Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Intensive care may account for more than one-third of the total hospital costs for cardiac surgery patients, and much of the short-term morbidity and mortality are based on perioperative events. The overall mortality after cardiac surgery is low. However, this ranges from less than 1% for elective coronary artery bypass grafting to more than 30% for more complex surgery in patients with significant myocardial dysfunction and significant comorbidities. Hence a comprehensive understanding of cardiovascular physiology and surgery is essential for a modern-day intensivist.
The initial days of care for a cardiac surgery patient present multiple challenges for an intensivist. The intensive care unit (ICU) stay for most patients lasts for 24–48 hours, but during this critical period, life-threatening problems such as low cardiac output (CO), arrhythmias, and coagulopathy may occur. After 48 hours, the problems encountered tend to become more like those experienced by other groups of critically ill patients.
The development of modern cardiac surgery is intimately related to the development of the ICU. Until the 1950s, cardiac surgery was limited to the control of traumatic injuries and the closed repair of valves. The development of the extracorporeal pump oxygenator in 1953 ushered in the era of open heart surgery. Heart valve replacement then became possible, and subsequently in the 1960s, coronary artery bypass grafting (CABG) for ischemic heart disease was developed and rapidly popularized.
Several studies have demonstrated that risk-adjusted mortality rates after CABG vary significantly among surgeons and hospitals and that mortality is related both to the number of surgeries performed by each surgeon and the total volume of procedures performed at the hospital. For high-risk surgical patients, survival is also related to the characteristics of the ICU care.
Over the past decade, the population of patients treated with cardiac surgery has changed dramatically. Advances in cardiology, including reperfusion therapy, angioplasty, stenting, and drug-eluting stents, have obviated the need for surgical approaches to treatment except for particularly complex problems or after failure of other, less invasive modalities. In the year 2000, 561,000 patients in the United States underwent percutaneous transluminal coronary angioplasty (PTCA), an increase of 262% relative to 1987. In the same year, 314,000 patients underwent CABG. Multiyear trends, represented in Fig. 144.1 , show a leveling off and subsequent decrease in the overall number of patients undergoing CABG. Studies comparing the use of stents versus CABG for left main disease have found no significant difference in rates of death, Q-wave infarction, or stroke; however, stenting was associated with higher rates of target vessel revascularization than was CABG.
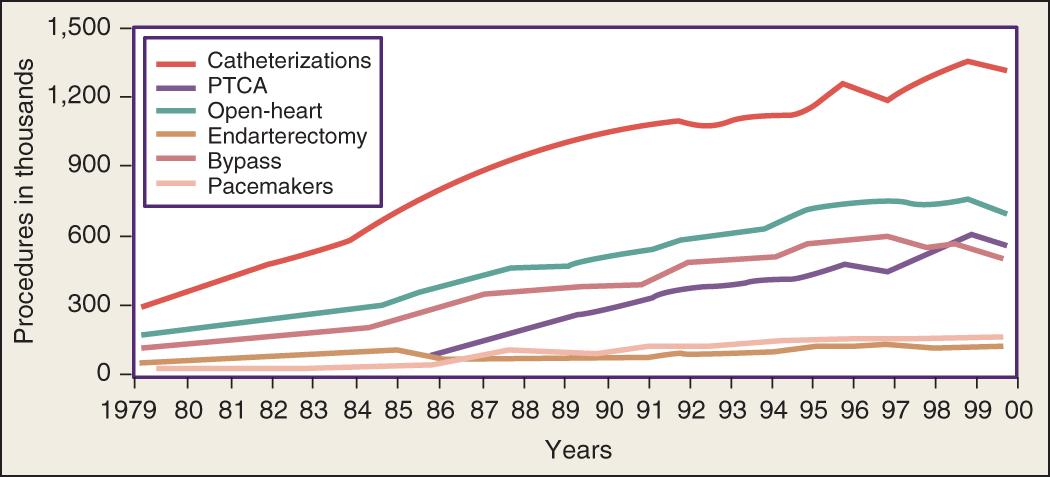
Even as younger patients are being treated with interventional techniques, the elderly are increasingly referred for operation. As the population ages and care becomes more sophisticated, cardiac surgery is being performed on older, sicker, and more complicated patients. Although these operations are successful even in most octogenarians, they are associated with increased hospital mortality and longer ICU and hospital stays. It is clear, however, that good results in terms of long-term survival and quality of life are achievable.
