Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The lymphoreticular system is composed of various organs and tissues which facilitate the interaction of lymphocytes with cells of monocyte–macrophage lineage in the generation of immune responses. The main organs and tissues of the system are the thymus ( E-Fig. 16.1 ), spleen ( E-Fig. 16.2 ), lymph nodes, bone marrow and mucosa- associated lymphoid tissue (MALT), such as tonsils ( E-Fig. 16.3 ) and Peyer’s patches of the gut ( E-Fig. 16.4 ).
Lymphocytes are produced in the bone marrow. Their number is selectively expanded, mainly in lymph nodes ( E-Fig. 16.5 ), in response to specific immunological requirements. Many lymphoid cells circulate through peripheral tissues via blood and lymphatic vessels in a constant search for antigens.
The monocyte–macrophage system includes tissue macrophages (histiocytes), which are found in virtually every organ. These become activated following tissue damage and, together with monocytes recruited from the blood, act as phagocytic cells in the process of organisation (see Ch. 2 ). These cells also process antigen for presentation to T lymphocytes. Some tissues include a population of specialised dendritic antigen-presenting cells that have a role in initiating specific immune responses.
We will consider disorders affecting some of the main organs of the haematolymphoid system as follows:
Lymph nodes
reactive processes
metastatic tumours
primary tumours (lymphoma).
Bone marrow
abnormalities of haematopoiesis
metastatic tumours
myeloma
leukaemia and lymphoma
myeloproliferative and myelodysplastic processes.
The lymphoreticular system is remarkably labile and quickly responds to the presence of infective agents or foreign material in the activation of an immune response. There are two main patterns of immune response:
The cell-mediated response : This involves the activity of T lymphocytes that are either directly or indirectly cytotoxic.
The humoral response : This involves the activation of B lymphocytes, which differentiate into antibody-secreting plasma cells; interaction of antibody with antigen leads to destruction or inactivation of the antigen.
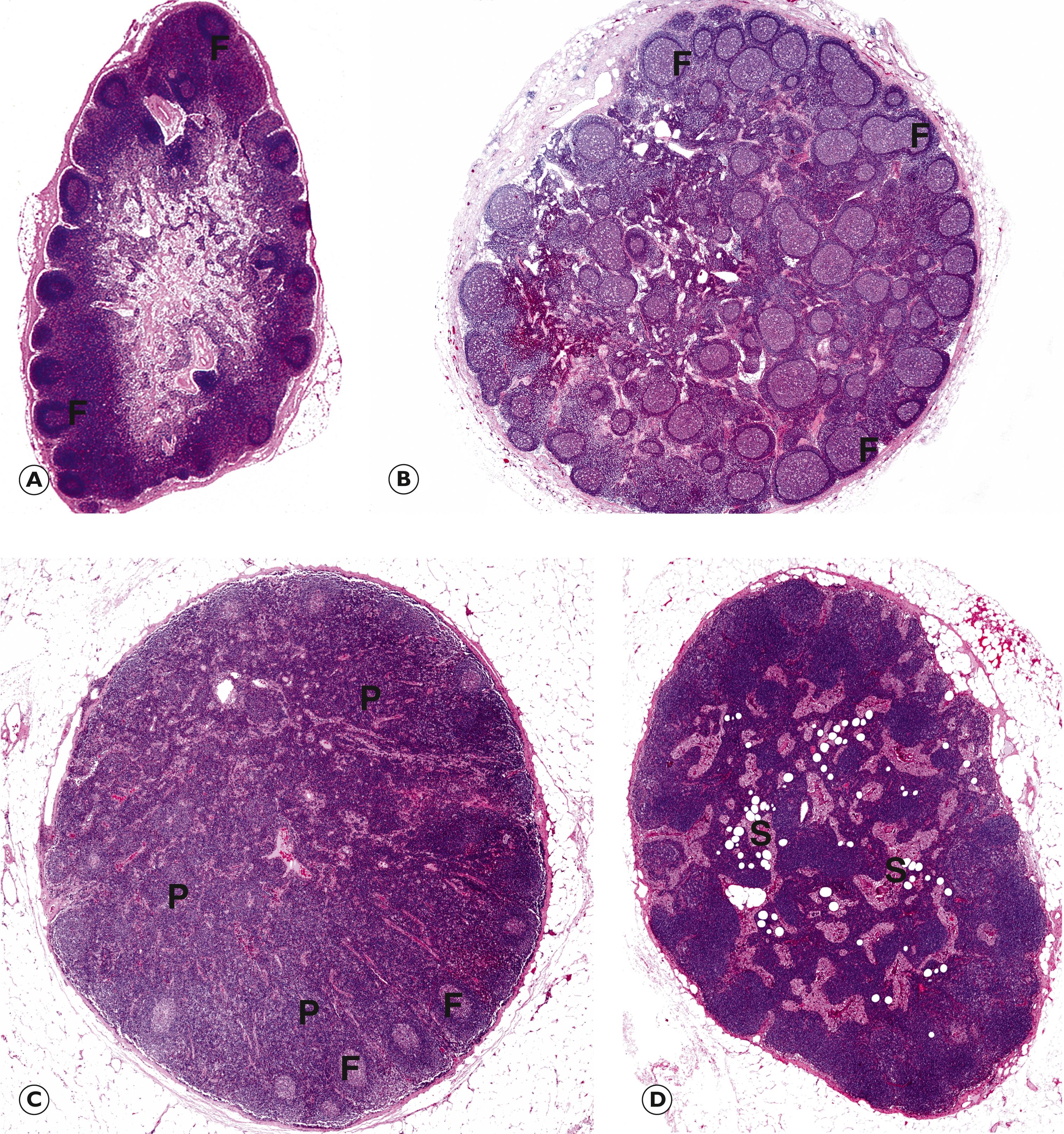
Lymphadenopathy (lymph node enlargement) is a common consequence of infection and other immune reactions. Various different histological appearances can occur. These are determined by the nature of the immune stimulus and the patient’s capacity to mount an immune response. In broad terms, three main patterns of non-specific lymphadenopathy are recognised which reflect reaction and expansion of different constituent areas of the lymph node ( Fig. 16.1 ):
follicular hyperplasia
paracortical hyperplasia
sinus hyperplasia/sinus histiocytosis.
Some infections and other immune stimuli give rise to more specific patterns of lymphadenopathy which can allow the pathologist to suggest a particular aetiology. Examples include the finding of multiple minute granulomas in cases of toxoplasmosis, granulomatous lymphadenitis with caseous necrosis in tuberculosis (see Ch. 5 ) and follicular lysis in HIV infection (see Ch. 5 ).
F follicle GC germinal centre N necrosis P paracortex S medullary sinus SS subcapsular sinus T tumour
This is less common than reactive lymphadenopathy, but is also a frequent finding in clinical practice. Although primary malignant tumours of the lymphoid system are the main focus of this section, it is important to recognise that metastatic spread of other tumours such as carcinoma and malignant melanoma ( E-Fig. 16.6 ) to lymph nodes is a regular occurrence (see Fig. 7.13 and Fig. 16.2 ), and this possibility must therefore always be considered before suggesting a diagnosis of lymphoma.
