Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Until very recently, obesity was not recognized as a disease, which confounded the ability of physicians to be compensated for treatment they delivered and to treat the condition effectively. The American Medical Association (AMA) officially recognized obesity as a disease in 2013 and in 2014 voted to approve the resolution “that our AMA, advocate for patient access to the full continuum of care of evidence-based obesity treatment modalities (including surgical interventions).”
Morbid obesity is defined as being 100 lbs above ideal body weight, twice ideal body weight, or body mass index (BMI; measured as weight in kilograms divided by height in meters squared) of 40 kg/m 2 . The last definition is more accepted internationally and has essentially replaced the former ones for all practical and scientific purposes. A consensus conference by the National Institutes of Health (NIH) in 1991 suggested that the term severe obesity is more appropriate for defining people of such size. This term is used interchangeably with morbid obesity in the remainder of this chapter.
The obesity epidemic in America continues to the point where nearly 40% of the U.S. adult population is obese, and prevalence of obesity in adolescents has increased to 18.5% in the most recent National Health and Nutrition Examination Survey (NHANES). The percentage of obese adults (BMI >30) in the United States increased 16 percentage points from 1980 to 2000, and increased by another 9 percentage points from 2000 to 2016. There are also significant differences in the prevalence of obesity in adults by sex, race, and Hispanic origin. Non-Hispanic Asians (12.7%) had significantly lower rates of obesity than all other race and non-Hispanic origin groups. Non-Hispanic whites (37.9%) had a lower prevalence of obesity than non-Hispanic black (46.8%) and Hispanic adults (47%). Hispanic men (43.1%) had a greater prevalence of obesity compared to non-Hispanic Asian (10.1%) and non-Hispanic black men (36.9%).
Worldwide, from 1975 to 2016, there has been a trend of increasing obesity in children and adolescents (age 5–19) in most regions of the world. The rate of increase in BMI has decreased in most high-income countries since 2000 albeit remaining at a high level. The rate of BMI increase, however, has accelerated in East, South, and Southeast Asia and is expected to surpass moderate to severe underweight children in those areas by 2022. Several countries (Nauru, Cook Islands, Palau, Niue, American Samoa) surpassed the United States in prevalence of obesity, while obesity was more than 20% or more in the United States, Polynesia, Micronesia, Middle East, and North Africa.
The prevalence of morbidly obese adults (BMI >40) has increased to 6.3% of the adult United States population and is the second leading cause of preventable death in the United States. Morbid obesity is second only to smoking on the list of preventable factors responsible for increased health care costs. It is a sobering thought to realize that a 40-year-old morbidly obese man has a 12.4% reduction in life expectancy, or 9.1 years of life lost, compared with a normal-sized man. Moreover, the cost of care is staggering and may be as high as 9% of annual medical expenditures or $147 billion per year. There appears to be significant population heterogeneity between BMI and mortality that is attenuated by increasing age of the individual. Mortality also increases significantly even for individuals with minimal increases in BMI greater than 30.0. Thus, it appears that age, gender, race, and income level all play a role in the development of obesity and obesity-related mortality.
The pathophysiology of severe obesity is multifactorial and has at its basis some genetic predisposition to obesity. There is a clear familial predisposition, and it is rare for a single-family member to have severe obesity. Scientists have identified specific genes that are associated with obesity, including the FTO gene (fat mass and obesity related) that plays a role in controlling feeding behavior and energy expenditure, the MC4R deficiency gene (melanocortin 4 receptor), which is associated with obesity, increased fat mass, and insulin resistance.
Single gene mutations causing obesity are rare, and expressed during early childhood ( Table 48.1 ). The most common single gene etiology of severe obesity is the MC4R, which induces appetite suppression (anorexigenic) effects on the hypothalamus in the regulation of energy homeostasis. Recent studies have suggested that the cilium of MC4R neurons in the hypothalamus is the most common pathway underlying the genetic causes of human obesity ( Table 48.1 ).
