Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The introduction of laparoscopy in the 1980s has revolutionized the field of complex abdominal surgery. Once highly morbid procedures requiring open exposure and prolonged recovery are now accomplished through minimally invasive techniques with comparable outcomes. In 1994 Gagner and Pomp described the first laparoscopic pylorus-sparing pancreaticoduodenectomy (PD) in a 30-year-old female with chronic pancreatitis (CP). The operative time was 10 hours, and her postoperative course was complicated by delayed gastric emptying, necessitating a 30-day hospital stay. Due to concerns of excessive morbidity, the introduction of laparoscopic pancreas surgery was challenged. However, since 2010, there has been growing momentum in laparoscopic surgery and it is now well integrated into routine pancreatic resections and reconstructions at high-volume centers, with equivalent rates of mortality compared with open procedures. Traditional operations for almost all pancreatic resections and reconstructions have been described and replicated through a laparoscopic approach.
Laparoscopic surgery can be generally described as pure laparoscopic, robotic assisted, or a combination. Conventional laparoscopic surgery permits smaller incisions and improved postoperative pain control. Patients benefit from expedited functional recovery and shorter hospital stays. Technically, improved visualization through a magnified view allows meticulous dissection and greater control of intraoperative bleeding. With the advent of robotic assistance, surgeons benefit from binocular three-dimensional vision, scaling, stabilization of tremor, reduced operator fatigue and improved ergonomics from the console-surgeon interface. This chapter will focus on the robotic approach, summarizing the data on the safety, efficacy, and technique of robotic approaches to minimally invasive pancreas surgery for both benign and malignant indications, showcasing the diversity of the platform for complex pancreatic operations.
Complications of acute pancreatitis, including infected pancreatic necrosis, were historically managed with laparotomy and open pancreatic necrosectomy, which were associated with high rates of mortality and morbidity at 56% and 78%, respectively. This has prompted less invasive techniques, such as percutaneous and endoscopic drainage. For infected necrotizing pancreatitis, the step-up approach as described by van Santvoort et al. in the PANTER (PAncreatitis, Necrosectomy versus sTEp up appRoach) trial recommends beginning with percutaneous or endoscopic drainage for sepsis control. After 72 hours, if there is no clinical improvement or a new collection forms that is inadequately drained, a second drain is recommended. If there is no clinical improvement after another 72 hours, a video-assisted retroperitoneal débridement (VARD) is recommended. This technique in stepwise escalation of invasive intervention was found to have equal rates of mortality compared with traditional necrosectomy. Furthermore, more than one-third of patients were successfully managed with drains alone and had fewer long-term complications of new-onset diabetes, incisional hernias, and pancreatic insufficiency.
The decision to pursue a minimally invasive approach for walled-off pancreatic necrosis (WOPN) depends on timing of presentation, anatomy of collection, and patient history. For patients with WOPN, we would recommend delaying surgical intervention for a minimum of 6 to 12 weeks following presentation, which allows the wall of the collection to mature and its contents to liquefy. Collections that consist of homogenous fluid may successfully be treated by endoscopic or percutaneous drainage, whereas those containing heterogeneous debris usually necessitate mechanical débridement. Patients who present with biliary pancreatitis will need a cholecystectomy, and a laparoscopic or robotic-assisted approach will permit a concurrent opportunity.
A minimally invasive cyst-gastrostomy (CG) allows en bloc débridement of infected necrotic pancreatic tissue and continued drainage via a connection with the stomach. Collections that are likely to be successfully treated by a CG are greater than 15 cm in size, possess a defined capsule, are medially located within the pancreas, and adherent to the posterior wall of the stomach.
Comparing laparoscopic with open CG, Khaled et al. found the laparoscopic approach to have shorter operative times, lower risk of postoperative morbidity, and shorter hospital stay. Although endoscopy and minimally invasive CG have been shown to have similar rates of complications, endoscopically managed patients typically require repeat interventions. Khreiss et al. compared 20 patients who underwent minimally invasive CG with 20 patients who underwent endoscopic drainage and found that 45% of endoscopic patients required repeat drainage for residual WOPN, compared with 15% of patients who received an operation. Similarly, Worhunsky et al. reported 90% of 21 patients with sterile pancreatic necrosis who underwent transgastric necrosectomy were successfully débrided without additional interventions.
