Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Listeria monocytogenes is a serious and sometimes lethal pathogen, especially for pregnant women and their fetuses, the newborn, and for older adults and immunocompromised individuals.
The pathogen was first described in 1926 by Murray and colleagues during an investigation of an epizootic outbreak of infection in laboratory rabbit and guinea pigs. Human disease caused by L. monocytogenes was identified in 1929 by Nyfeldt. The present name is derived from the fact that monocytosis develops in less than 24 hours after intravenous infection in experimental animals. Although there is some controversy over the name “Listeria,” it is commonly believed that it was named in honor of British surgeon Joseph Lister. Although Burn already recognized L. monocytogenes as a serious pathogen for human newborns in 1936, the first confirmed transmission from animals to humans was only documented in 1983. A recent upsurge in outbreaks in the human population has refocused attention on this organism.
Listeria organisms are regular, short, facultatively anaerobic, non–spore-forming, gram-positive rods that are motile and form bluish gray colonies on nutrient agar. They are non–acid fast, catalase-positive but do not branch or form capsules. Of six Listeria spp. ( L. monocytogenes, Listeria innocua, Listeria grayi, Listeria welshimeri, Listeria seeligeri, and Listeria ivanovii ), L. monocytogenes is the most important human pathogen ; the few case reports regarding other species causing infection typically involve immunocompromised hosts. Peritrichous flagella enable most Listeria species to exhibit tumbling motility at 25° C to 30° C; this motility helps clinical laboratories distinguish Listeria from morphologically similar microbes, such as Corynebacterium and Erysipelothrix . Inoculated semisolid motility media reveal a distinctive “umbrella” or “swarming” pattern at room temperature. Expression of flagellin (and thus motility) in most clinical isolates is turned off at 37° C. On sheep blood agar, L. monocytogenes exhibits a narrow zone of β-hemolysis (nonhemolytic strains exist) and a rectangular area of increased hemolysis when streaked on blood agar in proximity to Staphylococcus aureus (the Christie, Atkins, Munch-Peterson [CAMP] test). Listeria tolerate low temperatures, high salt concentrations, and high (alkaline) pH, which allows replication in soil, water, sewage, manure, animal feed, and, most importantly, refrigerated foods. The entire genome of L. monocytogenes has been sequenced.
Historically, L. monocytogenes strain typing has been based on somatic (O) and flagellar (H) antigens. This system discriminates 13 serotypes, many of which represent genetically diverse groups of strains. Only four serotypes (1/2a, 1/2b, 1/2c, and 4b) cause most human listeriosis. Multilocus sequence typing (MLST) is emerging as a genotyping tool, with both the power to discriminate different lineages and the ability to provide a standardized global approach to defining clones and clonal complexes. Epidemic clones (ECs) of L. monocytogenes are typically defined as isolates of a presumably common ancestor that are genetically related and involved in temporally and geographically unrelated outbreaks. Recent application of MLST to 300 isolates from five continents revealed a partitioning of genotypic diversity between clinical isolates, on one hand, and food or environmental isolates, on the other, while simultaneously identifying the existence of a few prevalent and globally distributed clones.
The United States Department of Agriculture projects approximately 2500 cases of serious Listeria infection per year in the United States, with a mortality of approximately 20% (compared with 13,000 cases of salmonellosis [0.4% mortality], Campylobacter [0.1% mortality], and Yersinia [0.01% mortality]). In the United States alone, illness caused by L. monocytogenes costs $2.6 billion annually and leads to loss of 9400 quality-adjusted life-years. Most infections are sporadic and have no known epidemiologic explanation, but Listeria is the leading cause of death resulting from foodborne infection in the United States as well as several European countries. Although promotion of preventative measures, as well as monitoring and voluntary recall of contaminated food products, has resulted in a reduction in the prevalence of listeriosis over recent decades, risk for foodborne infection remains high, and the incidence in older adults has in fact increased again.
Listeria species are ubiquitous in nature, with their natural habitat in decaying plant matter. Spoiled silage appears to be a source of infection for ruminant animals. Although direct transmission of L. monocytogenes to humans from infected animals has been described, most human infections are acquired through ingestion of contaminated food. L. monocytogenes isolates from clinical and food specimens are highly heterogenous; this suggests that infection does not simply reflect a few highly virulent subtypes of Listeria.
