Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Pterygium is a fibroproliferative condition of the ocular surface linked to chronic ultraviolet light exposure and is common in geographical regions with warm climates.
Simple bare sclera excision of a pterygium is associated with high rates of recurrence and should no longer be performed.
With reported low recurrence rates and minimal complications, pterygium excision with a conjunctival autograft is the recognized standard of treatment.
The most common complication of surgery is pterygium recurrence; this typically recurs within the first year following excision.
Rates of recurrence vary with different surgical techniques; adjuvant therapies can be used in an attempt to reduce recurrence.
Caution should be taken when using adjuvant therapies to reduce pterygium recurrence such as β-irradiation or mitomycin C, as they are related to long-term sight-threatening complications including scleral necrosis and endophthalmitis.
A pterygium is a triangular “wing-like” growth consisting of conjunctival epithelium and hypertrophied fibrovascular subconjunctival connective tissue that encroaches onto the peripheral cornea. It remains an ophthalmic enigma, since the actual pathogenic mechanisms of pterygium development are unknown.
Symptoms of pterygium range from mild ocular surface irritation and dryness to decreased vision from irregular astigmatism or, in severe cases, obscuration of the visual axis. Pterygia can also become inflamed.
The difficulty in treating this deceptively benign disease stems from our lack of understanding of this condition and its propensity for recurrence after surgical excision. There is a plethora of surgical and medical measures available in the management of pterygium. This chapter explores in detail our present understanding of the disease and its various forms of treatment.
Pterygium may be defined as primary or recurrent. Recurrent pterygia are generally more aggressive and can occur several weeks to months after the excision of a primary pterygium.
A pterygium consists of a “head” at its apex, usually with an avascular cap at the leading edge. The “body” represents the main bulk of the pterygium over the sclera and extends from the canthal region.
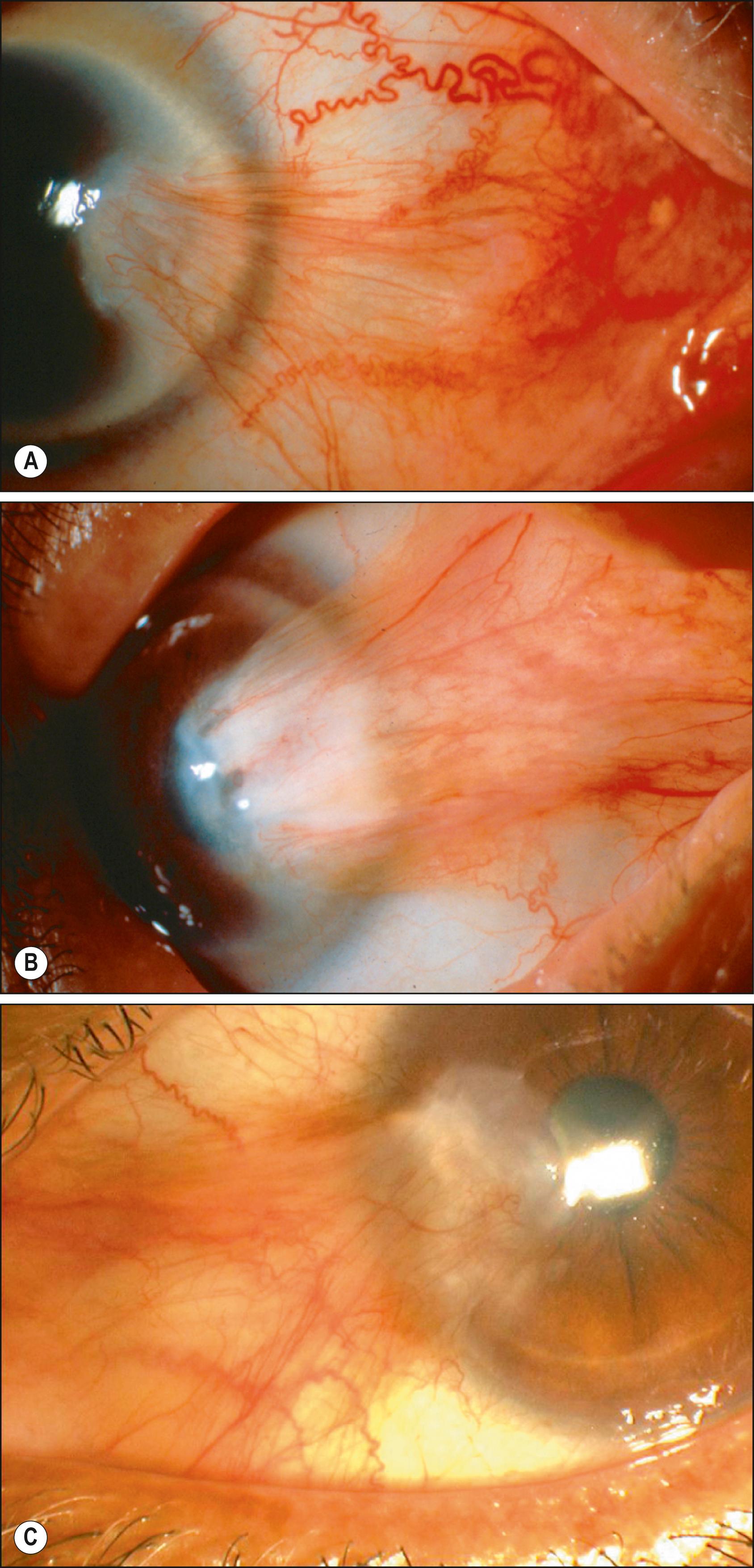