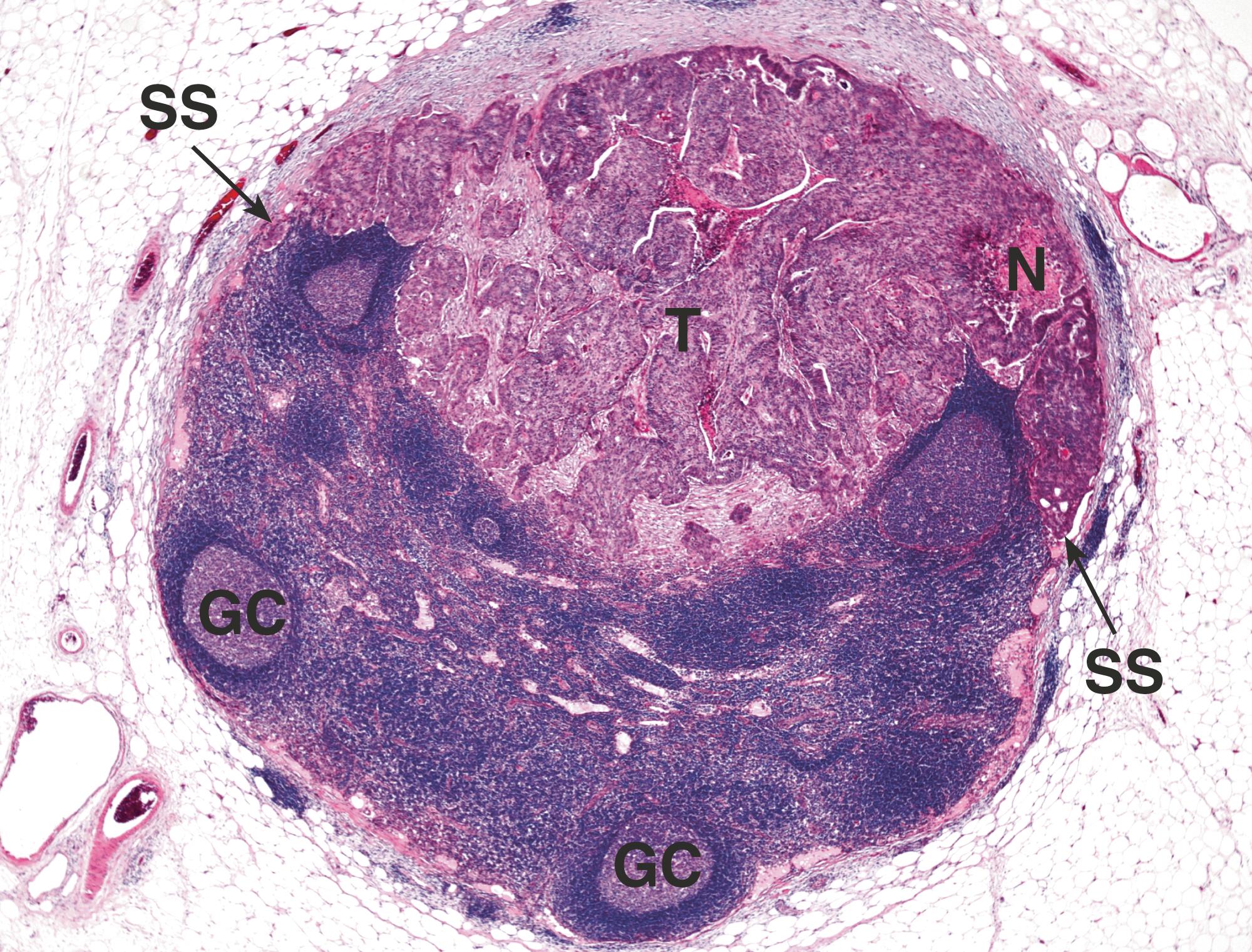
Lymphoma is a primary tumour of lymphoid tissue. This group of disorders is divided into two broad categories on the basis of the cell types present:
Non-Hodgkin lymphoma (NHL) may be derived from B or T lymphocytes.
Hodgkin lymphoma (HL), previously known as Hodgkin’s disease, is characterised by the presence of Reed–Sternberg cells.
Lymphoma usually presents with involvement of a group of lymph nodes and may then spread to involve multiple nodal groups, as well as other haematolymphoid organs such as bone marrow and spleen. NHL may arise in other organs or tissues such as skin, gut, thyroid, brain, salivary gland, lung and testis. Classification of lymphoma is complex and will be considered in the next section.
Other constituent cells of the lymph node may rarely proliferate and so give rise to lymphadenopathy. One example is a group of disorders known as Langerhans’ cell histiocytosis . This condition has previously been known as histiocytosis X and by a range of eponymous designations, reflecting different patterns and severities of disease ( Letterer–Siwe , Hand–Schüller–Christian disease , etc.). It is characterised by proliferation of histiocytic cells, which resemble the Langerhans’ antigen-presenting cells found in normal skin and lymph nodes. There is a spectrum of severity, ranging from benign and localised to disseminated and rapidly fatal disease, which can involve skin, viscera, bone marrow and lymph nodes.
Examination of lymph nodes draining many malignant tumours is a routine part of the pathological assessment of tumour stage (the extent of disease spread, important in planning treatment and in predicting likely prognosis). The surgeon will usually excise not only the main tumour mass for examination, but also any associated groups of regional lymph nodes that drain the site of the primary mass, e.g. axillary lymph nodes in breast carcinoma, hilar and mediastinal nodes in lung cancer.
On occasion, the pathologist may receive a lymph node that contains metastatic tumour from a patient who is not already known to have pre-existing malignant disease. In this setting, examination of the metastatic deposit, often with use of special staining techniques such as immunohistochemistry, may allow the pathologist to suggest a likely site of origin of the tumour and so guide further investigation and management of the patient.
Division of lymphomas into non-Hodgkin and Hodgkin types reflects important differences in both clinical behaviour and tumour morphology.
Non-Hodgkin lymphomas (NHLs ) form a large and diverse group of diseases, all of which are malignant, despite the suffix -oma. The main features common to this group are listed below:
Presentation is usually with painless lymphadenopathy.
Disease may appear to be localised to one nodal group or may be widely disseminated, with generalised lymphadenopathy and involvement of other organs.
Biopsy is usually performed to establish the diagnosis and to allow classification.
Definitive classification involves assessment of cell morphology, immunophenotype and molecular/ cytogenetic features.
A number of forms of NHL show characteristic chromosomal abnormalities that are useful in diagnosis and, in some instances, in assessing prognosis.
Most tumours are derived from a clonal proliferation of B or T lymphocytes and may be broadly divided into low- and high-grade types.
B-cell lymphomas are considerably more common than T-cell types.
B-cell lymphomas may arise at extranodal sites, most commonly within mucosa-associated lymphoid tissue (MALT), and these behave differently from nodal lymphomas.
T-cell lymphomas most frequently occur in skin and may present as a disease termed mycosis fungoides.
Hodgkin lymphoma (HL) is less common ( Fig. 16.13 )
Whilst most cases of NHL are widespread at the time of diagnosis, HL may be localised to a single nodal group.
Spread is predictable, usually by involvement of contiguous nodal groups and with dissemination to liver, spleen and bone marrow only late in disease.
The neoplastic cells (Reed–Sternberg cells and variants) often form only a small proportion of the mass, with a prominent reactive haematolymphoid cell population forming the bulk.
Extranodal involvement is uncommon.
Hodgkin lymphoma (HL) usually spreads by orderly involvement of contiguous nodal groups and staging of this disease is therefore very important in planning patient management. Whilst non-Hodgkin lymphoma generally requires systemic treatment with chemotherapy, the earlier stages of HL can often be treated effectively by localised irradiation of the affected nodal groups.
Various systems have been devised for the classification of haematolymphoid tumours. Classification of tumours is necessary to allow recognition of those that have similar features and facilitates improved prediction of behaviour, prognosis and response to treatment. Most earlier systems were based primarily upon the morphological appearances of the tumour cells, dividing lymphomas on the basis of architecture into diffuse (sheets of cells with no pattern of organisation) versus follicula r (arranged in nodules resembling the follicles of a normal node), on the basis of cell size into small cell and large cell types (broadly reflecting low-grade or high-grade behaviour), or on the basis of similarities to normal constituent cells within the lymph node (e.g. centrocytes, centroblasts, immunoblasts, etc). Although these classification systems were useful in allowing certain subtypes of lymphoma to be described and identified, it is now recognised that morphology alone is imperfect and is insufficient to allow reliable separation of some entities that appear histologically similar but behave differently.
The development of immunohistochemical staining methods and of newer molecular techniques has revolutionised the practice of haematolymphoid pathology. The most recent internationally recognised system of classification is that of the World Health Organization (WHO) . This system uses a combination of clinical, morphological, immunohistochemical and molecular techniques to recognise sub-groups of lymphoma with shared characteristics. The WHO classification includes a very large number of entities, but only those that are common or that illustrate an important pathophysiological concept will be considered here ( Fig. 16.3 ).