Asymptomatic fecal carriage in healthy humans after ingestion of contaminated foods in nonoutbreak settings has been reported, with a prevalence of 2% to 10%, but is higher during outbreaks ; carriage may be as high as 26% in household contacts of patients with listeriosis. Dissemination of L. monocytogenes from colonized humans can also occasionally play a role in transmission. Maternal colonization does not invariably lead to invasive illness of the fetus or newborn, but transmission of L. monocytogenes from mother to fetus is believed to occur in utero via infected amniotic fluid or the maternal circulation.
Although most cases of listeriosis occur in the community, nosocomial listeriosis in both neonates and adults has been described. Person-to-person transmission caused by suboptimal infection control is likely to be responsible for most of these small clusters. Among clusters reported in newborns, the index case may manifest as early-onset infection, and subsequent cases appear as late-onset listeriosis. Multiple cases of early-onset disease in the same hospital unit would suggest the possibility of foodborne or another common source.
Community-acquired listeriosis may occur spontaneously in adult subjects with no known underlying predisposing conditions but only at a relatively low frequency, ranging from 0.1 to 11 per 1,000,000 persons. In high-risk populations, such as the newborn, the elderly, and adults with immunocompromised function, incidence of infection and associated morbidity and mortality is high ( Box 13-1 ). The list of specific immune defects associated with an increased risk for severe Listeria infection is long and includes defects in gastrointestinal defense, cell-autonomous immunity, and innate as well as adaptive systemic immunity. Among patients with human immunodeficiency virus (HIV) infection in the era before antiretroviral therapy, a 400- to 1000-fold increased risk of suffering systemic invasive listeriosis, compared with HIV-negative subjects, has been documented. The clinical use of anti–tumor necrosis factor (TNF) or TNF receptor antibodies has more recently been identified as a risk factor for severe infection with L. monocytogenes. Alcoholism, diabetes, and cirrhosis also increase risk for infection.
The risk of listeriosis is about 20 times higher among pregnant women than among nonpregnant healthy adults.
About one third of listeriosis cases are diagnosed in pregnant women.
Pregnant women with listeriosis are at increased risk of spontaneous abortion, preterm delivery, or stillbirth.
Newborns are at greater risk of developing severe infection than pregnant women.
Newborns may present clinically with
Early-onset listeriosis (transmitted via placenta and usually diagnosed as sepsis in the first day of life).
Late-onset infection (≥7 days of life).
About 50% of all cases occur at age 60 years or older.
Immunocompromised patients.
People with cancer, diabetes, or kidney disease.
People with human immunodeficiency virus/acquired immunodeficiency syndrome: Listeriosis is up to 300 times more likely to occur in this group than in healthy adults.
Patients receiving immunosuppressive drug therapy (e.g., high-dose glucocorticosteroid, tumor necrosis factor inhibitor).
Transplant patients receiving antirejection drug therapy.
Listeriosis is 18 times more common in pregnant (12/100,000) than nonpregnant women (0.7/100,000), and overall 16% to 27% of all infections with L. monocytogenes occur in pregnancy. This occurs despite no differences in carriage rates of fecal or vaginal specimens between pregnant and nonpregnant individuals. The underlying factors leading to an increase in susceptibility to listeriosis during pregnancy remain unknown.
The most common and thus relevant predisposition to severe listeriosis, however, is age, in that the very young and very old are at highest risk. Perinatal and newborn infections represent 30% to 40% of the total caseload in humans. Furthermore, symptomatic listeriosis has an exceptionally high mortality rate in the unborn, the newborn, and the elderly, ranging from 20% to greater than 60%. The annual incidence of reported perinatal and neonatal listeriosis in nonoutbreak settings ranges between 2 to 13 per 100,000 live births ; during outbreaks, incidence can be as high 1% to 2% of all deliveries. However, the true incidence is probably much higher because abortion and stillbirth caused by Listeria go largely unrecognized because bacterial cultures are not obtained routinely in these cases. The reasons for this increased susceptibility at the extreme ends of the age spectrum have only partially been delineated.
As for most infectious pathogens, the risk of developing clinical disease depends on intensity of the exposure, virulence of the organism, and the ability of the host to defend against infection.