Pterygia most commonly occur over the nasal limbus, in the interpalpebral fissure, but may also occur temporally. Temporal pterygia can also coexist with a nasal pterygium (a “double” headed pterygium). The subconjunctival or fibrovascular component of pterygium may vary considerably in appearance, from thin and translucent to thick and fleshy. Tan et al. developed a simple clinical slit lamp grading scale based on the relative translucency of the body of the pterygium, which was predictive of recurrence if simple excision was performed. In this grading, T1 (atrophic) denotes a pterygium in which episcleral vessels underlying the body of the pterygium are unobscured and are clearly distinguished ( Fig. 144.1A ). Grade T3 (fleshy) denotes a thick pterygium in which episcleral vessels underlying the body of the pterygium are totally obscured by fibrovascular tissue (see Fig. 144.1B ). Pterygia in which the episcleral vessel details are indistinctly seen or partially obscured are categorized as grade T2 (intermediate; see Fig. 144.1C ). This grading system was used in a randomized clinical trial conducted by Tan et al. in which bare sclera pterygium excision was compared to conjunctival autografting. Investigators showed that the risk of recurrence correlated to the degree of fibrovascular tissue present in the pterygium when bare sclera pterygium excision surgery was performed; too few pterygia recurred in the conjunctival autograft group to determine the significance of the grading scale in this surgical technique. Surgical recurrence correlated well with the translucency of the pterygia, with fleshy pterygia having the highest capacity for recurrence and atrophic pterygia the lowest. These differences in recurrence rates were significant for both primary and recurrent pterygia.
Pterygium has a worldwide distribution but is more common in warm, dry climates. , Typically, pterygium occurs most commonly within the periequatorial “pterygium belt” (latitudes 37° north and south of the equator).
A recent meta-analysis of 68 studies with a total of more than 410,000 participants from 24 countries revealed a pooled prevalence of 12.0%. The commonest age of onset appears to be in the second and third decades of life, with the prevalence of pterygium increasing with age. , Large systematic reviews have indicated that men are more likely to develop pterygium compared to women, , which is in contrast to some studies that suggest that the male gender is a protective factor. Such gender differences have been explained by variations in lifestyles and occupations in different countries and cultures.
Most risk factors appear to be predominantly environmental in nature, but some studies implicate hereditary factors.
