Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Bacterial, viral, fungal, and parasitic organisms are all causative factors in central neurologic system (CNS) infection. Brain infection manifests as encephalitis, cerebritis, and meningitis. Encephalitis refers to diffuse infection of the brain parenchyma, whereas cerebritis is due to a more focal parenchymal infection. Meningitis refers to infection of the pia, arachnoid, and dural membranes as well as the cerebrospinal fluid (CSF). Ventriculitis is often present in cases of meningitis. Infectious complications most often include abscess and/or empyema. These infectious collections, in association with infectious symptomatology, are straightforward in their diagnosis. Infection in the setting of tumor or hemorrhage often presents a diagnostic conundrum.
Imaging of CNS infection is most often initially performed by means of computed tomography (CT) to assess for hydrocephalus or increased intracranial pressure before the performance of a lumbar puncture. Nonspecific parenchymal hypoattenuation indicative of edema on CT is sometimes appreciated in cases of more focal infection. In more complicated cases, CT is superior in the evaluation of bone erosion and destruction. More advanced imaging utilizing magnetic resonance imaging (MRI) is warranted in the assessment of infectious complications such as abscess, empyema, vasculitis, and/or ischemia in the setting of a worsening clinical condition or lack of clinical improvement. Infection is most often manifested on MRI by hyperintense signal abnormality on T2-weighted and proton density sequences. The most sensitive magnetic resonance (MR) sequence is fluid-attenuated inversion–recovery (FLAIR). There is often corresponding hypointense signal on T1-weighted sequences. Postcontrast T1-weighted images are essential in the evaluation for infectious collections and meningeal enhancement. Postcontrast images or magnetic resonance venography (MRV) sequences help to visualize suspected venous sinus thrombosis as a complication of infection. Diffusion-weighted imaging (DWI) and apparent diffusion coefficient (ADC) maps help in localizing abscess collections and infection associated with ischemia and can at times help distinguish lymphoma from abscess, especially in immunocompromised patients. DWI may also demonstrate lesions earlier than conventional sequences in detecting viral infections such as herpes and West Nile virus ( e-Fig. 34.1 ). MR spectroscopy (MRS) may be helpful in distinguishing pyogenic abscesses from those composed of more atypical organisms, with the former often demonstrating the presence of amino acids and lack of choline.
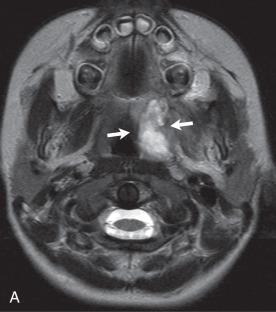
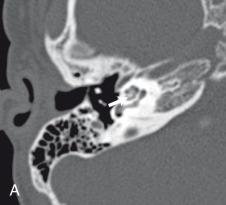
Nearly two-thirds of cases of bacterial meningitis in the United States occur in children. Routes of transmission include hematogenous, direct traumatic, congenital, as well as via direct extension from adjacent sinus or mastoid disease ( e-Fig. 34.2 , Figs. 34.3 and 34.4 , and e-Fig. 34.5 ). Imaging plays a key role in determining the course of treatment for bacterial infection. It is essential to distinguish between a focal cerebritis ( Fig. 34.6 ), in which surgery is contraindicated and tends to respond to antibiotics, and frank abscess that often requires surgical intervention in addition to antibiotic therapy.
