Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
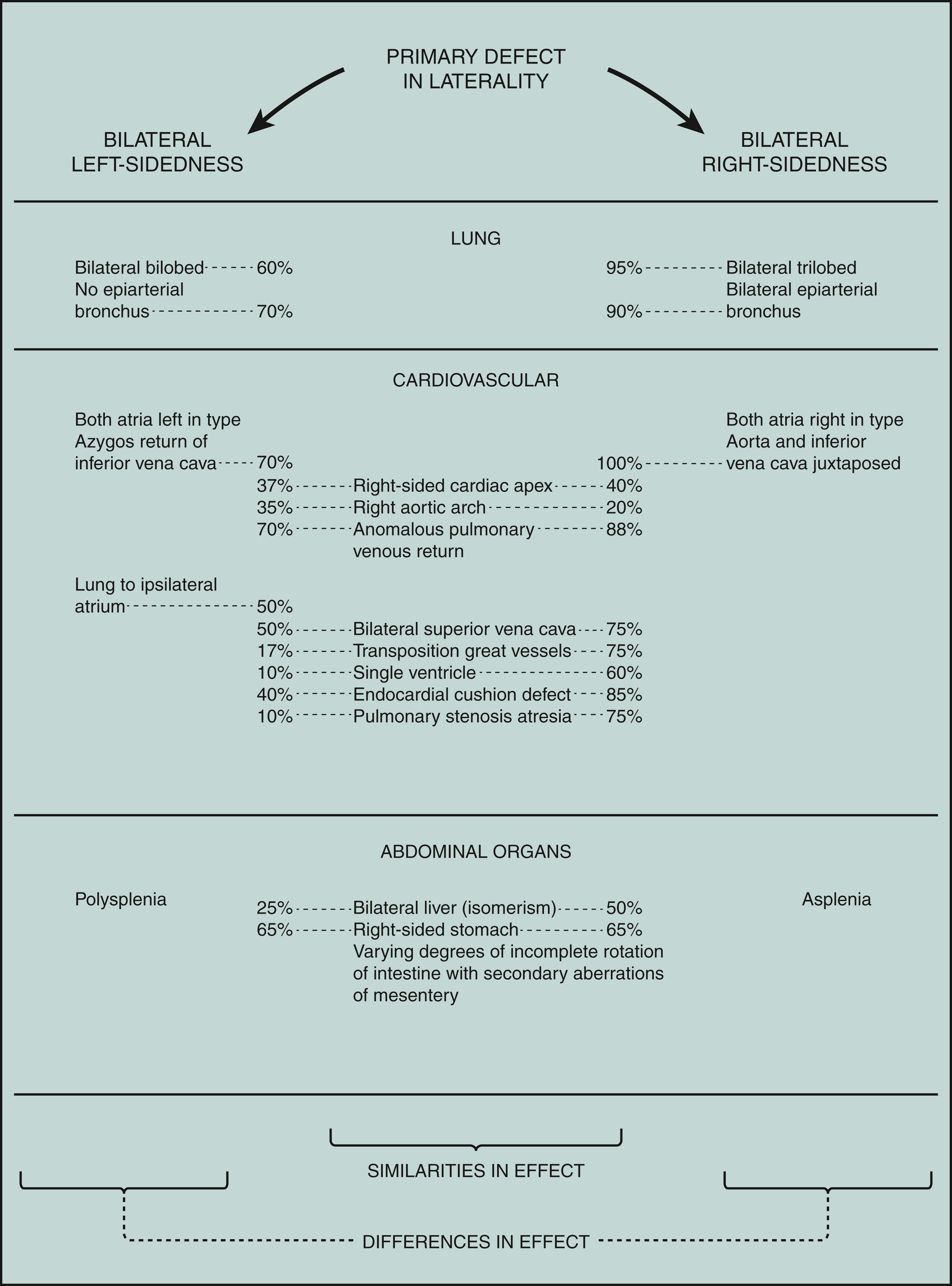
In addition to reversal of the sides, with partial to complete situs inversus, there can be bilateral left- or right-sidedness. The primary defect in both is a failure of normal asymmetry in morphogenesis. The basic problem would presumably be present before 30 days of development. Figure 1 sets forth the differences as well as the similarities between the patterns predominantly caused by left-sided bilaterality and by right-sided bilaterality. Among other differences, the spleen dramatically reflects the variant laterality in both. With left-sided bilaterality, there is polysplenia (usually bilateral spleens plus rudimentary extrasplenic tissue), and with right-sided bilaterality there is asplenia or a hypoplastic spleen.