The robotic transgastric CG as described by Khreiss et al. begins with six robotic ports: one 12-mm camera port, three 8-mm robotic arm ports, and two bedside assistant ports (12 mm and 5 mm) as depicted in Fig. 101.1 . Intraoperative ultrasound is used to localize the WOPN and plan the optimal location for CG. A 5-cm anterior gastrostomy is created using monopolar scissors, a minimum of 2 cm away from, and parallel to, the greater gastric curve. A 1-cm posterior gastrostomy is then made to reach the anterior wall of the collection. A suction irrigator is used until the effluent is dry. The CG is fashioned using either a linear cutting stapler or plication of posterior stomach wall to the anterior capsule using running suture. Under direct visualization, the necrotic material is removed through the CG, ideally as a large en bloc necroma. After cavity irrigation, the anterior gastrostomy is closed in 2 layers with a linear stapler or running absorbable suture followed by a second layer of silk sutures ( Fig. 101.2 ). At this point, such adjunct procedures as a cholecystectomy or jejunostomy feeding tube may be performed.
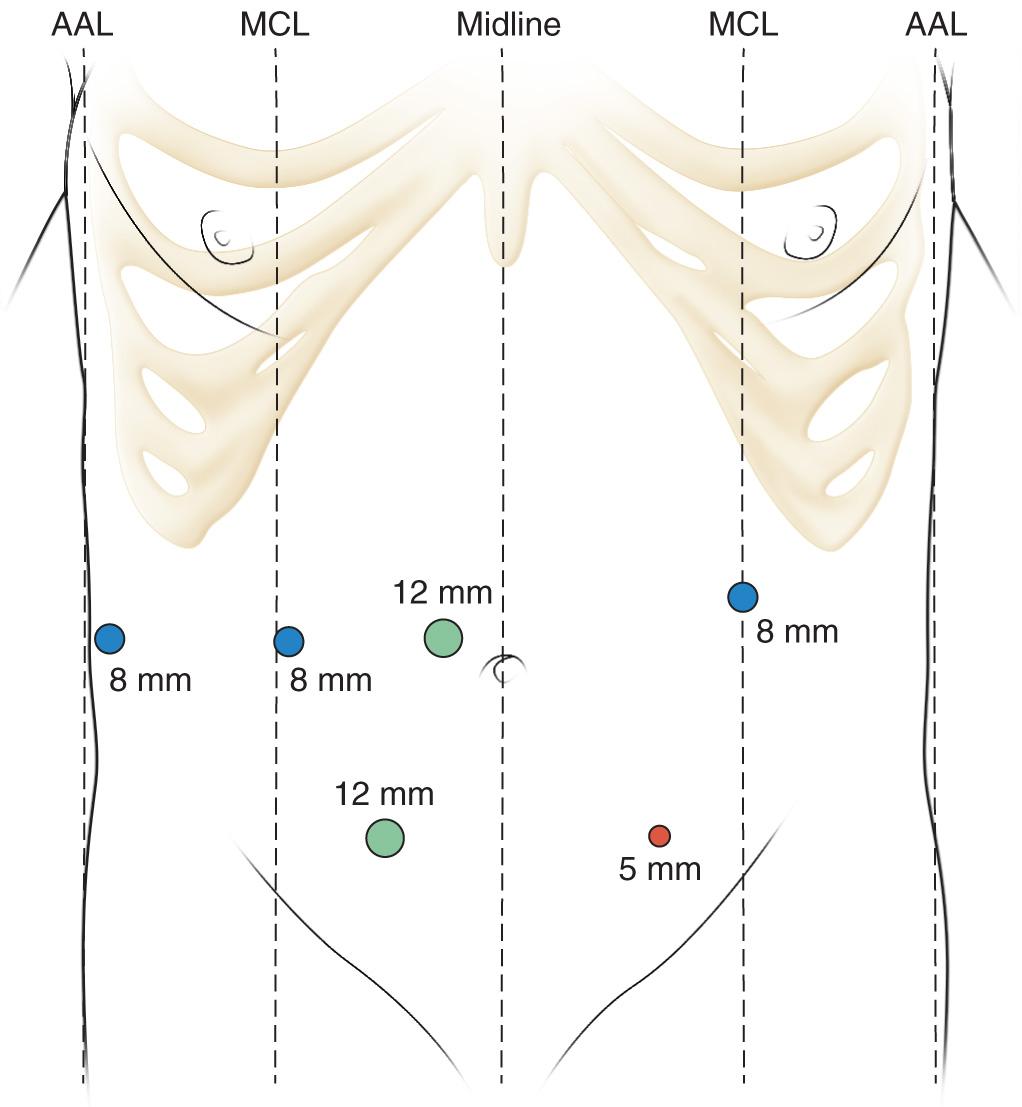
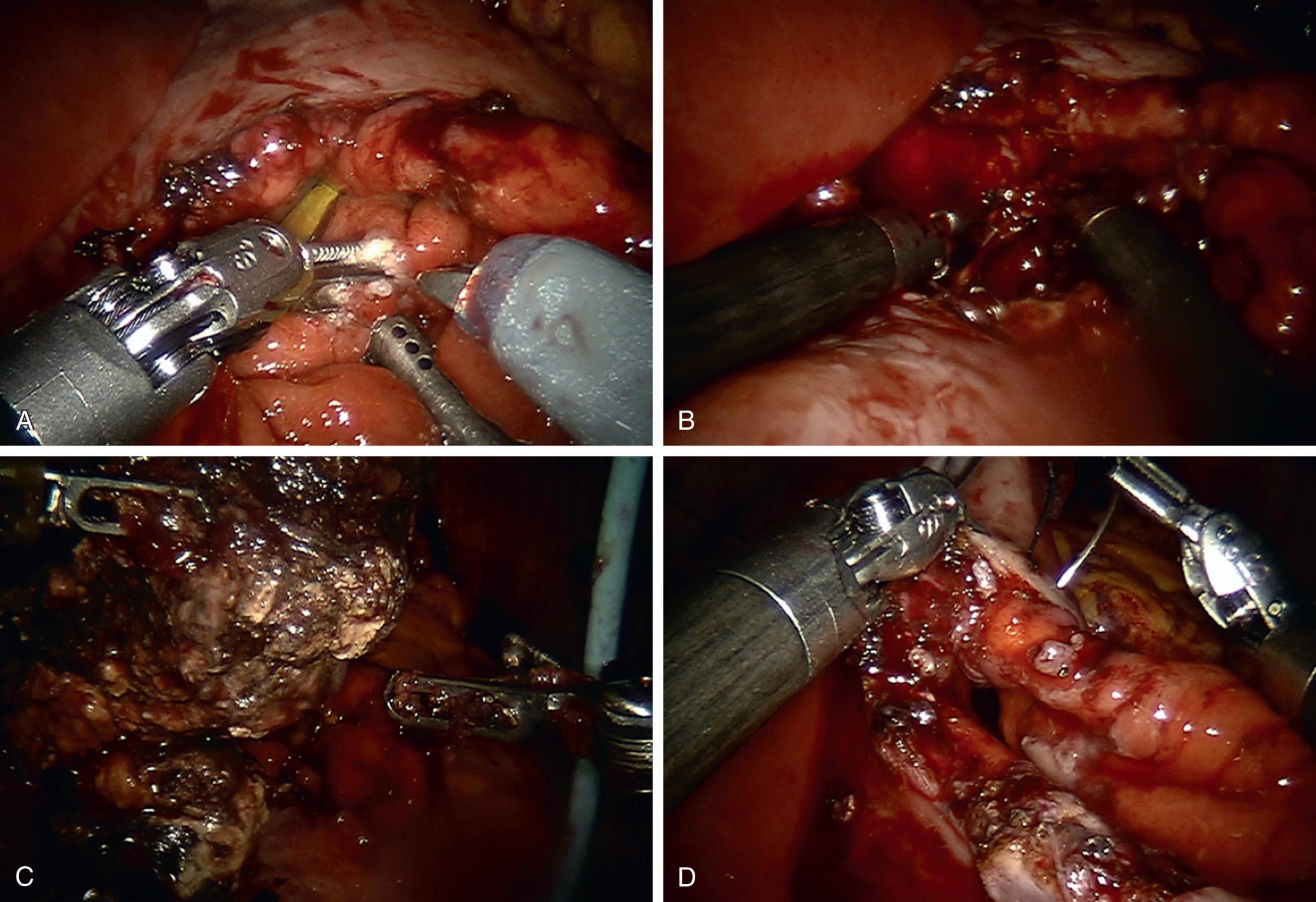
The VARD procedure allows pancreatic débridement under direct visualization through a retroperitoneal route. It is indicated for patients who have had a retroperitoneal drain in place. It may be ideal for patients with a significant abdominal surgical history by avoiding a transabdominal route or for patients who are septic and not expected to do well following a laparotomy.
