Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Coronavirus disease 2019 (COVID-19), the pandemic illness caused by a novel coronavirus, has been associated with stroke, possibly related to thrombophilia and severe inflammation.
Primary angiitis of the central nervous system (PACNS) is an inflammatory arterial disease restricted to the cerebral circulation that can afflict patients of any age, and often responds poorly to steroids alone.
Patients with PACNS commonly present with a combination of headache, encephalopathy, seizure, and ischemic or hemorrhagic stroke.
PACNS can be associated with lymphoma, acquired immunodeficiency syndrome, leukemia, sarcoidosis, varicella zoster virus infection, and cerebral amyloid angiopathy.
The most consistent cerebrospinal fluid abnormality in PACNS is an increase in protein, and many patients have a moderate lymphocytic pleocytosis.
Neurologic symptoms associated with giant cell arteritis include the new onset of a persistent headache, jaw claudication, visual symptoms (e.g., diplopia, scotoma, amaurosis fugax, or blindness), and rarely stroke.
Almost all patients with giant cell arteritis-associated strokes have a significant acute phase response with elevated erythrocyte sedimentation rate and C-reactive protein.
Visual loss is one of the most feared complications of giant cell arteritis, and anterior ischemic optic neuropathy is its most common cause.
Once the diagnosis of giant cell arteritis is suspected, the patient should be started on steroid therapy and biopsy performed as soon as possible.
Although vasculitis is frequently mentioned as an important cause of stroke in patients with systemic lupus erythematosus, documented vasculitic changes on postmortem examination are actually quite rare.
All patients with neurosyphilis should also be tested for human immunodeficiency virus.
The most common parasitic illnesses that cause stroke are Chagas disease and neurocysticercosis.
Aspergillus fumigatus is a highly angiotrophic fungus that causes stroke in immunocompromised patients, but it can affect immunocompetent patients as well.
Because not all patients with vasculitis due to varicella zoster virus (VZV) report prior exposure to VZV infection or rash, VZV should be considered in cases of cryptogenic arteriopathy and stroke.
Stroke is a common complication of many inflammatory, collagen vascular, and infectious diseases. This chapter reviews the clinical findings, diagnosis, and treatment of stroke associated with some of the most common inflammatory and infectious diseases.
Vasculitides comprise a heterogeneous group of diseases characterized by inflammation of the blood vessel wall. The central nervous system (CNS) can be a target of both primary and secondary CNS vasculitis. Causes of secondary CNS vasculitis include systemic vasculitides, such as giant cell arteritis and Takayasu arteritis (TA), as well as several infectious agents ( Table 36.1 ). Secondary causes of vasculitis need to be excluded before primary CNS vasculitis, also often called primary angiitis of the CNS, can be diagnosed.
| Large, medium, small, and variable vessel vasculitides | Giant cell arteritis Takayasu arteritis Polyarteritis nodosa Kawasaki disease Granulomatosis with polyangiitis (formerly Wegener disease) Cryoglobulinemic vasculitis Behçet disease Eosinophilic granulomatosis with polyangiitis (formerly Churg-Strauss syndrome) |
| Vasculitides associated with systemic diseases/connective tissue diseases | Systemic lupus erythematosus Rheumatoid arthritis Sjögren syndrome Scleroderma |
| Others | Drug induced (cocaine, amphetamine) Antiphospholipid antibody syndrome Lymphoma Graft-versus-host disease Paraneoplastic Bacterial endocarditis |
| Infectious Agents | |
| Viral | HIV, varicella zoster virus, cytomegalovirus, parvovirus B19, others |
| Bacterial | Treponema pallidum, Mycobacterium tuberculosis Borrelia burgdorferi, others |
| Fungal | Cryptococcus, aspergillosis, mucormycosis, coccidioidomycosis, candidiasis, others |
| Parasitic | Chagas disease, cysticercosis, malaria, schistosomiasis, others |
Primary angiitis of the CNS (PACNS) is an inflammatory arterial disease restricted to the cerebral circulation. PACNS afflicts patients of any age, preferentially involves smaller arteries and veins, and often responds poorly to steroids alone. The diagnosis is difficult, and because it has often been made without pathologic confirmation, the true extent and characteristics of the disease are poorly understood. , The incidence of PACNS has been estimated to be 2.4 cases per 1,000,000 person-years.
