Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Monoclonal gammopathy of undetermined significance (MGUS) is a premalignant clonal plasma cell disorder. It is characterized by the presence of a monoclonal (M) protein in the serum or urine that is commonly discovered during laboratory work-up of a variety disorders. MGUS is a precursor to various hematologic disorders including multiple myeloma (MM), Waldenström macroglobulinemia (WM), solitary plasmacytoma, and immunoglobulin light chain amyloidosis (AL). Smoldering MM (SMM) is a clinically-defined disorder similar to MGUS, but has a much higher risk of progression to MM or a related disorder. SMM is not a unique intermediate biologic stage between MGUS and MM, but it consists of a heterogeneous mixture of patients, some of whom have a biologic premalignancy and have a clinical course resembling MGUS, and some who have an asymptomatic malignancy that will develop end-organ damage typical of MM within a few years. Unfortunately, at the current time, there is no single pathologic or molecular feature that reliably distinguishes patients with SMM who have premalignancy from those with asymptomatic malignancy. MGUS and SMM are usually asymptomatic but can be causally related to a variety of systemic disorders. They also carry a lifelong risk of progression to MM (see Chapter 91 ) or a related malignancy (see box on Diagnosis of MGUS and SMM: Role of Screening .)
MGUS is defined by the presence of a serum monoclonal (M) protein of <3 g/dL (or increased serum free light chain (FLC) level with abnormal free light chain (FLC ratio), <10% clonal bone marrow plasma cells (BMPC) with no evidence of SMM, end-organ damage, or other myeloma defining events (MDEs) ( Table 90.1 ). There are three major subtypes of MGUS: IgM MGUS, non-IgM MGUS, and light-chain MGUS (LC-MGUS). IgM MGUS carries a risk of progression to WM (see Chapter 92 ), although patients can rarely develop IgM MM. Non-IgM MGUS is associated with a risk of progression to MM, and LC-MGUS is a precursor of light chain MM. All forms of MGUS can lead to AL amyloidosis (see Chapter 93 ).
| Disorder | Disease Definition |
|---|---|
| IgM monoclonal gammopathy of undetermined significance (IgM MGUS) |
|
| Non-IgM monoclonal gammopathy of undetermined significance (MGUS) |
|
| Light chain MGUS |
|
| Smoldering multiple myeloma |
|
| Multiple myeloma |
|
| Smoldering Waldenström macroglobulinemia (also referred to as indolent or asymptomatic Waldenström’s macroglobulinemia) |
|
| Waldenström macroglobulinemia |
|
| Solitary plasmacytoma |
|
| Solitary plasmacytoma with minimal marrow involvement b |
|
| Systemic AL amyloidosis |
|
| POEMS syndrome |
|
a A bone marrow can be deferred in patients with low-risk MGUS (IgG type, M protein <15 g/L, normal free light chain ratio) in whom there are no clinical features concerning for myeloma.
b Solitary plasmacytoma with 10% or more clonal plasma cells is considered as multiple myeloma.
With conventional testing, MGUS can be detected in approximately 4% of normal persons 50 years of age and older. Age-adjusted rates are greater in men than in women. The prevalence increases with age, exceeding 7.5% among those 85 years or older. Sensitive mass spectrometry studies show that the overall prevalence of MGUS is higher and is estimated at approximately 5% of the general population age 50 years and older. The prevalence of MGUS is higher in Blacks compared to Whites. In a large population-based study ( n = 12,482) of persons 50 years of age and older, using samples from the National Health and Nutrition Examination Survey (NHANES), the adjusted prevalence of MGUS was significantly higher in Blacks (3.7%) compared to Whites (2.3%) or Hispanics (1.8%) ( P < .001). A subsequent NHANES study found that the racial disparity in prevalence occurs even at younger ages, with MGUS having an onset approximately a decade earlier in Blacks compared with Whites. Table 90.2 gives the estimated prevalence of MGUS by race and age based on NHANES studies.
