Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Patients with neuromuscular diseases may present with cardiac dysfunction, arrhythmia, or respiratory failure. Information in the literature regarding intensive care for these patients continues to evolve and single institution results have been published. Standards of care have been established for neuromuscular diseases. Close interaction among the specialties of cardiology, critical care, neurology, and pulmonary medicine are necessary for optimal outcomes. This chapter will provide a brief overview of the management of life-threatening cardiopulmonary complications of pediatric neuromuscular disease.
In general, intensive care is not precisely defined. It is often characterized by the location of the patient and the services provided. Guidelines for pediatric transport and intensive care serve as a basis for the specialized care required by patients with neuromuscular disease. ICU care includes more frequent and detailed cardiorespiratory assessment, establishment of central venous and arterial access, assisted ventilation, administration of vasoactive medications, and acute therapy for arrhythmia, all in an environment with continuous monitoring and low nurse-to-patient ratios. Specific indications for critical care ( Box 44.1 ) include respiratory failure, heart failure, life threatening arrhythmia, and resuscitated sudden death. The ICU may also be used for recovery following sedation or anesthesia. For the neuromuscular patient, expectant admission is appropriate in the presence of rapidly progressive weakness, markedly reduced vital capacity, bulbar palsy, and autonomic instability.
Unstable congestive heart failure
Life-threatening arrhythmias
Bradycardia requiring cardiac pacing
Perioperative recovery in presence of chronic respiratory insufficiency
FiO 2 requirement greater than 50%
Rising pCO 2 (>70 or with symptoms)
Markedly reduced vital capacity
Compromised airway
Endotracheal intubation
Rapidly progressive weakness
Bulbar palsy
Severe autonomic instability
The care of the critically ill patient begins prior to admission to the ICU and continues after discharge to the inpatient unit. Individuals skilled in pediatric intensive care should transport the critically ill child to the ICU. Ideally, the patient should be stabilized before transport. During appropriate transport, the supportive measures routinely available in the ICU should be available, including oxygen, assisted ventilation, and antiarrhythmic and cardiovascular support medications. The key to successful transport is excellent communication between the referring and accepting facilities as well as the transport team. A pediatric intensivist, pulmonologist, and cardiologist, all of whom are familiar with the complications of neuromuscular disease, should be involved early, before transfer as well as during the hospitalization. Comprehensive discharge planning is necessary to successfully transition from the ICU to an inpatient unit and subsequently home.
The indication for pursuing intensive care support for any patient is a balance between the perceived risks and benefits. In the child with progressive disease, it is often difficult to be certain when aggressive interventions become futile. The patient, depending upon developmental age, and the family must be involved in all decisions regarding extraordinary support measures or withdrawal thereof. Ethics consultation is often appropriate. In a disease known to have progressive deterioration and limited therapies to impact the natural history, the physician should encourage frank discussions about end of life decisions before an acute event occurs.
A complex control mechanism modulates the pumping action of the heart. Interdependent determinants of systolic function are: preload, the degree to which the ventricle is filled in diastole; afterload, the resistance to forward blood flow; heart rate and contractility, the latter of which describes the ability of the cardiac fibers to shorten. Active relaxation and the resting stiffness of the heart modulate diastolic function.
Normal conduction proceeds from the sinus node to the atria, through the atrioventricular node to the bundle of His, which subsequently divides into the major bundle branches. The electrical impulse utilizes these bundle branches for rapid distribution to the working myocardium of the ventricles. Short- and long-term variation is modulated by the autonomic nervous system and by the local tissue environment including pH, oxygen level, potassium, and calcium .
Ventilation is regulated by the autonomic nervous system, with limited voluntary override. Inspiration is achieved with the contraction of the diaphragm and intercostal muscles, and is dependent upon chest wall compliance and lung volumes. Optimum gas exchange occurs when all alveoli have adequate opening and circulation (ventilation perfusion matching).
Equally important is effective clearance of secretions through an adequate cough, which requires good inspiration, glottic closure, and sufficient contraction of the expiratory muscles to generate high pressure.
Heart failure results when the pump function of the heart is inadequate to meet the metabolic needs of the body. The clinical signs and symptoms of heart failure are age dependent. In the infant, feeding intolerance, irritability, tachypnea, and tachycardia are most prevalent. The older child and adolescent are more likely to complain of activity intolerance or fatigue. In the patient with skeletal muscle weakness, activity related symptoms may be masked.
Once a specific neuromuscular diagnosis has been established, screening is directed to the associated type of cardiomyopathy. Conversely, if a patient has an unknown neuromuscular disorder, then the presence and the characteristics of a cardiomyopathy may aid in obtaining the correct diagnosis. The two cardiomyopathies associated with neuromuscular diseases that ultimately lead to heart failure are dilated and hypertrophic.
Dilated cardiomyopathy is the result of decreased contractility and loss of myocardium. Initially dilation is a compensatory mechanism that augments systolic function by increasing the preload. However, left ventricular dilation without increasing wall thickness increases wall stress, and oxygen supply and demand become mismatched. Mechanical stress may accelerate this process. It has been postulated that skeletal muscle weakness may be associated with slower progression of cardiac disease because the heart is never required to respond to exertion or physical stress. For example, motor dysfunction progresses more slowly in Becker muscular dystrophy (BMD) than in Duchenne muscular dystrophy (DMD), but symptoms of cardiomyopathy are common in carriers of the DMD gene. Thus, asymptomatic carriers of neuromuscular disease who have little or no limb weakness may present with cardiomyopathy or conduction disturbances.