The pathologic process in PACNS is segmental, and a negative biopsy therefore does not exclude the diagnosis, especially if larger vessels are involved, which typically are not targeted by biopsy. Any of the vessels of the brain and spinal cord may be involved, but there is a predilection for the small leptomeningeal vessels. Three main histopathologic patterns are seen: granulomatous, lymphocytic, and necrotizing vasculitis. The etiology of PACNS is unknown. Basic research suggests that memory T cells are associated with its pathogenesis, and that it can result from an antigen-specific immune response occurring in the wall of cerebral arteries. , Cases have been reported in association with Hodgkin lymphoma, acquired immunodeficiency syndrome (AIDS), primary intracerebral lymphoma, leukemia, sarcoidosis, varicella zoster virus (VZV) infection (especially in children), and cerebral amyloid angiopathy (CAA). , In a subset of patients with amyloid-β (Aβ) deposition, vascular inflammation is present, with features of a true vasculitis, most frequently granulomatous (Aβ-related angiitis or ABRA). It is likely that PACNS encompasses a spectrum of different diseases characterized by a vascular inflammatory reaction to antigens rather than a single nosologic entity.
The presentation of PACNS is heterogeneous. Patients range in age widely, from 3 to 96 years. The duration of illness is variable. Death may occur within days after presentation in some patients, whereas others have an indolent course that lasts for years. The two most common clinical manifestations at presentation are headache and altered cognition. Multifocal neurologic symptoms and signs may develop in a stepwise, progressive fashion. Stroke or focal symptoms develop in less than 20% of patients at the onset of disease and are uncommon in the absence of headache or encephalopathy, but during the disease course persistent neurologic deficits occur in 40% of patients. When rapidly progressive, PACNS may be fatal. Some patients have spinal cord involvement (either alone or with brain involvement), including progressive or acute myelopathy. ,