| Age Group (Years) | Blacks % (95%CI) | Whites % (95%CI) | Hispanic American % (95%CI) | Total a % (95%CI) |
|---|---|---|---|---|
| 10–19 | 0.00 (NA) | 0.00 (NA) | 0.11 (0.02–0.84) | 0.01 (0.00–0.07) |
| 20–29 | 0.26 (0.08–0.84) | 0.00 (NA) | 0.00 (NA) | 0.03 (0.01–0.11) |
| 30–39 | 0.81 (0.43–1.53) | 0.32 (0.11–0.99) | 0.23 (0.06–0.79) | 0.49 (0.27–0.91) |
| 40–49 | 3.26 (2.04–5.18) | 0.53 (0.20–1.37) | 2.20 (1.22–3.95) | 0.88 (0.53–1.47) |
| 50–59 | 2.19 (1.32–3.60) | 1.01 (0.62–1.66) | 0.85 (0.33–2.23) | 1.20 (0.83–1.72) |
| 60–69 | 3.48 (2.26–5.33) | 2.43 (1.72–3.43) | 2.41 (1.69–3.44) | 2.45 (1.85–3.23) |
| 70–79 | 5.67 (3.66–8.70) | 3.44 (2.55–4.63) | 2.51 (1.36–4.58) | 3.43 (2.64–4.47) |
| 80+ | 8.56 (4.92–14.47) | 4.42 (3.31–5.87) | 3.97 (1.33–11.22) | 4.58 (3.54–5.90) |
The increased risk of MGUS seen in African Americans has also been found in Black people living in Ghana. Furthermore, studies show that the racial disparity between Blacks and Whites persists even after adjusting for socio-economic status, suggesting that the disparity in incidence is more likely due to genetics rather than environmental factors. Recent studies show that the t(11;14) translocation type of MGUS may be responsible for most of the increased disparity in MGUS between Blacks and Whites. The probability of having one of three specific subtypes, namely t(11;14), t(14;16), or t(14;20) was significantly higher in the 120 individuals with the highest African ancestry (≥80%) compared with the 235 individuals with lowest African ancestry (<0.1%) (51% vs. 33%, respectively, P value = 0.008). Studies in other racial groups are limited, but so far an increased risk has not been seen in Japanese individuals, or in Hispanics living in the United States when compared to the White population of Olmsted County.
The annual incidence of MGUS in males is estimated to be 120/100,000 at age 50, and rises to 530/100,000 at age 90 years. The rates for women are 60/100,000 at age 50, and 370/100,000 at age 90. When MGUS is first diagnosed, it is estimated that the condition has already been present in an undiagnosed form for a median of over 10 years.
First-degree relatives of patients with MGUS and MM have a two- or threefold higher risk of MGUS compared to those with no known affected relatives. Obesity, exposure to pesticides, and immunosuppression are also known risk factors for MGUS.
Routine screening is not recommended for MGUS since there are no randomized trials to support such a strategy. There are also no interventions to offer to patients who are diagnosed with MGUS. However, we do not have interventions to prevent end organ damage for patients with high-risk SMM. Since these patients are asymptomatic, diagnosis is incidental. Thus many patients who can potentially benefit from early intervention will go unidentified. This dilemma has led to consideration of screening in high-risk populations, namely Blacks with one affected first-degree relative with MM, and in all others with two or more affected first-degree relatives. In these subsets of the population, the prevalence of MGUS is likely greater than 10%–15%, and the probability of finding patients with high-risk SMM is higher. In our practice, we recommend such selected screening, until we have results of a large, randomized trial in Iceland, which is investigating the role of screening as well as the role of more aggressive follow-up of monoclonal gammopathies.
MGUS likely originates when plasma cells are dividing in response to antigenic stimulation. During this period of antigenic stimulation they acquire a primary cytogenetic abnormality that established the MGUS clone ( Fig. 90.1 ). The primary cytogenetic abnormalities associated with the onset of MGUS consist of two major types: trisomies and immunoglobulin heavy chain (IgH) translocations ( Table 90.3 ). IgH translocations involve the immunoglobulin heavy chain switch region on chromosome 14q32, and one of five common partner chromosomes loci: 11q13 (CCND1 [cyclin D1 gene]), 4p16.3 (FGFR-3 and MMSET), 6p21 (CCND3 [cyclin D3 gene]), 16q23 (c-MAF), and 20q11 (MAF-B).