Left-right axis malformations are usually isolated, but can occur as one feature of a multiple malformation syndrome, the most common of which is immotile cilia syndrome. As a result of defective cilia and flagella, chronic respiratory tract infections occur commonly, and infertility in males, chronic ear infections, and decreased or absent smell occur variably. The cilia are functionally abnormal and, on electron microscopy, have absent or abnormal dynein arms connecting the nine pairs of microtubules. A subgroup of the immotile cilia syndrome is Kartagener syndrome, an autosomal recessive disorder associated with partial to complete situs inversus with gross defects in cardiac septation (50%) in addition to the other features of immotile cilia syndrome.
Bilateral left-sidedness sequence is also known as polysplenia syndrome. The gender incidence is about equal. The cardiac anomalies are usually not as severe as those with bilateral right-sidedness.
Bilateral right-sidedness sequence is also known as asplenia syndrome, Ivemark syndrome, triad of spleen agenesis, defects of heart and vessels, and situs inversus. The sequence is two to three times more common in males than in females. The complex cardiac anomalies, usually giving rise to cyanosis and early cardiac failure, are the major cause of early death. The possibility of gastrointestinal problems must also be considered, especially as related to the aberrant mesenteric attachments. Renal anomalies are also more frequent (25%). Survivors have had an increased frequency of cutaneous, respiratory, and other infections, possibly related to the asplenia. Tests to detect asplenia include evaluation of red blood cells for Howell-Jolly bodies and Heinz bodies.
Intestinal malrotation, biliary atresia, anomalous portal and hepatic vessels, intestinal obstruction, anal atresia/stenosis, urinary tract defects including renal agenesis/hypoplasia and ureteral malformations, meningomyelocele, cerebellar hypoplasia, arrhinencephaly.
The defect in lateralization leading to the failure of normal asymmetry in morphogenesis is etiologically heterogeneous, with both genetic and environmental factors involved. As such, although usually sporadic, autosomal dominant, autosomal recessive, and X-linked recessive inheritance have all been documented. At present, only 20% of cases are caused by mutations of known genes, although additional candidate genes have emerged from studies of left-right axis development in vertebrates. Laterality defects in humans can be caused by mutations in ZIC3 , an X-linked zinc-finger transcription factor located at Xq26.2, which are frequently associated with hindgut anomalies in addition to failure in normal symmetry. In addition, mutations of LEFTY 2 on chromosome 1q42, CFC1 , GDDF1 , NODAL which is associated with a higher frequency of pulmonary valve atresia, ACVR2B , MMP21 , and FOXH1 , all of which encode components or modifiers of nodal signaling have been documented. In addition, rare variants of other genes including CDCC11 , CRELD1 , MED13B , SHROOM3 , MEGF8 , NFX2 -5, NPHP4 , PKD2 , and PKD1L1, are responsible for rare cases in humans.
Both bilateral left-sidedness (polysplenia) and bilateral right-sidedness (asplenia) have been documented in different persons in the same family, indicating that the two conditions represent different manifestations of a primary defect in lateralization leading to failure of normal body asymmetry.
The molecular determinants of normal body asymmetry are beginning to emerge. In the mouse, motile embryonic cilia generate directional flow of extraembryonic fluid surrounding the node located at the tip of the embryo in the midline. This flow concentrates left-right determinants to one side of the node activating asymmetric gene expression at the node and beyond. In the chick, activin on the right side of the primitive streak represses expression of the gene sonic hedgehog ( Shh ). The remaining expression of Shh on the left induces nodal on the left, leading to the normal looping of the heart tube to the right.