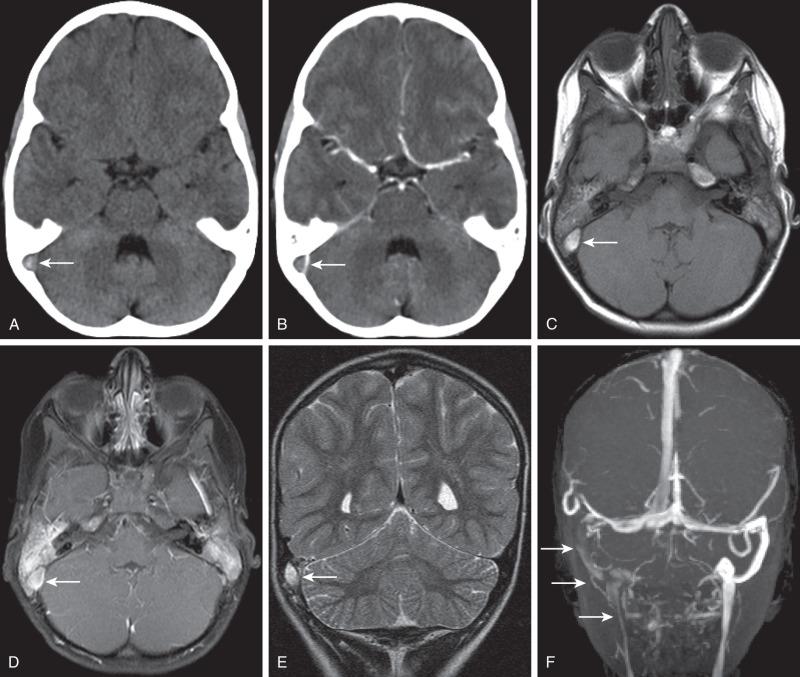
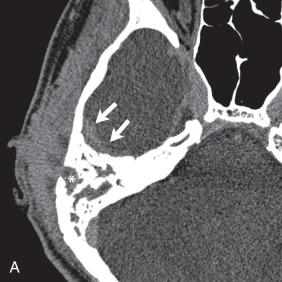
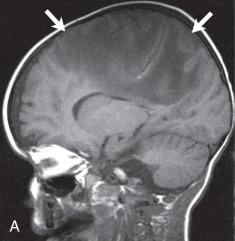
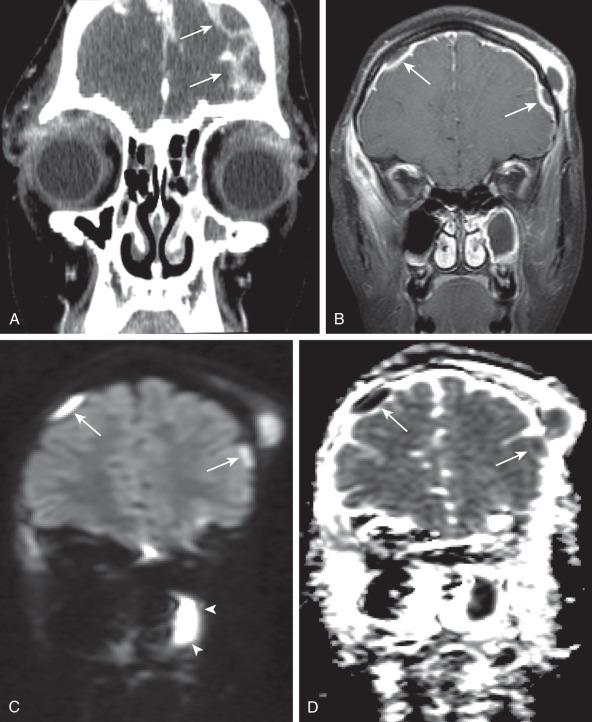
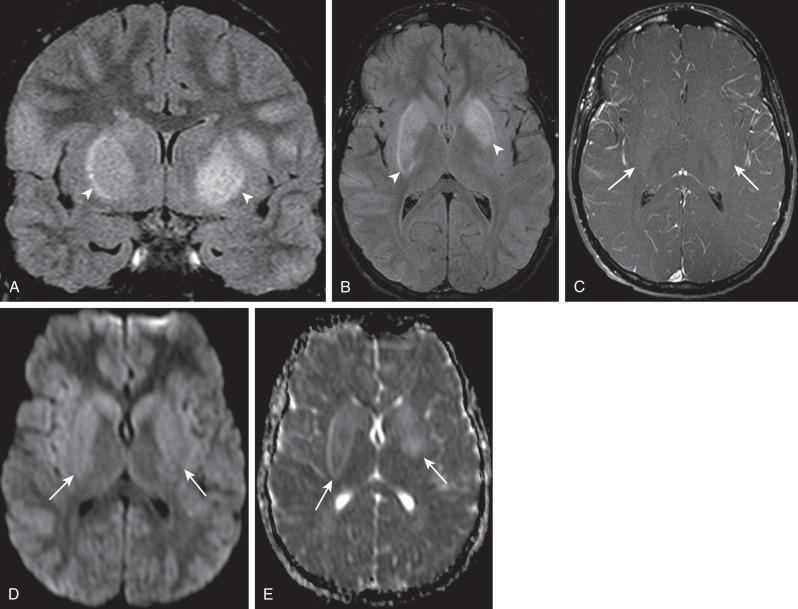
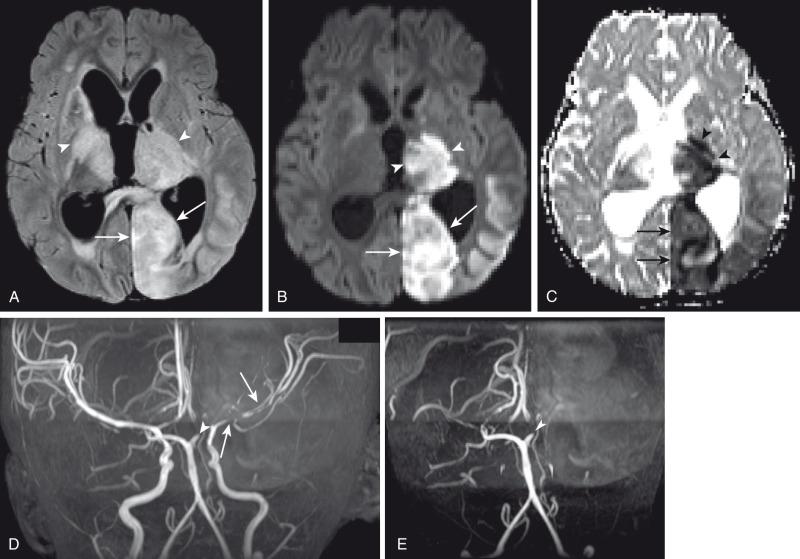
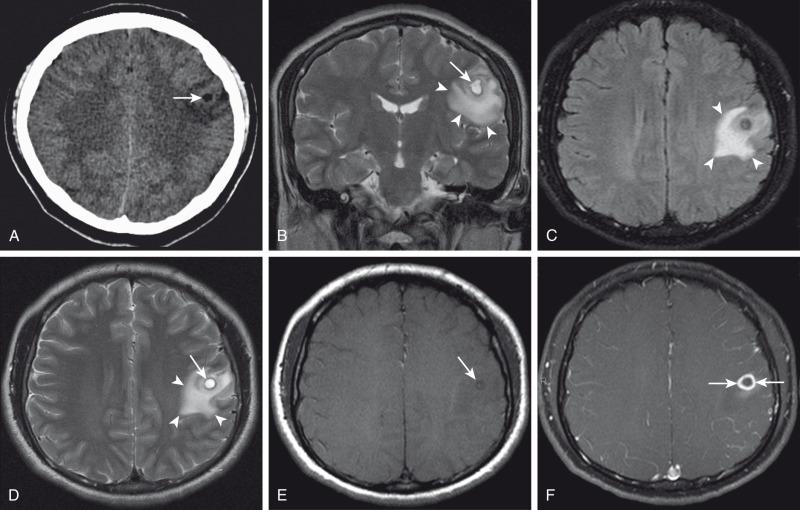
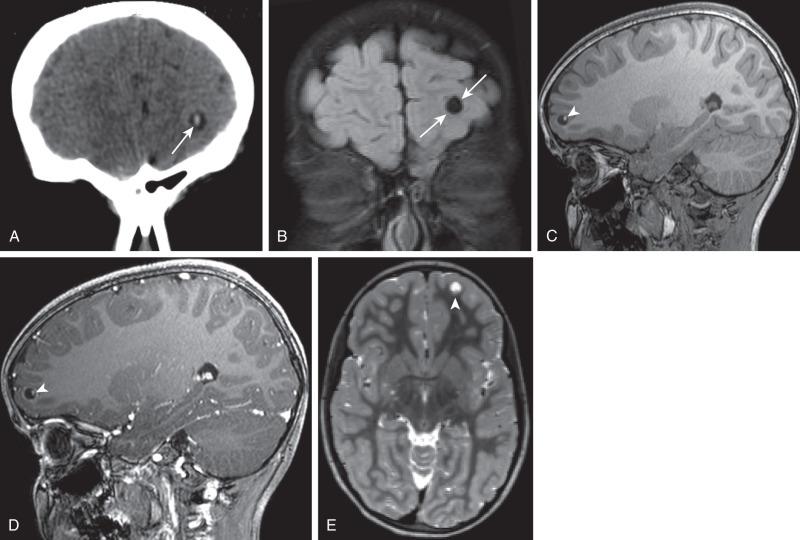
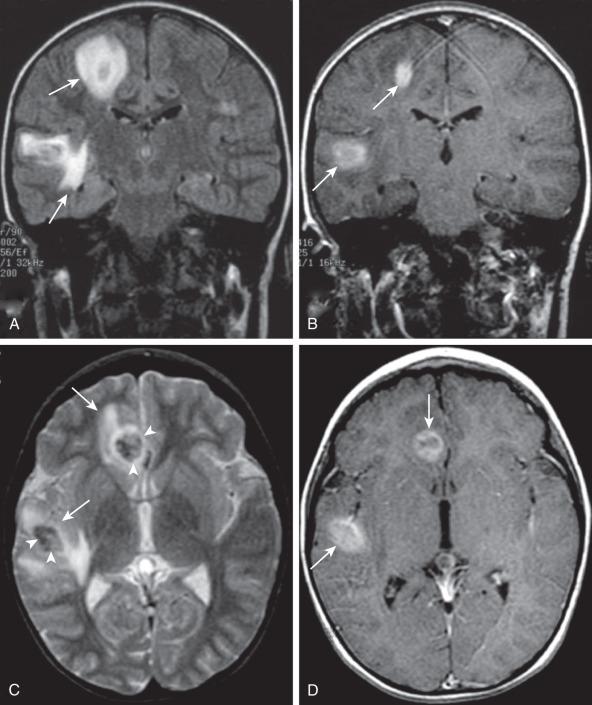
Hypoattenuation on CT, indicative of edema, and corresponding hyperintense T2-weighted and hypointense T1-weighted signal on MRI with patchy nonspecific postcontrast enhancement are typical imaging characteristics of cerebritis. Mild to moderate mass effect is often present. Sequential imaging is essential in the assessment of the response to antibiotics or progression to an abscess. Progression from cerebritis to an abscess generally takes 1 to 2 weeks but can progress more rapidly in the neonatal population. Citrobacter, Serratia, and Proteus are the most common causes of neonatal brain abscess. Citrobacter and Serratia infections may cause neonatal medullary vein thrombosis and associated hemorrhage ( Fig. 34.7 ). In general, abscesses tend to be situated at gray-white matter junctions in the region where the diameter of the end arterioles changes ( Fig. 34.8 ). Opportunistic organisms are common culprits in immunocompromised neonates. Differential considerations of a peripherally contrast-enhancing fluid-filled structure in the brain includes infectious abscess and tumor. DWI is often helpful when evaluating for an abscess, as lesions on DWI will appear hyperintense (and dark on ADC), indicating restricting material. On both MRI and CT, abscesses tend to have smooth regular inner margins and are often thinner-walled along their medial edge than along their lateral margins. Intraventricular rupture of an abscess portends a poor outcome. MRS of abscesses is notable for the presence of amino acids and lactate ( Fig. 34.9 ) and the absence of normal metabolite peaks. In neonates, ultrasound (US) may depict a hypoechoic abscess with peripheral hyperechogenicity, which at times may contain dependent echogenic debris.