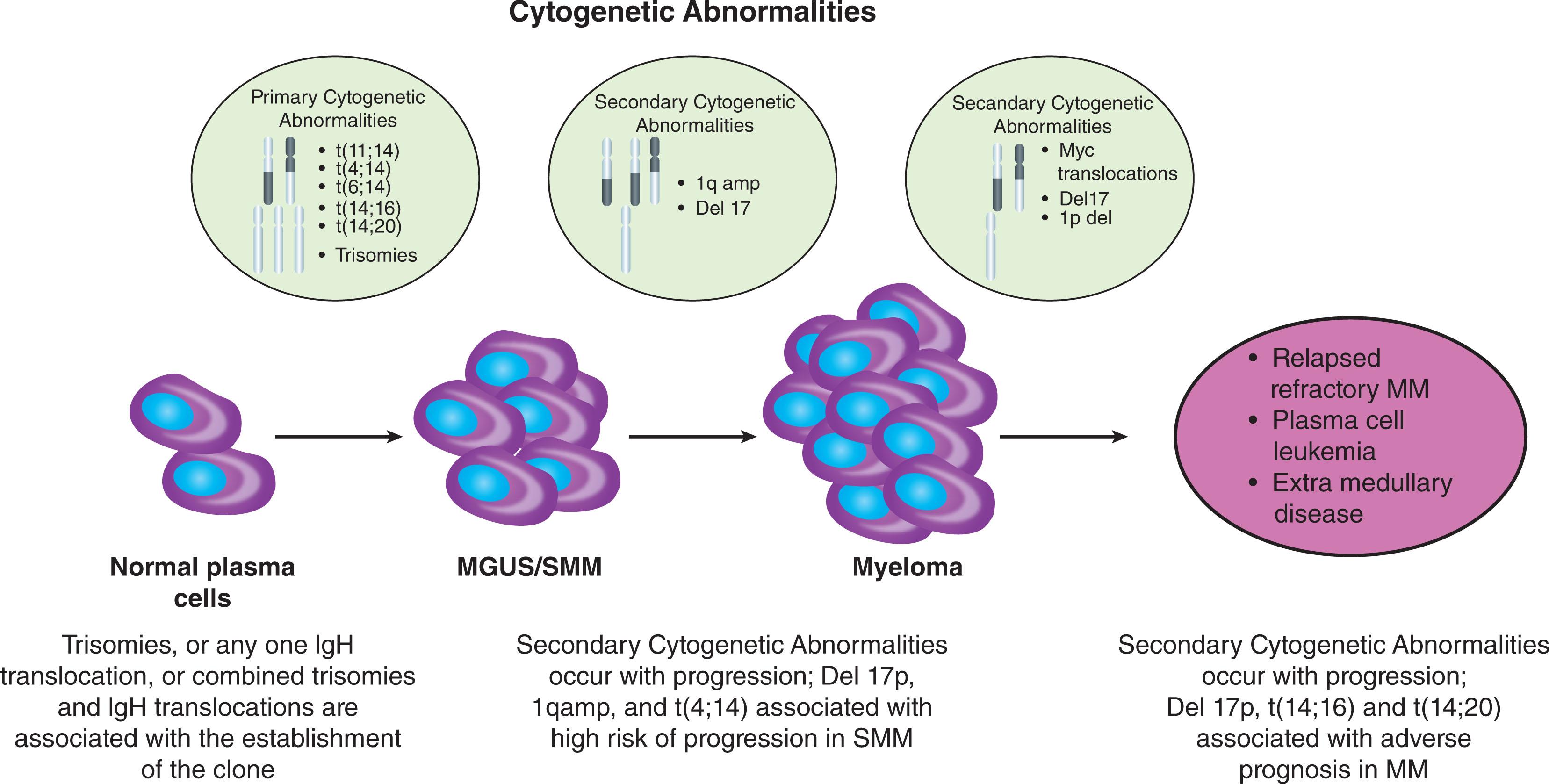
| Subtype | Gene(s) Affected |
|---|---|
| Trisomies | Usually involving odd-numbered chromosomes with the exception of chromosomes 1, 13, and 21. Multiple genes affected |
| Primary immunoglobulin rearrangements a | |
| t(11;14) (q13;q32) | CCND1 (cyclin D1) |
| t(4;14) (p16;q32) | FGFR-3 and MMSET |
| t(14;16) (q32;q23) | C-MAF |
| t(14;20) (q32;q11) | MAFB |
| t(6;14) (p21;q32) | CCND3 (cyclin D3) |
| Other IgH translocations | The genes affected vary depending on the partner chromosome |
a Other rare partner chromosomes have been described. Some patients have combined IgH translocations and trisomies, but they should be classified based on the IgH translocation.