| Gene | Effect | Action on | Inheritance | Linked To |
|---|---|---|---|---|
| Leptin/leptin receptor | Appetite stimulant | Hypothalamus | Autosomal recessive | Severe childhood obesity |
| Ghrelin receptor | Appetite stimulant | Hypothalamus | Autosomal recessive | Short stature and obesity |
| Melanocortin 4 receptor | Appetite inhibitor | Hypothalamus | Autosomal dominant | Increased fat mass, insulin resistance |
| Proopiomelanocortin (POMC) | Appetite inhibitor | Melanocortin 4 receptor in hypothalamus | Autosomal recessive | Severe early onset obesity by age 1 and excessive eating caused by insatiable hunger |
| Neuropeptide Y (NPY) | Appetite stimulant | Hypothalamus | Autosomal recessive | Hypertension, high low-density lipoprotein cholesterol, triglycerides, increased food intake and hunger |
Another theory suggests that bacteria within the gut, known as the microbiome, play an essential role in the metabolism and immune system. Simply giving subtherapeutic antibiotic treatment to mice for 4 weeks increases adiposity, plasma levels of insulin, leptin, and triglycerides when the mice are later fed a high-fat diet. The predilection to obesity is transferrable to other mice when the low-dose penicillin-selected gut bacteria are transferred to germ-free hosts, thus identifying that it is the action of the altered gut bacteria, not the antibiotics, that causes the obesity. Recent studies have demonstrated that the gut microbiome circadian rhythm is disrupted by lifestyle differences in developed countries (shift work or jet lag), which provokes the development of altered microbial community, thus predisposing the host to obesity and glucose intolerance. Other studies have shown that degradation of dietary flavonoids through the altered microbiome results in diminished energy expenditure, which leads to obesity. It is fascinating to hypothesize that the current epidemic of obesity relates to changes in the microbiome created by the lifestyle changes and increased use of antibiotics in childhood seen in people who reside in developed countries.
Although there is no definitive answer to the pathophysiology of severe obesity, it is clear that a severely obese individual has, in general, persistent hunger that is not satiated by amounts of food that satisfy the nonobese. This lack of satiety or maintenance of hunger with corresponding increases in calorie intake may be the single most important factor in the process. There appear to be fundamental differences in the satiety and appetite hormonal control of eating that have created the current epidemic. This is hypothesized to occur when the brain’s energy “set-point” rises to increase energy intake, through modulation of the individual’s appetite.
We know that hormones, peptides, and vagal afferents to the brain have a major influence on satiety, appetite, and energy intake. Ghrelin, the only known orexigenic gut hormone, is also known as the hunger hormone and is secreted by P/D1 cells of the gastric fundus. Ghrelin stimulates release of various neuropeptides, such as neuropeptide Y (NPY) and growth hormone, from the hypothalamus, which creates an orexigenic or increased appetite state. Increased levels of ghrelin produce increased food intake, and increased levels of ghrelin develop in individuals after low-calorie diets, thus suggesting that one possible mechanism for the failure of most diets after 6 months is the increase in the appetite hormone ghrelin.
One evolving concept is that the environment causes heritable change in gene function without modification of DNA sequences termed gene-environment interactions . The changes in the epigenome lead to the development of obesity and are much more common than either the monogenetic or the syndromic forms of obesity.
Morbid obesity is a metabolic disease associated with numerous medical problems, some of which are virtually unknown in the absence of obesity. Box 48.1 lists the most common. These problems must be carefully considered when one is contemplating offering a patient weight reduction surgery. The most frequent problem is the combination of arthritis and degenerative joint disease, present in at least 50% of patients seeking surgery for severe obesity. The incidence of sleep apnea is high. Asthma is present in more than 25%, hypertension in more than 30%, diabetes in more than 20%, and gastroesophageal reflux in 20% to 30% of patients. The incidence of these conditions increases with age and the severity and duration of severe obesity.
Hypertension
Sudden cardiac death myocardial infarction
Cardiomyopathy
Venous stasis disease
Deep venous thrombosis
Pulmonary hypertension
Right-sided heart failure
Obstructive sleep apnea
Hypoventilation syndrome of obesity
Asthma
Metabolic syndrome (abdominal obesity, hypertension, dyslipidemia, insulin resistance)
Type 2 diabetes
Hyperlipidemia
Hypercholesterolemia
Nonalcoholic steatotic hepatitis (NASH) or nonalcoholic fatty liver disease (NAFLD)
Gastroesophageal reflux disease
Cholelithiasis
Degenerative joint disease
Lumbar disk disease
Osteoarthritis
Ventral hernias
Stress urinary incontinence
End-stage renal disease (secondary to diabetes and hypertension)
Menstrual irregularities
Fungal infections
Boils, abscesses
Cancer of the thyroid, prostate, esophagus, kidney, stomach, colon, rectum, gallbladder, pancreas, female cancers of the breast, ovaries, cervix, and endometrium
Pseudotumor cerebri
Depression
Low self-esteem
Stroke
History of physical abuse
History of sexual abuse
Discrimination for employment
Social discrimination
The metabolic syndrome includes type 2 diabetes mellitus (insulin resistance), dyslipidemia, and hypertension. Patients with this constellation of problems are obese, with central body obesity being the primary essential feature (waist circumference >35 inches in women or >40 inches in men). The syndrome is characterized by impaired hepatic uptake of insulin, systemic hyperinsulinemia, and tissue resistance to insulin. Patients with metabolic syndrome are at high risk for early cardiovascular death.