There has been significant research into organizational factors in general ICU; however, there is less supporting information around issues such as open versus closed ICU and surgeon-led care versus intensivist-led care in the cardiac arena.
The increasing age of patients undergoing cardiac surgery and the relatively high incidence of adverse effects related to cardiopulmonary bypass (CPB) have led to the development of less invasive cardiac surgical techniques. These techniques are intended to decrease postoperative morbidity, reduce length of hospital stay, reduce costs, and hasten recovery of lifestyle ( Table 144.1 ). Three major techniques have been proposed.
| Technique | Incision Site | Cannulation Site | Advantages | Disadvantages |
|---|---|---|---|---|
| Conventional | Median sternotomy | Ascending aorta | Excellent exposure | Mediastinitis |
| CABG | Median sternotomy | Right atrium and Ascending aorta | Stable closure and extensive experience | Postoperative respiratory function limited by pain |
| MIDCAB | Left or paramedian or right thoracotomy or partial sternotomy | Ascending aorta and right atrium | Avoids median sternotomy |
|
| Port access | Right anterior thoracotomy, or | Ascending aorta via right paramedian port |
|
|
| Paramedian or left thoracotomy | Femoral vessels |
|
|
|
| OPCAB | Median sternotomy, or Right or left thoracotomy, Partial sternotomy | None |
|
|
Minimally invasive direct coronary artery bypass (MIDCAB) differs from conventional CABG mainly in the type of surgical incision. Instead of a median sternotomy, access is obtained via a left or right thoracotomy, a parasternal incision, or a partial sternotomy. Although the proposed benefit of such an approach is the reduction in morbidity related to median sternotomy, this advantage has not been demonstrated. MIDCAB grafting is a challenging technique and should be performed only in selected patients with favorable coronary anatomy. Both bare-metal and drug-eluting stenting have been shown to be inferior to MIDCAB for proximal left anterior descending coronary artery lesions, owing to higher reintervention rates and similar results in mortality and morbidity. ,
Off-pump coronary artery bypass (OPCAB) is performed on a beating heart without the benefit of CPB. The proposed advantage of this procedure is the reduction of morbidity related to hypothermia and CPB. The procedure is undertaken using partial to full heparinization. Extubation may be achieved earlier in these patients because they do not require rewarming and are less coagulopathic. A subset of patients cannot tolerate the extent of retraction of the heart required for the surgery and need to be urgently placed on CPB. These patients may suffer ischemic myocardial injury and require support with inotropes or intraaortic balloon pumping (IABP) during the postoperative period.
A third method of minimally invasive cardiac surgery is the port access technique. This operation entails obtaining access for CPB with the use of endovascular catheters. It allows surgery to be performed using CPB via either a left or right thoracotomy. The technique is particularly useful for mitral valve replacement through a right thoracotomy and for redo CABG (avoiding the complications associated with repeat sternotomy). The port access technique has been shown to be safe and is associated with shorter lengths of stay, reduced transfusion requirements, fewer infections, decreased incidence of renal failure, and less atrial fibrillation (Afib) compared with conventional techniques. Widespread adoption of this technique has been limited by the technical complexity of placing the required catheters, which requires both extra time and a specially trained, skilled operative team. As the techniques of minimally invasive cardiac surgery continue to evolve, the intensivist caring for cardiac surgical patients must continue to keep abreast of these new methods.