MGUS may stay dormant or acquire secondary cytogenetic abnormalities and progress to MM or a related malignancy. The rate of progression of MGUS is approximately 1% per year regardless of the known duration of antecedent MGUS, indicating a random hit model of progression. Although the precise pathogenetic steps are unclear, there appear to be two main pathways involved, namely, MYC translocations and mutations in the MAP kinase pathway. The presence of MYC structural variants is associated with a significantly shorter median time to progression (TTP) compared to the absence of such abnormalities. Other abnormalities associated with progression include chromosome 1q21 (gain 1q), p16 methylation, p53 mutations, and changes in the bone marrow microenvironment, including induction of angiogenesis and suppression of cell-mediated immunity, affecting things such as T-cell production.
Progression is also associated with increase in receptor activator of nuclear factor κB ligand (RANKL) expression by osteoblasts and possibly plasma cells accompanied by decreased stromal cell secretion of the RANKL decoy receptor, osteoprotegerin (OPG). There is also release of macrophage inflammatory protein–1α (MIP-1α) and MIP-1β by myeloma cells. Collectively these changes promote osteoclast activation through the nuclear factor κB (NF-κB) pathway. In addition, there is simultaneous inhibition of osteoblasts mediated by increased dickkopf 1 (DKK-1) expression by myeloma cells. The combination of osteoclast activation and inhibition of osteoblast differentiation is thought to be the mechanism behind the development of osteolytic lesions in MM, and may also explain increased risk of fractures seen in MGUS even in absence of progression to MM.
MGUS is an asymptomatic condition. It is typically detected as an incidental finding when electrophoresis and immunofixation of the serum and/or urine, or the serum FLC assay are performed during the work-up of suspected MM or WM, including symptoms such as unexplained weakness or fatigue, increased erythrocyte sedimentation rate, anemia, unexplained back pain, osteoporosis, osteolytic lesions or fractures, hypercalcemia, proteinuria, renal insufficiency, or recurrent infections. MGUS may also be detected during work-up of patients with symptoms suggestive of AL amyloidosis such as unexplained sensorimotor peripheral neuropathy, carpal tunnel syndrome, refractory congestive heart failure, nephrotic syndrome, orthostatic hypotension, malabsorption, weight loss, change in the tongue or voice, paresthesias, numbness, increased bruising, bleeding, and steatorrhea.
Although most patients diagnosed with MGUS are asymptomatic, some can have disorders that are causally related to the M protein, without any evidence of overt progression to MM or related malignancy ( Table 90.4 ). In fact, increasingly MGUS is diagnosed in such situations, during work-up of unexplained renal, neuropathic, or other organ dysfunction. Over a hundred different medical conditions have been reported to be associated with MGUS, but most such reported associations are coincidental rather than causal. The important causal related disorders are discussed below.
| Primary Organ Involved | Clinical Presentation | Role of Monoclonal Protein/Pathophysiology |
|---|---|---|
| Schnitzler syndrome | Chronic urticaria, fever, bone pain, IgM-MGUS | Unclear |
| Insulin autoimmune syndrome | Episodic confusion, diaphoresis, dizziness, lethargy, palpitation, seizure | Antibody to insulin causing its inactivation |
| TEMPI | Telangiectasias, erythrocytosis, elevated erythropoietin level, MGUS, perinephric fluid collections, and intrapulmonary shunting | Unclear |
| Rheumatologic | ||
| scleromyxedema | Waxy papules or plaques, arthralgia, restrictive lung disease, seizure | Unclear |
| Progressive glomerulonephritis with monoclonal immunoglobulin deposits | Hematuria, hypertension, proteinuria, renal impairment, mostly IgG3κ-MGUS | Granular deposition of immunoglobulin in glomeruli |
| Sporadic late-onset nemaline myopathy | Muscular weakness and atrophy frequently resulting in “head drop,” respiratory insufficiency, congestive heart failure | Unclear |
| Crystalline keratopathy | Decreased visual acuity, corneal opacity, IgGκ-MGUS | Corneal deposition of antibody forming a crystalline structure |
| Crystal-storing histiocytosis | Mass or tissue infiltration, which may involve the bone marrow, breast, gastrointestinal tract, kidneys, lymph node, skin, or spleen | Accumulation of light chain crystals in histiocytes |
Approximately 3% to 5% of patients with monoclonal gammopathy have a peripheral neuropathy, and approximately 3% to 5% of patients with a peripheral neuropathy have evidence of an M protein. IgM MGUS associated neuropathy presents as a chronic distal, acquired, demyelinating, symmetric sensory neuropathy (DADS-M). In approximately half of these patients, the IgM M protein binds to myelin-associated glycoprotein (MAG). In contrast, patients with non-IgM associated neuropathy have a varied presentation, but the clearest association is in patients who present with features similar to chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) with predominantly motor involvement with distal and proximal involvement. MGUS neuropathy must be differentiated from two other plasma cell disorders that cause neuropathy that have been well characterized and have strict diagnostic criteria, namely immunoglobulin light chain (AL) amyloidosis, and neuropathy associated with POEMS (polyneuropathy, organomegaly).
