Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Periprosthetic infection with a total hip arthroplasty occurs at a rate of approximately 0.5% to 1%; the prevalence will increase substantially as the volume for this procedure grows to meet the projected demand.
Prevention relies on optimizing patient selection and other host factors, improving the surgical suite environment, and administering prophylactic antibiotics.
The most common infecting organisms are Gram-positive cocci (most notably Staphylococcus species) but some infections may be polymicrobial.
Diagnosis is made by a high level of suspicion, a thorough history and physical examination, screening with an erythrocyte sedimentation rate and C-reactive protein, and selective use of hip joint aspiration if these screening laboratory tests are abnormal or if the clinical suspicion for infection is high. Synovial fluid obtained from an aspiration should be sent for a synovial fluid white blood cell count with differential and culture.
Successful treatment is predicated on the duration of the infection; it is reasonable to attempt debridement and component retention in acute postoperative and acute hematogenous infections (although staphylococcal infections seem to do worse with this strategy).
Chronic infections are treated most commonly using a 2-stage exchange protocol, although there is increasing interest in the use of a 1-stage exchange technique.
Despite the great success of total hip arthroplasty (THA), deep infection remains one of its most devastating complications. This diagnosis almost inevitably leads to prolonged and complex treatments involving reoperation and, ultimately, poorer outcomes. The cost to the patient is enormous with regard to the impact on both physical and mental health states. Treatments usually are costly and place significant burden on any health care system. The economic cost to society when accounting for direct and indirect costs averages approximately $400,000 for a single patient with a periprosthetic hip infection based on Markov models.
In a review of the Healthcare Cost and Utilization Project Nationwide Inpatient Sample, infection was the third most common reason (after instability and loosening) for revision surgery following THA and was by far the most common reason for removal of the prosthesis. Although prevention, diagnosis, and treatment have improved as surgical technique and patient management have evolved, the risk of developing this feared complication has not been eliminated. The unfortunate state is that as the number of THAs expected to be performed in the coming years significantly increases, so too will the prevalence of deep infection.
There are innumerable factors in infinite permutations that may contribute to or directly result in deep infection following THA. To simplify and thus best address this complex clinical scenario, these factors can be organized into 3 groups: host, local wound environment, and microbiome. By investigating and addressing how these entities contribute singularly and interact with one another, a better understanding of the disease process and its treatment will emerge.
The incidence of deep infection following THA in the early experience of Charnley was 9%; fortunately, this was reduced to 1.3% in his next cohort of patients. The reduction was attributed to the routine use of prophylactic antibiotics, air cleanliness, and body exhaust attire. Although this dramatic reduction was realized over the course of the initial 10 years of his experience into the late 1960s, the acknowledged rate of infection is currently approximately 1% at large-volume centers. This relatively small but important improvement in incidence can be attributed to many advances, including better patient selection, the use of prophylactic antibiotics, and operating room measures.
Improved patient selection has been predicated on the identification of host risk factors, both systemic and local, predisposing to infection following THA. Although the physiologic stress of surgery itself may predispose to infection, multiple factors should be recognized and, if possible, addressed to reduce risk. Malnutrition and advanced age are independent factors known to affect innate immunity and normal immune response by altering both humeral and cell-mediated immunity, thus increasing infection risk. Systemically immunocompromised patients, because of underlying disease or pharmacologic treatment, are at increased risk for infection. Patients with rheumatoid arthritis and those being treated with steroid therapy are at increased risk for infection. Obesity increases the odds of infection 4.2 times with super-obese patients (body mass index [BMI] > 50 kg/m 2 ) having the highest risks. Patients with diabetes mellitus appear to experience higher rates of infection compared with nondiabetic patients, with some studies suggesting an astonishingly high rate of nearly 11% in matched groups. In patients with diabetes mellitus, preoperative glycemic control appears to reduce the risk of infection. While no strict cutoff has been established for hemoglobin A1C, patients with hemoglobin A1C values below 7.5 mg/dL appear to have lower rates of deep infection. Improved perioperative glycemic control appears to reduce the risk of infection in thoracic surgery. Although definitive prospective studies have not been completed yet, suggestions have been put forth that strict glycemic control in the perioperative period may reduce the rate of infection in THA patients. Likewise, metabolic syndrome (the combination of obesity, diabetes, and hypercholesterolemia) also appears to be a significant risk factor for complications after THA. Chronic renal failure patients on dialysis and those with liver or kidney transplants are at increased risk for infection. Patients with malignancy and those with human immunodeficiency virus, particularly patients with CD4 counts below 240 cells/mm 3 , are also at increased risk.