Optimal results from cardiac surgery require a skilled, dedicated, and multidisciplinary ICU team. Patients undergoing cardiac surgery are usually admitted to the hospital on the day of surgery and arrive in the ICU directly from the operating room (OR). A typical patient is transferred to a step-down unit on the morning after surgery. This unit allows continued monitoring with telemetry for an additional 24–48 hours. Patients remaining in the ICU beyond 48 hours tend to become similar to a standard ICU population, as they develop secondary complications such as sepsis, pneumonia, and acute respiratory distress syndrome (ARDS).
Guidelines developed by the American Heart Association and the American College of Cardiology outline the requirements for cardiac surgical ICUs. These include the development of protocol-driven care, a minimum number of cardiac surgical ICU beds that is half the number of surgeries performed per week, and one-to-one nursing care during the first night in the unit. The ICU coverage by a dedicated intensivist has been shown to improve outcomes in other types of major surgeries and should be recommended after cardiac surgery as well.
Cardiac surgery involves a continuum of care from presentation to postdischarge management and rehabilitation. The intensive care specialist must be involved in this continuum, rather than functioning in isolation from surgeons, anesthetists, and cardiologists.
Successful management of a postoperative cardiac surgery patient begins by understanding what occurs in the OR because operative problems often persist after transfer to the ICU. An understanding of the technical and pathophysiologic aspects of CPB may help an intensivist better manage the postoperative cardiac surgical patient.
The goal of CPB is to separate the heart and lungs from the systemic circulation so that the heart may be arrested while the surgical repair is performed. Blood is drained from the right side of the heart via a cannula in the right atrium or via the femoral vein advanced into the right atrium. The blood is collected in a reservoir and then pumped through an oxygenator that contains a membrane where the blood is oxygenated and carbon dioxide is removed ( Fig. 144.2 ). A perfusionist controls both the fraction of inspired oxygen and the rate of oxygen flow through the circuit, thereby controlling the patient’s arterial oxygen and carbon dioxide levels, respectively. The treated blood then passes through an air filter and is returned to the patient via an arterial cannula placed in either the ascending aorta or the femoral artery. The perfusionist controls the amount of systemic flow provided to the patient (i.e., CO). Mild to moderate systemic hypothermia (28°C–34°C) is used during bypass to minimize oxygen consumption by both the body and the brain. After adequate CPB is established, an aortic cross-clamp is applied to the ascending aorta, between the aortic cannula and the heart. The interval when the cross-clamp is applied is referred to as ischemic time, because no blood is circulated through the heart during this period. The heart is arrested by infusing a high-concentration potassium solution into the native coronary arteries (antegrade cardioplegia) via a cannula placed between the aortic cross-clamp and the heart. Cardioplegia may also be given “backward,” through the venous system of the myocardium (retrograde cardioplegia) via a catheter placed in the coronary sinus. Potassium is used as the arresting agent because it stops the heart from beating and minimizes myocardial oxygen consumption.