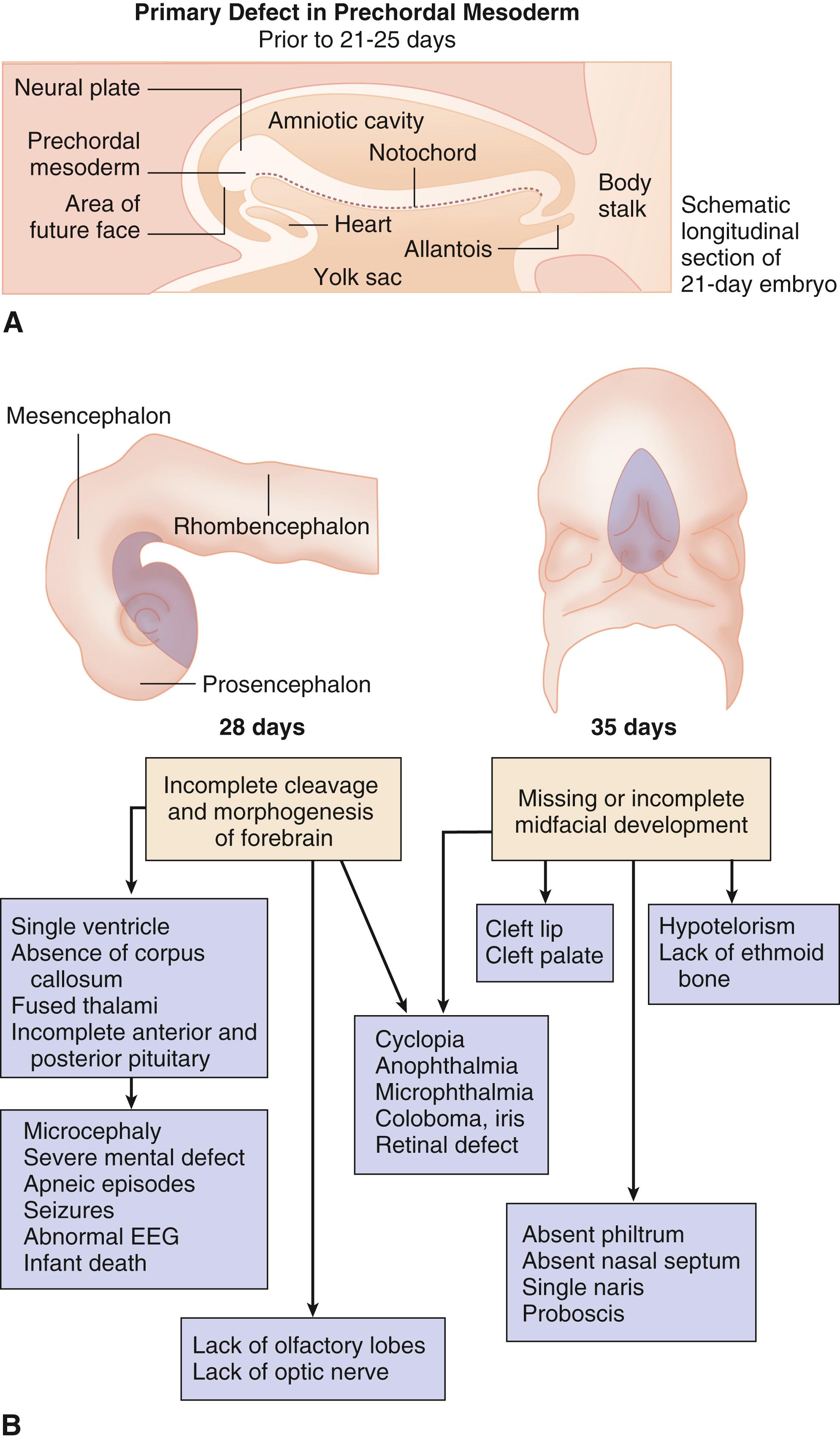
Holoprosencephaly results from incomplete cleavage of the prosencephalon between the eighteenth and twenty-eighth day of gestation. Neuroanatomically it is divided—based on the extent to which the forebrain has failed to separate—into alobar (the most severe type), semilobar, lobar, and middle interhemispheric type (the mildest type). In the alobar type the prosencephalon fails to cleave sagittally into cerebral hemispheres, transversely into telencephalon and diencephalon, and horizontally into olfactory tracts and bulbs. As a consequence of, and associated with, these severe defects in brain development, varying degrees of midline facial development occur. Cyclopia represents a severe deficit in early midline facial development. The eyes fuse, the olfactory placodes consolidate into a single tube-like proboscis above the eye, and the ethmoid and other midline bony structures are missing. Less severe deficits result in hypotelorism and varying degrees of inadequate midfacial and incomplete forebrain development that are more common than cyclopia and frequently include midline cleft lip and palate. The important clinical point is that incomplete midline facial development—such as hypotelorism, absence of the philtrum or nasal septum, a single central incisor, congenital nasal pyriform aperture stenosis, and/or a missing frenulum of the upper lip—suggests the possibility of a serious anomaly in brain development and function. Endocrine disorders, including diabetes insipidus, adrenal hypoplasia, hypogonadism, thyroid hypoplasia, hypoglycemia, precocious puberty, growth hormone defects, and short stature, are common. In addition, seizures and autonomic instability affecting temperature control, heart rate, and respiration have been reported. Finally, neural tube closure defects, ptosis, coloboma, choanal atresia, cleft lip and palate, genitourinary defects including micropenis, cryptorchidism, and ambiguous genitalia, and renal anomalies, as well as thymic hypoplasia, common mesentery, single umbilical artery, diaphragmatic hernia, gallbladder agenesis, esophageal atresia, intestinal stenosis, a variety of skeletal abnormalities, and cardiac defects occur. Nonalcoholic fatty acid disease has recently been associated.