Local host factors that appear to increase the risk of infection include previous infection of the native hip that has not been quiescent for longer than 10 years and revision hip arthroplasty for any cause of failure. Delayed wound healing and persistent wound drainage often lead to infection. Remote sources of infection, including skin lesions, dental caries or procedures, and urinary retention with subsequent bladder instrumentation can lead to infection at the site of THA. Systemic predispositions should be optimally minimized and potential local and remote sources of infection should be addressed before surgery.
Administration of an appropriate prophylactic antibiotic is considered the single most important factor in reducing postoperative wound infection. The choice of a first-generation cephalosporin is often made on the basis that this agent is bactericidal, therapeutic against commonly infecting pathogens, and inexpensive while possessing an appropriate half-life. Antibiotic prophylaxis with cefazolin before and after surgery was demonstrated to reduce the risk of infection after THA compared with placebo from 3.3% to 0.9% in a double-blind controlled study. For patients with hypersensitivity to penicillin, clindamycin or vancomycin should be considered as the alternative. The preoperative dose of prophylactic antibiotic appears to be most effective in reducing infection when administered within 2 hours preceding the incision, and peak bone concentrations are reached within 35 to 40 minutes after intravenous administration. A multicenter prospective trial observed that when the preoperative antibiotic was given within 30 minutes before incision, the rate of surgical site incision was 1.6%, compared with 2.4% when the antibiotic was given between 31 and 60 minutes. In addition, this trial found that intraoperative re-dosing in cases lasting longer than 4 hours appears to reduce the risk of infection. The postoperative duration of prophylactic treatment is debatable; a prospective double-blind multicenter trial found no difference in treatment for 24 hours versus 3 days. Multiple other studies have confirmed that prolonged prophylactic treatment does not appear to be of benefit.
Modifications of the surgical suite environment, including limiting operating room personnel or “traffic,” surgical time, and the use of iodophor-incorporated drapes, have been shown to reduce the risk of infection. Use of ultraclean-air operating rooms, vertical laminar flow, and body exhaust suits have been demonstrated to favorably affect the rate of infection; however, these points have been debated. Gore-Tex gowns may be more appropriate than cotton in preventing dissemination of shed bacteria by the surgical team. The use of double gloves and the choice of the optimal antiseptic remain controversial. Frequent changing of the suction tip may also decrease the introduction of bacteria into the wound. Pulsatile lavage and antibiotic irrigation solutions are effective in reducing the wound bacterial load. Further, a study by Brown et al. showed a significant reduction in the rate of deep infection when a dilute Betadine lavage was performed prior to wound closure.
Although rates of bacteremia following oral procedures and diagnostic procedures of the genitourinary and gastrointestinal tracts have been documented, and even though bacteremia may lead to bacterial introduction to the hip arthroplasty, antibiotic prophylaxis given before these procedures remains controversial. The most recent statement by the American Academy of Orthopaedic Surgeons and the American Dental Association has reversed their previous position on standard antibiotic prophylaxis, now recommending against routine prophylaxis.