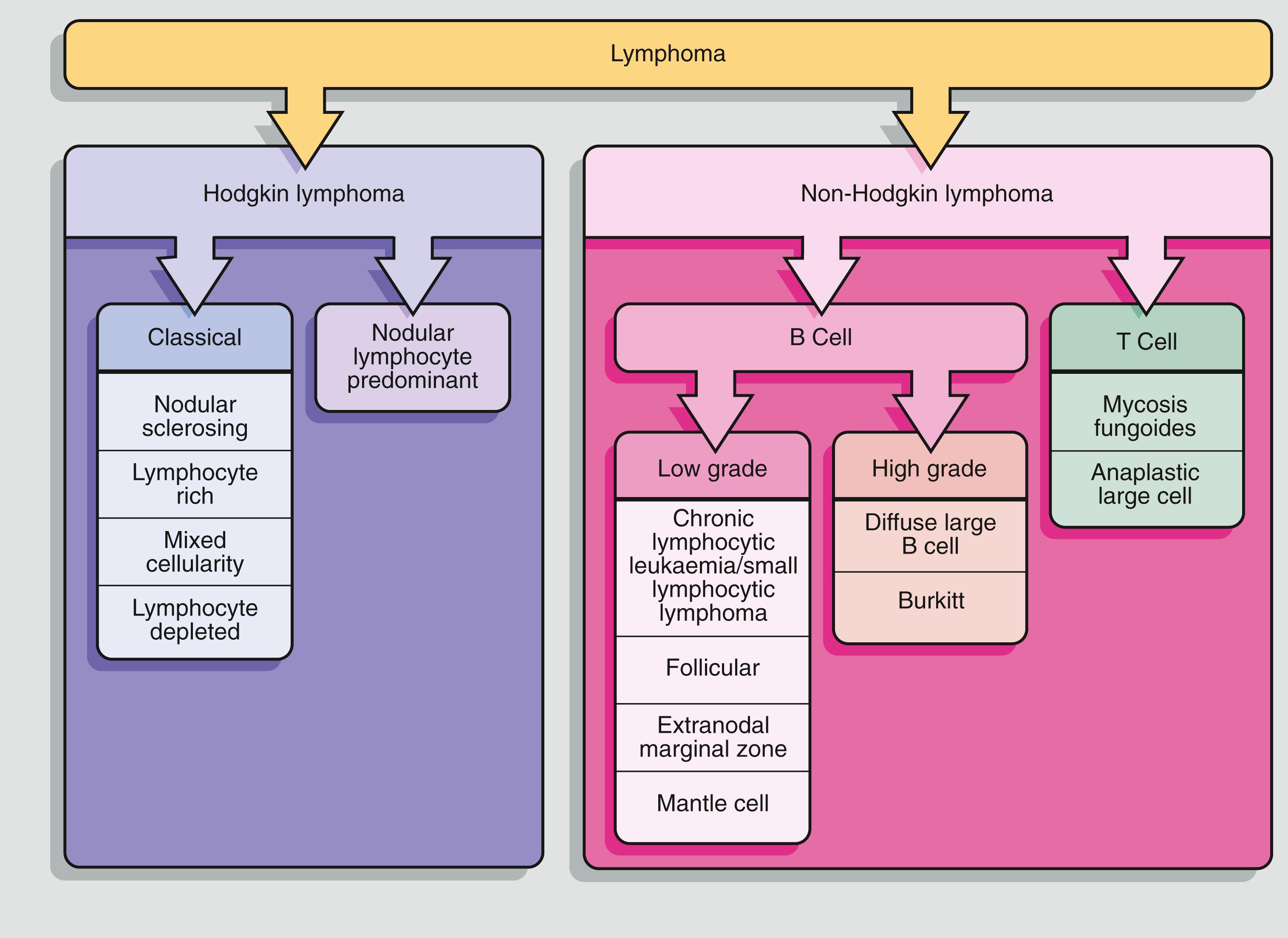
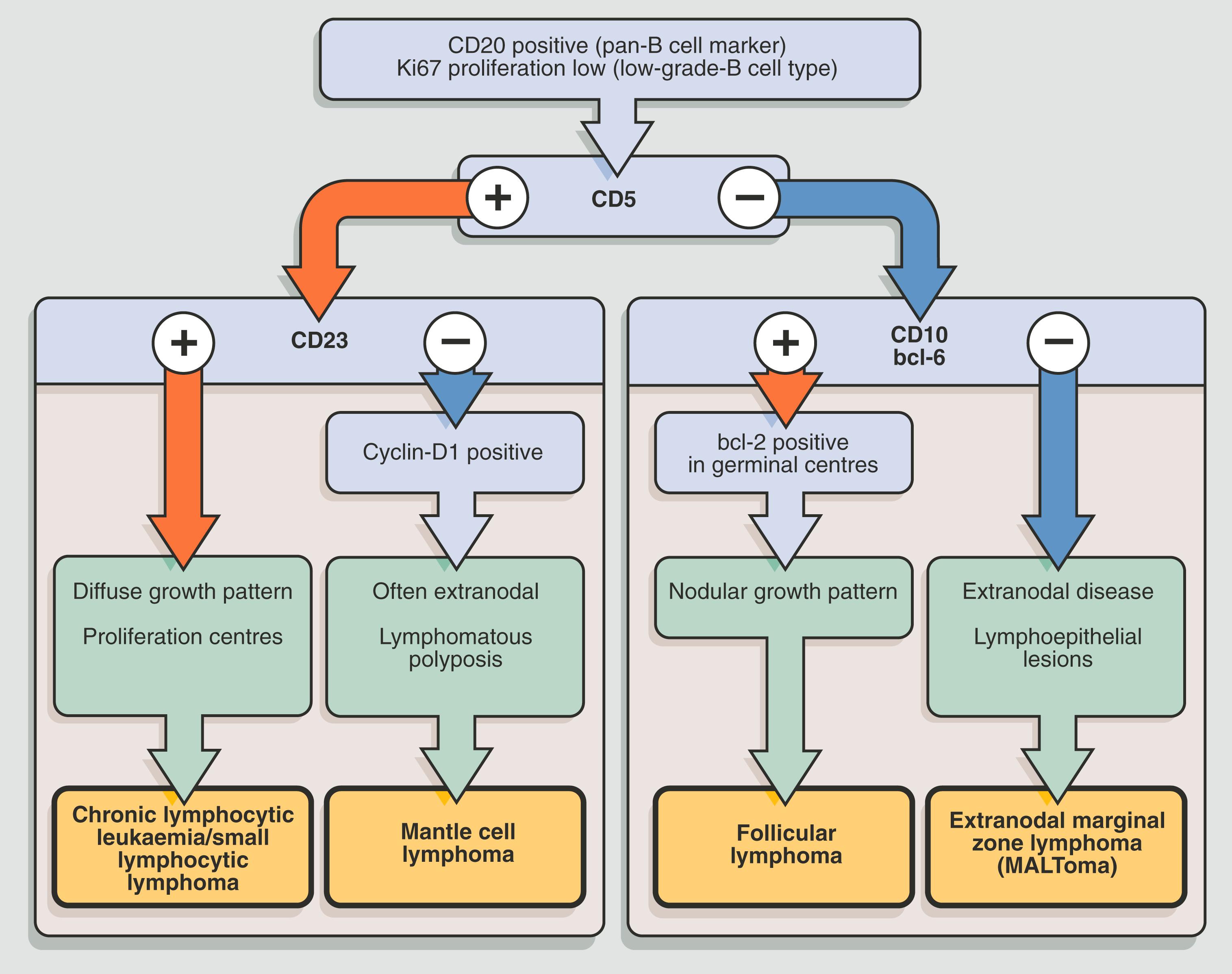
In clinical practice, the diagnostic pathologist first considers the architecture of the involved lymph node and notes the cytological features of the constituent cells, including cell size, nuclear shape, the presence, type and position of nucleoli and the amount and distribution of cytoplasm. This initial assessment guides the selection of appropriate immunohistochemical stains which can identify whether the tumour is of B or T cell lineage, as well as other markers which characterise specific subtypes of lymphoma (see Fig. 16.10 ).
This examination is often sufficient to achieve a diagnosis but may be supplemented, particularly in difficult cases, by molecular investigations . Molecular studies can confirm clonality of the cell population where there is uncertainty about whether a lymphoid cell population is reactive or neoplastic (by showing that all tumour cells have the same immunoglobulin or T cell receptor gene rearrangement) or can be used to look for non-random chromosomal translocations that characterise a particular tumour type, e.g. a t(8;14) translocation is typically present in cases of Burkitt lymphoma.
All of these findings should then be correlated with the clinical and radiological information so that an appropriate plan can be formulated for optimal patient management. From the pathologist’s point of view, this may involve further staging of the tumour by sampling of tissue from other sites, e.g. bone marrow biopsy, or by the assessment of subsequent biopsies for residual disease following treatment. Some newer therapies use specific molecular targeting systems and in some instances, pathological examination to look for immunopositivity within the tumour is needed to predict whether a certain drug will be effective, e.g. rituximab targets the CD20 antigen of B cell tumours and so will lack efficacy in CD20 negative tumours. In addition to their diagnostic value, some markers also provide important prognostic information. For example, high grade B cell NHL may be divided into distinct prognostic and therapeutic groups using a combination of immunohistochemical and molecular methods (see clinical box ‘WHO classification of lymphoid neoplasms’).
The most recent revision of this classification emphasises the importance of combining clinical, histological, immunohistochemical and genetic features to allow reliable recognition and diagnosis of disease. This is essential so that all pathologists use the same, reproducible criteria to diagnose disease and therefore we know that we can make meaningful observations regarding how different groups of tumours behave and respond to treatment. Advances in molecular diagnostic methods have been used to refine and subclassify some tumour groups, aiming to give better prognostic information and guide treatment. An example of this is seen in the revised classification of high grade B cell non-Hodgkin lymphomas, where rearrangements of the MYC, BLC2 and BLC6 genes are now used diagnostically to define aggressive ‘double hit’ and ‘triple hit’ subtypes of the disease.