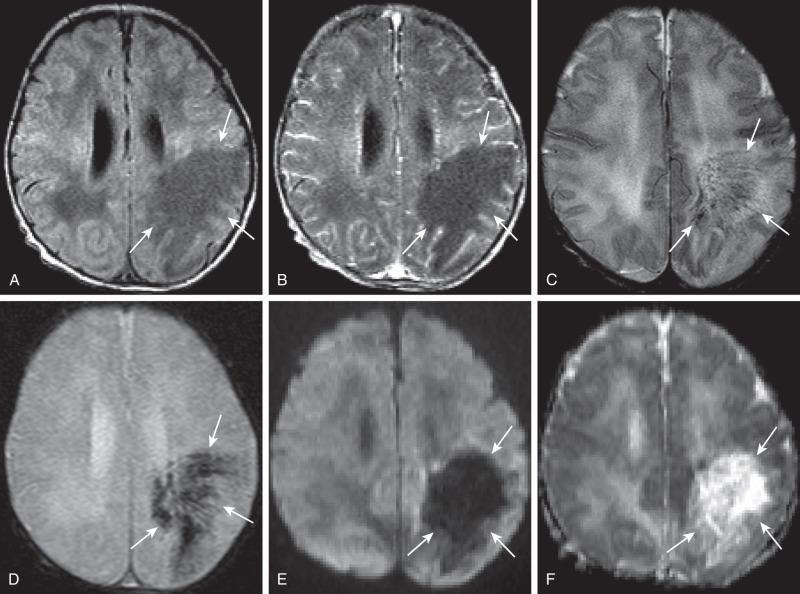
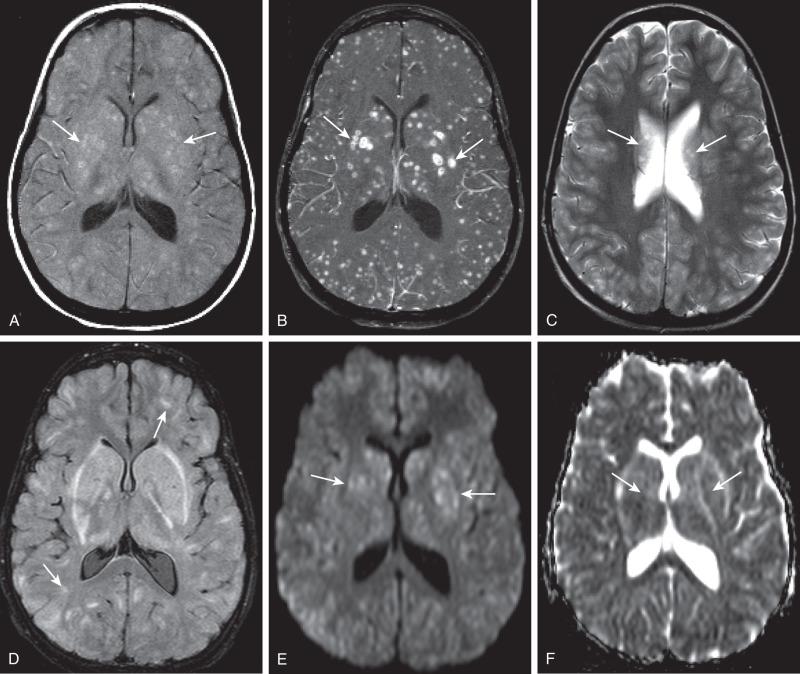
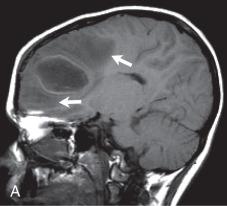
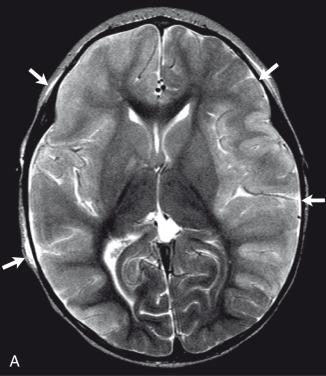
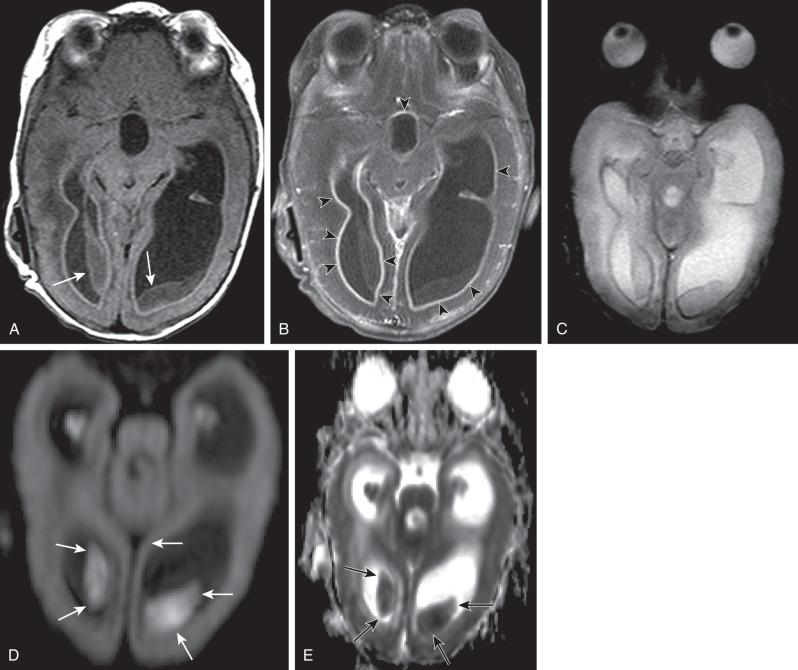
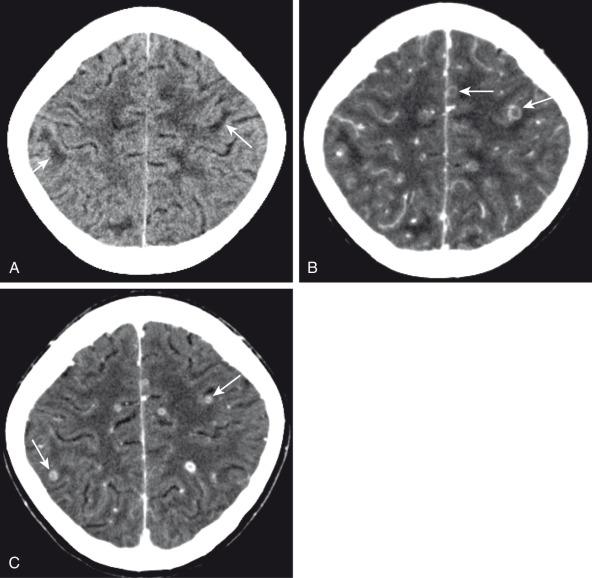
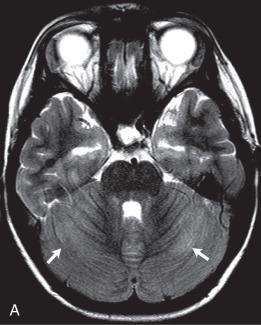
Bacterial meningitis is the most common form of pediatric CNS infection ( Fig. 34.10 ). Although not diagnosed by imaging, imaging is warranted if a diagnosis is unclear, persisting seizures are present, and/or symptoms persist despite treatment. It is more common in preterm infants as well as full-term infants within the first month of life. The subarachnoid space tends to resist infection in older normal children, making meningitis in this age group a rarity. Most cases of neonatal meningitis in the United States are caused by group B streptococcus (GBS) ( Fig. 34.11 ) and Escherichia coli . Other less common organisms (e.g., Serratia, enterococci, and Listeria ) tend to inflict more extensive destruction. In infants greater than 1 month of age, the most common causative organisms are Haemophilus influenza type b, Streptococcus pneumonia ( Fig. 34.12 ), Neisseria meningitides , and E. coli . Complications of meningitis include cerebritis, abscess, empyema, hydrocephalus, venous thrombosis, infarction (venous and/or arterial), ventriculitis ( e-Fig. 34.13 ), mycotic aneurysms ( e-Fig. 34.14 ), and sensorineural hearing loss ( Fig. 34.15 ).