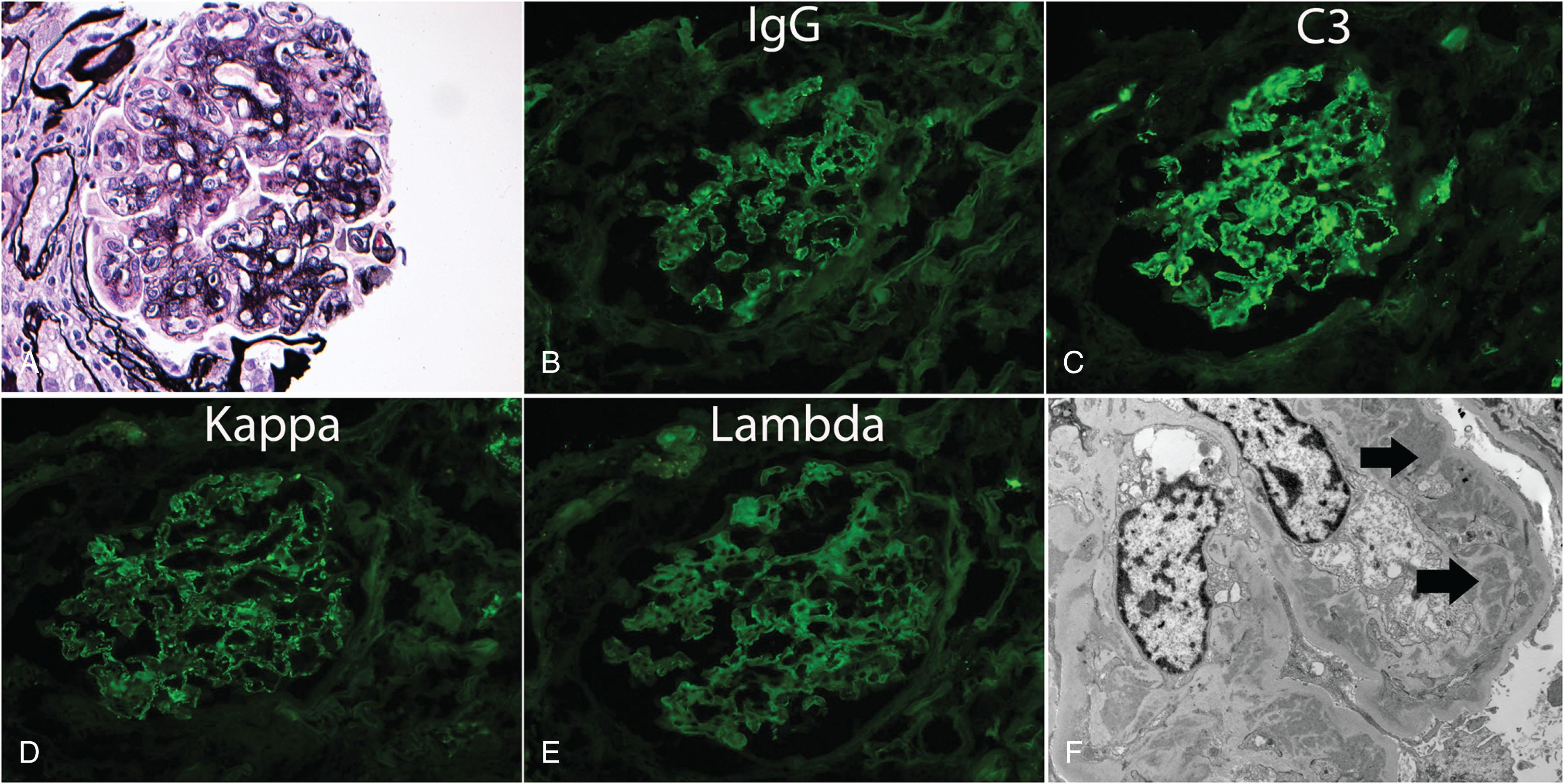
A substantial proportion of cases with “idiopathic” immune-complex mediated membranoproliferative glomerulonephritis (MPGN) are now felt to be related to MGUS. MGUS can cause proliferative glomerulonephritis by direct deposition of M protein in the mesangium and along capillary walls, resulting in an immune complex-mediated proliferative glomerulonephritis. It can also cause glomerulonephritis by, indirectly, activating the alternative pathway of complement resulting in deposition of complement factors in the glomeruli, resulting in C3 glomerulopathy. The term monoclonal gammopathy of renal significance (MGRS) is used to describe renal diseases such as those caused by a premalignant MGUS clone, and the aforementioned M protein-related disorders such as AL amyloidosis and monoclonal immunoglobulin deposition disease.