Systemic symptoms, such as fever and weight loss, are uncommon, distinguishing this entity from most systemic inflammatory diseases that cause stroke, such as giant cell arteritis (GCA) or systemic lupus erythematosus (SLE). The erythrocyte sedimentation rate (ESR) is usually not increased, and when abnormal, it is not as high as in GCA. Blood cell counts, electrolyte levels, and results of serologic tests for collagen vascular disease are usually normal. PACNS patients present with a highly diverse cerebrospinal fluid (CSF) immune cell repertoire. The most consistent (CSF) abnormality is an increase in protein (abnormal in 80%–90% and frequently >100 mg/dL), a sign of a disruption of the blood/CSF barrier, although CSF may be normal. Many patients have a moderate lymphocytic pleocytosis (usually <150 cells/mL) and thus manifest as if they have chronic meningitis.
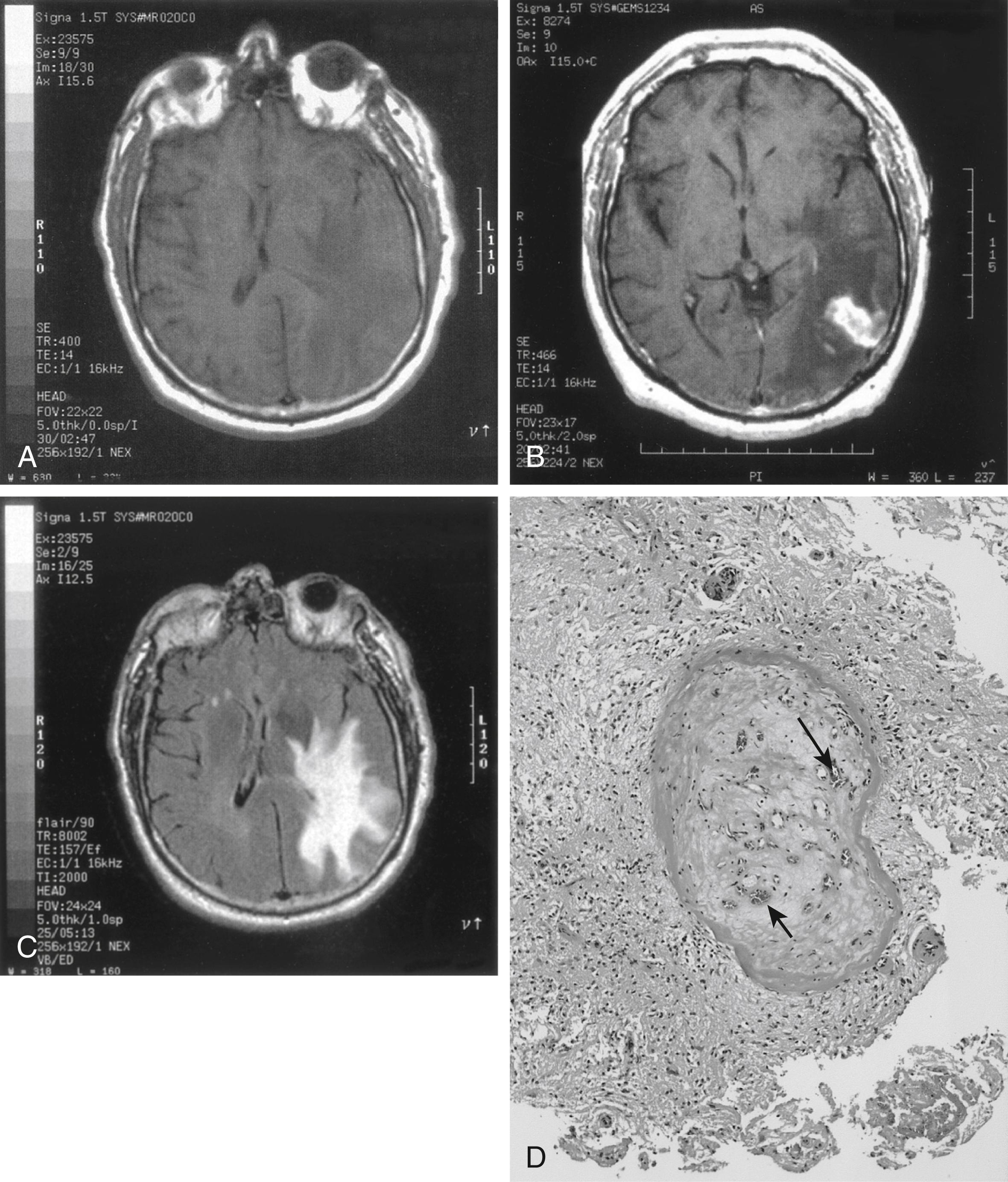
Neuroimaging is a critical component of the evaluation of PACNS patients. Computed tomography (CT) and magnetic resonance imaging (MRI) findings are heterogeneous and nonspecific, but PACNS is unlikely if MRI is normal ( Fig. 36.1A –C). Ischemic strokes on diffusion-weighted imaging (DWI) sequences, vascular fluid-attenuated inversion recovery (FLAIR) hyperintensities, as well as parenchymal and/or meningeal hemorrhages can be seen. Cerebral angiographic results are usually abnormal, with alternating segments of concentric arterial narrowing and dilatation. However, cerebral angiographic findings may be normal when smaller blood vessels are affected, and abnormal constrictions found in vessels may also be due to a host of other causes, making reliance on angiography alone for the diagnosis problematic. High-resolution vessel wall magnetic resonance angiography (MRA) for the assessment of CNS vasculitis is a noninvasive diagnostic method that has emerged in recent years..
One increasingly recognized mimic of primary CNS vasculitis is the reversible cerebral vasoconstriction syndrome (RCVS), which may also present with severe headache, subarachnoid hemorrhage, focal deficits and infarcts, and evidence of vasculopathy on angiography (see Chapter 37 ). However, it may be distinguished by the normal CSF and a benign clinical course. In addition, the vessel wall MRA changes may be less pronounced in RCVS.