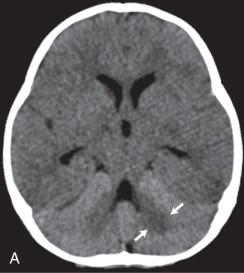
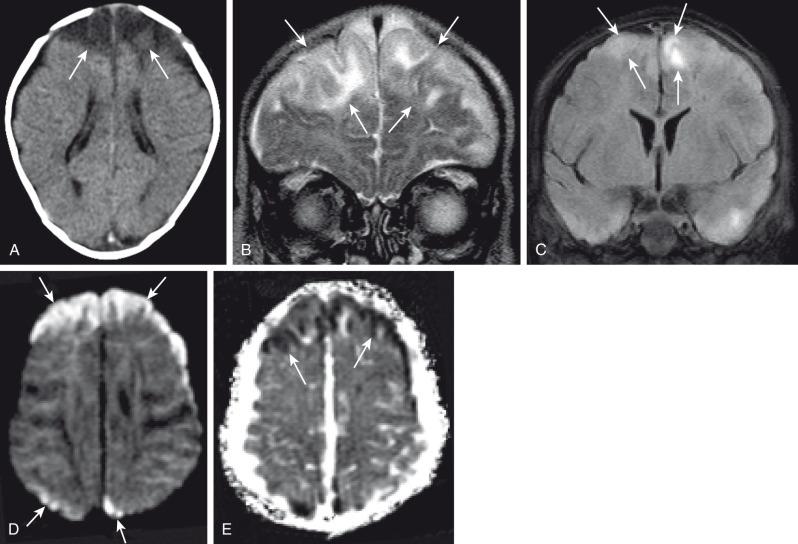
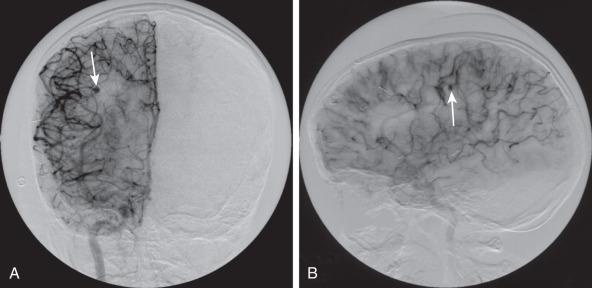
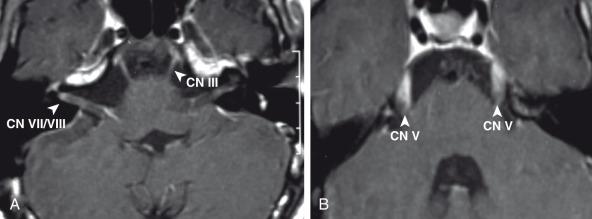
CNS infection with Mycobacterium tuberculosis (TB) differs clinically and radiographically from pyogenic infection. Other granulomatous and fungal organisms are rare in the pediatric age group and often present with similar imaging characteristics as TB. The pathogenesis of tuberculous meningitis is different from that of other bacterial meningitides. Hematogenous spread to the CNS may produce miliary tuberculosis (rare in children) but more typically produce tuberculomas in the meninges and/or brain at the gray-white junction, spinal cord, or rarely in the choroid plexus. After a variable period of time, organisms from one or more of these foci are discharged into the CSF/subarachnoid space and causing meningitis. Isolated meningeal tuberculomas tend to localize in the region of the Sylvian fissures, producing meningitis in the basal cisterns. This leads to secondary complications including cranial nerve palsies, infarction consequent to vasculitis, and hydrocephalus secondary to blockage of fourth ventricular outlet foramina.
Hematogenous dissemination of tuberculous bacilli is believed to occur about 1 week after the infected inhalation. Three stages of tuberculous meningitis have been described that progress over approximately a 3-week period. Clinical presentation ranges from personality change and anorexia to coma and death, with nuchal rigidity being the most common presenting clinical sign. Pathologically, tuberculous exudate fills the subarachnoid spaces. Most affected patients demonstrate hydrocephalus. On CT, exudate within the cisterns is manifested by attenuation greater than CSF. On contrast-enhanced MRI there is marked subarachnoid and cisternal enhancement on T1-weighted sequences ( Fig. 34.16 ), with T2-weighted sequences demonstrating obscuration of the cisterns due to inherent hyperintense signal within these affected regions. Microinfarctions are common. Tuberculomas often manifest at gray-white junctions and are more common above the tentorium cerebelli. On CT, they demonstrate high attenuation and ring enhancement ( Fig. 34.17 ), yet on MRI they demonstrate hyperintense T1-weighted and hypointense T2-weighted signal. When less than 2 cm, tuberculomas enhance uniformly, and when greater than 2 cm, they show more peripheral enhancement. The rare tuberculous abscess can be differentiated from a tuberculoma by its central hyperintense T2-weighted signal and more pronounced associated vasogenic edema. While DWI is not particularly helpful in making a diagnosis, lipid detection on MRS has been reported as helpful in certain cases.
