Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Diagnosis is based on the anatomic location of ocular inflammation: vitreous and peripheral retina with macular edema.
Intermediate uveitis is frequently associated with a systemic or infectious disease.
Pars planitis is a subtype of intermediate uveitis.
Complications include macular edema, epiretinal membrane, vitreous hemorrhage, retinal detachment, and glaucoma.
Uveitis in patients with multiple sclerosis may be associated with a shorter time to first disease recurrence and a higher annualized relapse rate.
Treatment involves corticosteroids and immunosuppressive agents, including biologic agents.
Cryoretinopexy and vitrectomy may be beneficial for some patients.
The diagnosis of intermediate uveitis is based on the anatomic location of ocular inflammation; it is not a distinct clinicopathologic condition. The diagnosis is made when ocular inflammation primarily involves the vitreous and the peripheral retina. Intermediate uveitis was first described as chronic cyclitis by Fuchs in 1908. The clinical description of intermediate uveitis was further elucidated in 1950 by Schepens, who described patients with a peripheral uveitis characterized by inflammation centered on the vasculature and exudative changes in the retinal periphery. In 1960, Brockhurst et al. described additional patients with peripheral vascular abnormalities and exudate along the ora serrata and pars plana, and Welch et al. used the term pars planitis to describe inflammation characterized by the white accumulation on the pars plana. Other terms used in the literature to describe patients with intermediate uveitis include vitritis, peripheral exudative retinitis, cyclochorioretinitis, chronic posterior cyclitis, and peripheral uveoretinitis. Unfortunately, there is still confusion and controversy, even among uveitis specialists, regarding how to classify patients with this anatomic distribution of intraocular inflammation. Grouping conditions with the anatomic features of intermediate uveitis does provide value because it helps with developing a differential diagnosis and in determining response to therapy and long-term prognosis. The Standardization of Uveitis Nomenclature (SUN) Working Group has defined intermediate uveitis as disease where the vitreous is the major site of inflammation.
We use the term intermediate uveitis, as suggested by the International Uveitis Study Group, to classify patients with intraocular inflammation predominantly involving the vitreous and the peripheral retina. Some specialists use this term only if a more specific diagnosis cannot be made. Specific diseases, such as sarcoidosis and multiple sclerosis, are known to cause intermediate uveitis, and we often classify patients as having intermediate uveitis associated with sarcoidosis or associated with multiple sclerosis, when an underlying disease can be identified. The reason for this is that some diseases, such as sarcoidosis, can also present with other manifestations, such as predominant retinitis, and these patients will respond differently to therapy. We also use the term pars planitis to describe a subset of patients with intermediate uveitis when a white opacity (commonly called “snowbank”) occurs over the pars plana and the ora serrata. Although pars planitis probably does not represent a clinical entity distinct from intermediate uveitis, patients with pars planitis often have worse vitritis, more severe macular edema, and a worse prognosis compared with patients with intermediate uveitis who do not have a pars plana exudate.
Intermediate uveitis accounts for approximately 4% to 12% of cases of uveitis in a referral practice. In approximately 15% of patients with uveitis referred to us at the National Institutes of Health (NIH), intermediate uveitis is diagnosed. In a retrospective, population-based cohort study, the incidence of pars planitis was 2.077 per 100,000 persons (95% confidence interval [CI]: 1.43–2.62). Although the incidence of uveitis in children is low, intermediate uveitis may account for up to 25% of cases. Intermediate uveitis has been reported in both young children and older adults, but it tends to mostly affect people from the teens to the 40s. There appears to be no sex or race predilection.
There have been occasional reports of intermediate uveitis occurring in families, suggesting that either hereditary or environmental factors may predispose one to the disease. Human leukocyte antigen (HLA) associations have been identified in patients with multiple sclerosis and intermediate uveitis, but there is debate about the possibility of other HLA haplotypes being associated with intermediate uveitis. In 1963, Kimura and Hogan first reported multiple family members with intermediate uveitis. With a focus on the genetic associations of inflammatory diseases, a number of these families have been reported. , In one instance of familial pars planitis, the two affected brothers were not twins but had identical HLA haplotypes. There have also been cases of intermediate uveitis of the pars planitis subtype in identical twins. Some of the familial occurrences of intermediate uveitis are associated with demyelinating disease. , Studies have indicated a possible association of intermediate uveitis with HLA-A28, HLA-DR15, HLA-DR51, and HLA-DR17 haplotypes. IL2RA gene variants were found to be associated with intermediate uveitis but not with HLA-B27 acute anterior uveitis.