The major environmental risk factor for the development of pterygium is chronic exposure to ultraviolet (UV) light. , Individuals with more than 5 hours of daily exposure to sunlight are more likely to develop pterygium compared to those with under 5 hours of exposure (odds ratio [OR] 1.24). Compared to indoor occupations, outdoor occupations are also associated with pterygium (OR 1.46). The use of sunglasses has been shown to be protective. Furthermore, rural-dwellers have been reported to be more likely to develop pterygium compared to urban residents. This was explained by the differences in lifestyles and occupations associated with sun exposure.
UV radiation absorbed by the cornea and conjunctiva promotes cellular damage and subsequent cellular proliferation. The precise molecular mechanism through which UV radiation causes pterygium to develop is unclear. It is thought that UV radiation causes either direct phototoxic effects (e.g., inflammation, fibrovascular proliferation, DNA damage) or generates reactive oxygen species (ROS), which damage cellular DNA, proteins, and lipids.
The human papilloma virus (HPV) is an oncogenic virus associated with squamoproliferative lesions of the skin, oropharynx, and ano-genital regions. Published literature suggests that HPV can be detected in a subgroup of pterygia and that HPV may be an associated factor in both the pathogenesis and clinical behavior of pterygium. However, HPV as a causative factor for pterygium remains controversial in current medical literature.
Early reports have suggested that other environmental factors such as dust or sand acted in combination with solar light exposure, leading to chronic ocular surface inflammation and subsequent pterygium development. , Surprisingly, smoking has been reported to have a protective effect against the development of pterygium. On the contrary, the risk of pterygium is higher in individuals who consume alcohol. ,
Not all individuals exposed to the same environmental influences develop pterygium. It is therefore likely that external factors only play a role to a certain extent. Underlying genetic predisposition may be an important factor in the pathogenesis of pterygium. Isolated case reports have described clusters of family members with pterygium, and a hospital-based case-control study showed family history to be significant, suggesting a possible autosomal dominant pattern. However, the general lack of consistent familial association suggests that the inheritance of pterygium is multifactorial. More recently, genes associated with important biologic functions, including DNA repair (e.g., p53, RAD50, CYP1A1), cell proliferation, migration, and angiogenesis (e.g., various growth factors, matrix metalloproteinases, S100), have been shown to be associated with pterygium. , This is not surprising, since the disruption of cell remodeling and proliferation is likely to contribute in the multifactorial pathogenesis of pterygium.
The exact pathophysiologic mechanisms underlying pterygium, its progression, and recurrence are still being elucidated. Chronic irritation or inflammation occurring at the limbal area or the peripheral cornea has been suggested by many proponents of the “chronic keratitis” theory, , resulting in focal limbal deficiency and subsequent pterygium development. The presence of a “pterygium angiogenesis factor,” and more recently, high vascular endothelial growth factor (VEGF) levels, have been shown to be associated with pterygia development.
Pterygium has long been considered to be a chronic degenerative condition based on histologic examination. Classically described as an “elastotic degeneration”, pterygium tissue is characterized by abnormal subepithelial tissue containing altered collagen fibers demonstrable with elastic stains. ,
There are, however, behavioral and etiologic features of pterygium that suggest a proliferative growth disorder, not unlike properties seen in benign tumors. As mentioned, pterygium has been linked to dysfunctions in DNA repair mechanisms, cellular proliferation, migration, and angiogenesis. UV light, the major environmental risk factor in pterygium, has also been shown to induce mutations in solar keratosis, Bowen disease, and skin carcinomas. , Moreover, wide excision is advocated, as pterygia have a high propensity for recurring aggressively after surgical excision, and adjunctive treatment with antimetabolites reduces its recurrence. Of note, pterygia is associated with a malignant predisposition, although this occurrence is rare, with the incidence varying between different geographical locations.