Obesity has been shown to increase the risk of developing cancer of the thyroid, colon, rectum, esophagus, stomach, kidney, prostate, gallbladder, pancreas, breast (postmenopausal), endometrium, ovaries, and cervix. There are several mechanisms that may be responsible for the increased risk of cancer. Obesity increases chronic inflammation, which is linked to the development of esophageal adenocarcinoma through chronic inflammation from gastroesophageal reflux disease (GERD). Fat produces excess levels of estrogen, which is linked to increased risk of endometrial, ovarian, and postmenopausal breast cancer. Increased levels of insulin and insulin-like growth factor 1 is hypothesized to be linked to the development of colon, prostate, kidney, endometrial, and postmenopausal breast cancer.
Not listed in Box 48.1 are the associated societal discriminatory problems that severely obese individuals face. Public facilities in terms of seating, doorways, and restroom facilities often make access to events held in such settings unavailable to a severely obese person. Travel on public transportation is sometimes difficult, if not impossible. Employment discrimination clearly exists for these individuals. Finally, the combination of low self-esteem, a frequent history of sexual or physical abuse, and these social difficulties coalesce to create a very high incidence of depression in the population of morbidly obese patients.
Medical therapy for severe obesity has limited short-term success and almost nonexistent long-term success. Once severely obese, the likelihood that a person will lose enough weight by dietary means alone and remain at a BMI below 35 kg/m 2 is estimated at 3% or less. The NIH consensus conference recognized that for this population of patients, medical therapy has been largely unsuccessful in treating the problem. Review of the clinical trials of lifestyle interventions for prevention of obesity demonstrated that the majority of trials were completely ineffective, and the few that were marginally effective had an extremely small impact on BMI.
One of the most remarkable stories in modern medicine has been the absolute superiority of bariatric surgery over medical therapy for the treatment of morbid obesity and its comorbidities. Multiple long-term follow-up trials comparing morbidly obese diabetics who underwent bariatric surgery with those who did not have shown decreased mortality long term after bariatric surgery as shown in Table 48.2 .
| Study | Surgery Mortality Rate | Medical Mortality Rate | Odds Ratio Or Hazard Ratio Surgical Reduction in Mortality | Notes |
|---|---|---|---|---|
| Swedish Obesity Study | 5.0% | 6.3% | 0.71 HR | Prospective trial of 2010 bariatric surgery and 2037 matched control patients, 15-year follow-up |
| Adams | 2.7% | 4.1% | 0.63 HR | Retrospective matched cohort of 7925 RYGB and 7925 severely obese control patients, mean follow-up 7.1 years |
| Guidry RYGB versus propensity-matched controls | 6.5% | 12.7% | 0.48 Odds ratio | Retrospective matched cohort of 401 RYGB and 401 matched control patients, 10-year follow-up |
| Arteburn | 13.8% | 23.9% | 0.47 HR | Veterans Affairs multisite cohort of 2500 bariatric surgery patients and 7462 matched control patients, 10-year follow-up |
| Kauppilia | 3.6% | 15.2% | 0.74 HR | 5 Nordic countries, population-based study of 49,977 bariatric surgery patients and 494,842 who did not have surgery, 15-year follow-up |
The Swedish Obese Subjects (SOS) study is the first prospective controlled trial to provide long-term data on the effects of bariatric surgery on diabetes, cardiovascular events, cancer, and overall mortality. The study enrolled 2010 bariatric surgery subjects (gastric bypass, 13%; banding, 19%; vertical banded gastroplasty, 68%) and 2037 matched controls who received standard medical treatment and observed the subjects for 10 to 20 years. The SOS study was able to obtain follow-up on 98.9% of the subjects and found at 15 years after initiation that the surgery patients had lost 18% of their body weight, whereas the control group had only a 1% weight loss at 15 years. The long-term sustained weight loss and reduction in comorbid conditions after bariatric surgery resulted in a 29% reduction in mortality in the bariatric surgery patients (adjusted hazard ratio [HR], 0.71; 95% confidence interval, 0.54–0.92; P = 0.01), as shown in Fig. 48.1 . The most common cause of death in the SOS study was cancer (47 in the control group and 29 in the surgery group). The incidence of myocardial infarction was significantly reduced in the surgery group compared with the control group (HR, 0.56), and the surgery group had a lower number of first-time cardiovascular events (HR, 0.67) compared with the control group. Most strikingly, the SOS trial showed an 80% decrease in the annual mortality of diabetic individuals in the surgical weight loss group versus the matched control patients (9% mortality in the surgery group vs. 28% mortality in a control group).