The causes of intermediate uveitis and pars planitis have not been elucidated. Evidence suggests that all patients with intermediate uveitis probably do not have the same disease. Although intermediate uveitis is a commonly encountered condition in uveitis clinics, the limited amount of pathologic material that has been studied has prevented a thorough clinicopathologic classification of the disease process.
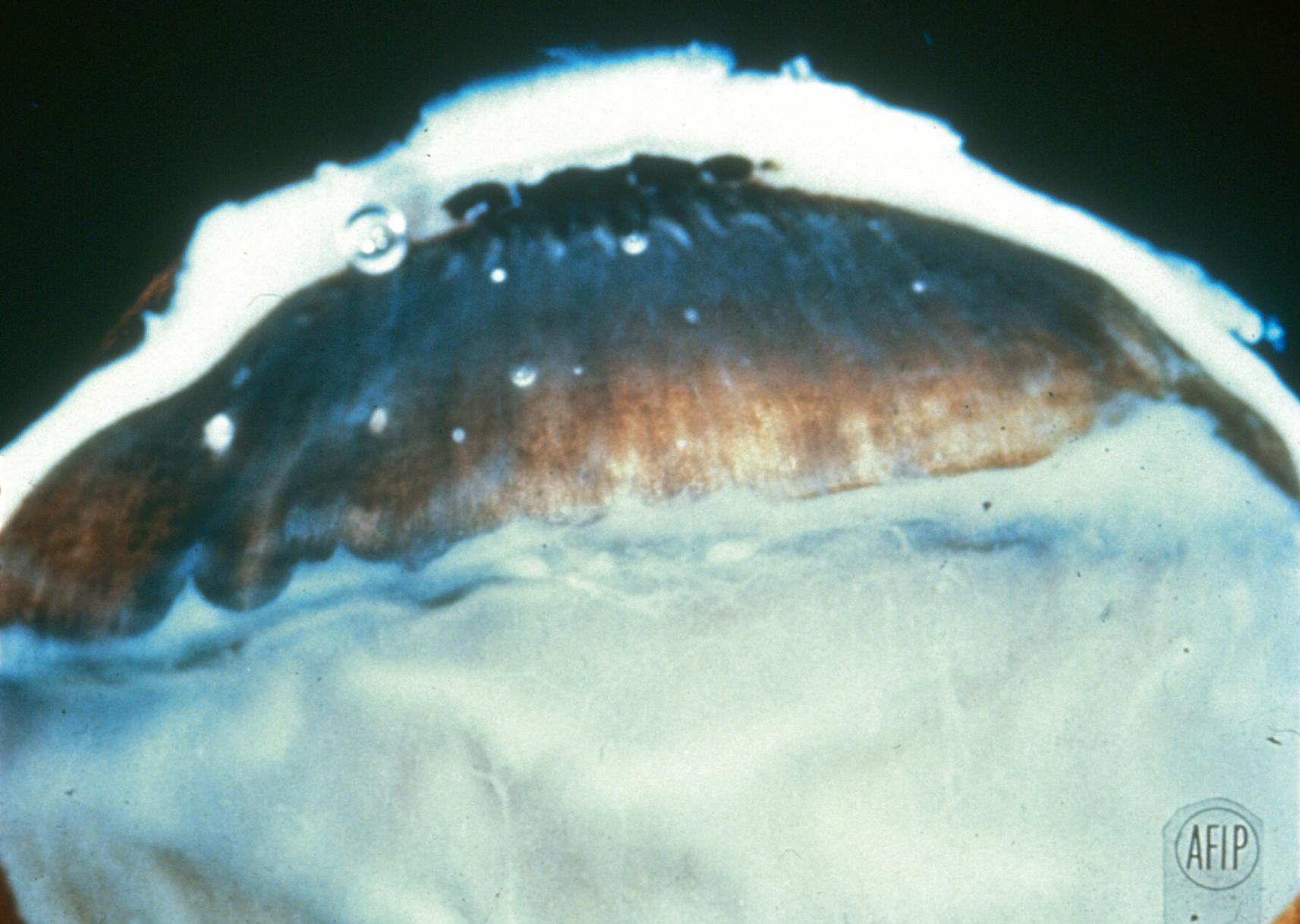
Exudate deposited in eyes with intermediate uveitis of the pars planitis subtype typically localizes to a broad area of the inferior pars plana (see Fig. 3.17 ). The inferior location of exudate is attributed to gravity. A diffuse phlebitis leads to breakdown of the blood–ocular barrier and release of inflammatory cells, cytokines, and other inflammatory mediators that settle inferiorly. Gross pathologic examination of the peripheral snowbank in pars planitis shows exudate deposited on the peripheral retina and pars plana ( Fig. 22.1 ). Histologic examination of this snowbank reveals collapsed vitreous; blood vessels; fibroglial cells, including fibrous astrocytes; and scattered inflammatory cells, predominantly lymphocytes ( Fig. 22.2 ). Peripheral veins show lymphocytic cuffing and infiltration. The vascular component of the snowbank is continuous with the retina in some cases; this suggests that the neovascularization associated with pars planitis may be the result of retinal vessels extending through breaks in the inner limiting membrane of the vitreous base. Only mild inflammation is noted in the choroid and the ciliary body, suggesting that the inflammatory process in intermediate uveitis primarily involves the vitreous base and the peripheral retina, and not the uvea.
