Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Unlike the coronaviruses causing severe acute respiratory syndromes, namely SARS-CoV and SARS-CoV-2 (the cause of Coronavirus Disease-2019 [COVID 19]), which developed rapid and aggressive person-to-person spread, the coronavirus that causes Middle Eastern respiratory syndrome (MERS-CoV) has yet to develop significant person-to-person spread. It remains a highly dangerous pathogen nonetheless. Roughly 2500 cases have been reported to date, with a 35% case fatality rate; however, actual rates of disease are uncertain, as up to 80% of cases may be asymptomatic. Bats are the major reservoir of MERS-CoV in nature, although camels appear to be the key reservoir host for human cases. This may have limited the spread of MERS-CoV, largely confining disease to the Arabian Peninsula. A high index of suspicion based on local epidemiology is essential to making the diagnosis. MERS-CoV has a predilection for DPP4 receptor–positive cells of the lower respiratory tract, in contrast to the ACE-2 receptor–positive cells in the upper respiratory tract used by SARS-CoV and SARS-CoV-2 for viral invasion. This difference in pathogenesis may also serve to limit the respiratory spread of MERS-CoV, although nosocomial transmission to health workers has been well documented. There are no confirmed or licensed effective treatments to date, although there are a few promising candidates. There are at least two experimental vaccines in clinical-stage development and multiple preclinical candidates. Supportive care, including intensive care unit (ICU) admission and mechanical ventilation, remains the mainstay of survival.
A 56-year-old man fell ill in his home country a week after taking a pilgrimage to Mecca for the Hajj. His illness initially began with a mild cough but within a few days quickly progressed to fever and respiratory distress, prompting admission to a local hospital. He was initially diagnosed with acute pneumococcal pneumonia and treated empirically with antibiotics. The facility lacked an ICU or mechanical ventilation. The patient’s condition worsened over 36 hours, at which point he deteriorated rapidly and died. No autopsy was performed. A week later, a healthcare worker who had helped to look after the patient fell ill with a mild respiratory illness, and missed a few days of work.
COMMENT: MERS-CoV can present as a range of illness severities from asymptomatic to severe with fulminant respiratory failure. Transmission has been focused in the Arabian Peninsula, likely limited by the geographic distribution of camels as the primary human reservoir host. As such, with a little over 2500 cases in 8 years, it has not emerged with nearly the severity of SARS-CoV-2 (COVID-19), which has become a global pandemic with more than half a million deaths over 6 months in 2020. However, visitors to the Arabian Peninsula have brought cases back to their home countries, even leading to extensive nosocomial and eventually person-to-person transmission, as in South Korea in 2015. Specific therapies remain in experimental clinical development. A high index of suspicion is needed to diagnose MERS-CoV.
MERS is caused by a novel, highly pathogenic coronavirus strain termed MERS-CoV that first appeared in Jordan in 2012. Within 5 years more than 2000 cases had been reported, with approximately 600 deaths. Although the spread of MERS has not been nearly as dramatic or extensive as the current pandemic of COVID-19 (caused by SARS-CoV-2), there were documented cases in 21 countries within 3 years. To date, 2500 cases of MERS have been reported, with a 35% case fatality rate. Many of those who died had underlying medical conditions. The origins of nearly all human cases have been traced to the Arabian Peninsula. However, in 2015 there was a significant outbreak in South Korea, where more than 80 infections occurred within a few days and 186 over 3 months. This was thought to be due to a single “super spreader” event originating from an elderly man who had traveled to Saudi Arabia. The infection is believed to have spread beginning with the index case through healthcare workers at a network of hospitals.