The most common pathologic finding in PACNS is multiple small foci of infarction, followed by multiple foci of hemorrhage (“brain purpura,” “petechiae,” Fig. 36.1D ). Large infarcts and, less often, large confluent intraparenchymal hemorrhages may be present. Vessel wall thickening and intramural inflammation of large arteries is quite specific. Brain and leptomeningeal biopsy demonstrating vasculitis remains the gold standard for the diagnosis of PACNS. , A biopsy, ideally taken from a region of brain enhancement on neuroimaging, is the best test for the diagnosis of PACNS , and is necessary to distinguish among tumor (especially lymphoma or intravascular malignant lymphomatosis), infection, and other mimics. However, biopsy results have been negative in several subsequently autopsy-proven cases, presumably due to the segmental nature of the disease. ,
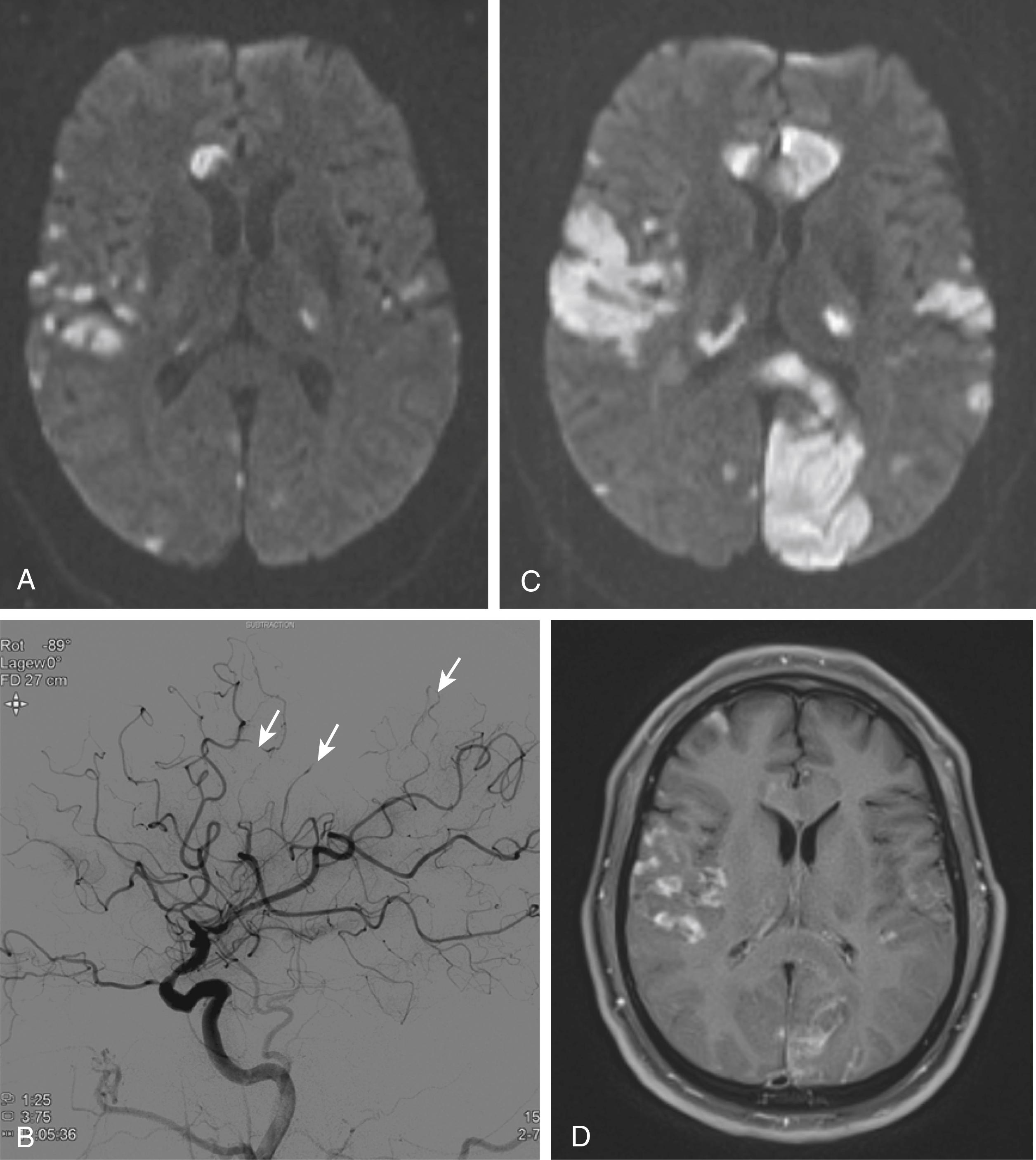
No controlled therapeutic trials for CNS angiitis have been performed, and there is no generally accepted standard treatment. In one of the largest cohorts of patients with PACNS ( n = 101), glucocorticoids alone, or the combination with cyclophosphamide resulted in a favorable outcome in most, although 26% of patients had recurrences ( Fig. 36.2A –D). PACNS is most likely a heterogeneous condition composed of clinical subsets that differ in terms of outcome and response to treatment. Patients with angiographic evidence of multiple bilateral large-vessel lesions and MRI evidence of multiple cerebral infarctions have higher mortality, whereas patients with more small-vessel involvement appear to have less severe disease (including patients with amyloid-related angiitis). Treatment therefore may need to be based on clinical progression and the size of the vessel involved at diagnosis. In a cohort of 52 patients, those with relapsing disease more often had gadolinium enhancement and seizures at diagnosis, and they needed therapy for longer than did those with nonrelapsing disease. Azathioprine as well as mycophenolate mofetil (MMF) have been used as steroid-sparing agents, and there have been reports of successful treatment with methotrexate, tumor necrosis factor-α (TNF-α) blockers, and rituximab.