A thorough understanding of the microbiologic environment that leads to infection after THA is crucial to successful prevention and treatment. The introduction of a biomaterial into the body has been likened to a race to inhabit the surface of the material between host tissue and bacterial pathogens. Should the bacteria win this race, the result is often deep infection of the prosthesis. Introduction of bacteria to the prosthetic interface may occur at the time of surgery through direct inoculation, or it may occur at a later time through hematogenous seeding. Certain bacteria may have a predilection for seeding different biomaterials. For example, Staphylococcus aureus seems to preferentially seed metallic implants and Staphylococcus epidermidis shows a preference toward polymers such as polyethylene and polymethylmethacrylate. Many bacteria form a mucopolysaccharide biofilm that isolates and protects organisms from the host immune response, antibiotic penetration, and even mechanical debridement. Even though approximately 75% of cultures in a single study revealed a single organism, retrieval and microbiologic analysis have demonstrated that the bacterial glycocalyx may harbor multiple different bacterial species. This makes treatment of such patients difficult without removal of the prosthesis because the appropriate antibiotic regimen cannot be determined. Recent investigation into methods of better diagnosis through molecular diagnostic techniques, as well as more effective antibiotic biofilm penetration, has demonstrated that rifampin appears to improve treatment protocols when used synergistically with other antibiotic agents.
In multiple large studies, the most common offending organisms are Gram-positive aerobes, ranging between 64% and 74% of all infecting organisms following THA, with the Staphylococcus species (mainly S. epidermidis and S. aureus ) representing approximately half of these Gram-positive cocci. Other identified Gram-positive organisms include Enterococcus , Streptococcus viridans , and Streptococcus groups A, B, and G. There may exist a trend toward an increasing prevalence of cultures positive for S. epidermidis and a decrease in Gram-negative bacteria. Gram-negative organisms known to cause periprosthetic hip infection range from 11% to 14% of all isolated organisms and include Pseudomonas aeruginosa , Enterobacter cloacae , Serratia marcescens , Proteus mirabilis , Escherichia coli , Klebsiella pneumoniae , Acinetobacter species, Moraxella nonliquefaciens , and Salmonella choleraesuis. Other more rare organisms that have been reported include anaerobes, mycobacteria, and fungi.
A trend that has generated extreme concern has been the emergence of antibiotic-resistant bacterial strains. Over a 5-year period, nosocomial infection rates in intensive care unit (ICU) patients had increased by 31% for methicillin-resistant S. aureus and by 55% for vancomycin-resistant Enterococcus . Fortunately, these alarming trends have not been observed to this same magnitude in periprosthetic infection of the hip, but they do exist. The rate of methicillin-resistant S. epidermidis in hip replacement patients was observed to be as high as 40% of 30 positive cultures and 48% of 56 S. epidermidis cultures. In a single study of 35 infected cases following THA, 54% were infected with a resistant organism, and results of treatment for these resistant organisms were far inferior to that given for sensitive organisms, with successful retention of the prosthesis in only 16%. In a second study comparing the treatment of methicillin-resistant and methicillin-sensitive S. aureus joint infections, patients with methicillin-resistant organisms had significantly longer hospital stays and higher risks of treatment failure and removal of the prosthesis. This further emphasizes the critical need for accurate diagnosis and identification of the infecting species and its sensitivities in providing optimal care for these patients.
The diagnosis of infection following THA requires a high level of suspicion and clinical acumen. These findings should be supplemented with serologic testing, radiographic evaluation (conventional and occasionally advanced imaging), synovial fluid analysis, microbiologic cultures, and/or histopathology. The diagnosis of periprosthetic hip infection should be based on the criteria outlined by the Musculoskeletal Infection Society. These criteria included either 1 of the following 2—sinus tract communicating with the joint or 2 identical positive cultures from 2 different specimen—or 4 of the following 6—elevated erythrocyte sedimentation rate (ESR)/C-reactive protein (CRP), elevated synovial white blood cell count (threshold 3000 WBC), elevated percentage of synovial neutrophils (threshold 80%), single positive culture, positive histology, or gross purulence. The diagnosis should be established expeditiously because the treatment algorithm may shift depending on the chronicity of the infection.