Horvath et al. conducted the largest review of VARD procedures in 25 patients with necrotizing pancreatitis across six tertiary care centers. There were four (16%) primary complications, including bleeding and enteric fistula, and nine (36%) secondary complications, including pneumonia, deep vein thrombosis, renal failure, and pancreatic fistula. There were no 30-day mortalities and 1 mortality at 3- to 6-month follow-up. Although their rates of morbidity and mortality compare favorably with open débridement, 19% required a second VARD for additional débridement. Garcia-Urena describes seven consecutive patients who underwent VARD for a variety of indications and reported similar results: four (57%) complications, the most common of which being pancreatic fistula, with no mortalities. A second VARD was required in two (29%) of seven patients.
The technique involves using the percutaneous drain as a guide to enter the pancreatic collection. One method is using the Seldinger technique, in which a flexible guidewire is inserted through the drain followed by an introducer sheath, and a 5-mm laparoscopic port through the introducer. Alternatively, direct cutdown can also be used. Under videoscopic guidance, irrigation, suction, and manual débridement is used to judiciously excavate the purulent necrotic material. A large sump drain is left in the cavity to allow scheduled lavages postoperatively until the effluent becomes clear.
The decision to pursue a minimally invasive approach for CP depends on patient factors, including underlying pathophysiology, age, comorbidities, and history of prior pancreatic interventions. The specific operation of choice for CP depends on anatomic considerations, including the presence of a pancreatic head mass, a dilated pancreatic duct, and peripancreatic fluid collections. Outcomes of the published literature are summarized in Table 101.1 .
| Author | Procedure | Approach | Patients | OR Time (min) | EBL (mL) | Complications | Mortality | LOS (Days) | Symptom Improvement (%) |
|---|---|---|---|---|---|---|---|---|---|
| Galvani et al. (2014) | TP + AIT | Robotic (1 with AIT) | 6 | 712 | 630 | 2 (33%) | 0 | 12.6 | 100 |
| Zureikat et al. (2015) | TP + AIT | Robotic | 10 | 560 | 650 | 2 (20%) | 0 | 10 | NR |
| Cuschieri et al. (1996) | DP | Lap | 5 | 240–360 | 400 | 1 (20%) | 0 | 6 | 100 |
| Fernandez-Cruz et al. (2002) | DP | Lap | 5 | 240 | 450 | 1 (20%) | 0 | 4.5 | 100 |
| Khaled et al. (2011) | LPJ | Lap | 37 | 218 | NR | 5 (13.5%) | 0% | 5.5 | 89 |
| Meehan et al. (2011) | LPJ | Robot | 1 | 390 | NR | NR | 0 | 8 | 100 |
| Khaled et al. (2014) | LPJ | Lap | 5 | 278 | 150 | 1 (25%) | 0 | 5 | 80 |
| Khaled et al. (2014) | CG | Lap | 30 | 62 | NR | 3 (10%) | 0 | 6.2 | 97 resolution |
| Worhunsky et al. (2014) | CG | Lap | 21 | 170 | 50 | 12 (57%) | 1 (5%) | 5 | NR |
| Khreiss et al. (2015) | CG | 14 Robot 6 Lap |
20 | 167 | 30 | 4 (20%) | 0 (0%) | 7 | NR |
| Horvath et al. (2010) | VARD | Lap | 25 | 135 | NR | 9 (36%) | 0 | 48 | NR |
| Garcia-Urena et al. (2013) | VARD | Lap | 7 | 63 | NR | 4 (57%) | 0 (0%) | 50 | NR |
| Cooper et al. (2014) | Frey | Lap | 1 | NR | NR | 0 | 0 | 6 | 100 |
| Tan et al. (2015) | Frey | Lap | 9 | 323 | 57 | 1 (14%) | 0 | 7 | 100 |
| Khaled et al. (2014) | Beger | Lap | 1 | 285 | NR | 0 | 0 | 5 | 100 |
The majority of surgical interventions for CP are typically executed through an open approach. Therefore data on outcomes following minimally invasive procedures are rare. However, open techniques of pancreatic resection, drainage, and combination procedures have been replicated through conventional laparoscopic and robotic-assisted techniques and have been shown to be safe and feasible. Given the infrequent indication for surgical interventions for CP, they are best delivered at high-volume tertiary centers by multidisciplinary teams. Therefore approach for surgical treatment will largely be dictated by the local expertise.