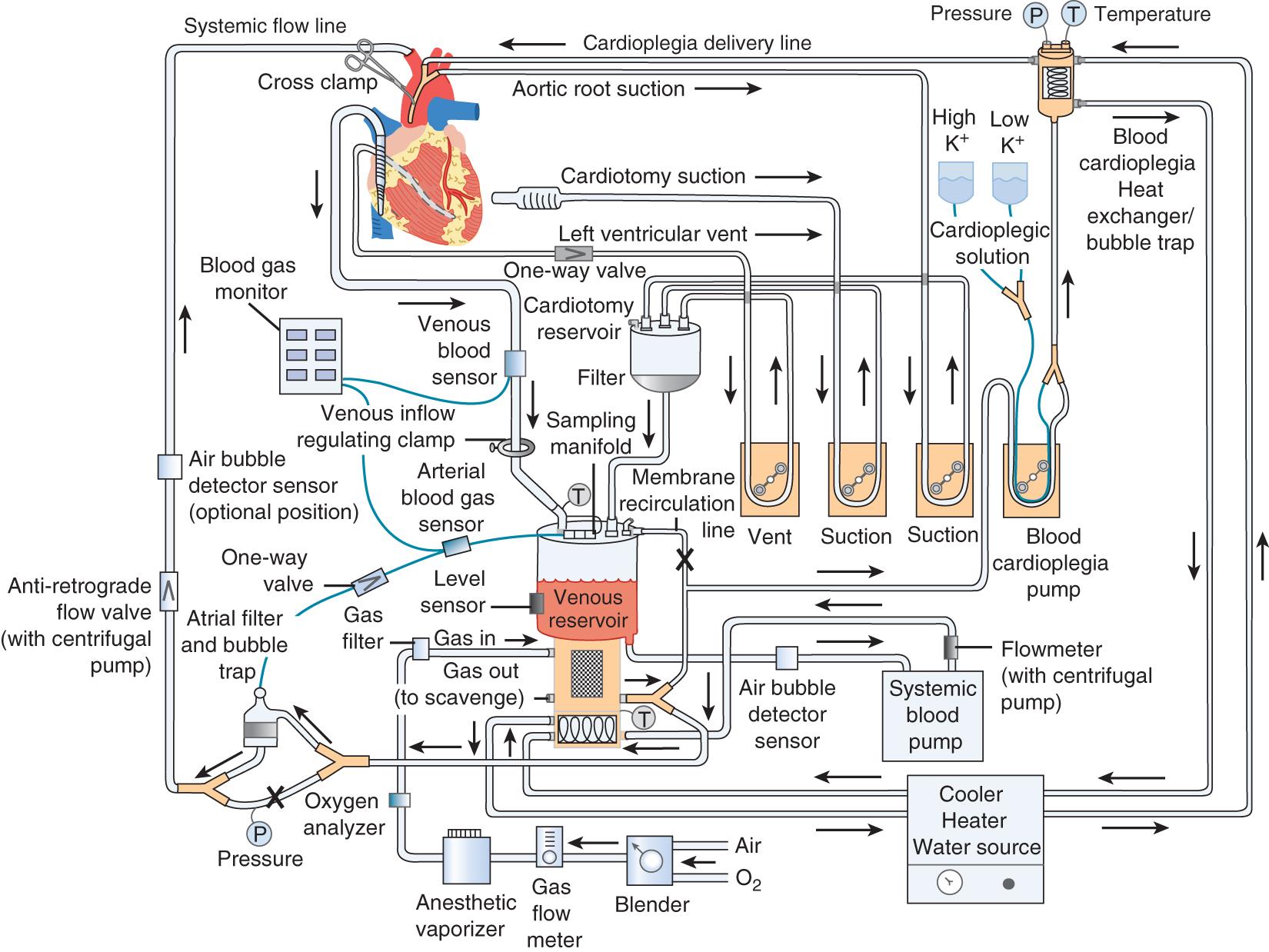
Several measures are taken to protect the heart during ischemic time, because irreversible myocardial damage may otherwise occur. Electromechanical arrest is the most important protective measure, because the beating action of the heart accounts for about 85% of the heart’s total oxygen consumption. The heart is usually cooled to about 10°C with a cold cardioplegia solution (4°C) supplemented with topical ice slush. Additionally, the left ventricle is “vented” to prevent distention, which could lead to subendocardial ischemia. Finally, various additives are included in the cardioplegia solution to minimize myocardial edema, maintain normal intramyocardial pH, and provide substrates for anaerobic metabolism. The adequacy of intraoperative myocardial protection is critical for determining the subsequent course and final outcome for a patient.
Weaning from CPB is the process whereby cardiopulmonary function is transferred from the bypass system back to the patient’s own heart and lungs. Successful separation from CPB requires that the metabolic, cardiac, and respiratory parameters are as close to normal as possible. Separation from CPB implies that the native circulation will be required to support the body’s metabolic demands. The surgical team manipulates the heart rate and rhythm, preload, afterload, and myocardial contractility to achieve this goal.