Infection with L. monocytogenes begins with the ingestion of the bacteria. Bacterial load, measured as a function of the inoculum of L. monocytogenes, has been mathematically linked to increased risk for infection and confirmed by finding of a significantly elevated concentration of Listeria in contaminated foods during outbreaks. All of this suggests that dose likely matters. The infectious dose is estimated to be 10 4 to 10 6 organisms (colony forming units [CFU])/g of ingested food, but may be as low as 10 CFU/g in immunocompromised hosts and patients who have diminished gastric acidity or have an injured intestinal lining.
Clinical risk for invasive listeriosis is influenced by the pathogenic potential of the infecting strain. For example, among the many L. monocytogenes strains, serovars 1/2a, 1/2b, and 4b are responsible for 95% of human infections, and most outbreaks are caused by strains of serovar 4b. In tracing the journey of the organism from ingestion through to successful replication in the host, an array of virulence factors comes into play. Survival of Listeria in the human gastrointestinal tract has been tightly linked to bacterial sigma factor σ B . σ B drives expression of enzymes that permit bacterial survival in the acidic upper gastrointestinal tract ; the alkaline, bilious environment of the proximal small intestine, as well as the high salt concentrations throughout the gastrointestinal tract. The transit across the mucosal barrier begins with adhesion of L. monocytogenes to the mucosal lining via the Ami surface protein. After adhesion, L. monocytogenes enters intestinal target cells either through phagocytosis (for M cells) or through actions of additional listerial surface proteins (for intestinal epithelial cells) called internalins. Among the 25 internalin-like proteins, InlA and InlB are the most important. InlA binds to the host adhesion protein E-cadherin. E-cadherin is transiently exposed to the luminal surface of the intestinal villi during remodeling of epithelial junctions and goblet cells (GCs), thereby offering an entry point to L. monocytogenes in the gut. In addition to its established role in crossing of the intestinal barrier, InlA also appears to be involved in the crossing of the maternofetal placental barrier by binding to E-cadherin–expressing syncytiotrophoblasts. Listeria rapidly transcytoses across the intestinal epithelium and is released in the lamina propria by exocytosis, from where it disseminates systemically without histologic evidence of significant inflammation in the bowel wall.
Infection of the liver is facilitated by the bacterial adhesin FbpA, which binds to human fibronectin expressed on the surface hepatocytes. Listerial internalin B (InlB) then engages the host hepatocyte growth factor receptor (a tyrosine kinase receptor, Met) and complement component C1q receptor, which mediates hepatocyte entry via endocytosis. In addition to intestinal enterocytes and hepatocytes, Listeria can also infect, grow, and spread in fibroblasts, epithelial cells, vascular endothelial cells, and renal tubular epithelial cells. However, although invasion efficiency varies with host cell type, it never reaches the entry rate observed in phagocytes. M cells, monocytes, macrophages, and dendritic cells are infected in all organs and are the main carriers of L. monocytogenes during its systemic spread. Entry of L. monocytogenes into these cells occurs via phagocytosis, initiated after lipoteichoic acid, a component of the listerial cell wall, is bound by type I/II scavenger receptors and/or complement receptors.
During the early course of infection, L. monocytogenes resides within a vacuole (for nonphagocytic cells) or a phagosome (in monocyte/macrophage-derived cells) ( Fig. 13-1 ). The escape of L. monocytogenes from the single-layer membrane vacuoles/phagosomes is assisted by two virulence-associated bacterial molecules: listeriolysin O (LLO) and phosphatidylinositol–phospholipase C (PlcA). LLO is encoded in the hly gene cluster, and in mice, but not humans, is essential for bacterial escape from vacuoles. LLO is a secreted cholesterol‐dependent cytolysin (CDC) toxin that binds the phagosomal membrane as cholesterol-associated monomers that oligomerize into large complexes and then penetrate the membrane, forming a pore. The pores also lead to increased pH and decreased calcium ion (Ca 2+ ) concentration within the phagosomal compartment, inhibiting phagosome maturation. Because the activity of LLO is optimal at the acidic pH of the phagosome, it is less active at the neutral pH of the cytoplasm, preventing excessive cell damage. As with other CDCs, LLO is a potent signaling protein that can activate important pathways, such as nuclear factor kappa B, mitogen-activated protein kinase, and protein kinase C. In animal models, LLO is required in vivo for bacterial growth in placental trophoblastic cells and for subsequent fetal invasion.