Resection procedures for CP include total pancreatectomy, with or without auto islet transplantation (TP ± AIT), PD, distal pancreatectomy (DP), and the Puestow, Frey, and Beger procedures. TP ± AIT is a rarely performed procedure with few reports of a minimally invasive approach. It is indicated for patients with small-duct disease without diabetes. The largest published case series includes 10 patients who underwent a robotic-assisted TP, one of whom received AIT. There were no mortalities at 90 days, and two Clavien-Dindo grade III complications, including one that required reoperation. Galvani et al. retrospectively reviewed a single-institutional series of six patients with CP who underwent a fully robotic-assisted TP + AIT. There were no major complications or mortalities, and all patients were in the process of weaning off chronic narcotics at 1-month follow-up. These outcomes are comparable with a series of 12 patients who underwent open TP + AIT.
The robotic approach as described by Zureikat et al. first places seven laparoscopic ports as depicted in Fig. 101.3 , composing one 12-mm camera port, three 8-mm robotic ports, two assistant ports (12 mm and 5 mm), and one 5-mm port for a self-retaining liver retractor. The lesser sac is entered, and the posterior stomach is freed from the surface of the pancreas. A medial visceral rotation is performed by mobilizing the right colon, and a Kocher maneuver is used to mobilize the duodenum. The jejunum is transected with a linear cutting stapler approximately 10 cm from the ligament of Treitz and is sutured to the stomach by a single stitch to mark the future gastrojejunostomy, approximately 50 cm downstream. The distal stomach is transected with a linear cutting stapler. The robot is then docked directly over the head of the table.
