Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Thoracic aortic aneurysms (TAAs) are potentially lethal due to rupture or dissection. They represent the 19th most common cause of death in the United States. Only about 5% of patients present with symptoms before an acute event occurs, and for the other 95% the first “symptom” is often death.
For this reason, the diagnosis is often only made at the time of aortic dissection or death.
Arteriosclerotic disease is the most common cause of TAA However, aneurysmal degeneration can also occur in the elderly population. Aging results in changes in collagen and elastin, which lead to weakening of the aortic wall and aneurysmal dilation. According to the Laplace law, luminal dilation results in increased wall tension and the vicious cycle of progressive dilation and greater wall stress. Pathologic sequelae of the aging aorta include elastic fiber fragmentation and cystic medial necrosis.
Other risk factors are all of the conditions that increase aortic wall stress, such as hypertension, cocaine abuse, extreme weightlifting, trauma, and aortic coarctation.
The etiology of aortic aneurysms is multifactorial and is correlated to the presence of multiple risk factors.
TAA is also related to genetic conditions that lead to aortic medial weakness or destruction such as Loeys-Dietz syndrome and Marfan syndrome. , ,
The thoracic aorta plays a vital role not only as a conduit of high-flow and high-volume blood but also to control systemic vascular resistance and heart rate. Specific pressure-responsive receptors localized at the level of the ascending aorta and in the aortic arch can modify the heart function according to the aortic pressure. Moreover, thanks to its elasticity, the aorta acts as a second pump during the diastolic phase.
Hemodynamic factors play a role in the formation of aortic aneurysms. The human aorta is a relatively low-resistance circuit; the lower extremities have higher arterial resistance. The repeated trauma of a reflected arterial wave on the distal aorta may injure a weakened aortic wall and contribute to aneurysmal degeneration. Systemic hypertension compounds the injury and contributes to aneurysm formation.
The normal diameter of the aortic segments differ depending on age, sex, and body surface area. In general, the aortic diameter does not exceed 40 mm in adults. By definition, an aneurysm is a localized or diffuse dilation of an artery with a diameter at least 50% greater than the normal size of the artery. An expansion of 0.9 mm in men and 0.4 mm in women for each decade of life is reported.
In patients with Marfan syndrome, the aorta may grow at a rate of 0.5–1 mm/yr. In patients with Loeys-Dietz syndrome the thoracic aorta can grow faster than 10 mm/yr, resulting in death at a mean age of 26 years.
TAAs of the descending aorta grow faster (3 mm/yr) than those localized in the ascending aorta (1 mm/yr).
When the aortic diameter exceeds 60 mm in the ascending aorta and 70 mm in the descending, a higher risk of dissection and rupture is reported.
TAAs are divided according to their location in the ascending aorta, aortic arch, and descending aorta. Ascending aortic aneurysms may extend as proximally as the aortic annulus and as distally as the innominate artery. Descending thoracic aneurysms commence beyond the left subclavian artery.
TAA can also be correlated with the presence of aneurysms of the abdominal aorta (AAA); 27% of patients with an AAA also have a concomitant TAA. Another study reported that larger ascending aorta and aortic valve disease are more frequent in those patients with AAA.
Crawford introduced the first anatomic thoracic (TAA) and thoracoabdominal aneurysm classification in 1986:
Type I: involves the descending aorta from the left subclavian artery to the suprarenal abdominal aorta.
Type II: extends from the left subclavian artery to the aortoiliac bifurcation.
Type III: involves the distal thoracic aorta to the aortoiliac bifurcation.
Type IV: thoracoabdominal aortic aneurysms restricted to the abdominal aorta below the diaphragm.
Type V (added by Safi’s group): extends from the distal thoracic aorta and includes the celiac and superior mesenteric origins, but not the renal arteries.
The most common complications of TAA are acute rupture and dissection.
In most cases, TAAs are asymptomatic and they are usually found incidentally on chest radiographs or other imaging studies. Although the value of screening in Marfan syndrome patients is well recognized, for patients with a bicuspid aortic valve there is controversy over whether first-degree relatives should be screened.
Cough, shortness of breath, or difficult or painful swallowing can be present in patients with large TAAs. In the presence of these specific symptoms, a clinical examination should be conducted. An evaluation of the blood pressure in both arms can exclude the presence of an aortic dissection.