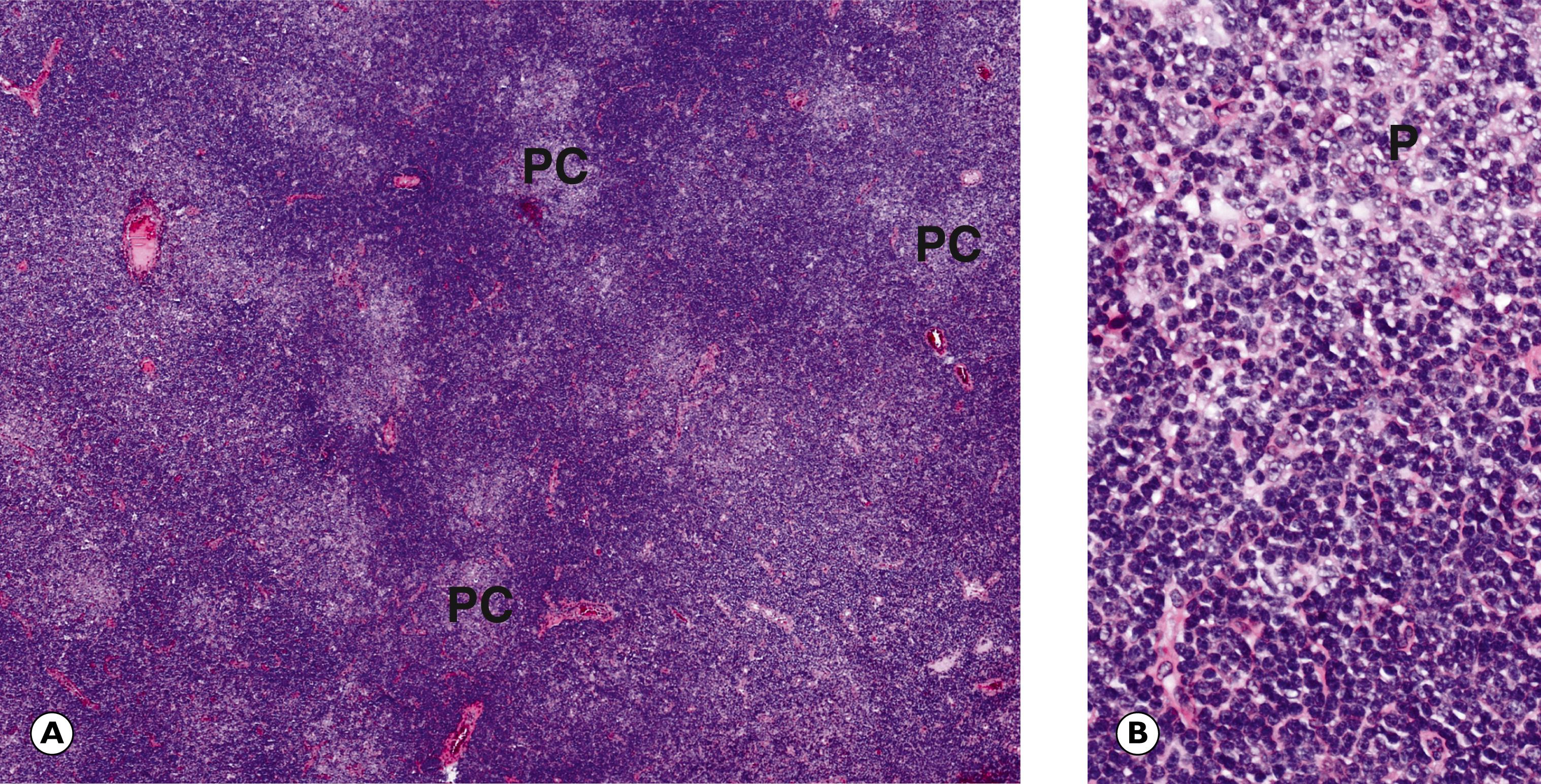
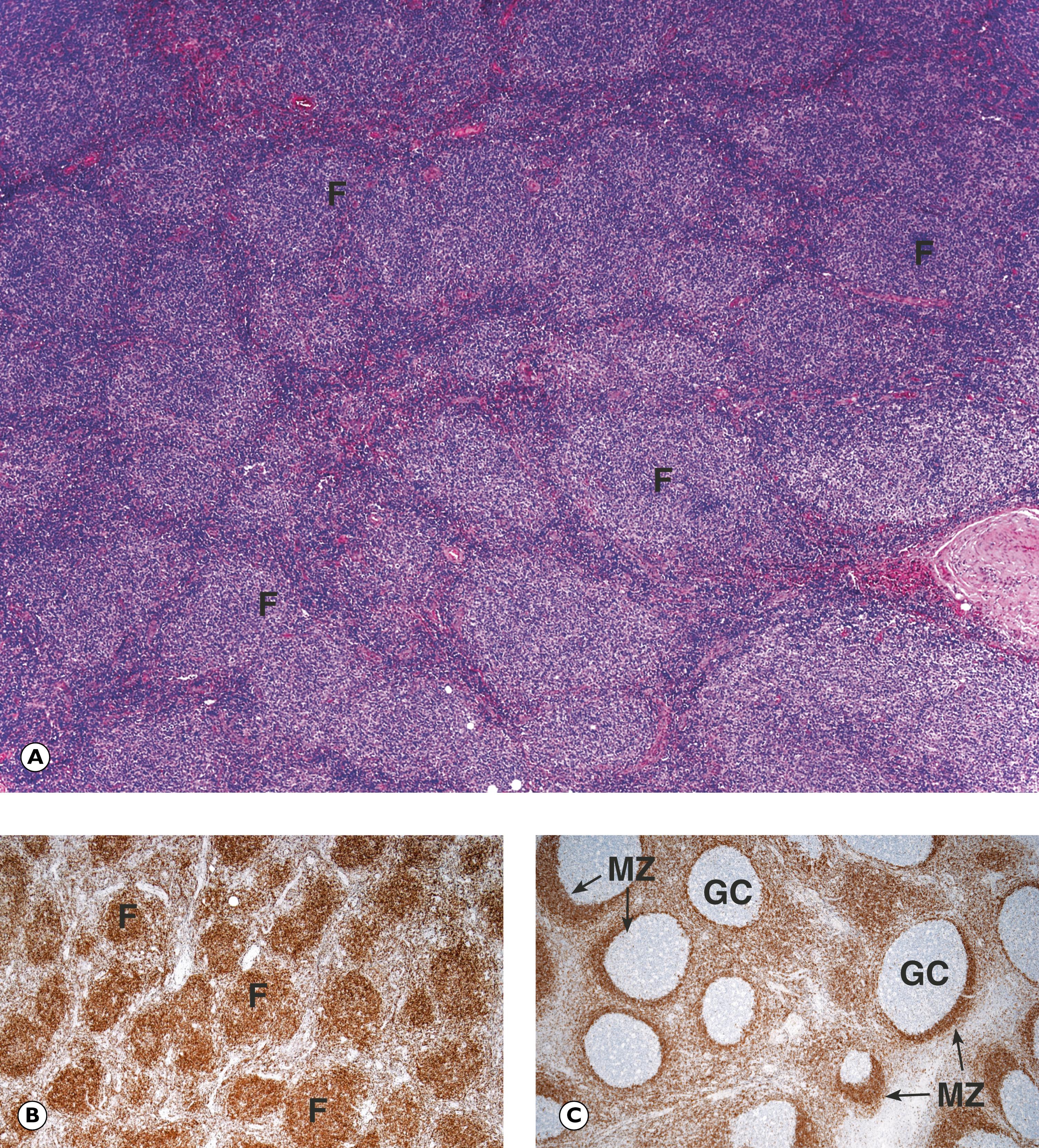
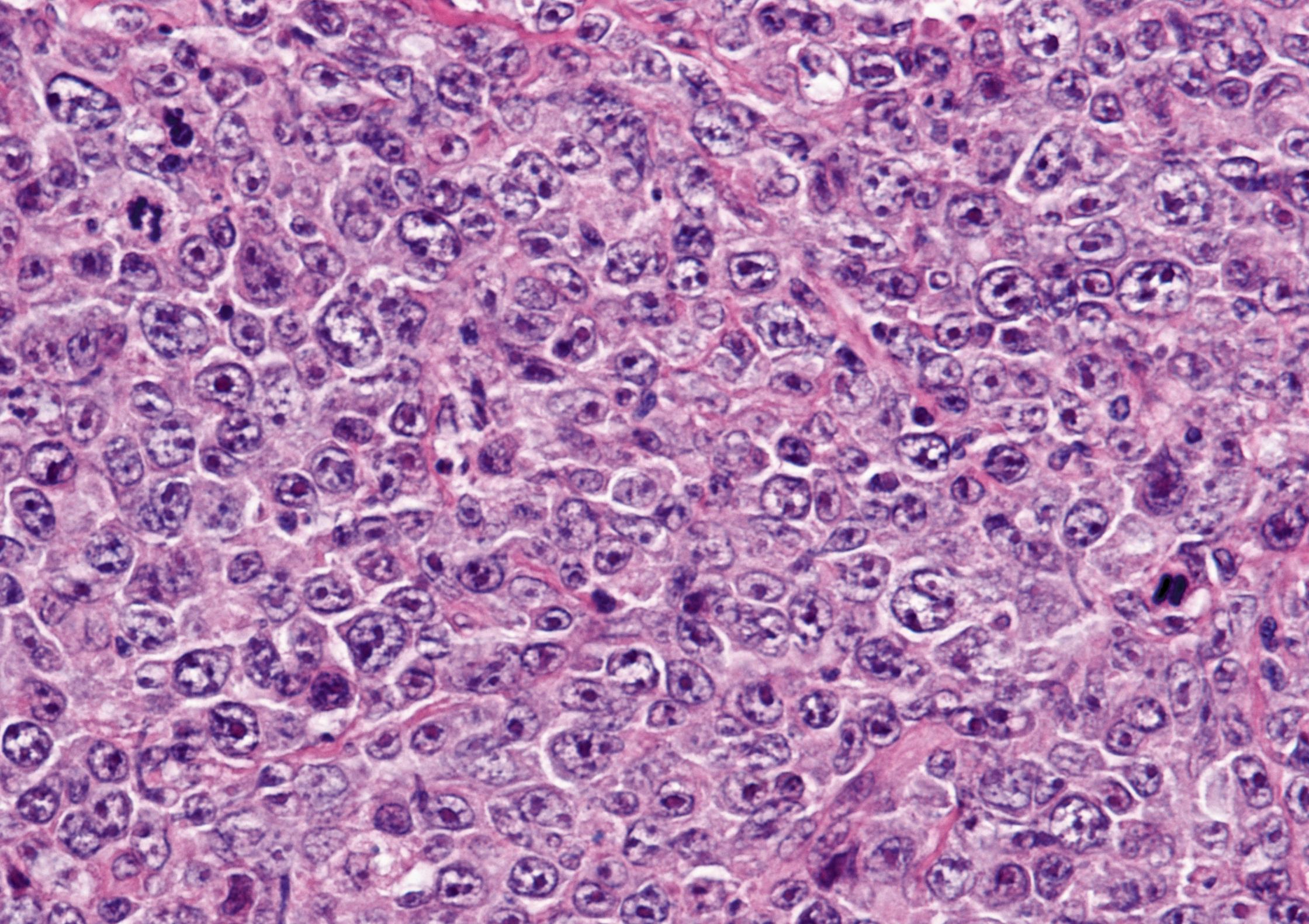
F follicle GC germinal centre M mitotic figure MZ mantle zone P prolymphocytes PC proliferation centre
Various forms of low grade B cell lymphoma can appear very similar on routine histology. Immunohistochemistry allows us to distinguish many of the common subtypes. For example, chronic lymphocytic leukaemia/ small lymphocytic lymphoma cells are typically CD5 and CD23 positive, whilst mantle cell lymphomas are typically CD5 and cyclin D1 positive but are CD23 negative. Immunohistochemical stains are always used as a panel in this setting because individual markers may be misleading or unhelpful if interpreted in isolation. The features must also be correlated with clinical and molecular findings.
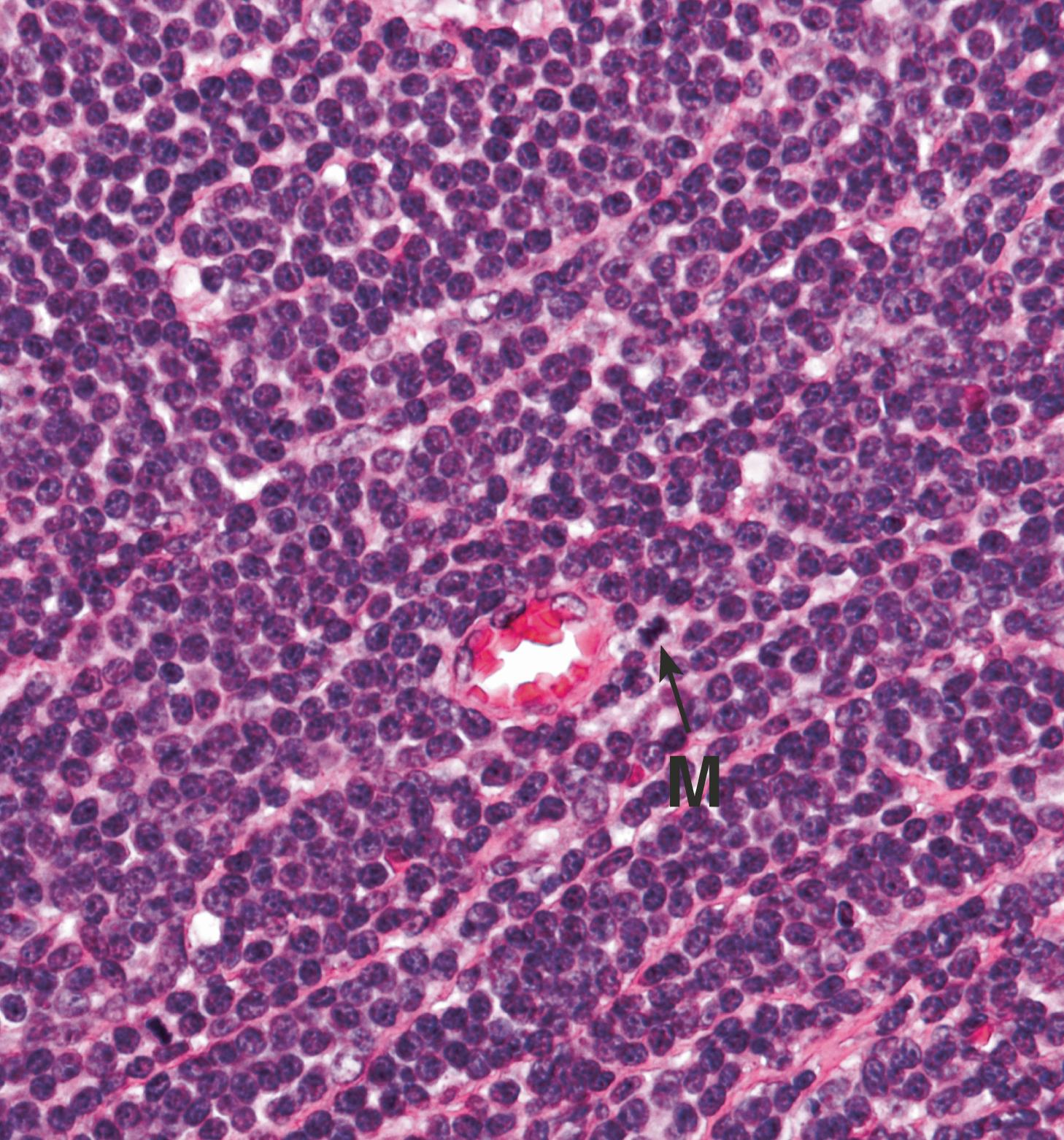
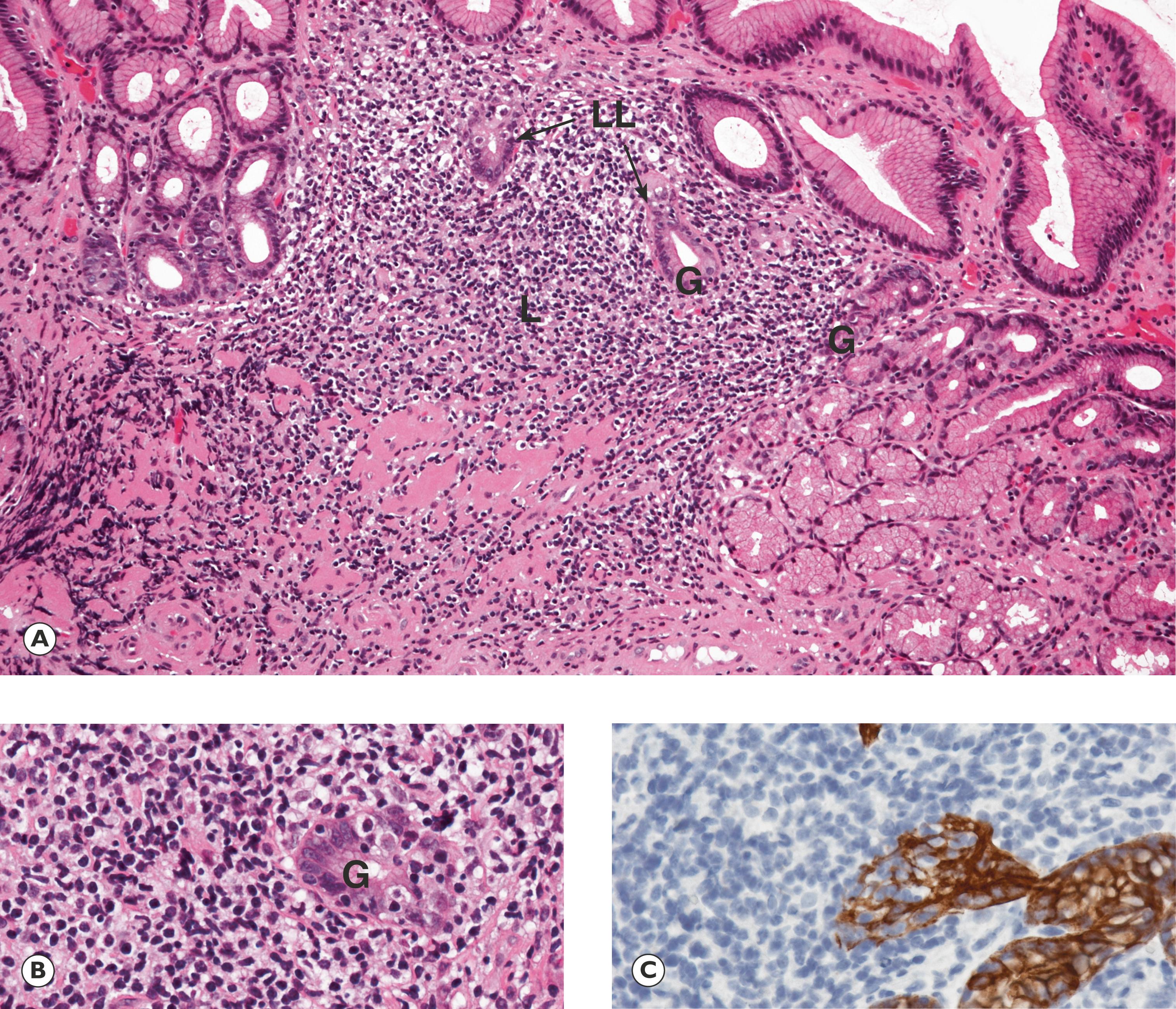
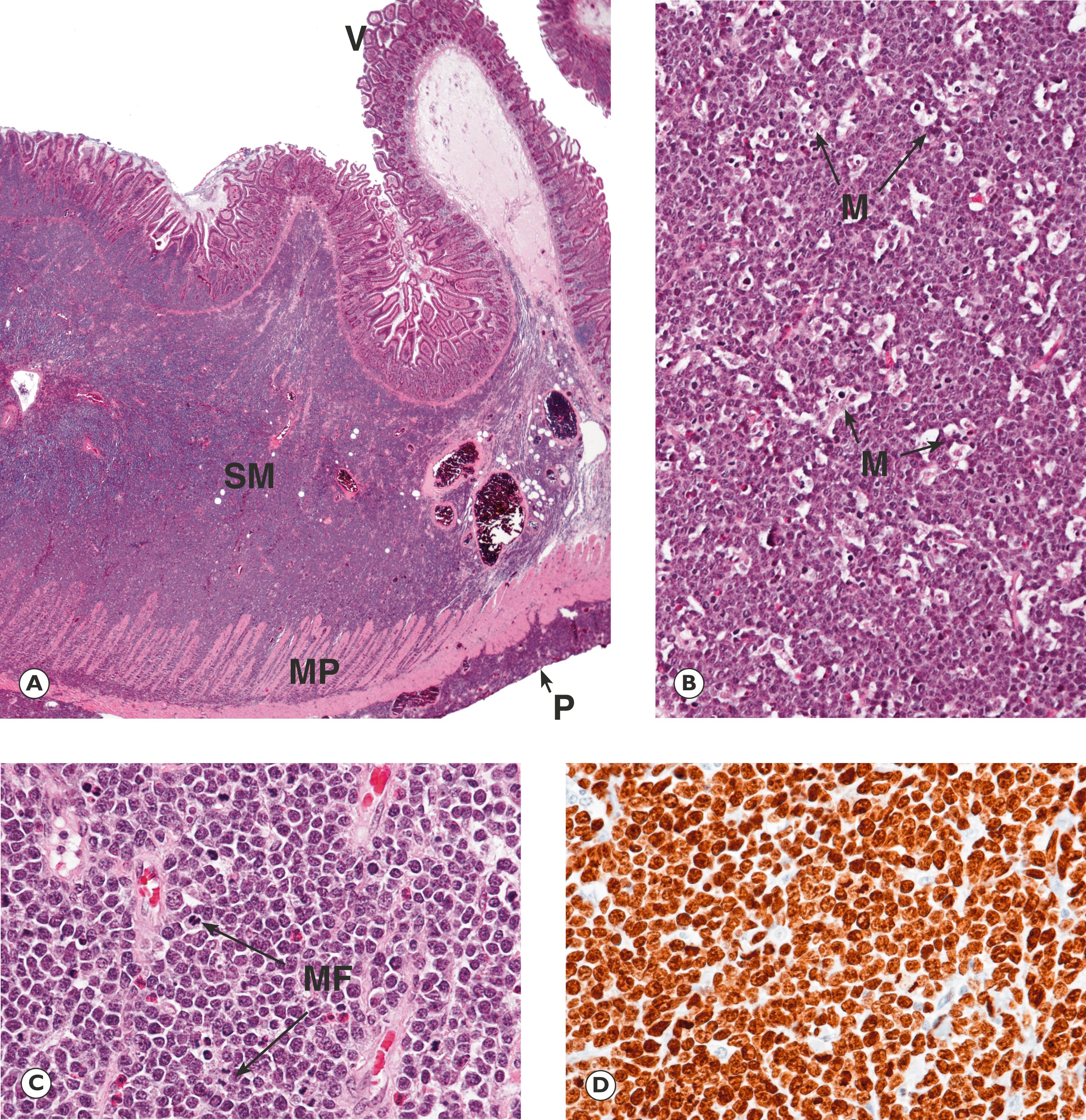
G gastric gland L lymphoid infiltrate LL lymphoepithelial lesion M tingible body macrophage MF mitotic figure MP muscularis propria P peritoneal surface SM submucosa V villi
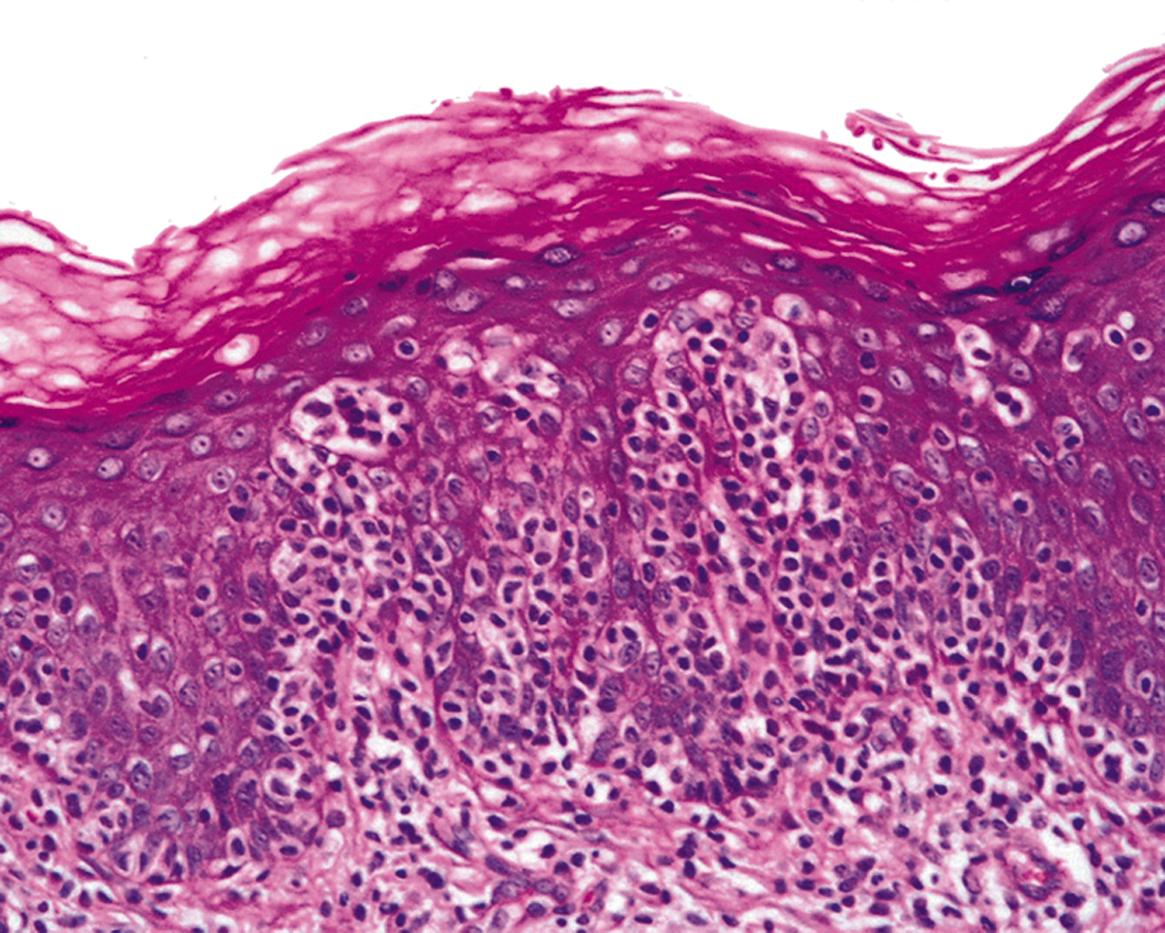