The classic signs or clinical hallmarks of limbal deficiency include conjunctival ingrowth, vascularization, chronic inflammation, destruction of basement membrane, and fibrous ingrowth. These signs are clearly present in pterygium and, therefore, many researchers today have suggested that it is a manifestation of localized, interpalpebral limbal stem cell dysfunction or deficiency, perhaps as a consequence of UV light-related stem cell destruction. ,
Pterygium tissues also exhibit intrinsic abnormalities in DNA repair as a result of UV radiation, as seen by a high incidence of microsatellite instability and loss of heterozygosity. Studies have shown that basal epithelial layers of both primary and recurrent pterygia exhibit overexpression of the p53 tumor suppressor gene, an oncogene that acts as a transcription factor to activate or repress the expression of growth-controlling genes and thus explaining the classic histologic findings of epithelial hyperplasia. As p53 has an integral role in the transcriptional regulation of apoptosis-controlling genes, aberrant apoptosis has been found to occur in the epithelium of primary and recurrent pterygium. Studies of pterygia have also shown increased fibroblast activity with deregulated transforming growth factor β (TGF-β) signaling and an overexpression of matrix metalloproteinases. ,
The management of pterygium primarily involves surgical excision and approaches to reduce its recurrence after surgery. Techniques vary from the simplest procedure of bare sclera excision to the more complex and extensive conjunctiva and Tenon excisions coupled with conjunctival autografts. Recurrence is the commonest complication following pterygium surgery. Reported rates of recurrence vary widely from 0% to 89% depending on the techniques used. , Adjuvant therapies have also been used to reduce the rates of pterygium recurrence, although complications arising from some of these adjunctive therapies are increasingly being recognized.
Pterygium surgery can be divided into four main groups, in order of increasing complexity:
Bare sclera excision
Excision with conjunctival primary closure or transposition
Bare sclera excision with antimitotic adjunctive therapies
Ocular surface transplantation techniques, including conjunctival or conjunctival-limbal autografts and amniotic membrane transplantation (AMT).
With low reported recurrence rates and complications, pterygium excision with a conjunctival autograft is currently the gold standard for primary pterygium surgery. In general, the approach to pterygium surgery may differ according to: (1) the extent, size, and number of pterygium (single or double headed), (2) whether it is a primary or recurrent pterygium, and (3) whether there is available conjunctiva for conjunctival autograft. These approaches are discussed below.
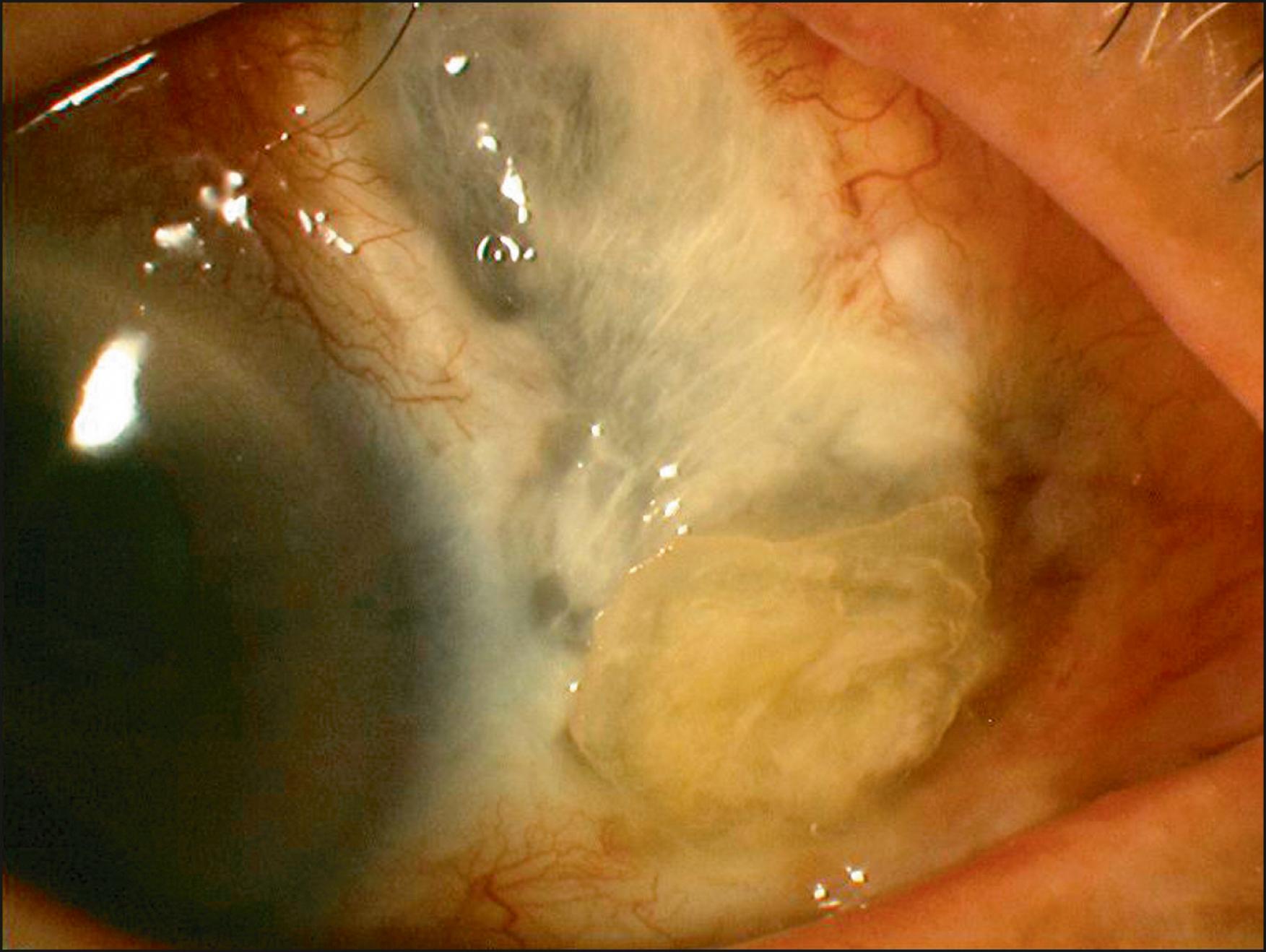