A variety of dermatologic diseases have been associated with MGUS including lichen myxedematosus (papular mucinosis, scleromyxedema), scleredema (Buschke disease), pyoderma gangrenosum, diffuse plane xanthomatosis, and subcorneal pustular dermatosis. Other skin disorders associated with MGUS are necrobiotic xanthogranuloma and Schnitzler syndrome, a rare disorder characterized by chronic urticaria and an IgM monoclonal gammopathy.
MGUS has been associated with an increased risk of fractures and deep vein thrombosis, and has been linked to chronic lymphocytic leukemia, hairy cell leukemia, T-cell lymphoma, and other malignancies, but the significance of these associations is not known. MGUS has also been associated with acquired von Willebrand disease, Gaucher disease, pure red cell aplasia, connective tissue disorders, various neurologic disorders. and can occasionally be the cause of systemic capillary leak syndrome.
The hallmark of MGUS is the presence of an M protein in the serum and/or urine. M proteins are detected using protein electrophoresis of the serum and urine. The M protein appears as a narrow peak (like a church spire) on the densitometer tracing or as a dense, discrete band on agarose gel ( Fig. 90.2 ). Protein electrophoresis also permits quantitation of M proteins. Urine electrophoresis requires a 24-hour urine collection. Immunofixation of the serum and urine is more sensitive than protein electrophoresis and is used to identify the type of M protein heavy and light chains (IgM kappa, IgG lambda, etc.).

The FLC assay is a nephelometric assay that measures free kappa (κ) and lambda (λ) light chains that are not bound to intact immunoglobulin. Patients with abnormally low kappa/lambda FLC ratios are considered to have a monoclonal lambda FLC, and those with abnormally high ratios are defined as having a monoclonal kappa FLC. The serum FLC assay is more sensitive than electrophoresis or immunofixation in detecting free monoclonal light chains, and must be done at diagnosis in conjunction with those tests.
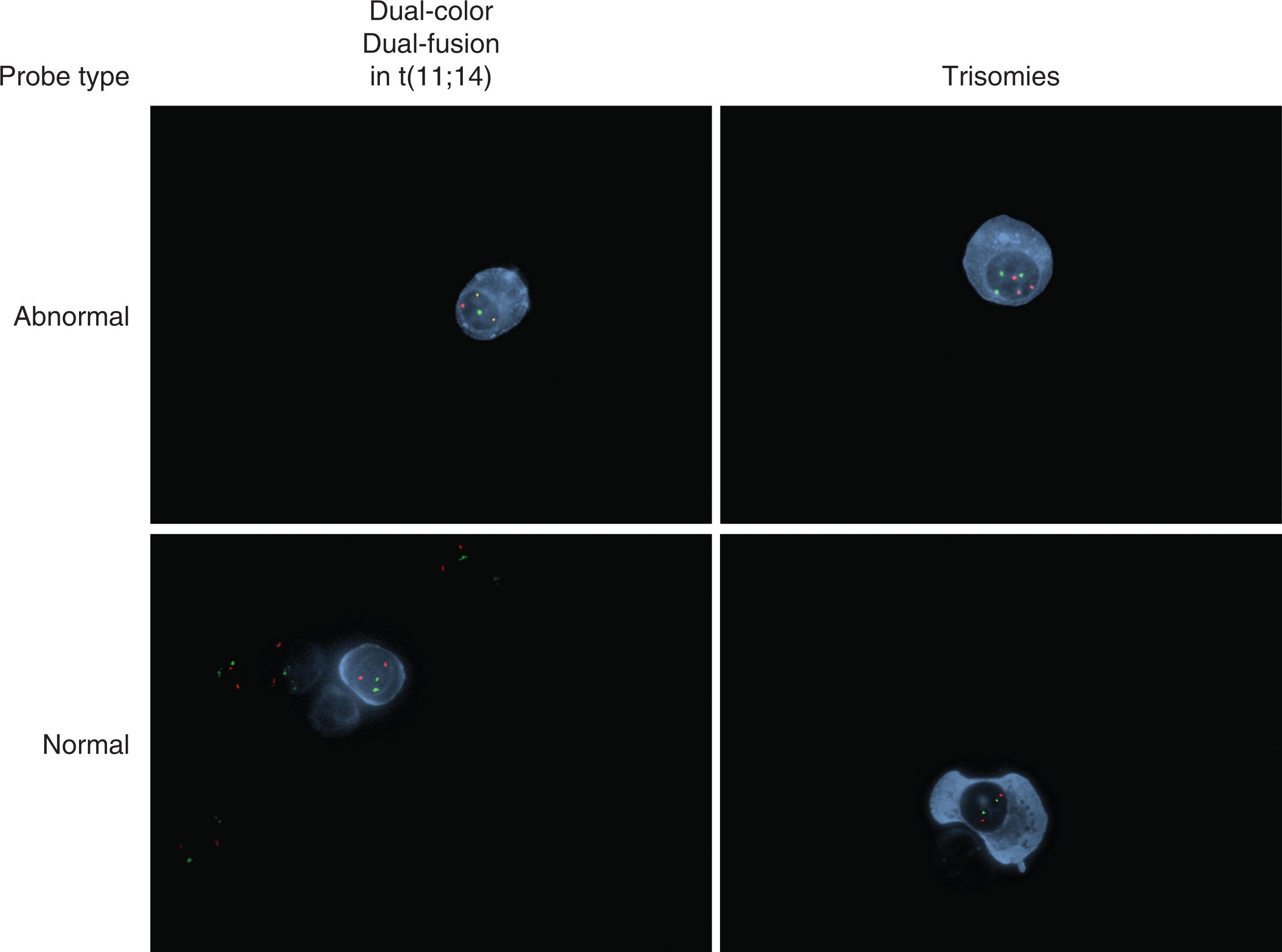