Some other types of T-cell lymphoma are of particular clinical interest. Enteropathy-associated T-cell lymphoma is seen in some patients who present with symptoms of malabsorption . Clinically, there is steatorrhoea (fatty diarrhoea due to failure of absorption of dietary lipid), often associated with anaemia and various vitamin deficiencies. This presentation closely resembles coeliac disease (gluten-sensitive enteropathy, see Fig. 13.15), but patients fail to respond to dietary gluten restriction and it is thought that the disorder known as refractory coeliac disease or refractory sprue is a precursor of overt lymphoma. In its earliest stages, the histological features of this disease also resemble coeliac disease, but the intraepithelial lymphocytes have an abnormal immunophenotype and sensitive molecular techniques can demonstrate clonality of the intraepithelial lymphoid cells. Over time, this may evolve into an aggressive lymphoma that often forms multiple tumour masses in the small intestine. Patients may then present with features of small intestinal obstruction or even with acute peritonitis due to perforation through a necrotic tumour mass.
A later section in this chapter deals with bone marrow disease and with leukaemias, including acute lymphoblastic leukaemia (ALL) . As we have previously noted, leukaemia and lymphoma are not always entirely distinct entities and the same tumour may produce either a solid tissue mass or may diffusely involve bone marrow, giving rise to a leukaemic picture. Most ALL presents as childhood leukaemia and is derived from precursor B lymphoblasts. The T-cell form of ALL is less frequent but usually occurs in adolescent boys, forming a mediastinal lymphomatous mass involving the thymus (the site of normal T-cell maturation). These patients may therefore present with symptoms related to compression of vital structures within the mediastinum such as the superior vena cava
E eosinophil GC tumour giant cell M atypical mitotic figure
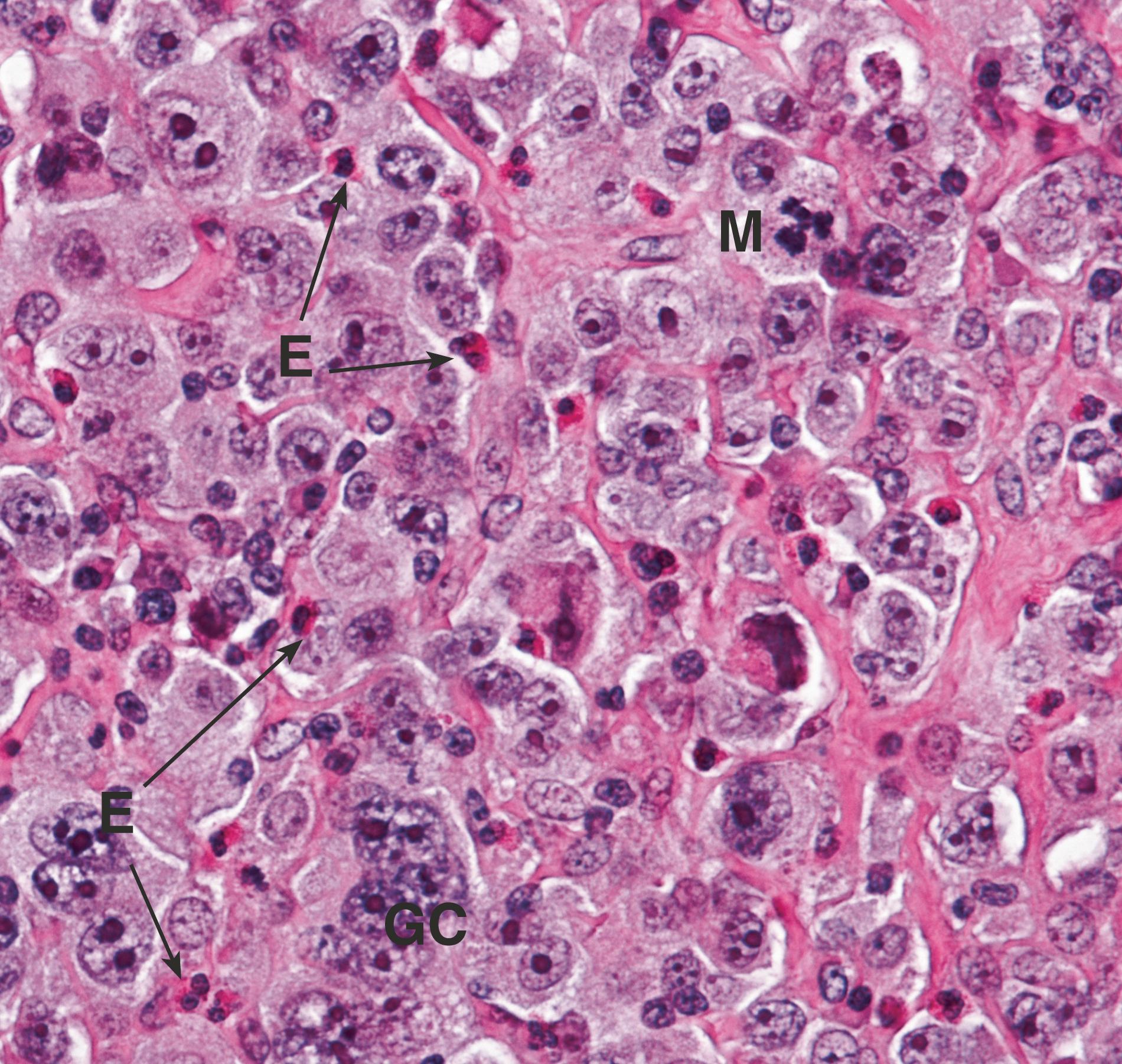
These large cells are essential for diagnosis of Hodgkin lymphoma (HL). The classical form has two nuclei or a single bi-lobed nucleus with two prominent, inclusion-like, eosinophilic nucleoli, usually as large as normal small lymphocytes. This gives rise to the appearance of ‘owl’s eyes’. Mononuclear variants appear similar but have a single nucleus. Lacunar cells are associated with nodular sclerosing HL and have a folded nucleus with surrounding pale, delicate cytoplasm. This cytoplasm is often disrupted during histological processing, leaving the nucleus within a clear space (the lacuna). Lymphohistiocytic variants (L&H cells) are also known as ‘popcorn’ cells as they have abundant cytoplasm and highly convoluted nuclei, resembling popcorn kernels. These are found in nodular lymphocyte predominant HL.
The bone marrow spaces between the trabeculae of spongy bone are occupied by adipose tissue with intervening collections of haematopoietic cells which are responsible for the production of red blood cells, white blood cells and platelets ( E-Fig. 16.8 ). These cell lines are derived from a reservoir of haematopoietic stem cells , which differentiate into the various specialised cell lines in response to specific growth factors. The bone marrow is able to respond rapidly to increased demands for the various blood cells by increased proliferative activity of the precursor and stem cells.
The main diseases involving the bone marrow are:
abnormalities of haematopoiesis
metastatic tumours
myeloma
leukaemia and lymphoma
myeloproliferative and myelodysplastic processes.