Large ascending aortic aneurysms may cause superior vena cava obstruction manifesting as distended neck veins. Ascending aortic aneurysms also may develop aortic insufficiency, with widened pulse pressure or a diastolic murmur. Arch aneurysms may cause hoarseness, which results from stretching of the recurrent laryngeal nerves. Descending thoracic aneurysms may compress the trachea or bronchus and cause stridor, wheezing, or cough. Compression of the esophagus may result in dysphagia. Erosion into surrounding structures may result in hemoptysis, hematemesis, or gastrointestinal bleeding. Erosion into the spine may cause back pain or instability. Spinal cord compression or thrombosis of spinal arteries may result in neurologic symptoms of paraparesis or paraplegia. Descending thoracic aneurysms may thrombose or embolize thrombus and atheromatous debris distally to visceral, renal, or lower extremity arteries. ,
Chest x-ray or ultrasound performed for other indications may incidentally detect abnormalities of aortic contour or size. Chest radiography has a very low sensitivity for the diagnosis of aortic dissection. However, pooled data from 10 studies indicates predictive sensitivity for thoracic aortic disease of 64% in case of a widened mediastinum and 71% for abnormal aortic contour.
Once dilation of the aorta is suspected, complete visualization of whole aorta is needed to assess the extension of the dilation and possible complications.
Computed tomography angiography (CTA) is the method of choice for correct diagnosis and accurate evaluation of the thoracic aorta (pooled sensitivity 100%, pooled specificity 88%).
It can be performed both electively, in case of suspicious findings or symptoms, or as an emergency.
CTA allows a complete visualization of the whole aorta and peripheral vessels. A complete examination of the thoracic aorta is based on axial images acquired before and after intravenous injection of contrast media. Delayed images are recommended after stent-graft repair to detect the presence of endoleaks.
Electrocardiogram-gated acquisition protocols in elective examinations can reduce motion artifacts of the aortic root and thoracic aorta. ,
With CTA we can evaluate the maximum aortic diameter, the presence of calcifications or thrombus, and the extent of the aneurysm disease. Contrast extravasation is correlated with aortic rupture.
A complete examination must always include the abdominal aorta, the ileofemoral axis (common femoral artery), and assessment of the coronary artery status to exclude the presence of significant coronary artery disease before treatment. This information is essential for preoperative planning.
Postenhanced images are acquired after the injection of 150 mL of iodinated contrast media injected at a flow rate of 3–5 mL/s, followed by a saline bolus using a power injector. , ,
CTA has some drawbacks, such as the administration of iodinated contrast media (allergies, renal impairment) and x-ray exposure.
Because of the intrinsic contrast between blood and the vessel wall, magnetic resonance imaging (MRI) is a valuable tool for detecting vessel wall disease and in general for the assessment of aortic disease.
However, MRI cannot be performed in the emergency setting because of its longer acquisition time and high movement artifact susceptibility.
MRI has some advantages in not requiring ionizing radiation or large volumes of iodinated contrast. This is particularly valid in young patients with connective tissue disorders. However, there are limitations in patients with pacemakers, other metallic implants, or reduced renal function (nephrogenic systemic fibrosis). , ,
Some sequences could be useful to assess aortic disease, such as spin echo black-blood sequences to evaluate the aortic dissection flap and gradient echo sequences to demonstrate changes in aortic diameters during the cardiac cycle. Non electrocardiogram-gated contrast-enhanced MRI with intravenous injection of gadolinium is useful and can be performed rapidly with the option to evaluate the aortic arch anatomy. Standard techniques include injection of gadolinium at flow rates of 3–5 m s −1 with a bolus tracking technique. It is mandatory to evaluate both the source and the maximum intensity projection images. Newer time-resolved 3D flow-sensitive MRI of the entire thoracic aorta allows visualization and measurement of blood flow patterns.
A limiting factor in performing MRI is the difficulty of evaluating aortic valve calcification of the anchoring zones, which is important for avoiding endoleaks post endografting.
Several workers use fluorodeoxyglucose-positron emission tomography to assess aortic involvement with inflammatory disease and to detect graft infections. However, these indications are debated.
The potential advantage of positron emission tomography may be when it is performed in combination with CT to improve imaging resolution.