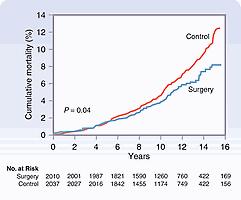
As can be seen in in Table 48.2 , there are multiple studies with long-term follow-up comparing bariatric surgery to matched control groups and found the surgical groups have significant survival advantage (all cause, cancer, cardiovascular), associated with improvement in diabetes, obstructive sleep apnea, dyslipidemia, and hypertension. These studies are convincing evidence that bariatric surgery provides long-term weight loss, resolution of comorbidities, and improvement in mortality. While the SOS study found improvement in all-cause mortality after 10 years of follow-up, the majority of procedures performed were vertical banded gastroplasty but Roux-en-Y gastric bypass (RYGB), a much more effective procedure comprised only 13% of the operations performed. Since RYGB is a more effective procedure, it is not surprising that the Adams and Guidry studies, which compared RYGB to medical treatment, showed convincing mortality differences as early as 3 to 5 years after surgery, much earlier than the Swedish Obesity Study. Similarly, Kauppila studied 49,977 patients undergoing bariatric surgery, of which RYGB made up 73.4% of the operative procedures, and compared to a cohort of obese persons in five Nordic countries and found that all-cause mortality was reduced by 4 years and improved further at 15 years later.
Moreover, in the Adams study, the mortality for the first year was equal in the surgery and control groups (0.53% vs. 0.52%, respectively). The Arterburn study was from the Veterans Affairs (VA) hospitals in the United States, so it had a predominance of males (74% male) as opposed to all of the other reported trials having a majority of female subjects, but it also identified all-cause reduction in mortality at 5 years. The VA study is also remarkable for the higher first year mortality rate in the 2000 to 2005 period (HR, 1.66) than in the 2006 to 2011 period (HR, 0.88), which points to the improvements in surgical care occurring during this period. Despite the higher first year, mortality rate the all-cause mortality rate was lower for the surgery patients than the medical treated group after 5 to 14 years (HR, 0.47).
Adams and colleagues have recently updated the results of the trial reported in 2007 with outcomes measured 12 years after gastric bypass ( N = 418) compared to 417 matched patients who sought but did not undergo surgery and to 321 patients who were matched but never sought to undergo surgery. The improvements in weight and comorbidity were continued at 12 years post-RYGB, as seen in Table 48.3 .
| RYGB | Control Group 1 | Control Group 2 | |
|---|---|---|---|
| Weight loss | −26.9% | −2.0% | 0.0% |
| Body mass index | −11.5 | +0.1 | +1.2 |
| Glucose, mg/dL | −8.0 | +14.4 | +10.5 |
| Systolic blood pressure | +0.1 | +10.1 | +8.3 |
| LDL cholesterol | −11.0 | +19.3 | +16.5 |
| HDL cholesterol | +12.9 | −2.3 | −3.3 |
| Triglycerides | −62.8 | +11.2 | +11.7 |
The data from multiple studies comparing medical to surgical treatment strongly supports that bariatric surgery reduces long-term mortality. But does this long-term effect hold true for older patients who have a higher operative mortality and shorter time to see long-term benefits? Surprisingly, a cohort of 7925 morbidly obese patients matched to an equal number of matched control patients not undergoing surgery showed that long-term mortality was lower in the surgery groups for patients age 35 and older. Patients younger than 35 had a significant increase in externally caused deaths (HR = 2.53, P = 0.009), which was even higher in young women (HR = 3.08, P = 0.005). Nevertheless, this study shows that older patients aged 55 to 74 years old had a significant reduction in long-term mortality and the highest reduction of long-term mortality was in men aged 55 to 74 years.
One perception of bariatric surgery is that it may induce profound unalterable changes in eating that negatively affect the patients’ health-related quality of life (HRQOL). A well-done 12-year prospective study evaluated HRQOL changes after gastric bypass surgery compared to two nonsurgical groups matched for similar demographics. The patients who underwent bariatric surgery had greatly improved QOL in the physical component from before surgery. There were also significant differences between the surgery patients and both nonsurgical groups for both the weight-related HRQOL and the physical HRQOL. The magnitude of improvement after 12 years after gastric bypass surgery from before surgery and between matched control groups supports the conclusion that bariatric surgery improves the patient’s quality of life (QOL). However, this study and the SOS study identified an increase in suicides and self-harm in patients undergoing bariatric surgery. The SOS study suicide and nonfatal health self-harm events were greater in the surgery group than in the matched controls ( N = 87 and 49, respectively, out of 68,528 person-years, adjusted HR = 1.78). Analysis of the Utah long-term study revealed that suicide and nonfatal self-harm events were more frequent after gastric bypass than in the intensive lifestyle group ( n = 341 and 84, respectively, out of 149,582 person-years). The authors concluded that bariatric surgery, in particular gastric bypass, was associated with increased risk of suicide or self-harm, but the absolute risk and numbers of patients do not support not offering bariatric surgery to patients. They recommend that preoperative psychiatric mental health assessment and postoperative monitoring for mental health particularly substance abuse is needed.