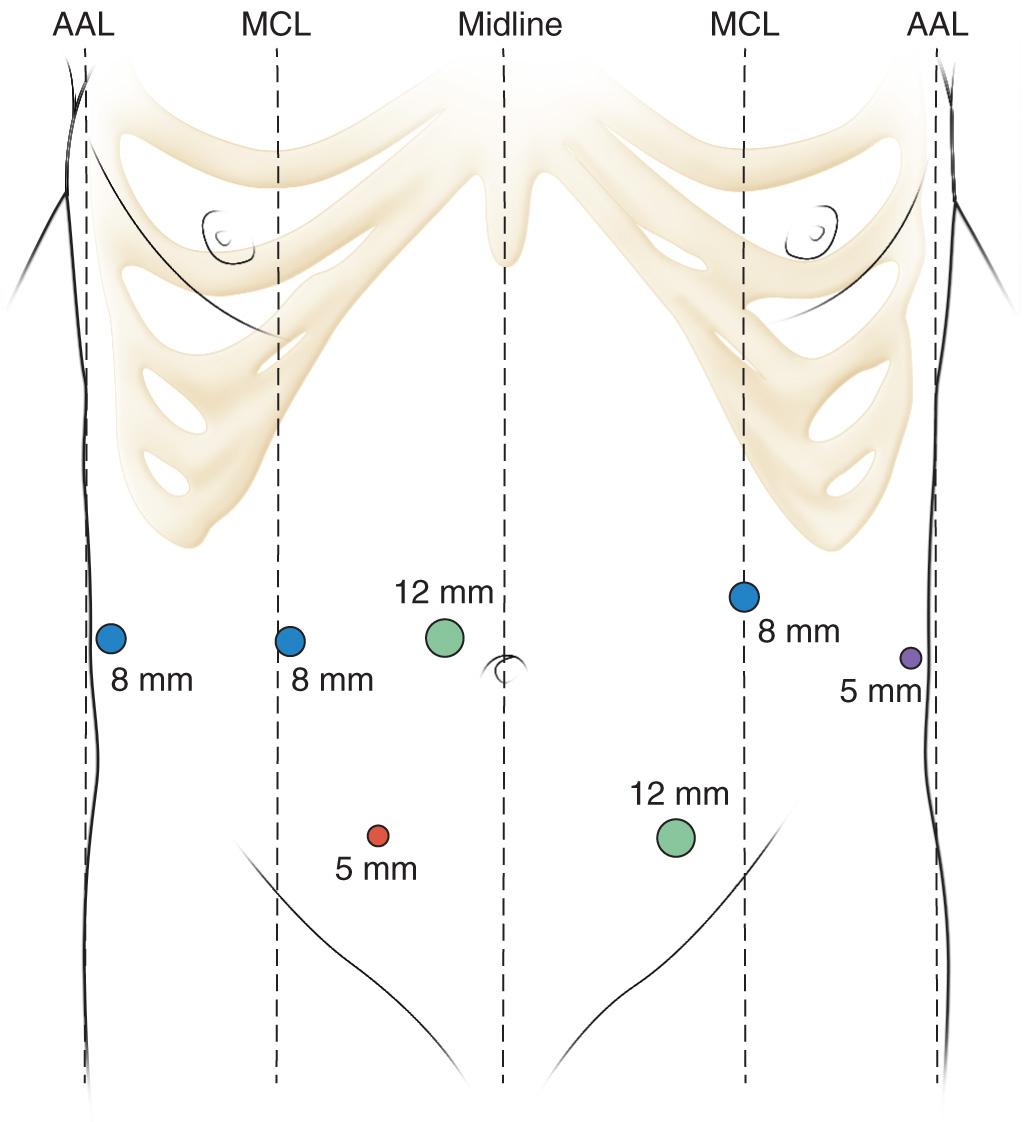
Dissection of the porta hepatis is performed using robotic hook cautery with isolation of the gastroduodenal artery (GDA), portal vein (PV), and common bile duct (CBD). In a TP the GDA is transected with a vascular linear stapler with enforcement of the stump with a 10-mm clip ( Fig. 101.4 ). With a planned AIT, the GDA transection is delayed until just prior to pancreatic extraction to reduce the warm ischemia time. The CBD is then transected with a linear stapler. The division of the pancreas is delayed to preserve islet cell yield but can be transected in a TP alone for ease of dissection. Similarly the splenic vein (SV) and artery are transected with vascular staplers but preserved until specimen extraction in the case of an AIT. The retroperitoneum is incised laterally, and the retropancreatic space is developed to include the SV, splenic artery (SA), and pancreatic body and tail. The entire pancreaticosplenic unit is mobilized by dividing the splenic flexure, splenorenal and splenocolic attachments. The pancreas is dissected from the superior mesenteric vein (SMV) and PV to expose the superior mesenteric artery (SMA) posteriorly. This requires dividing the inferior and superior pancreaticoduodenal vessels.
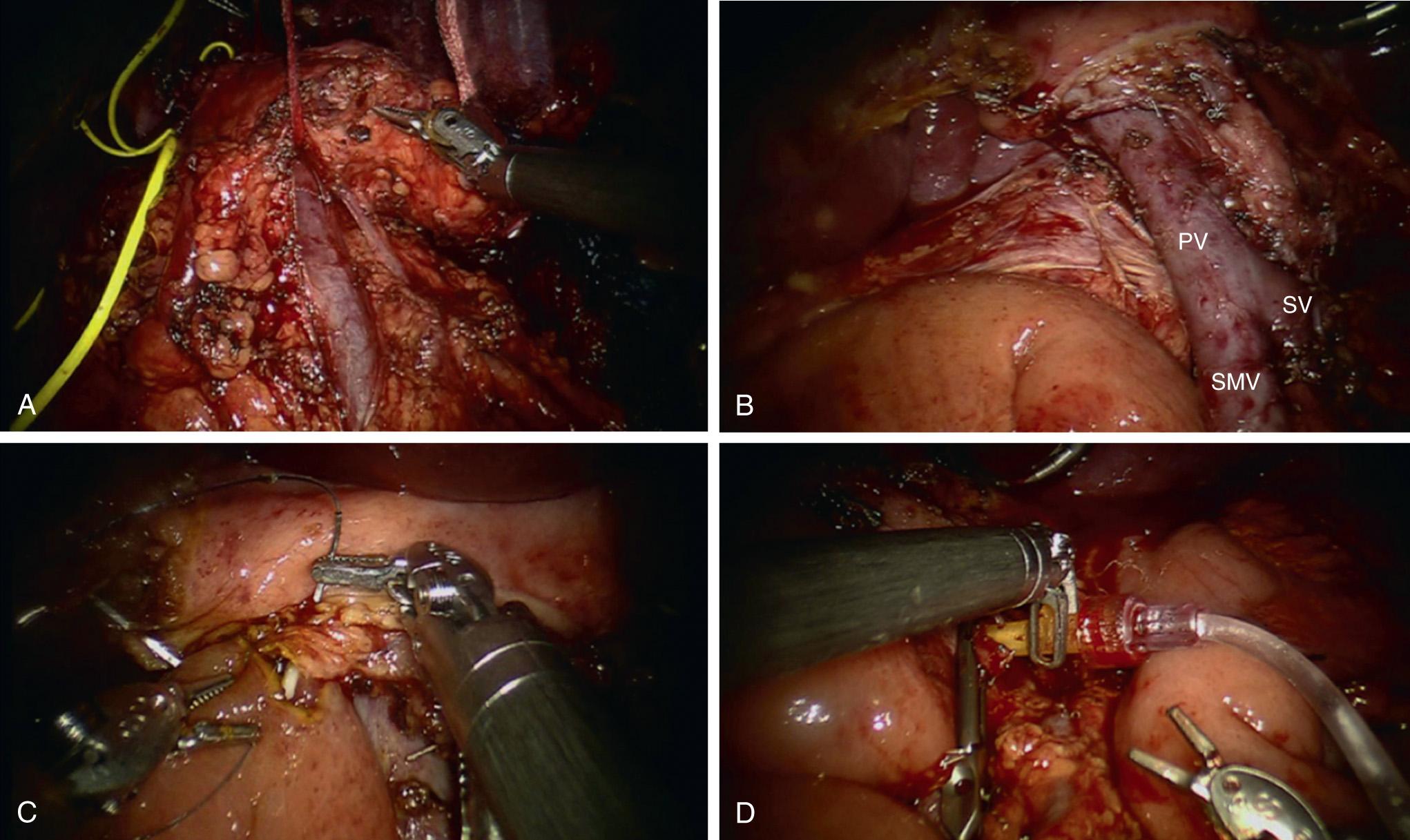
To perform the AIT, the pancreas is elevated from the retroperitoneum, only attached by the GDA, SA, and SV. Heparin is administered (50 IU/kg), and all three vessels are transected with a vascular stapler in the following order: SA, GDA, SV. A generous SV stump facilitates the introduction of a 14-gauge catheter for islet cell infusion. The specimen is then retrieved with an