In most cases, normal sinus rhythm is restored after discontinuation of cardioplegia and rewarming of the heart. Occasionally, discontinuation of cardioplegia and rewarming lead to the onset of ventricular fibrillation; in such cases, electrical defibrillation is required. Other dysrhythmias commonly encountered are atrioventricular dissociation and Afib. An attempt should be made to convert these to sinus rhythm by pharmacologic means. Bradyarrhythmias are treated by pacing, using temporary epicardial wires placed by the surgeon after completion of the repair. A heart rate of 70–90 beats/min usually is optimal. Pharmacologic support of the circulation may be needed to provide appropriate afterload or systemic vascular resistance (SVR) during separation from CPB. Most patients are vasodilated to some extent, possibly as a result of a systemic inflammatory response to CPB or the effects of rewarming, or both. As a consequence, the infusion of a vasoconstrictor is often required. Care must be taken to strike a proper balance so that increased SVR maintains adequate arterial blood pressure without excessively increasing left ventricular afterload and compromising CO.
Most often, myocardial function is adequate, and the infusion of an inotrope is not necessary. However, inotropic support is often needed for patients with a poor preoperative ventricular function or inadequate myocardial protection or revascularization during CPB. The optimal inotrope in this situation is a matter of considerable debate, and data are lacking to support a strong recommendation for a specific agent. Epinephrine, norepinephrine, dopamine, dobutamine, amrinone, and milrinone have all been used successfully. Intraoperative monitoring using transesophageal echocardiography (TEE) is particularly useful for titration of inotropic therapy.
Once all preparations for separation from CPB have been made, the perfusionist begins to wean the patient from bypass. This is done by slowly decreasing the amount of blood drained from the right atrium while simultaneously reducing flow into the aorta. Once the patient is off CPB (i.e., no blood is being drained from the right atrium into the circuit), the perfusionist, at the direction of the anesthesiologist or surgeon, may continue to infuse through the aortic cannula. This maneuver allows optimization of ventricular filling or preload. However, care must be taken not to overdistend the heart; also, during this period, TEE is extremely useful.
After weaning from CPB, protamine is given to neutralize any residual heparin. Dosing may be based on the patient’s weight, the total amount of heparin given, or an assay of residual heparin activity. Institutional preference governs the technique employed, but all have been proven effective. Several adverse responses to protamine administration are possible, including histamine-induced systemic hypotension, immunoglobulin E–mediated allergic reactions, and complement-mediated catastrophic pulmonary hypertension. The clinical sequelae of cardiopulmonary bypass are summarized in Table 144.2 .
| Intraoperative Event | Sequelae | Postoperative Manifestation |
|---|---|---|
| Aortic cannulation and clamping |
|
|
| Right atrial cannulation |
|
|
| Femoral artery cannulation |
|
|
| Femoral vein cannulation |
|
|
| Graft twisting |
|
|
| High-dose heparin |
|
|
| Crystalloid priming of CPB circuit |
|
|
| Extracorporeal circulation |
|
|
| Cardioplegic arrest |
|
|
| Hypotension on CPB |
|
|
| Hypothermia |
|
|
| Hypothermic circulatory arrest |
|
|
| Reaction to protamine |
|
|
| Coronary air embolism | Coronary ischemia | Myocardial injury and dysfunction |
After chest closure, confirmation of hemodynamic stability, and adequate medical and surgical hemostasis, the patient is transferred to the ICU. Transport of a critically ill patient is a potentially dangerous process and requires extreme vigilance. Transport between the OR and the ICU should be done with the same degree of monitoring as would be available at either end. This usually includes continuous monitoring of arterial blood pressure, pulmonary artery pressure and/or central venous pressure (CVP), electrocardiogram (ECG), and pulse oximetry. The transport bed should be equipped with a full oxygen tank, a bag valve mask, intubation equipment, resuscitation drugs, and defibrillator. Care must be taken to ensure that infusions of vasoactive drugs are not interrupted.