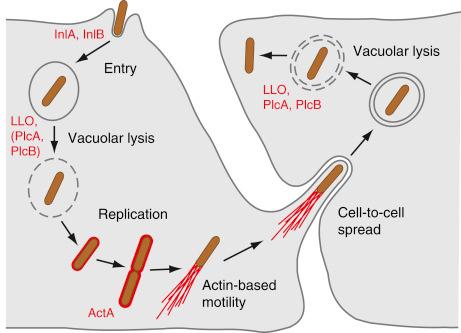
The C-type phospholipases, phosphatidylinositol-specific (PI-PLC) and a broad-range phosphatidylcholine (PC-PLC), mediate L. monocytogenes escape from the vacuole/phagosome by digesting the phagosomal membrane. Although the two phospholipases act synergistically with LLO to lyse primary and secondary vacuoles, allowing L. monocytogenes to escape into the cytoplasm, The C-type phospholipase PlcB promotes lysis of the human primary vacuole even in the absence of LLO and directly inhibits downstream signaling. L. monocytogenes also requires host factors to assist in bacterial escape from the phagosome. Although Listeria activates host antimicrobial nitric oxide (NO), normally important in the degradation of phagocytosed bacteria, in the case of L. monocytogenes infection, NO instead leads to downregulation of the vacuolar-type H + –adenosine triphosphatase (ATP)ase (V-ATPase), which in turn inhibits maturation of the secondary phagolysosome and aids bacterial escape into the cytoplasm.
Once free in the cytoplasm, L. monocytogenes co-opts host-provided nutrients necessary for its intracellular multiplication. In the cytoplasm L. monocytogenes also induces polymerization of host actin filaments to move within the cytoplasm and to spread from cell to cell. The bacterial surface protein ActA is the only bacterial determinant necessary for host actin-based motility of L. monocytogenes. It binds and activates the host Arp2/3 complex, inducing actin polymerization. ActA is primarily involved in driving Listeria toward the cytoplasmic membrane, where the bacterium is then enveloped in filopodium-like structures (also termed a listeriopod) that are engulfed by adjacent cells, leading to the formation of secondary double-layer membrane vacuoles. In human cells, PlcB-mediated lysis of the secondary (now double-layer membrane) vacuoles initiates a new cycle of infection. L. monocytogenes does not always have to escape the phagosome but can replicate inside modified vacuoles called SLAPs (spacious Listeria -containing phagosomes) ; however, the rate of bacterial replication inside SLAPs is reduced (>8 hours) compared with bacterial replication in the cytosol (<30 minutes).
Invasion of the placenta is a crucial element in the pathogenesis of congenital listeriosis. E-cadherin is expressed on villous cytotrophoblasts and in localized areas of the basal plasma membrane of syncytiotrophoblasts. Syncytiotrophoblasts are directly exposed to maternal blood in the intervillous space and are considered a form of specialized endothelium. InlA is required for L. monocytogenes to cross the human maternofetal barrier, which in part explains the specific tropism of the organism for the placenta. InlB facilitates the transfer of L. monocytogenes across placenta, a process that also involves ActA and the host mannose receptor C type 1.
The mechanisms by which Listeria accesses the central nervous system (CNS) remain poorly understood, but two major routes of infection have been proposed. A retrograde neural route is likely to occur in ruminants upon crossing of the oral epithelium, and this probably accounts for the observation that Listeria induces almost exclusively rhombencephalitis in these animals. In contrast, the hematogenous route is likely the more frequent in humans, in whom bacteria circulating in the blood, either free or associated with phagocytes, are thought to breach the blood-brain barrier. Only In1B and In1A have been clearly implicated in facilitating the transfer of L. monocytogenes across the blood–brain barrier.
The genes that encode the most prominent virulence-associated proteins—LLO, ActA, PlcA, and PlcB—are situated adjacent to each other in a 9.6 kb virulence gene cluster that is principally regulated by a pleiotropic virulence regulator PrfA. PrfA’s function is regulated by temperature : At a temperature lower than 30° C, the untranslated region of prfA mRNA adopts a stable secondary structure that prevents binding of the ribosome and blocks translation. In the host, the temperature of 37° C induces melting of the secondary structure; consequently, PrfA is translated and activates virulence gene expression. There are yet other mechanisms involved in regulating the virulence of Listeria; for most of these, the mechanisms are not fully understood.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here