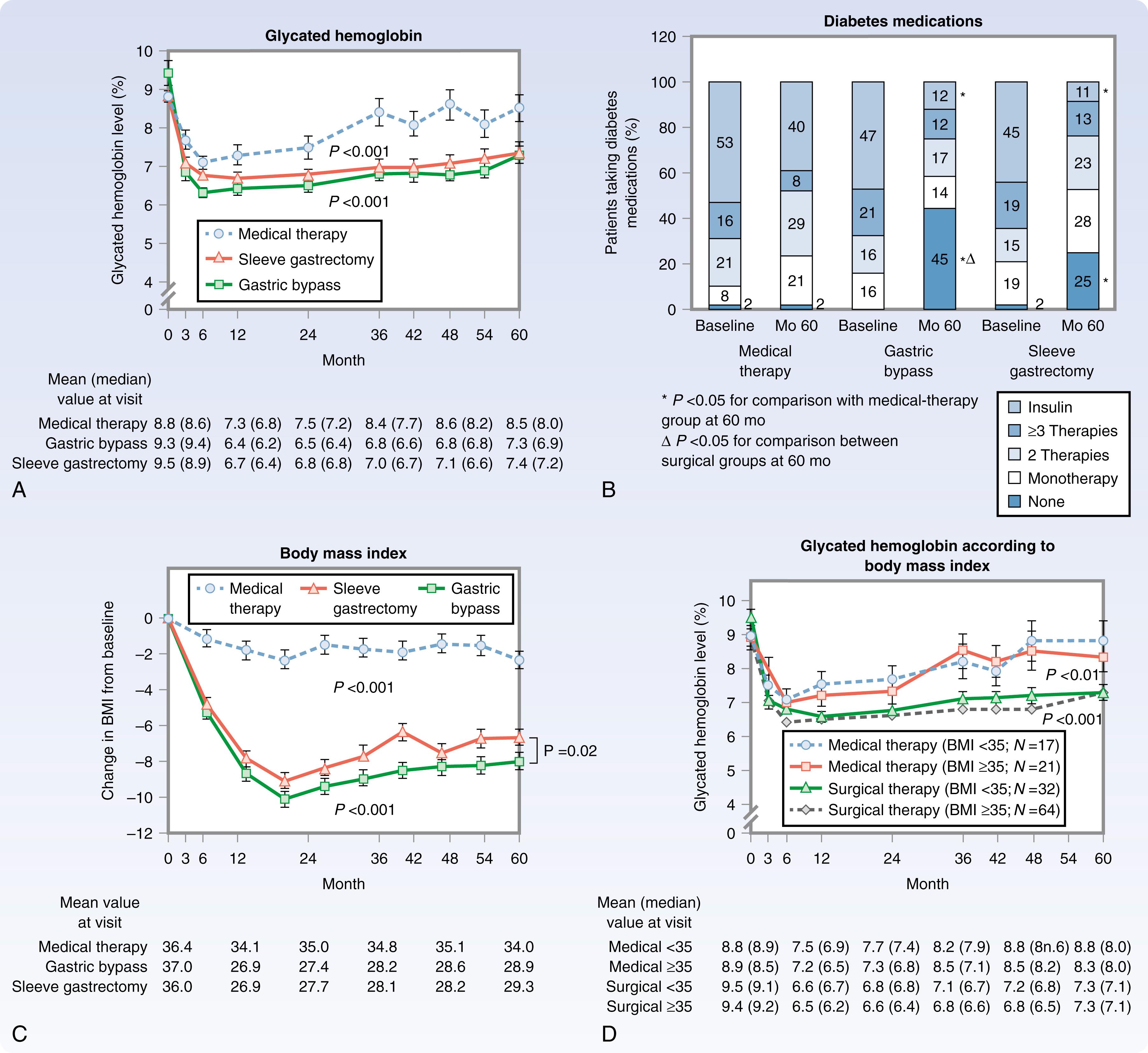
The Surgical Treatment and Medications Potentially Eradicate Diabetes Efficiently (STAMPEDE) trial is 1 of 11 randomized controlled trials (RCTs) that have demonstrated the superiority of bariatric surgery over intensive medical therapy in type 2 diabetes. These trials have shown bariatric surgery to be more effective in glycemic control, weight loss, medication reduction, improvement in lipids, and in QOL, as shown in Fig. 48.2 . Metabolic surgery reduced hemoglobin A1c (HgbA1c) by 2% to 3.5% while medical therapy was only able to reduce HgbA1c by 1% to 1.5%. Short- and long-term (2- to 5- year) results shown in Table 48.4 demonstrate that a significant number of patients (28%–43%) undergoing laparoscopic RYGB (LRYGB) achieved HgbA1c levels less than 6 compared to only 5% to 7% of medically treated patients. It is even more remarkable that the medically treated patients still required intensive medical therapy while the surgery patients had reduced or eliminated diabetic medications. Weight loss for both LRYGB and laparoscopic sleeve gastrectomy (LSG) was far superior to medical therapy. In the case of RYGB, the effect of duodenal bypass on the improvement in diabetes appears to be partially unrelated to weight loss, although in the STAMPEDE trial, the percentage weight loss attained at 1-year postoperative LRYGB was significantly associated with achieving HgbA1c less than 6 at 5 years postoperatively. Thus, whereas there may be a benefit to bypassing the duodenum, the sustained long-term weight loss appears to be an essential element of the salutatory effects of RYGB on type 2 diabetes. LRYGB also improves the other associated medical conditions of the metabolic syndrome, including improvements in hypertension and high-density lipoprotein (HDL) cholesterol, as shown in Table 48.3 , which results in reduced cardiovascular events/deaths. Specifically, diabetic patients who undergo bariatric surgery have a lower rate of incident microvascular disease (16.9% surgery vs. 34.7% medical), with an adjusted odds ratio (AOR) of 0.41. The reduced incidence rate of microvascular disease was largest in the reduction of nephropathy (59% reduction) but also present in diabetic neuropathy (63% reduction) and diabetic retinopathy (45% reduction). The authors of this multi-institution study concluded that bariatric surgery not only improves glycemic control but also significantly reduced the incidence of microvascular disease, which improves survival. The authors argue that this additional evidence of improvement in microvascular disease should push primary care providers to talk to their obese diabetic patients about the benefits of bariatric surgery.