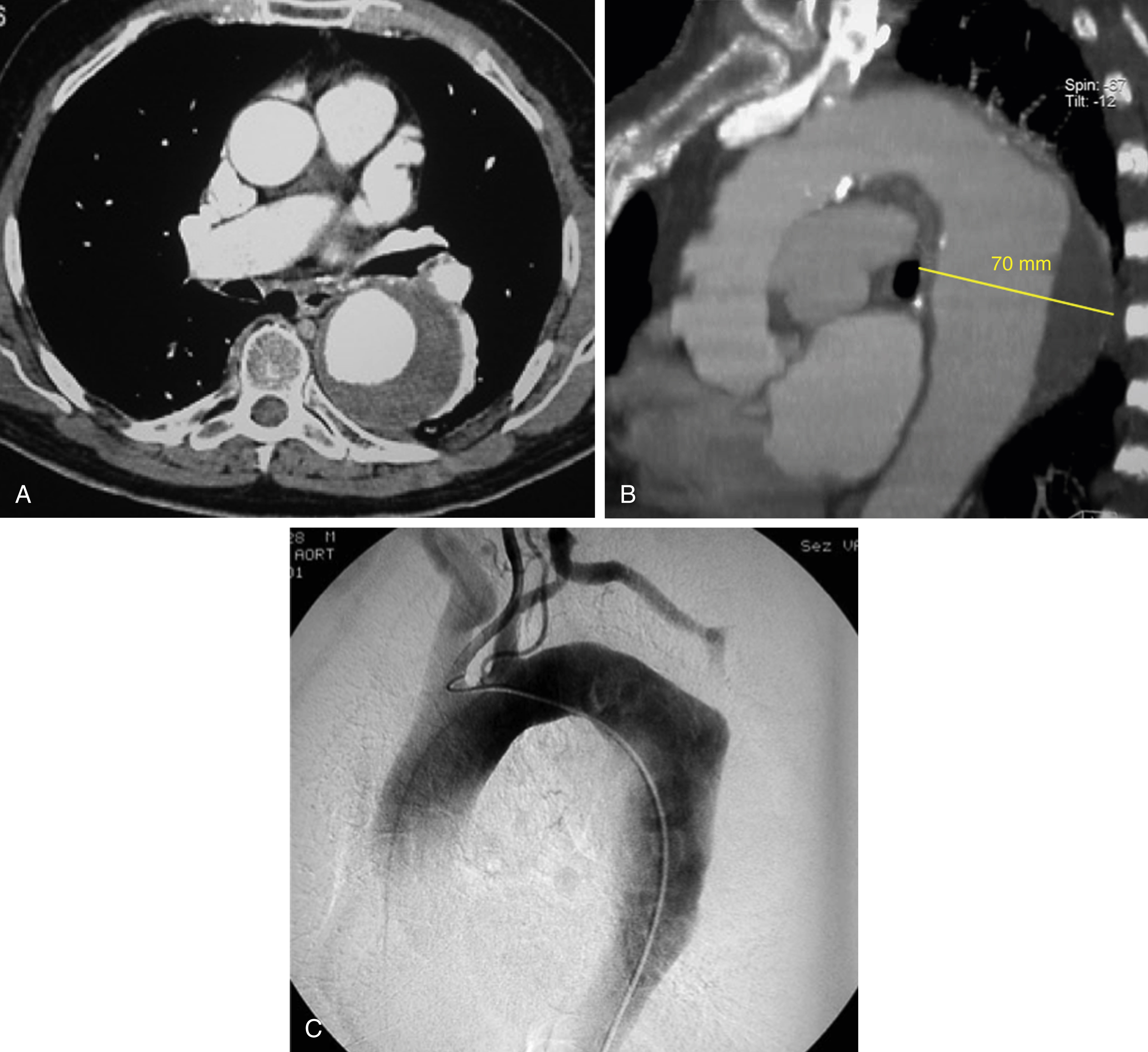
Angiography was once the gold standard technique for evaluating the aorta, but has been replaced by CTA. Catheter-based invasive angiography is now generally reserved for situations where a therapeutic intervention is planned or as part of the interventional procedure to visualize the aortic lumen, aortic side branches, and collaterals ( Fig. 51.1 )
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here