On arrival in the ICU, the intensivist-led team assumes patient care. A detailed sign-out from the operative team ensures continuity of care. The sign-out should include a detailed history, including an assessment of preoperative cardiac functional status, a list of preoperative medications, and a detailed description of the surgery. Key facts are the type of repair performed, target vessels (if the patient has undergone CABG), duration of CPB and cross-clamping, difficulties encountered in separation from CPB, presence of abnormal bleeding, and postoperative assessment of cardiac function. All treatments administered in the OR should be detailed, in particular, fluids, blood products, and vasoactive drugs.
Handover should be structured to minimize disruption and maximize communication. The priorities are to confirm surgical procedure and any complications, confirm integrity and position of the endotracheal tube, re-establish mechanical ventilation of both lungs, re-establish all patient monitoring, access details of premorbid conditions, confirm venous, arterial and central access, heart rhythm and presence and mode of any pacing devices. The handover should also include details of the chest drains, establishing a blood pressure target, document results of any intraoperative echocardiography regarding ventricular function, provide details of intraoperative transfusion of blood or factors together with the results of any benchtop or laboratory tests of hemostasis and getting an early 12-lead electrocardiograph to exclude or identify acute ischemia.
Once care has been handed over to the ICU team, a thorough examination of the patient should immediately follow. This examination should include verification of endotracheal tube placement, type and position of arterial or central venous lines, chest tube position and patency, and presence and location of any epicardial pacing wires.
All patients admitted to the ICU after cardiac surgery will have their blood pressure continuously monitored using an intraarterial line, which is usually placed in either a radial or a femoral artery. Accuracy of the measurements depends on strict attention to calibration, leveling, and removal of air from the tubing. After CPB, femoral arterial pressure may more accurately reflect central aortic pressures, but this problem is usually resolved by the time the patient arrives in the ICU. If the radial artery is cannulated, the hand should be examined for signs of ischemia. Vascular complications of femoral arterial lines are extremely rare, but femoral catheters may be associated with an increased incidence of infection.
Central venous access is required in all patients for drug administration and hemodynamic monitoring. In low-risk patients, a CVP catheter may be all that is needed, particularly if echocardiography is available as a backup. Pulmonary artery catheters have the advantage of allowing measurement of pulmonary artery occlusion pressure (PAOP), thermodilution, and CO, in addition to sampling of the mixed venous blood saturation (SvO 2 ). Use of the pulmonary artery catheter remains controversial. Improved outcome resulting from the use of a pulmonary artery catheter for monitoring cardiac surgical patients has not been demonstrated. Some studies showed an increased risk of death or adverse outcome when treatment was guided by the use of a pulmonary artery catheter. , However, many of these studies have been criticized on methodologic grounds, and the use of a catheter in cardiac surgery remains widespread. Current guidelines recommend the use of a pulmonary artery catheter in high-risk patients undergoing surgery (severely decreased left ventricular [LV] function [ejection fraction <30%], right ventricular [RV] failure, pulmonary hypertension, severe renal insufficiency, or thoracic transplantation) in an appropriate practice setting. Such a setting is the one in which the physician and nursing staff are familiar with the catheter and trained to properly interpret the information obtained. If echocardiography is readily available, it is possible to manage even high-risk patients using a CVP catheter.
On admission to the ICU, the patient is connected to a continuous ECG monitor, and a formal 12-lead ECG is obtained. The cardiogram is examined for rate, rhythm, QRS complex morphology, and signs of myocardial ischemia. For patients who are being paced postoperatively, the type of pacing and the degree of capture should be assessed.
Continuous ECG monitoring allows detection of arrhythmias. If an arrhythmia is detected, a 12-lead ECG should be obtained and serum electrolyte concentrations should be measured. Treatment of arrhythmias should be carried out using established protocols. If a malignant arrhythmia occurs, myocardial ischemia should be considered as a possible precipitating cause.