Disorders of haematopoiesis are common, but are largely outside the scope of this book. Cytological examination of smear preparations of aspirated bone marrow and histological examination of trephine biopsy specimens of bone marrow form an important method of investigating some diseases.
Most anaemias (lack of red cells) are the result of lack of iron, vitamin B 12 or folate or are due to excessive red cell breakdown (haemolytic anaemia). One type (leukoerythroblastic anaemia) is due to destruction of the haematopoietic tissue by tumour or fibrosis and histological examination of a bone marrow biopsy is vital to establish the diagnosis.
C collagen E eosinophil H histiocyte L lymphocyte N neutrophil PC ‘popcorn’ cells RS Reed–Sternberg cell
The bone marrow is a common site for blood-borne metastasis of certain malignant tumours. The tumours that most commonly spread to bone marrow are carcinomas and the most frequent primary cancers include:
breast carcinoma
lung carcinoma ( Fig. 16.14C and D )

carcinoma of the kidney
carcinoma of the thyroid (particularly the follicular type)
carcinoma of the prostate ( Fig. 16.14A and B ).
In most cases, the growth of metastatic carcinoma in the bone marrow leads to destruction of trabecular bone, leading to osteolytic deposits which appear as lucent areas on x-ray. Carcinoma of the prostate often stimulates the active formation of new woven bone, producing osteosclerotic deposits that appear as increased areas of bone density on X-ray examination. Another tumour which frequently metastasises to bone marrow is neuroblastoma, an aggressive tumour which usually occurs in childhood.
B bone C carcinoma
This group of disorders falls within the WHO category of mature B-cell neoplasms but is considered here because its most frequent presentation is as a disseminated disease, characterised by widespread bone marrow involvement. Myeloma is a neoplastic proliferation of terminally differentiated B cells that have developed the features of plasma cells and so can produce large amounts of immunoglobulin. The features of myeloma are diverse and are a product of various interrelated processes, including displacement of normal haematopoietic elements by the tumour (leading to anaemia, thrombocytopenia and immune dysfunction), bone destruction ( E-Fig. 16.9 ) (causing pain, lytic lesions on X-ray and increased risk of fracture), renal disease and hypercalcaemia, with its associated metabolic consequences. In some patients, the excess immunoglobulin molecules produced may be deposited in the tissues in the form of amyloid , an abnormal fibrillar protein with a ß-pleated sheet configuration (see Ch. 15 and E-Fig. 16.10 ). These insoluble deposits may cause end-organ damage, particularly in the kidney.
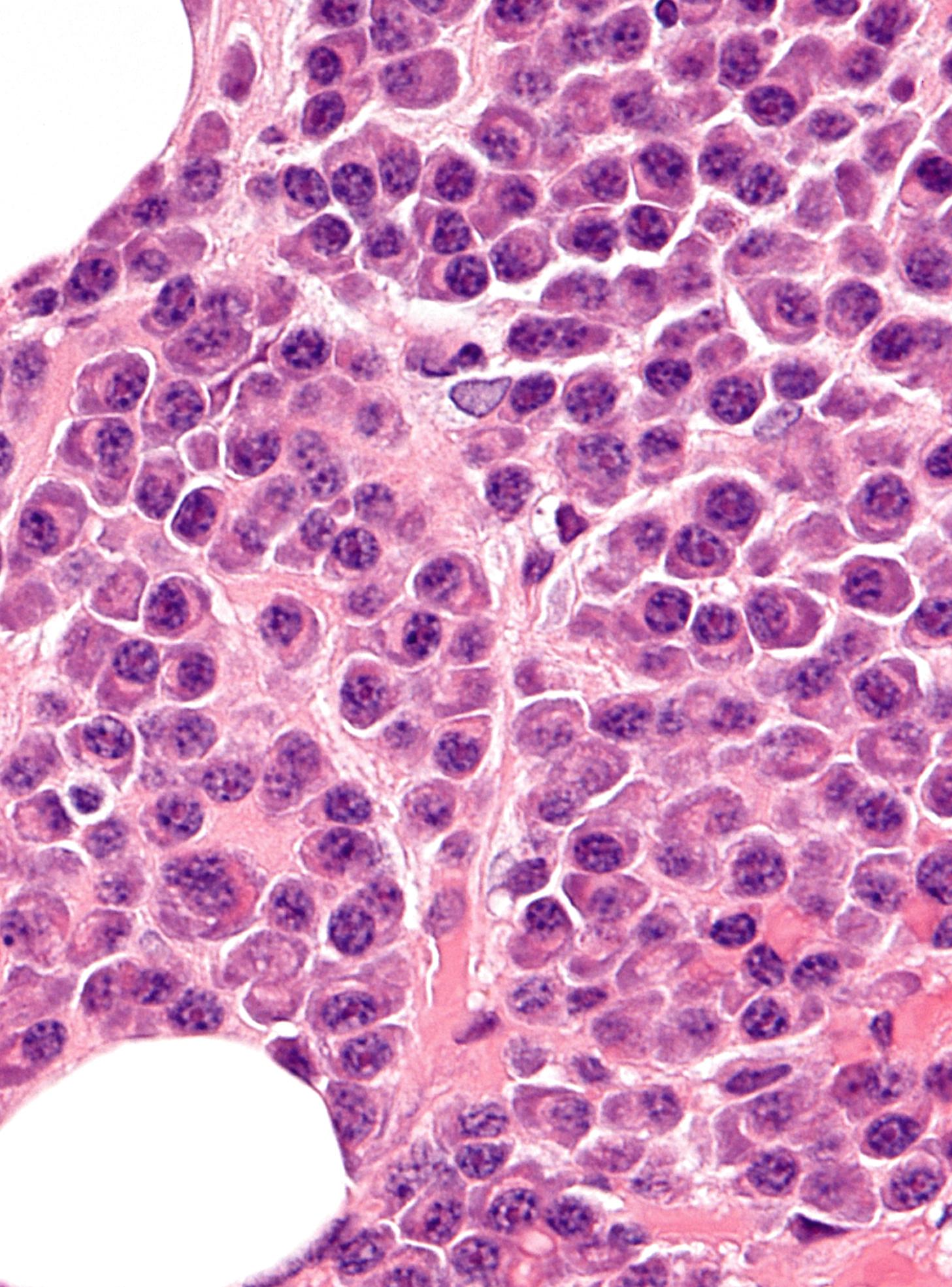
Leukaemias are characterised by neoplastic proliferation of white cells within the bone marrow, which then circulate within the peripheral blood. General characteristics of leukaemias include:
neoplastic proliferation of marrow derived cells, forming one or more neoplastic cell lines
circulation of the neoplastic cells in the peripheral blood
suppression of normal haematopoietic activity by the expanding leukaemic cell population.
Because the neoplastic leukaemic cells circulate in the blood, the cells are usually deposited in other tissues and organs, particularly lymph nodes, liver and spleen. Leukaemias are classified according to the cell of origin, but a useful general division is into two main types: chronic leukaemias and acute leukaemias.
Chronic leukaemias are characterised by neoplastic proliferation of relatively mature white cells. Typically, the clinical course is indolent, with a long natural history. Although the neoplastic cells predominate in the marrow, other haematopoietic cells can survive alongside and adequate numbers of red cells and platelets are produced. Chronic leukaemias are mainly seen in adults over the age of 40 years, but some may eventually transform into a more aggressive acute leukaemia.
Acute leukaemias are characterised by neoplastic proliferation of very immature white cells ( blast cells ). The course of the disease is very rapid, with destruction of all other haematopoietic cell lines in the marrow. Without treatment, acute leukaemias are rapidly fatal.
Chronic lymphocytic leukaemia (CLL) is considered in the previous section owing to its overlap with small lymphocytic lymphoma ( Fig. 16.5 ), but its appearance in the bone marrow is illustrated here ( Fig. 16.16 ).

Chronic myeloid leukaemia (CML) is a neoplastic proliferation of the more mature precursors of neutrophils (for example myelocytes and metamyelocytes) and occurs most commonly in adults.
Acute lymphoblastic leukaemia (ALL) is a tumour of immature lymphocyte precursors (lymphoblasts), and occurs most commonly in children (see also clinical box ‘Clinical features of some T-cell non-Hodgkin lymphomas’).
Acute myeloid leukaemia (AML) is a tumour of immature precursor cells in the myeloid series (mainly myeloblasts) and is most common in adults ( Fig. 16.17 ). It is the most common of a group of acute leukaemias derived from various myeloid precursors, collectively called acute non- lymphoblastic leukaemias.

Some lymphomas that originate in lymph nodes may also eventually spread to involve bone marrow and there may be enough circulating lymphoma cells in the blood to be detected in a blood film, highlighting again that there are areas of overlap between leukaemia and lymphoma.
B bone
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here