At diagnosis, osteolytic bone disease must be excluded in order to distinguish MGUS from MM. The preferred method to detect bone disease is a whole-body low-dose computed tomography (CT). Positron emission tomography-CT (PET-CT) is an equally sensitive alternative to whole body low-dose CT. A unilateral bone marrow aspiration and biopsy, if indicated, must show less than 10% clonal BMPCs. For flow cytometry, plasma cells in MGUS typically stain positive for CD38 and CD138, and are positive only for one of the two light chain isotypes, indicating they are monoclonal (either kappa or lambda). Baseline cytogenetic abnormalities are best ascertained by fluorescent in situ hybridization (FISH) testing on the bone marrow sample at diagnosis ( Fig. 90.3 ). In patients suspected of monoclonal protein related disorders, confirmatory testing is needed, such as renal biopsy in patients with monoclonal gammopathy associated proliferative glomerulonephritis ( Fig. 90.4 ).
MGUS is differentiated from MM and SMM by the size of the M protein, the BMPC percentage, and the presence or absence of anemia, renal failure, hypercalcemia, or lytic bone lesions (see Table 90.1 ). Because anemia and renal insufficiency are relatively common in the elderly population with MGUS, the causes of these conditions should be carefully investigated with adequate laboratory studies.
MGUS is associated with a life-long risk of progression to MM or a related disorder. In a study of 1384 patients from Southeastern Minnesota, the risk of progression of MGUS to MM or related malignancy was 1% per year. The true life-time probability of progression is lower when competing causes of death are taken into account, approximately 11% at 25 years. The risk of progression with MGUS does not diminish with time, even after over 30 years of follow-up. Progression events include MM, WM, solitary plasmacytoma, and AL amyloidosis. The risk of progression from MGUS has been estimated in several other studies, and the results mirror those seen in the Southeastern Minnesota study. No findings from the diagnosis of MGUS can reliably distinguish patients whose condition will remain stable indefinitely from those in whom MM or related malignancy develops. However, there are several known prognostic factors that assist in estimation of the risk of progression for appropriate counseling and management.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here