| Schauer LRYGB | Schauer LSG | Ikramuddin Lrygb | Mingrone Lrygb | Simonson Lrygb | |
|---|---|---|---|---|---|
| HgbA1c | |||||
| Medical therapy | 5% ∗ | 5% ∗ | 7% | 27% † | 0% † |
| Surgery | 29% ∗ | 23% ∗ | 38% | 42% † | 42% † |
| Change in Weight (Percentage or kg) | |||||
| Medical therapy | −5% | −5% | −7% | −6.9% | −5.2 kg |
| Surgery | −23% | −19% | −24% | −28.4% | −24.9 kg |
The first study reporting on the effectiveness of surgery in treating obesity and related comorbid conditions, published by Surgery, Gynecology & Obstetrics, in 1955 reported observing “the amelioration of diabetes mellitus following subtotal gastrectomy.” A few decades later, the wide acceptance of bariatric surgery as treatment for severe obesity has given significant momentum to study the physiology of weight loss surgery. The initial prevailing theory behind bariatric surgery was based on two primary mechanisms for surgically induced weight loss: caloric restriction and nutrient malabsorption. There is little question that the reduction in caloric intake and resulting weight loss are responsible for much of the improvement in comorbidit-comorbidities after bariatric surgery. However, after taking into account the more recent scientific evidence, the concepts of restriction and malabsorption do not fully explain the metabolic effects of bariatric surgery. In fact, the mechanisms seem to extend beyond the magnitude of weight loss alone to include effects on central nervous system regulation of appetite and metabolism and improvements in insulin secretion and insulin sensitivity. Long-term weight loss, improvement in glucose metabolism, and other metabolic effects clearly result from significant postoperative changes in the enteroencephalic and the enteroinsular endocrine axes, as shown in Table 48.5 .
| Restrictive | Malabsorption | Enteroencephalic Endocrine Axis | Enteroinsular Axis | |
|---|---|---|---|---|
| Vertical banded gastroplasty historical | ++++ | 0 | 0 | 0 |
| Lap adjustable gastric banding | ++++ | 0 | 0 | 0 |
| Laparoscopic sleeve gastrectomy | ++++ | + | ++++ | ++ |
| Roux-en-Y gastric bypass | ++++ | +++ | ++++ | +++ |
| Biliopancreatic diversion/duodenal switch | ++ | ++++ | +++ | ++++ |
RYGB and LSG are known to increase satiety and reduce hunger. During the periprandial phase, the interplay between the gastrointestinal tract and the regulatory centers of the brain has been shown to activate complex neural networks to modulate food intake. In the central nervous system, the hypothalamus is the key area involved in the regulation of appetite. The hypothalamus regulates energy intake using a complex system of anorexic and orexigenic neuronal signaling. NPY is the dominant hormonal signal regulating energy intake. Release of NPY is modulated by hormones secreted into the circulation by gastrointestinal cells, such as ghrelin and peptide YY (PYY).
Ghrelin is secreted into the circulation by P/D1 cells located in the fundus of the stomach. Ghrelin levels increase significantly before a meal and levels quickly diminish postprandially. Ghrelin activates receptors in the arcuate nucleus and the lateral hypothalamus, stimulating the synthesis of NPY and agouti-related protein. This activation creates an orexigenic signal. The role of ghrelin in the postbariatric surgery regulation of appetite has been extensively studied, sometimes with controversial results. Most studies show that ghrelin levels fall significantly following LSG. In patients who undergo RYGB, the results of various studies are contradictory, especially when they compare fasting levels. A recent cross-sectional study performed by Svane and colleagues has finally provided more definitive data regarding ghrelin levels during the periprandial period in postbariatric surgery patients. In this study, the postprandial areas under the curve (AUCs) for total and acylated ghrelin were significantly lower after LSG as compared with RYGB and controls. In addition, total ghrelin AUC was significantly lower after RYGB as compared with controls. This study provides significant evidence that anatomic changes after bariatric surgery reduce the postprandial secretion of ghrelin and thereby reduce hunger signals.