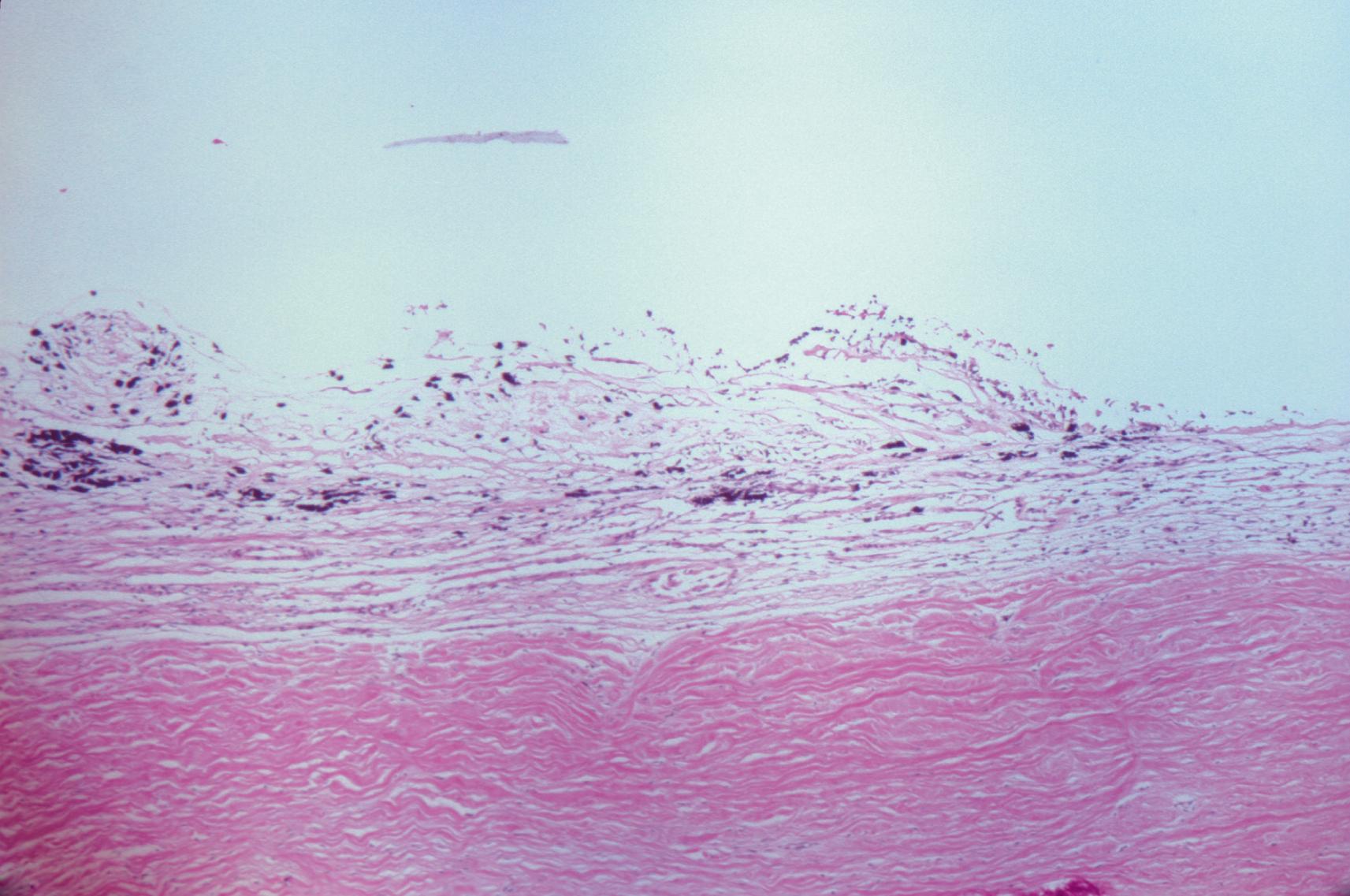
The vitreous snowballs in patients with intermediate uveitis are composed of epithelioid cells and multinucleated giant cells. Green et al. noted that these cells are absent from the uvea, suggesting that the vitreous may be the primary site of inflammation. We had the opportunity to study an eye from a patient with familial pars planitis. Histologic examination revealed glial tissue in the snowbank and extensive peripheral vascular disease, with perivascular infiltration of lymphocytes and expression of major histocompatability complex (MHC) class II antigen on the vascular endothelium. There was no substantial choroidal inflammation, again suggesting that pars planitis is a type of peripheral retinitis or vitritis, and not choroiditis. Studies of eyes with intermediate uveitis without pars plana exudate are sparse; this makes clinicopathologic correlation between the two disease types a difficult task.
The association between intermediate uveitis and autoimmune diseases, such as multiple sclerosis, suggests that at least some occurrences of the disorder are autoimmune in nature. Intermediate uveitis is thought to be a type of primary peripheral retinal perivasculitis. Once initiated, the process of vascular occlusion and inflammation may lead to vitritis and snowbank formation in some patients. Although others, such as Khodadoust et al., have hypothesized that intermediate uveitis is primarily vitreal inflammation, they have noted that the