endoscopic bag. An end-to-side hepaticojejunostomy is fashioned in a “neo-duodenal” fashion using a running suture. An antecolic end-to-side gastrojejunostomy is handsewn in two layers. Lastly, a 14-gauge angiocatheter is inserted into the SV stump, and islet cells are infused by gravity, followed by a restapled closure of the SV stump.
Despite an abundance of literature on single-institution experience with minimally invasive PD and DP, many studies do not stratify between benign and malignant conditions. Therefore the efficacy of minimally invasive approaches for CP cannot be generalized and warrants further study. However, it can be inferred from large studies that minimally invasive PD and DP are safe and feasible in high-volume centers. For example, the largest single-institution review of 132 robotic PDs, including 8% with benign indications, found outcomes similar to the open Whipple. Overall mortality at 30-days was 1.5%, with 21% Clavien-Dindo grade III or IV complications.
Cruz et al. compared a series of five patients with CP who underwent laparoscopic DP with 41 patients who received an open DP. The laparoscopic group had 1 (20%) complication (duodenal perforation) versus 20 patients with complications in the open group (48%). There were no mortalities in either group. At 13-month follow-up, all five laparoscopic patients were pain free versus 80% in the open group. There are also data to suggest that robotic DP is associated with higher rates of splenic preservation and lower rates of open conversion when compared with conventional laparoscopy.
The techniques of robotic-assisted PD and DP are similar for benign or malignant conditions. Description of the approach is summarized later for pancreatic adenocarcinoma.