PYY is another hormone that acts on NPY neurons. L-cells found throughout the small and large intestines secrete PYY in response to the presence of nutrients within the lumen of the distal gut. PYY exerts an anorexigenic effect on NPY neurons. Svane and colleagues showed that postprandial PYY levels are significantly affected by bariatric surgery. Patients after RYGB have significantly higher PYY peak and AUC than LSG and matched controls. No difference in PYY AUC was observed between LSG and matched obese controls.
The remission/amelioration of type 2 diabetes mellitus is considered one of the major benefits after bariatric surgery. Although the mechanisms involved in type 2 diabetes mellitus remission are not completely understood, the improved insulin action, beta-cell function, and modulative effect of gut hormones in the enteroinsular axis appear to play a significant role. Among these hormones, glucagon-like peptide-1 (GLP-1) appears to be the major player. GLP-1 is released by L-cells in the distal gastrointestinal tract, and levels increase in response to the presence of nutrients in the lumen of the distal or hindgut intestines. In obese subjects, there is a delay in the postprandial release of GLP-1 and overall significantly reduced circulating levels of the peptide. GLP-1 is known to exert anorexigenic effects in obese subjects secondary to a reduction in gastric emptying. However, GLP-1 action is more predominant in the enteroinsular axis. GLP-1 is part of a family of peptides involved in the synthesis, secretion, and regulation of insulin: the incretins. In the enteroinsular axis, GLP-1 has many physiologic functions, including the stimulation of insulin secretion by the pancreas, an increase in the insulin sensitivity of pancreatic cells (α-cells and β-cells), and inhibition of glucagon secretion. Many studies have shown that GLP-1 is a major driver of insulin secretion after bariatric surgery. GLP-1 has been found to be consistently elevated (peak and postprandial AUC) in response to both RYGB and LSG, with significant higher magnitude in RYGB than LSG. More important, the effect of both RYGB and LSG on postprandial GLP-1 secretion is not observed in subjects with an equivalent degree of weight loss achieved only by caloric restriction. These results provide further evidence of a metabolic effect for bariatric surgery that it is independent of weight loss.
Selection of patients for bariatric surgery is based on currently accepted NIH and American Heart Association/American College of Cardiology/The Obesity Society (AHA/ACC/TOS) guidelines. Patients must have a BMI greater than 40 kg/m 2 without associated comorbid medical conditions or a BMI greater than 35 kg/m 2 with an associated comorbid medical problem. They must have also failed dietary and behavioral therapy. Several criteria must also be used as guidelines for indications for surgery, including psychiatric stability, motivated attitude, and ability to comprehend the nature of the operation and its resultant changes in eating behavior and lifestyle. Criteria for eligibility for bariatric surgery are given in Box 48.2 . An inability to fulfill these criteria is a contraindication to bariatric surgery.
Patients must meet the following criteria for consideration for bariatric surgery:
Body mass index (BMI) >40 kg/m 2 or BMI >35 kg/m 2 with an associated medical comorbidity worsened by obesity
Failed dietary therapy
Psychiatrically stable without alcohol dependence or illegal drug use
Knowledgeable about the operation and its sequelae
Motivated individual
Medical problems not precluding probable survival from surgery
One criterion not listed in Box 48.2 that unfortunately is often a significant issue for a severely obese patient is insurance coverage for the operation. The Affordable Care Act (ACA) mandates that patients covered under the ACA Marketplace health plans must receive obesity screening and counseling without charging of copayment or coinsurance even if the patient has not met the yearly deductible. Remarkably, the ACA does not mandate coverage for bariatric surgery, and most Federal Marketplace insurance coverage does not cover bariatric surgery, despite that the writers of the ACA wanted to prevent bias against preexisting conditions and the overwhelming evidence that bariatric surgery is the only effective modality of long-term treatment in this population. Meanwhile, the medical societies recognize the need to refer severely obese individuals to bariatric surgeons particularly for the care of patients with type 2 diabetes. In the most recent guidelines, the ADA states: “Metabolic surgery should be recommended as an option to treat type 2 diabetes in appropriate surgical candidates with BMI >40 kg/m 2 (BMI >37.5 kg/m 2 in Asian Americans) who do not achieve durable weight loss and improvement in comorbidities (including hyperglycemia) with reasonable nonsurgical methods.”
The Centers for Medicare and Medicaid Services (CMS), the federal agency that sets Medicare guidelines, established criteria for coverage of bariatric surgery in 2006. The ruling required that only surgeons in hospitals that are designated Centers of Excellence perform bariatric surgery. The unique requirements for Medicare beneficiaries were at least partly due to concern by policymakers that the morbidity and mortality associated with bariatric surgery were high and that the explosive growth in the number of hospitals and surgeons performing the operations did not match the hospital oversight of these procedures and the resulting complications.