Although the defect is isolated in the vast majority of cases, holoprosencephaly is etiologically heterogeneous with both genetic and environmental causes identified. Aneuploidy syndromes—including trisomies 13 and 18, as well as several structural chromosome aberrations, including del2p21, dup3pter, del7q36, del13q, del18p, and del21q22.3—should be considered. Autosomal dominant mutations in a number of genes have been identified in “nonsyndromic” holoprosencephaly. Mutations in four genes that have been identified in both sporadic and familial cases are responsible for most of these nonsyndromic cases. These four genes are SHH, ZIC2, SIX3, and TGIF. Sonic hedgehog ( SHH ), responsible for 12% of cases, is associated with wide variability of expression and is located at 7q36. Mutations in ZIC2, located at 13q32, occur in 9% of cases and are associated with a specific facial phenotype, including bitemporal narrowness, upslanting palpebral fissures, a flat nasal bridge, a short nose with anteverted nares, a broad and deep philtrum, and large ears. Mutations in SIX3, located at 2p21 and associated with an increase in renal anomalies, occur in up to 5% of cases, and mutations in TGIF, located at 18p11.3, occur in 1% to 2% of cases. Mutations in a number of genes involved in signaling pathways important for brain development, including PATCHED1 and GLI2 , as well as TDGF1 and FAST1 , which are involved in the Nodal/transforming growth factor β (TGF-β) pathway, occur far less frequently. Parents of an affected child should be checked for mild manifestations, such as a single central incisor, a missing upper lip frenulum, and absence of the nasal cartilage. Finally, holoprosencephaly has been seen as one feature of multiple malformation syndromes, such as Meckel-Gruber syndrome and Smith-Lemli-Opitz syndrome, in the offspring of diabetic women, and as an occasional feature in fetal alcohol spectrum disorder. The prognosis for central nervous system function in individuals with this type of defect is very poor.
A significant number of infants with holoprosencephaly survive into childhood and even adulthood. Factors associated with long-term survival include holoprosencephaly subtype (lobar, semilobar, and middle interhemispheric), female gender, and a milder facial phenotype.