The lateral pancreaticojejunostomy (LPJ) or “modified Puestow” procedure permits drainage of the pancreatic duct and is commonly indicated for patients with an obstructed or dilated pancreatic duct. It offers the advantage of preserving the head of the pancreas along with exocrine and endocrine function.
In limited case reports of minimally invasive approaches, the laparoscopic approach appears to be safe, feasible, and effective. In a review of five patients with CP who underwent laparoscopic LPJ, there were no reported mortalities, one complication of pancreatic bleeding requiring reoperation (20%), and ultimately four out of five (80%) patients pain free at a median 15-month follow-up. In a single case report of a robotic LPJ, there was no mortality or morbidity, and the patient remained asymptomatic at 2 years postoperatively.
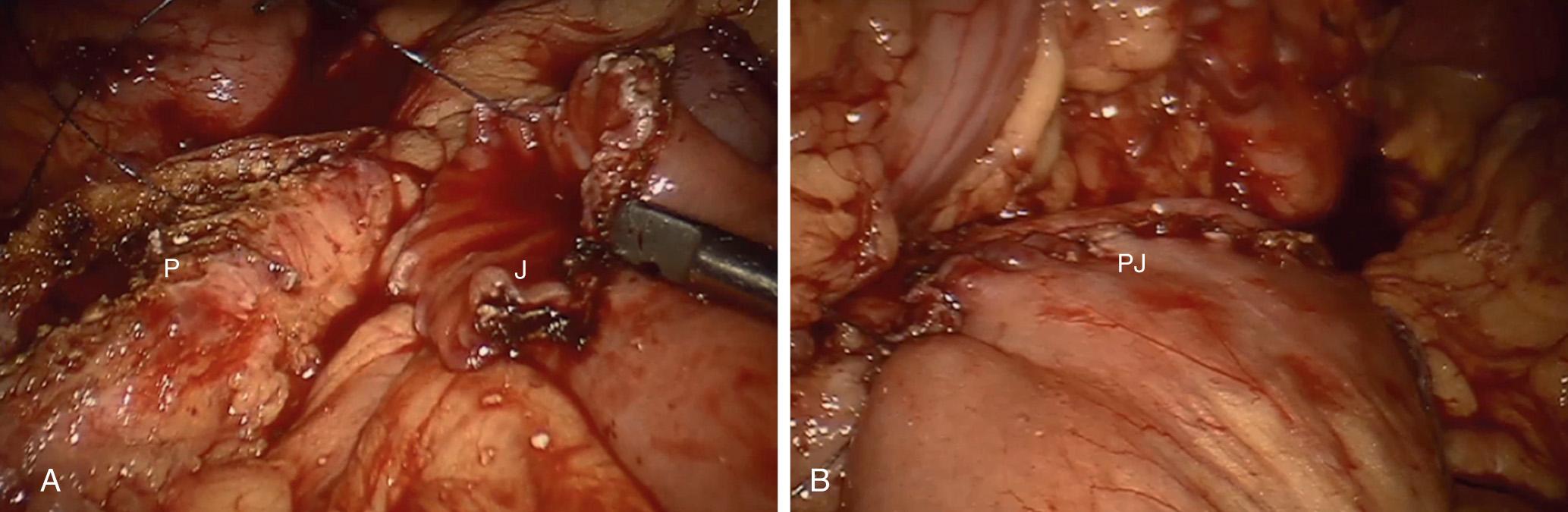
The robotic approach as described by Meehan and Sawin begins with five ports: one 12-mm camera port, two robotic arm ports, and two assistant ports. The lesser sac is entered using the robotic hook cautery, and the endoscopic ultrasound is used to localize the dilated pancreatic duct. The pancreatic duct is opened longitudinally along the entire length of the pancreas using the hook cautery. To create the Roux limb, the bowel is divided approximately 20 cm beyond the ligament of Treitz using the endoscopic stapler. The Roux limb is aligned along the pancreatic duct, and the distal bowel anastomosis is created on the antimesenteric surface of the bowel, using the endoscopic stapler. The Roux limb is brought up in a retrocolic fashion, and the mesenteric defect is closed. The LPJ is robotically sewn using absorbable 3-0 suture ( Fig. 101.5 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here