In DMD, clinically apparent cardiomyopathy is first evident after 10 years of age and increases in incidence with age; it is present in all patients over 18 years of age. Cardiac dysfunction typically manifests as left ventricular wall motion abnormalities and dilated cardiomyopathy. Histologic studies indicate that heart muscle from DMD and BMD patients is deficient in dystrophin affecting membrane integrity, calcium movement, and stretch receptor activation. Additional factors contributing to cardiomyopathy may include an abnormal coronary reserve, such that patients are unable to augment myocardial oxygen at times of increased demand.
Barth’s syndrome presents in infancy and is an example of rapidly fatal dilated cardiomyopathy with skeletal muscle weakness, neutropenia, 3-methylglutaconic aciduria, and abnormal mitochondria.
In hypertrophic cardiomyopathy , there is abnormal wall thickness that commonly impairs relaxation and ventricular filling; less commonly there is sufficient hypertrophy to obstruct ventricular emptying. Emery-Dreifuss muscular dystrophy (EDMD) and limb girdle muscular dystrophies, myofibrillar and metabolic myopathies, and Friedreich’s ataxia (FA) have been associated with hypertrophic cardiomyopathy. The hypertrophy of the left ventricle in FA appears to correlate with a large number of trinucleotide repeats.
The two broad mechanisms of arrhythmia are abnormal impulse formation (automaticity), and abnormal impulse conduction (reentry or block). All clinical arrhythmias involve one or both of these mechanisms. Arrhythmia classification is further subdivided into tachycardia and bradycardia as well as the involved chambers of the heart.
Examples of reentrant tachycardia that are seen in neuromuscular disease include atrioventricular nodal reentrant tachycardia, and atrial flutter. Atrial fibrillation may be due to abnormal automaticity or reentry.
Ventricular tachycardia arises below the level of the AV node and is characterized by a QRS complex that is wider than normal. This also may be due to automatic or reentrant mechanisms.
Bradycardia is due to sinus node dysfunction (abnormal automaticity) or failure of the electrical impulse to conduct to the ventricle (block). Sinus bradycardia has been reported in muscular dystrophy and mitochondrial disease.
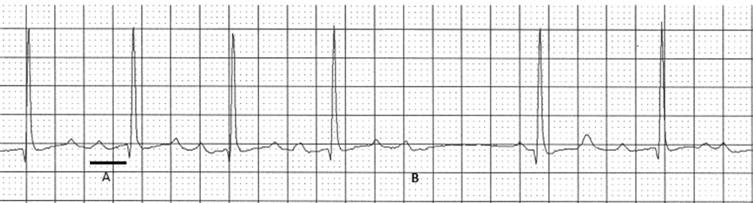
Heart block may occur anywhere within the cardiac specialized conductions system. Block can be intermittent, known as second degree ( Figure 44.1 ), or permanent, known as complete or third degree atrioventricular block.
Arrhythmias associated with neuromuscular disease are extensive. Significant arrhythmia frequently coexists with asymptomatic left ventricular dysfunction and wall motion abnormalities. In EDMD, cardiac abnormalities become apparent in the teenage years and are characterized by cardiac conduction defects and infiltration of the myocardium by fibrous and adipose tissue. Usually atrial paralysis is the first manifestation. Subsequent atrioventricular nodal dysfunction may result in progressive heart block and the potential for sudden death. Treatment with ventricular pacing is usually needed and is instituted with the onset of conduction system impairment. Female carriers of EDMD are also at risk of sudden death. Relatives of affected patients should be offered screening with electrocardiography (ECG) and echocardiography. Patients with myotonic dystrophy often develop progressive ECG changes without obvious clinical symptoms until the development of complete heart block when, again, there is the risk of sudden death. The severity of the rhythm disturbance seems to correlate with the expansion of unstable repeats. Some clinicians recommend prophylactic placement of a pacemaker in the adult myotonic dystrophy patient.
Sudden death may be arrhythmic, circulatory, or noncardiac. Noncardiac causes are numerous, but include neurologic events, respiratory failure, and trauma. Arrhythmia and congestive heart failure account for the vast majority of sudden deaths in neuromuscular patients. Tachyarrhythmia (ventricular tachycardia or fibrillation) or bradyarrhythmia (atrioventricular block or asystole) may cause sudden death. Complex ventricular ectopy, left ventricular dysfunction, and dilated cardiomyopathy are risk factors for sudden death. Specific risk factors for sudden death in myotonic dystrophy include increased PR interval or QRS duration, abnormal QT intervals, atrial arrhythmia, and a positive family history.
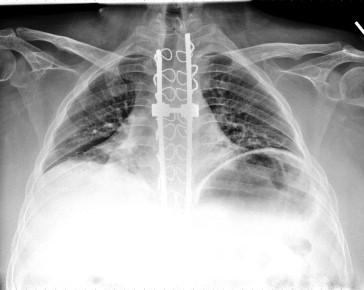
Acute respiratory illness leading to respiratory compromise was the leading cause of unplanned admission to the ICU in children with neuromuscular disease ( Figure 44.2 ). The number of patients placed on home mechanical ventilation has increased dramatically, as has the need for ICU care. For the majority, initiation of mechanical ventilation was unplanned.
There are three primary causes responsible for respiratory failure in neuromuscular diseases. Respiratory muscle weakness (pump failure) impairs ventilation. Inefficacy of cough is due to expiratory muscle weakness, upper airway (glottic) muscle weakness, and inspiratory muscle weakness. Glottic muscle weakness may lead to aspiration and pneumonia. Abnormal chest wall and lung compliance contribute to respiratory insufficiency.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here