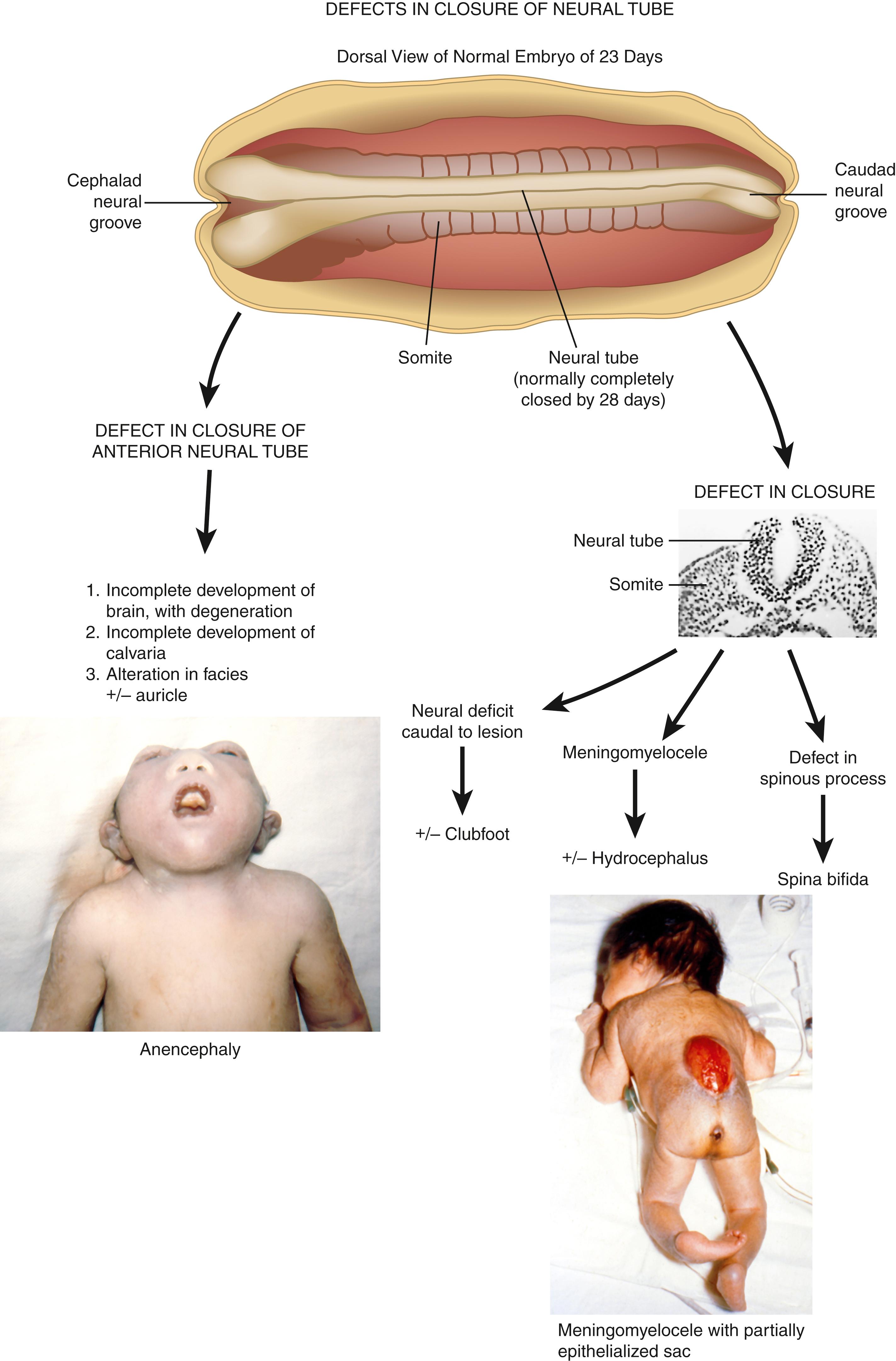
The initiating malformation appears to be a defect in closure of the neural groove to form an intact neural tube, which is normally completely fused by 28 days. Anencephaly represents a defect in closure at the anterior portion of the neural groove. The secondary consequences are these: (1) the unfused forebrain develops partially and then tends to degenerate; (2) the calvarium is incompletely developed; and (3) the facial features and auricular development are secondarily altered to a variable degree, including cleft palate and frequent abnormality of the cervical vertebrae.
