infection. In one study, M. hominis was recovered from samples of ureteral urine collected during surgery from 7 of 80 patients (4 in pure culture) with acute pyelonephritis, and in a second study from 3 of 18 patients with acute exacerbation of chronic pyelonephritis. M. hominis was not found in the upper urinary tract of 22 patients with chronic pyelonephritis without acute exacerbation or in 60 patients with noninfectious urinary tract disease.
M. hominis is considered an etiologic agent of pelvic inflammatory disease. Inoculation of M. hominis into fallopian tubes of primates induces parametritis and salpingitis within 3 days, whereas inoculation of human fallopian tube explants produces ciliostasis. The organism has been isolated in pure cultures from the fallopian tubes of approximately 8% of women with salpingitis diagnosed by laparoscopy but not in any women without salpingitis. The organism also can be isolated from the endometrium. A role for this organism in cases of pelvic inflammatory disease not associated with either Neisseria gonorrhoeae or Chlamydia trachomatis is supported by significant increases in specific antibodies to M. hominis . Ureaplasma is not considered to be a cause of pelvic inflammatory disease.
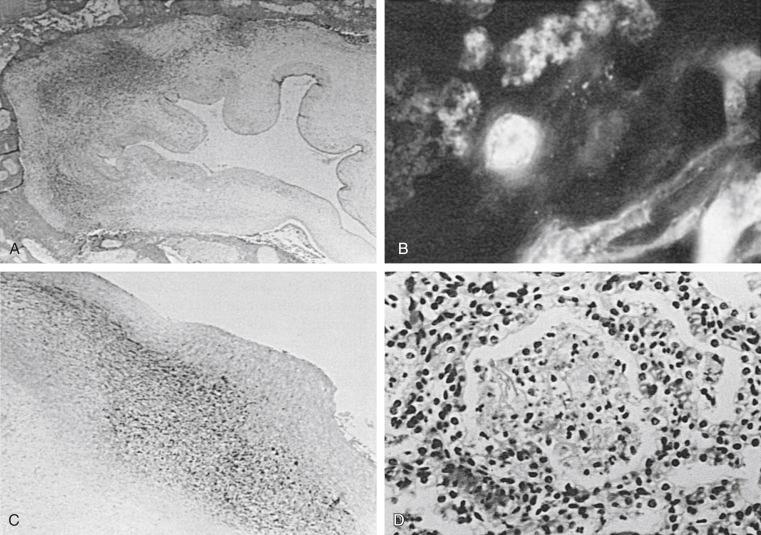
Isolation of Ureaplasma, but not M. hominis, from the chorioamnion uniformly has shown a significant association with histologic chorioamnionitis. Studies in which extensive culture for other agents was performed reported that women whose amniotic membranes contained Ureaplasma were more likely to have histologic evidence of chorioamnionitis than women without Ureaplasma, even after adjusting for duration of labor, premature rupture of membranes, duration of membrane rupture, and presence of other bacteria. Ureaplasma in the chorioamnion was found to be significantly associated with histologic chorioamnionitis in the presence of intact membranes when delivery was by cesarean section. In some cases, Ureaplasma was the only organism isolated. Case reports indicate that Ureaplasma can persist in the amniotic fluid for 7 weeks in the presence of an intense inflammatory response and in the absence of ruptured membranes or labor and can be isolated as a single microorganism when cultures for multiple agents are performed. These findings show that ureaplasmas can produce histologic changes consistent with chorioamnionitis.
Although Ureaplasma and M. hominis can invade the amniotic fluid at 16 to 20 weeks of gestation in the presence of intact membranes and in the absence of other microorganisms, these infections tend to be clinically silent and chronic ( Fig. 20-1 ). Ureaplasma and M. hominis have been isolated more frequently from the chorioamnion than from the amniotic fluid. Isolation of organisms from the chorioamnion or amniotic fluid has been significantly associated with histologic evidence of chorioamnionitis but not with clinical amnionitis. Ureaplasmas can be detected in the amniotic fluid in 50% of asymptomatic and symptomatic individuals. Based on these reports, the role of Ureaplasma in clinical amnionitis remains unclear.
In an investigation by Yoon and colleagues, amniocentesis was performed in 154 patients with preterm premature rupture of membranes. Amniotic fluid was cultured for aerobic and anaerobic bacteria and for mycoplasmas. Polymerase chain reaction (PCR) assay for Ureaplasma also was performed on the fluid. These investigators found that amniotic fluid culture for mycoplasmas missed 42% of cases identified as positive by Ureaplasma PCR assay. Patients with a negative result on amniotic fluid culture for Ureaplasma but a positive result on PCR assay had a significantly shorter interval from amniocentesis to delivery, higher amniotic fluid interleukin-6 (IL-6) concentrations, and higher white blood cell counts compared with patients without detection of Ureaplasma by culture or PCR assay. Subsequently, in a similar investigation by the same group in 257 patients with preterm labor and intact membranes, significant findings were similar except that the prevalence of Ureaplasma was lower.
The detection of M. hominis does not correlate with clinical symptoms. M. hominis commonly invades the chorioamnion and amniotic fluid, but such invasion rarely occurs in the absence of other organisms, particularly ureaplasmas. It is unclear whether this organism alone is a cause of histologic chorioamnionitis or clinical amnionitis
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here