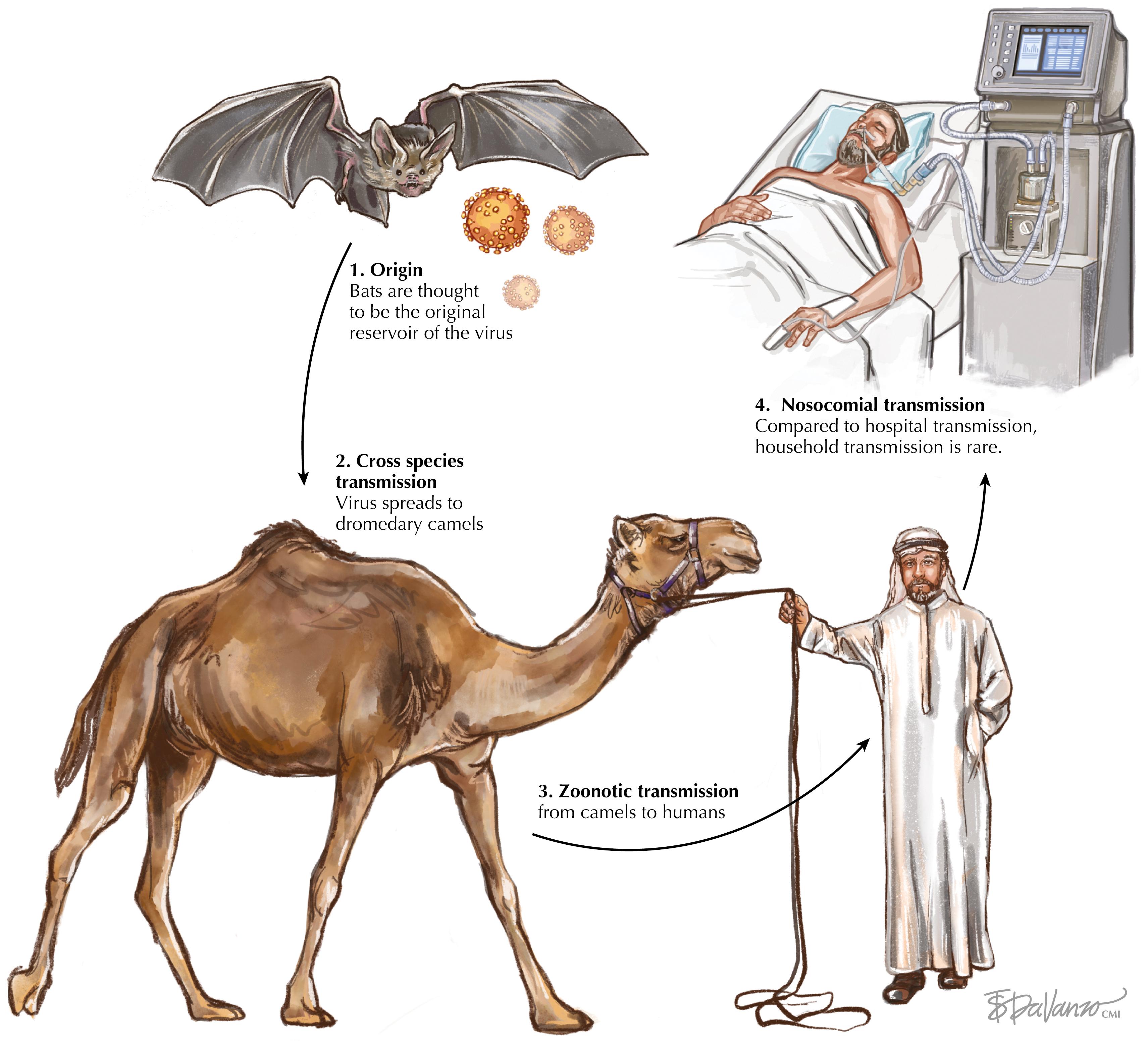
MERS-CoV is a Betacoronavirus in the Coronaviridae family. Coronaviruses are positive-sense single-stranded RNA viruses. MERS-CoV is thought to be more closely related to bat coronaviruses than to SARS-CoV. Although bats are believed to be the ultimate reservoir of MERS-CoV, the virus also infects camels, which appear to be the primary reservoir of human infections. The relatively limited geographic distribution of camels may be a factor that has limited the spread of MERS-CoV thus far compared with that of SARS-CoV and SARS-CoV-2. The virus appears to transmit asymptomatically among camels, and has been widespread in the herds studied. Sustained person-to-person transmission risk is thought to be low, although there has been documented nosocomial spread to healthcare workers from infected patients. The potential for mutation, allowing more efficient human-human transmission, remains a possibility. MERS-CoV has a relatively unique tropism for nonciliated bronchial epithelium, unlike most other respiratory viruses, which target ciliated epithelium. More detail on coronavirus infections can be found in Chapter 95 on severe acute respiratory syndrome coronaviruses (SARS and COVID-19).
Based on limited data from known transmissions, the estimated incubation period is 2 to 14 days (median 5 days). The time from clinically apparent symptoms to hospitalization is 4 to 5 days, with death occurring within 12 days for severe cases. It remains unclear as to whether MERS-CoV is transmitted primarily through contact or respiratory transmission, but sustained person-to-person transmission has not been documented. MERS-CoV was initially thought to use the angiotensin-converting enzyme 2 (ACE2) receptor to gain entry to bronchial epithelial cells, as in the case of SARS-CoV and SARS-CoV-2, but it is now thought to use the DPP4 receptor, which localizes to the lower respiratory tract. A relatively small population of lung cells are affected, supporting the hypothesis that a high infectious dose may be needed to cause infection, in contrast to SARS-CoV and SARS-CoV-2. The immune response to MERS-CoV infection appears to vary as well, with limited reports of poor outcomes associated with lack of a type-1 interferon response (interferon alpha), which in turn hindered a sufficient Th-1 immune response to clear the viral infection.
Details regarding viral transmissibility remain murky. As mentioned earlier, other viral respiratory pathogens may infect via receptor cells generally located in the upper respiratory tract, but MERS-CoV can infect via receptor cells located in the lower respiratory tract: this difference can have bearing on viral loads expelled by coughing and sneezing. It has yet to be established whether infections represent single zoonotic transmission with subsequent human transmission or multiple zoonotic events in different locations. The virus was likely circulating undetected for 10 to 15 years prior to its discovery based on molecular clock analyses. At least seven separate zoonotic transmissions were reported prior to 2012. Although it is not thought to transmit effectively from person to person, the possibility remains that mutations could allow transformation into a more readily communicable respiratory illness.
The environmental stability of coronaviruses has been studied extensively. Although they are inactivated by heat and light, they can persist in the environment for significant periods of time, particularly in cold climates. MERS-CoV has been detected in and isolated from numerous sources including multiple bat species and dromedary camels but not sheep, goats, cattle, or other camels ( Fig. 96.1 ). Undercooked camel meat or unpasteurized milk can be sources of spread. Camels can develop an upper respiratory infection similar to the common cold in humans, with viral shedding demonstrated in oral and nasal secretions and milk but not other fluids. This differs from viral shedding in humans, which occurs in both the upper and lower respiratory tracts and can persist for up to 1 month after infection.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here