Secondary causes of CNS vasculitis include systemic large, medium, and small-vessel vasculitides due to connective tissue diseases, drugs, neoplasms, and some infectious agents. In a recent national epidemiologic study in Sweden, several immune-mediated diseases increased the risk of hospitalization for both ischemic and hemorrhagic stroke. The relative risk of ischemic and hemorrhagic stroke during the first year after hospitalization with systemic inflammatory diseases (including systemic vasculitides but also other diseases) was even higher than the risks associated with many traditional risk factors for ischemic and hemorrhagic stroke. Identification of these secondary causes of CNS vasculitis is important, since they may be treated differently, including removal of the offending antigen, and antimicrobial therapy. Prognosis in these cases also depends on the underlying disease, and other causes, especially infectious causes, must be excluded before treating patients with immunosuppressants.
GCA is the most common of the vasculitic syndromes and, being a disease of the elderly, its incidence is increasing with the aging of the population. GCA, also known as temporal arteritis, is an inflammatory disease affecting medium and large arteries throughout the body, including the aorta and most of its major branches. ,
Giant cell arteritis is a disease that affects the elderly, with a peak incidence at the age of 70–80 years; age (50 years or older) is considered a criterion for the diagnosis. Current incidence rates vary between 1 and 30/100,000 people aged ≥50 years. The highest frequencies have been reported from Scandinavian countries and populations of predominantly Scandinavian descent; rates in southern Europe are intermediate, and they are still lower in people of African, Asian, and Hispanic descent. There is an almost threefold greater incidence in women than in men. The incidence may be up to seven times higher among whites than blacks, possibly in relation to the lower frequency of HLA-DR4 (D-related human leukocyte antigen) among blacks. ,
Inflammation of the vessel wall is characterized by infiltration of T-cells and macrophages, the presence of giant cells, granulomatous lesions, intimal hyperplasia leading to occlusion of the lumen, and destruction of elastic fibers. The cause of GCA has not been clearly identified, but it is thought that GCA occurs on a genetic background and is triggered by environmental factors such as infectious agents that could activate and lead to the maturation of dendritic cells localized in the adventitia of normal arteries. ,
Clinically, GCA may present with symptoms related to the involved cranial vessels, with the signs of a systemic illness (fever, malaise, and weight loss), or with polymyalgia rheumatica (PMR). Neurologic symptoms include the new onset of a persistent headache (most common), jaw claudication, visual symptoms such as diplopia, scotoma, amaurosis fugax, or blindness—or, rarely, stroke. , Almost all patients with GCA-associated strokes have a significant acute phase response with elevated ESR and C-reactive protein (CRP).
Visual loss is one of the most feared complications of GCA. Involvement of ophthalmic vessels (ophthalmic artery and/or posterior ciliary arteries) is frequent and may be responsible for permanent or transient visual loss, especially because of consecutive acute optic nerve ischemia, occurring in 10%–15% of patients. Before fixed visual deficits develop, 8%–28% of patients experience transient loss of vision (transient monocular blindness). Bilateral involvement occurs in 20%–62% of patients. Afferent pupillary defects are common. Field defects are usually altitudinal and inferior.
Less often, the visual loss takes the form of homonymous hemianopia, or even cortical blindness, as a result of posterior circulation infarction. Overall, strokes have been reported to occur at disease onset in 2.8%–7.2% of patients. ,
In a recent cohort study of 781 patients suffering from PMR, which is associated with GCA, 113 had a stroke within 3 years follow-up. The hazard ratio (HR) of stroke for patients with PMR was 2.09 times that of age- and sex-matched controls (95% CI, 1.63–2.66; P < .001), after adjusting for sociodemographic characteristics and comorbidities. Mortality with GCA has been reported to be as high as 75%.
Postmortem examinations usually document involvement of the extradural segments of the vessels only, although some have shown evidence of intradural involvement as well. Thrombotic occlusion due to arteritis in internal carotid arteries has also been described, although GCA-associated stroke is more common in the vertebral artery territories. Other patients have had artery-to-artery embolism with hemorrhagic infarction.
When GCA is suspected, ESR and CRP should be obtained to confirm evidence of inflammation, but biopsy of an artery showing the typical histologic features provides the definitive diagnosis. Because of its easy accessibility and frequent involvement, the temporal artery is usually chosen as the biopsy site. The sensitivity and specificity of temporal artery biopsy have been reported to be approximately 75% and 90%, respectively. However, due to the possibility of skip-lesions, sensitivity depends on obtaining a sufficient length of the biopsy sample, and bilateral biopsies may be performed. In one study, CRP levels greater than 2.45 mg/dL and platelet counts greater than 400,000 were the strongest laboratory predictors of a positive temporal artery biopsy, whereas ESR was not significant in models already including CRP and platelet count.