Lyme disease is the most common tick-borne disease in the United States and Europe. More than one in five cases have been reported in children or adolescents. This multisystemic disorder involves the neurologic system in approximately one-fifth of affected children, manifesting as lymphocytic meningitis, meningoencephalitis, pseudotumor cerebri-like syndrome, or cranial neuropathy. It is unclear whether the mechanism of CNS involvement is a result of direct invasion by the Borrelia burgdorferi organism or due to an autoimmune phenomenon.
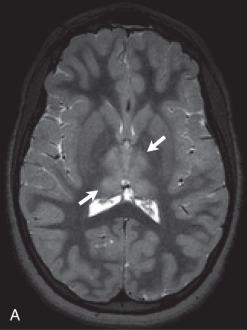
CT scans are usually normal in Lyme disease, although focal areas of hypoattenuation have been reported. Focal white matter lesions, which are hyperintense on T2-weighted images, are seen in about one in four neurologically affected patients. Contrast-enhanced MRI may show leptomeningeal enhancement, with or without enhancement of the affected cranial nerves, in children with cranial neuropathy ( Fig. 34.18 ).
In the United States, parasitic CNS infection in children is rare. While cysticercosis is not very common per se, it often presents in immigrants from endemic regions. The most common presenting symptoms of pediatric neurocysticercosis include seizures, developmental delay, and hydrocephalus. The most common form, parenchymal cysticercosis, is caused by inflammatory reaction secondary to the death of the parasite. Focal lesions are cystic and solid and present with peripheral enhancement and calcification, respectively. They are most often situated at the cortex and/or gray-white matter junction ( Fig. 34.19 ). Parenchymal lesions follow CSF signal intensity on DWI, which helps differentiate them from pyogenic abscesses. The typical calcification seen in adults is not always seen in children. Surrounding inflammatory reaction demonstrates hyperintense T2-weighted signal. Intraventricular cysterci often cause obstructive hydrocephalus. Thin-slice T1-weighted imaging best depicts the causative intraventricular scolex ( e-Fig. 34.20 ). The leptomeningeal form of cysticercosis mimics the radiographic appearance of TB, with marked postcontrast subarachnoid enhancement. Subarachnoid granulomata mimic their parenchymal counterparts. Hydrocephalus and vasculitis are common associated findings. Racemose cysts are large cysts that are most often found in the cerebellopontine angle, Sylvian fissures, as well as the basilar and suprasellar cisterns. Multiple coexisting forms of cysticercoids often suggest the diagnosis. On MRI, clusters of cysts are most commonly depicted.
Viruses affecting the brain ( e-Fig. 34.21 ) all produce inflammation and neuronal necrosis to a greater or lesser extent. Some viruses such as herpes simplex virus (HSV) and Coxsackieviruses may also produce white matter necrosis, whereas the primary inflammatory process involves the parenchyma. There is usually a meningeal inflammatory reaction or even ventriculitis. In general, acute viral infection typically causes marked edema ( e-Fig. 34.22 ). Hyperechogenicity on US, hypoattenuation on CT, as well as hyperintense signal on T2-weighted and FLAIR sequences are typical imaging findings of CNS viral infection. Restricted diffusion on DWI is the earliest sign of viral encephalitis. Fortunately differences in presentation and location help suggest one viral etiology over another. Viral-like syndromes with autoimmune associations, such as acute disseminated encephalomyelitis (ADEM) and posttransplantation lymphoproliferative disorders (PTLD) ( e-Fig. 34.23 ), often demonstrate imaging characteristics similar to viral infections.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here