In 1948, D’Ombrain first described the idea of excising the head and body of the pterygium back to the nasal canthal region, and laying the scleral bed bare to reepithelialize. It is now generally recognized that simple bare sclera excision is not adequate as it is associated with a high risk of recurrence. Simple excision should not be performed even for small primary lesions. Recent studies using bare sclera excision as the surgical control for more advanced procedures report unacceptably high recurrence rates ranging from 24% to 89%. , , , , Pterygium recurrence after bare sclera excision may also be highly aggressive, and in severe cases ocular morbidity may far exceed the primary lesion, with the development of symblepharon, ocular motility restriction, and gaze-dependent diplopia. Scleral necrosis and infectious scleritis following bare sclera excisions without adjunctive treatment have also been reported ( Fig. 144.2 ).
As far back as the 1940s, many surgeons have felt that laying the sclera bare after excising a pterygium does not comply with the general principles of wound healing and closure. Hence many procedures describing conjunctival wound closure of the pterygium bed have been reported. Wounds may be closed by simple approximation of undermined conjunctival margins, with or without relaxing incisions, or by conjunctival transposition using a rotational pedicle flap from above or below. However, recurrence rates of rotational flaps do not appear to be significantly lower than the high recurrence rates observed after bare sclera excisions, probably related to the problem of flap retraction. ,
A number of adjunctive therapies have been described to reduce the risk of recurrence after surgical removal of a pterygium. This remains a popular procedure in many countries due to the relative simplicity of the surgical procedure.
Strontium-90 was introduced for the treatment of neoplastic disease in the 1950s. It has been used as an adjunct after pterygium excision to reduce recurrence, with reported recurrence rates around 10%; however, the popularity of its use plummeted in the 1990s as a result of the delayed-onset complications encountered over the areas of application.
The adverse events associated with β irradiation are believed to be dose related and may have a latency period of more than 10 years. Serious complications include scleral necrosis and melts, infectious scleritis with or without ensuing endophthalmitis, cornea perforation, cataract formation, iris atrophy, secondary glaucoma, and calcific scleral plaques. , Milder complications include conjunctivitis, conjunctival scarring, keratitis, ptosis, and evidence of early limbal stem cell dysfunction that may ultimately progress to more severe ocular surface disease. In current clinical practice, β irradiation is now rarely used.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here