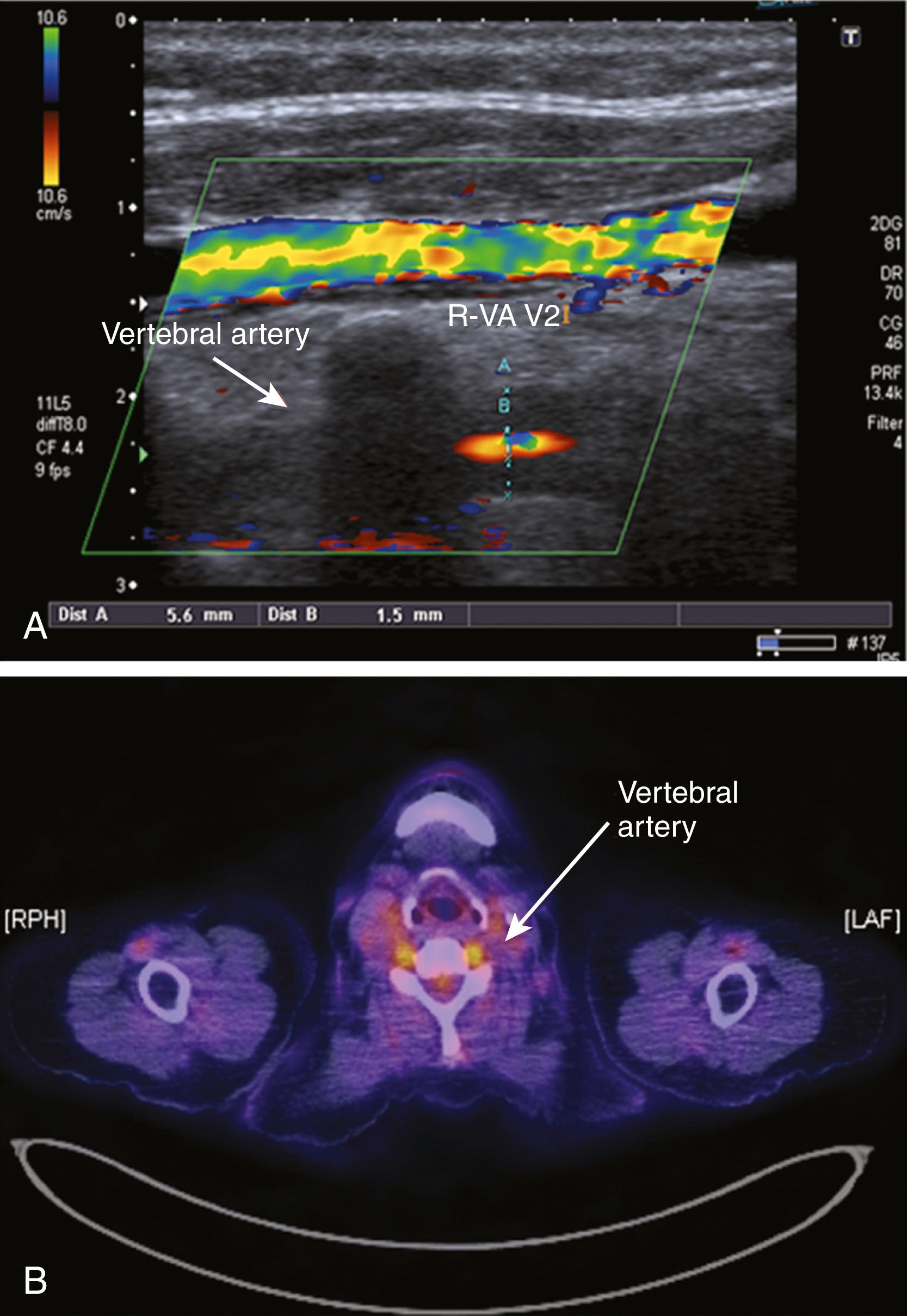
Imaging—including ultrasound, positron emission tomography (PET), and MRI—has begun to play a role as a broadly applicable and noninvasive tool to establish the diagnosis of GCA. Using color duplex ultrasonography, a typical dark halo has been reported around the lumen of the superficial temporal artery in 73% of patients, an abnormality that disappears with treatment. The vertebral arteries may also show this abnormality ( Fig. 36.3 ). Assessing 18F-fluoro-2-deoxy-D-glucose (FDG) uptake by PET appears helpful to assess vasculitic involvement of intrathoracic vessels that cannot be readily visualized by duplex ultrasound, and in cases of nonspecific signs and symptoms compatible with large-vessel vasculitis. Sensitivity and specificity of PET/CT to detect large-vessel vasculitis in untreated patients are about 85% and 95%, respectively. Brain MRI may show multifocal dural enhancement and temporalis muscle enhancement. ,
Once the diagnosis of GCA is suspected, the patient should be started on steroid therapy and biopsy performed as soon as possible. The diagnostic sensitivity of temporal-artery biopsy remains high even after glucocorticoid therapy has been initiated; the sensitivity declines after several weeks of therapy. , Symptoms usually respond promptly to steroids, although visual loss and stroke may occur after initiation of treatment. When ischemic complications—such as visual impairment or stroke—are present, 0.5–1 g of methylprednisolone can be administered intravenously for 3 days. Based on a meta-analysis of the published evidence, current EULAR (European League Against Rheumatism) guidelines suggest that the initially prescribed dose of prednisolone should be maintained for 4 weeks and tapered gradually thereafter. In the absence of evidence for relapsing disease, the dose should reach 10–15 mg/day by month 3 and 5 mg/day by month 6; this dose should be maintained for at least 12 months after diagnosis.