After the imposition of the CMS mandate in 2006, CMS removed the requirements for facility and surgeon certification to perform bariatric surgery in September 2013 partly based on improvements in bariatric surgery outcomes since the 2006 ruling. Today, greater than 88% of bariatric procedures are performed at Metabolic and Bariatric Accredited and Quality Improvement Programs (MBSAQIP).
Accreditation, improvement in communication, and structured quality programs have been important in the improvements in quality of care. The American Society for Metabolic and Bariatric Surgery (ASMBS) has published recommendations on credentialing of bariatric surgeons by hospitals recently that include the need for active participation within a structured bariatric surgery program and quality improvement program. Although hospital-wide coordination of care, communication, and multidisciplinary teams must function well to achieve best results inbariatric surgery, the technical skill of the individual surgeon was highly correlated with significantly fewer complications, readmissions, reoperations, and visits to the emergency department. There is considerable variation in serious complications within 30 days on the index bariatric procedure even among Bariatric Centers of Excellence. The differences in outcomes could not be totally explained by volume, case mix, or procedure type rates that the authors suggest are related to the skill of the individual surgeon and to inconsistent adherence to the pathways promulgated by the Centers of Excellence. In summary, these studies show that the surgeon not only must achieve technical proficiency but also must engage the entire operative and hospital team to achieve excellent outcomes.
Bariatric operative mortality has declined around the world to remarkably low levels. In France, the mortality after 6056 laparoscopic adjustable gastric banding (LAGB) operations was zero in 2012 and LRYGB mortality has decreased threefold from 0.33% to 0.11% from 2008 to 2012. In France, the independent factors associated with operative mortality were male sex (AOR, 1.94), age older than 50 (AOR, 3.69), BMI < 50 kg/m 2 (AOR, 2.05), and type 2 diabetes (AOR, 1.6). There was also a graded relationship between increased hospital volume and decreased operative mortality.
Medical contraindications to bariatric surgery are relative, and all patients with comorbid conditions are at greater risk. The surgeon must ensure that these risks are well understood by all patients before bariatric surgery, especially those at high risk. Ideally, several family members are included in these discussions. There are certain individuals who have end-stage organ dysfunction of the heart, lungs, or both. These patients are unlikely to gain the benefit of longevity and improved health.
Surgery is contraindicated in patients who are unable to ambulate because their level of debility precludes recovery during the rapid weight loss phase after surgery. Prader-Willi syndrome is another absolute contraindication because no surgical therapy affects the constant need to eat by these patients.
The U.S. Food and Drug Administration expanded the use of the LAP-BAND to include patients with BMI between 30 and 34 kg/m 2 who have an existing condition related to their obesity. Other contraindications to LAGB include cirrhosis, portal hypertension, autoimmune connective tissue disorders, chronic inflammatory conditions such as inflammatory bowel disease, and need for chronic administration of steroids.
Patients who weigh more than 500 lbs are at increased risk for mortality and have more complications. Many options for diagnostic testing, such as computed tomography (CT), are exceeded by this weight limit. At this weight, operating room tables, moving and lift equipment and teams, blood pressure cuffs, sequential compression device (SCD) boots, and any sort of invasive bedside procedures such as central venous catheters become extraordinarily difficult. It has been my practice to require patients weighing more than 500 lbs to lose weight down to that level by nonoperative methods.
Age is a controversial contraindication to bariatric surgery. For adolescents, most pediatric and bariatric surgeons recommend that the operation be performed after the major growth spurt (mid to late teens), thus allowing increased maturity on the part of the patient. The Teen–Longitudinal Assessment of Bariatric Surgery (Teen-LABS) demonstrated that severely obese teens (<19 years) had multiple comorbid conditions and could undergo one of three commonly performed operations (LRYGB, LSG, and LAGB) with no mortality and a favorable short-term complication profile. Three-year follow-up after LRYGB or LSG in Teen-LABS showed excellent and sustained weight loss and resolution of diabetes in 95% of patients who had type 2 diabetes at baseline, a figure far higher than the 50% to 70% of adult bariatric surgery patients. The authors hypothesize that adults accumulate pounds-years that are less reversible than in the adolescent population. They conclude LRYGB and LSG are effective at 3 years in adolescents.
Although in our practice, we have generally set the age of 65 years as a rough cutoff for performing gastric bypass and 70 years for LSG, patients older than 70 years have been individually evaluated. Such evaluations focus on the patient’s relative physiologic age and potential for longevity rather than chronologic age. The duration and degree of obesity are the most important factors in evaluating an older patient. In general, the duration and the severity of obesity and the number of comorbid medical problems that exist lower the potential for such individuals to benefit from bariatric surgery.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here