inflammation appears to start adjacent to the involved vascular tissue. Supporting the vascular etiology of the disease, Thorne et al. demonstrated that current smoking was associated with a dose-dependent increase in cystoid macular edema complicating intermediate uveitis. Interestingly, in a study in monkeys, injection of hyaluronic acid into the vitreous induced a clinical picture similar to that of pars planitis. Furthermore, Gartner demonstrated that the vitreous contains cellular remnants that may be the antigenic stimulus in pars planitis. Bora et al. , noted a 36-kDa protein with sequence homology to a yeast nucleopore complex, which may be a marker for the disease, in the serum of patients with active intermediate uveitis.
However, none of these theories fully explains the familial association in intermediate uveitis; the association of this disorder with certain systemic diseases, such as sarcoidosis, inflammatory bowel disease, and multiple sclerosis; or the cause of cystoid macular edema. We think that the periphlebitis and breakdown of the blood–ocular barrier are the initiating events in many cases. Increased levels of immune markers, such as soluble interleukin (IL)-2 receptors and intercellular adhesion molecule-1 (ICAM-1), have been found in the serum of patients with intermediate uveitis. , Elevated levels of IL-6 in the vitreous humor of patients with pars planitis confirms the local immune activation in this disease. However, the specific cause of this vascular disease is multifactorial and probably varies from patient to patient.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here