Because complications due to steroid therapy are very common, steroid-sparing/disease modifying antirheumatic drugs have been tested in various studies aiming to optimize outcome while reducing glucocorticoid toxicity. Methotrexate was shown in an individual patient meta-analysis based on three trials to be an effective adjunctive therapy in GCA. A randomized controlled trial has also shown that tocilizumab, an interleukin (IL)-6 receptor α inhibitor, taken weekly or every other week, combined with a 26-week prednisone taper, was superior to either 26 or 52 weeks of prednisone tapering plus placebo with regard to sustained glucocorticoid-free remission in patients with giant cell arteritis.
Randomized, controlled, clinical trials are needed to clarify the usefulness of aspirin in primary prevention of GCA-associated vascular complications, but low-dose aspirin should be considered for all patients without contraindications.
TA (pulseless disease, idiopathic aortitis) is a large-vessel arteritis that affects the aorta, its main branches, and, occasionally, the pulmonary artery. Although pathologic changes in the arteries are similar to those found in GCA, TA affects younger people, particularly women. The disease is found worldwide, with the greatest prevalence in Asia, and women are affected in 80%–90% of cases, with an age of onset that is usually between 10 and 40 years. As in GCA, constitutional symptoms and increased ESR are common in the acute phase. Brachial pressures and pulses are frequently asymmetric, and there may be differences between blood pressures in the arms and legs. Symptoms, such as arm claudication and syncope, occur later during the disease as do cerebrovascular complications. Cerebral infarction and retinal ischemia occur subsequent to stenosis or occlusion of the extracranial carotid or vertebral arteries, but the intracranial arteries are rarely involved. Among a retrospective cohort of 190 TA patients, hemodynamic compromise due to large-artery stenosis and thromboembolic mechanisms were the most important causes of ischemic stroke associated with TA.
Conventional angiography generally provides clear outlines of the lumen of involved arteries and is clearly indicated if therapeutic intervention is anticipated, but to assess arterial wall thickening and as a first-line diagnostic approach MRA, PET/CT, and high-resolution ultrasound are being used more frequently. Gadolinium-enhanced MRI shows delayed hyperenhancement in the aortic wall in patients with TA, and this correlates with ESR and CRP. , FDG-PET can detect metabolic activity in vasculitis because of increased FDG uptake in inflammatory cells in the vessel wall, including larger vessels such as the aorta, its main branches, and the femoral and pulmonary arteries.
Treatment consists of corticosteroids, cytotoxic agents (cyclophosphamide), anti-TNF agents, surgery, or a combination of these modalities. Fifty percent of patients respond to corticosteroids alone. Rituximab has been associated with a reduction in disease activity in small numbers of patients, including those refractory to anti-TNF treatment. In a recent small randomized placebo controlled trial, tocilizumab showed a promising effect in the reduction of time to relapse, but only in the intention-to-treat population. Antiplatelet agents are recommended in secondary prevention. A recent retrospective multicenter study found that in 79 consecutive patients with TA who underwent 166 vascular procedures, the overall 5-year arterial complication rate was 44%, and biological inflammation at the time of revascularization increased by seven times the likelihood of complications.