Defects of closure at the mid or caudal neural groove can give rise to meningomyelocele and other secondary defects, as depicted. Of greatest concern relative to outcome for independence and survival is the hydrocephalus and other manifestations of the Chiari II malformation, which is present in virtually all cases.
Defects of closure in the cervical and upper thoracic region can culminate in the iniencephaly sequence, in which secondary features may include retroflexion of the upper spine with short neck and trunk, cervical and upper thoracic vertebral anomalies, defects of thoracic cage, anterior spina bifida, diaphragmatic defects with or without hernia, and hypoplasia of lung and/or heart. Evidence suggesting that there may be four sites of anterior neural tube closure explains the variations observed in their location, recurrence risk, and etiology. Most commonly, no specific etiology is appreciated. The recurrence risk is 1.9% for parents who have had one affected child. Measurement of alpha fetoprotein (AFP) in maternal serum in conjunction with detailed ultrasonography allows prenatal diagnosis in the vast majority of cases.
Encouraging results have been reported from the Management of Myelomeningocele Study (MOMS), a multicenter, randomized, controlled trial that compared in utero prenatal closure to postnatal closure. The study was published in 2011 and long-term outcomes continue to be documented. The study so far has demonstrated a decrease in hindbrain herniation resulting in a decreased need for ventriculoperitoneal (V-P) shunting for hydrocephalus, decreased pressure on the hindbrain, and improvement in motor function in the prenatal repair group compared with those who received surgery postnatally.
The United States Public Health Service has recommended that women of childbearing age should consume 0.4 mg of folic acid daily to reduce their risk of conceiving a child with a neural tube defect. For women who previously have had an affected infant, it has been recommended that 4.0 mg daily of folic acid should be consumed from 1 month before conception through 3 months of pregnancy.
Following closure of the neural groove at approximately 28 days, the cell mass caudal to the posterior neuropore tunnels downward and forms a canal in a process that gives rise to the most distal portions of the spinal cord—the filum terminale and conus medullaris. Failure of normal morphogenesis in this region leads to a spectrum of structural defects that cause orthopedic or urologic symptoms through tethering or compression of the sacral nerve roots, with restriction of the normal cephalic migration of the conus medullaris. Defects involve structures derived from both mesodermal and ectodermal tissue and include mesodermal hamartomas, sacral vertebral anomalies, hyperplasia of the filum terminale, and structural alterations of the distal cord itself. In most situations there is a cutaneous marker at the presumed junction between the caudal cell mass and the posterior neuropore in the region of L2-L3. Markers consist of tufts of hair, skin tags, dimples, lipomata, and aplasia cutis congenita. Cutaneous markers, such as a pit at the tip of the coccyx, are extremely common and are not usually associated with a tethered cord.
The recognition of the surface manifestations of such a malformation sequence at birth should ideally lead to further evaluation and management. Roentgenograms of the spine may or may not show any abnormality. Ultrasound to document normal movement of the spinal cord with respiration, followed by magnetic resonance imaging in questionable cases, is usually sufficient to document the defect. Early management will prevent neuromuscular lower limb or urologic problems, such as retention, incontinence, or infection secondary to continued tractional tethering of the cord and nerve roots. If the physician waits for signs of such serious complications, the neurologic damage may not be reversible. A 4% incidence of open neural tube defects has been documented in first-degree relatives of probands.
De Morsier recognized the association between the absence of the septum pellucidum and hypoplasia of the optic nerves and called it septo-optic dysplasia. The midline brain defects have subsequently been expanded to include hypoplasia of the corpus callosum. The clinical spectrum of altered development and function arising from this defect has in addition, been reported by Hoyt and others to include hypopituitarism. The diagnosis is primarily clinical and requires two or more features of the triad. The presumed developmental pathogenesis is depicted in Fig. 1A .
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here