Monitoring of trends in ST-segment elevation or depression allows early detection of postoperative myocardial ischemia. Although transient ST-segment changes are relatively common and of unclear significance, persistent changes should be investigated by obtaining a 12-lead ECG and measuring circulating levels of creatine kinase myocardial band (CK-MB), troponin-T, or troponin-I. , If ischemia is strongly suspected, echocardiography followed by coronary angiography should be considered. Findings from these studies may indicate the need for further coronary revascularization.
The postoperative chest radiograph should be systematically evaluated. Proper placement of the endotracheal tube and any central lines inserted should be confirmed. If a pulmonary artery catheter is placed, the location of its tip should be noted and adjusted as needed. The lung fields should be examined for the presence of pneumothorax (PTX) or consolidation. Additional air may be noted as subcutaneous emphysema or pneumopericardium, although these findings are of little clinical significance. Further examination of the lung fields commonly shows small areas of atelectasis and pleural effusion. The cardiac silhouette is often enlarged after surgery as a result of myocardial edema and accumulation of fluid in the open pericardial sac. Increasing size of the cardiac silhouette or pleural effusions on serial chest radiographs may be evidence of ongoing mediastinal bleeding. The admission radiograph detects abnormalities in up to 35% of patients, although few of these result in a change in therapy.
Echocardiography is an excellent tool for evaluating chamber size and function and the adequacy of valve repair or replacement. Indications include postoperative assessment of LV function, assessment of unexplained sudden hemodynamic deterioration, evaluation to rule out pericardial tamponade, and workup of new cardiac ischemia. Limitations to transthoracic echocardiography include inadequate windows early after operation because of air and edema in the soft tissues and wound dressings.
TEE is increasingly being used as a tool to facilitate decision making in managing critically ill patients, including cardiac surgical patients. In the cardiac surgical ICU, this modality may have a particularly high yield when used to establish the cause of postoperative hypotension. In one large series, a new diagnosis was established or an important pathology was excluded in 45% of TEE examinations performed in the ICU. Pericardial tamponade was diagnosed in 34 cases (11%) and excluded in 36 cases (12%). Other diagnoses included severe LV failure and the presence of large pleural effusions. The results of TEE had an impact on therapy in 220 cases (73%) by leading to a change of pharmacologic treatment and/or fluid administration, reoperation, or a decision that reoperation was unnecessary.
However, the early postoperative period after cardiac surgery presents unique challenges to image optimization because of mechanical ventilation and surgical dressings and drains and to image interpretation because of diagnostic findings unusual in other clinical settings.
Pericardial hematoma can cause focal cardiac chamber compression; thus every cardiac chamber must be clearly visualized. Rarely, massive hemothorax can cause tamponade. Dynamic left ventricular outflow tract (LVOT) obstruction should be suspected in the patient with mitral valve repair, hypertrophic left ventricle, or aortic valve replacement for aortic stenosis who develops shock that fails to respond to catecholamines or intraaortic balloon counterpulsation.
Echocardiography in these patients demonstrates a high LVOT flow velocity and mitral regurgitation. RV dysfunction may occur in isolation because of inadequate intraoperative cardioplegia. Increased intrathoracic pressure may present with hemodynamic features similar to pericardial tamponade, but should be recognized from assessment of ventilation. On echocardiography there are features of acute cor pulmonale. Patient prosthesis mismatch results in a high transvalvular pressure gradient and difficultly weaning from mechanical ventilation. Other causes of prosthetic valve dysfunction (e.g., leaflet entrapment or paravalvular leak) warrant expert and TEE evaluation.