CNS involvement is infrequent (<15%) in small- to medium-size vessel vasculitis, including polyarteritis nodosa (PAN), Kawasaki disease, antineutrophil cytoplasmic antibody (ANCA)-associated vasculitides, eosinophilic granulomatosis with polyangiitis (formerly Churg-Strauss syndrome), immune complex vasculitis (such as cryoglobulinemic vasculitis [CV]), and variable vessel vasculitis (such as Behçet disease [BD]).
PAN is a necrotizing angiitis of the medium to small muscular arteries throughout the body. The peripheral nervous system is more commonly involved than the CNS. In a series of 348 patients diagnosed with PAN over a 42-year period, only 4.6% presented with CNS-related abnormalities. Neurologic symptoms of PAN-related CNS involvement can be acute (e.g., stroke or seizures) or more chronic and insidious, such as headaches or encephalopathic symptoms (cognitive and vigilance disorders or psychiatric manifestations).
Lacunar infarcts were the most frequent (73%) stroke type, complicating the disease course among patients with PAN in a small series of patients. The authors argued that hypertension-associated microangiopathy and a prothrombotic state may be more frequent components of a CNS event in these patients than a vasculitic process. However, among early-onset PAN the deficiency of adenosine deaminase 2 (ADA2) is associated with an early onset vasculopathy, hemorrhagic, and ischemic strokes. ,
Treatment of PAN varies according to cause (e.g., hepatitis B virus [HBV] positivity or not, deficit of adenosine deaminase-2 [DADA2]) and severity. First-line treatments are corticosteroids and sometimes immunosuppressants in the absence of HBV infection, or corticosteroids, then antiviral treatment like lamivudine (for HBV) or interferon-α and ribavirin (for hepatitis C). In emergency situations plasmapheresis may be tried. In patients with DADA2 anti-TNF therapy is recommended.
Wegener granulomatosis is an ANCA-associated necrotizing granulomatous vasculitis involving the respiratory tract, kidney, and other organ systems. The prevalence of CNS manifestations ranges from 3% to 9% in large series of patients. Neurologic involvement may occur through three major mechanisms: (1) vasculitis involving CNS vessels; (2) granulomatous lesions located in the brain, meninges, or cranial nerves; and (3) direct extension of destructive granulomatous tissue from nasal or paranasal structures.
Granulomatosis with polyangiitis should be suspected when stroke patients have evidence of other manifestations of neurologic inflammatory disease, such as meningeal disease, cranial neuropathies, and mononeuritis multiplex; concomitant constitutional symptoms, such as fever, malaise, weight loss, or migratory arthralgias; or involvement of the ear and nasal passages, pulmonary airways or parenchyma, or kidneys, especially glomerulonephritis. Diagnosis can be challenging, as the signs and symptoms are variable and may mimic infectious or malignant disease. Antineutrophil cytoplasmic antibodies should be tested, as approximately 90% of patients are ANCA-positive. Patients with proteinase 3 ANCA may have a reduced vascular risk compared to myeloperoxidase ANCA or negative ANCA status (about 10% of patients). In addition, the diagnosis should be confirmed by biopsy of involved tissue, often lung or kidney. Chest CT and renal evaluation should be undertaken to assess for involvement of those organs.
Therapy for granulomatosis with polyangiitis includes initial immunosuppressive therapy followed by maintenance therapy. The mainstays of initial therapy are glucocorticoids and either cyclophosphamide or rituximab. Plasma exchange may be used in severe disease. Once remission has been achieved, maintenance therapy with less toxic immunosuppressive medications, such as azathioprine, MMF, or methotrexate, may be used. Patients should also receive prophylaxis against opportunistic infections.
Eosinophilic granulomatosis with polyangiitis (EGPA) is a rare systemic ANCA-associated vasculitis. The CNS is involved in 8%–14% of patients, and cerebral infarction is one of the most frequently reported manifestations. In a series of 88 reported cases with EGPA-related CNS manifestations, four distinct neurologic pictures were identified: (1) ischemic lesions (52%), (2) intracerebral hemorrhages (24%), (3) cranial nerve palsies (821%), and (4) loss of visual acuity (33%), with 28% having more than one of these manifestations.
Cyclophosphamide and glucocorticoids are the main therapy for induction of remission in patients with generalized or severe ANCA-associated vasculitides. Dose reductions and avoidance of prolonged use of these agents have also been successfully implemented. For patients without organ-threatening disease, methotrexate can be used. Rituximab is as effective as cyclophosphamide for remission induction. In a randomized controlled trial, therapy with mepolizumab, an anti-IL-5 monoclonal antibody, resulted in significantly more weeks in remission and a higher proportion of participants in remission than did placebo, thus allowing for reduced glucocorticoid use.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here