Patients are typically admitted to the ICU intubated and ventilated. Sedation with a short-acting agent, typically propofol, is continued until the patient is ready for extubation. ,
Hypothermia (<36°C) is used intraoperatively to preserve cardiac and neurologic function postoperatively. Unfortunately, hypothermia carries the risk of adverse effects, including increased SVR, increased risk of arrhythmias, shivering, and coagulopathy. Shivering is of particular concern, as it increases oxygen (O 2 ) consumption while also increasing carbon dioxide (CO 2 ) production, sometimes precipitating acidosis. Pethidine and dexmedetomidine have been shown to treat shivering. , The increase in SVR can mask hypovolemia and increase afterload, which increases myocardial demand. The dangerous combination of increased SVR and coagulopathy can also lead to mediastinal bleeding. Without active rewarming with forced air, hypothermia and its complications, particularly the prolongation of anesthetic time half-life, can also prolong intubation times. There is evidence that the risk of wound infection also increases with hypothermia as well, likely owing to the immunosuppressive effects of hypothermia. Therefore upon arrival to the ICU, steps should be taken to increase core body temperature to normal using forced air, as that has been shown to be more effective at expeditiously increasing core body temperature than radiant heaters or warming blankets.
Once hemodynamic stability is ascertained and chest tube drainage is judged to be under control, the patient is allowed to awaken. There is no need for prolonged weaning from mechanical ventilation. A short trial of spontaneous ventilation is sufficient to determine whether respiration will be adequate without mechanical support. Chest tubes are commonly removed on the first postoperative day. The pulmonary artery catheter, if present, is discontinued, and the patient may be transferred to a step-down unit.
The current trend in cardiothoracic surgery is toward early extubation, variably described as extubation within 6 hours or up to 12 hours. Extubation criteria may differ among cardiothoracic groups, but, at a minimum, patients should be awake with stimulation and following commands, moving all extremities (i.e., without neuromuscular blockade), minimal chest tube drainage (<50 or 100 mL/hr), core temperature above 35.5°C, hemodynamically stable with a CO >2.2 L/min per meter squared, free of arrhythmias, with ventilator stability with respiratory rate between 12 and 30 breaths/min, and reassuring arterial blood gas (ABG) levels.
Fast-tracking of cardiac surgical patients refers to a comprehensive program designed to reduce both length of stay and hospital costs. , As a part of this program, multiple anesthetic techniques designed to allow earlier postoperative extubation have been proposed, studied, and shown to be safe. The key to proper use of this technique is patient selection. Patients who are at risk of prolonged intubation are those with increasing age, chronic obstructive pulmonary disease, tobacco use, chronic kidney disease, peripheral vascular disease, depressed LV fraction (ejection fraction <30%), myocardial infarction within the past 90 days, preoperative ventilation, prior cardiac surgery, or urgent or emergent surgery. Intraoperative risk factors also exist, such as deep hypothermic circulatory arrest, coagulopathy, and long pump runs (>4 hours). Postoperatively, mediastinal bleeding, hemodynamic instability, respiratory failure, and stroke are the major risk factors for prolonged intubation. It is estimated that about 5%–10% of patients are unable to be extubated early and remain intubated for 48 hours or longer. The fast-track group has been shown to have shorter extubation times, shorter ICU or postanesthesia care unit stays, and a lower incidence of low CO syndrome.
Low CO is the most common problem encountered in the postoperative cardiac surgical patient. A hallmark of low CO is low blood pressure. However, a patient may have a low CO with tissue hypoperfusion and still maintain what appears to be an adequate blood pressure. In the postoperative state, the physician must continuously examine and monitor the patient for signs of hypoperfusion. Physical signs of inadequate tissue perfusion include altered mental status; cool, pale, or even cyanotic extremities; diaphoresis; and low urine output. Global measures of hypoperfusion include increased base deficit, elevated blood lactate concentration, and decreased SvO 2 . Although the clinician must consider CO in terms of adequacy of perfusion, blood pressure per se is still important. Both the brain and kidneys depend on adequate blood pressure to maintain tissue perfusion. Additionally, coronary artery blood flow is dependent on the diastolic blood pressure.
When assessing a patient with hypotension or signs of hypoperfusion, it is useful to consider the problem in relation to the components of CO—namely, preload, contractility, afterload, and rate and rhythm.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here