A thorough history should identify any risk factors for infection, including immunocompromised states, history of prior hip surgery, or history of infection. Persistent wound drainage postoperatively or superficial wound infection raises concern for persistent deep infection. Any abnormalities in the recovery period, including persistent postoperative or even new-onset deep pain, are causes of concern for infection. A history of fever can indicate infection; however, this finding can be unreliable. Any recent procedures that may have caused bacteremia or remote infection should be investigated.
The range of motion of the hip should be determined and compared with previous examination findings. Guarding, pain with passive motion, pain at rest, and diminishing motion are matters of concern regarding infection. A thorough neurovascular examination and examination of the skin for any remote lesions should be performed. The wound should be carefully examined for drainage, erythema, fluctuance, or an active or healed sinus tract. Wound drainage should not be cultured because these results are highly inconsistent and may only confuse the clinical picture. Empirical antibiotics for wound drainage should be avoided because they suppress and delay the definitive diagnosis.
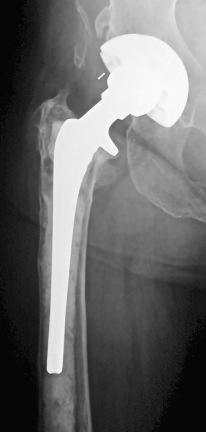
Plain radiographs should be evaluated and compared with prior studies for the presence of rapid or progressive loosening, osteolysis, periosteal or endosteal reaction, and osteopenia. These findings, however, may have other causes. Implant loosening or osteolysis within the first 2 years is particularly suspicious for infection ( Fig. 105.1 ). Plain radiographs can be helpful should any of these findings be observed, but the lack of radiographic changes does not exclude infection.
The serum CRP level and ESR should be the initial screening laboratory tests when infection is suspected. In a recent study of preoperative testing for more than 200 revision THAs, 100% specificity for a hip not to be infected was noted when both CRP and ESR were normal. An elevated ESR (> 30 mm/h) was shown to have a sensitivity of 97%, a specificity of 39%, a positive predictive value of 42%, and a negative predictive value of 96%. A CRP value greater than 10 mg/L was shown to have a sensitivity of 94%, a specificity of 71%, a positive predictive value of 59%, and a negative predictive value of 96%. These values are consistent with the findings of previous studies, which also showed that when both the ESR and CRP are elevated, the probability of infection was 83%.
If the ESR and CRP are elevated, or if the clinical suspicion for infection is high based on the history and physical examination, aspiration of the hip is recommended and the fluid obtained is sent for a synovial fluid WBC count, differential, and culture. Patients should be off of antibiotics for a minimum of 2 weeks before aspiration to maximize culture yield and accuracy. Table 105.1 provides recommended diagnostic criteria from several previously reported studies of periprosthetic infection of the hip and knee using ESR, CRP, synovial WBC, and differential. Although these synovial tests alone may provide valuable information, the combination of these test results with ESR and CRP results can greatly improve diagnostic accuracy. Bacterial growth on solid media and gross purulence visualized within the joint are generally considered diagnostic for infection, although the clinician should be aware that cultures (even on solid media) can be falsely positive, and alternative causes may be proposed for purulent-appearing fluid around a prosthetic hip. These include wear of a polyethylene or metal-on-metal bearing surface and implant corrosion; thus, the results of multiple tests should be analyzed together to allow the most accurate conclusion to be drawn.
| Authors | WBCs/µL | PMN, % | CRP, mg/L | ESR, mm/h | Population | Time from Index Surgery, y (mean, range) |
|---|---|---|---|---|---|---|
| Spangehl et al. | 50,000 a | 80 a | 10 a | 30 a | 202 hips | Mentioned anecdotally, up to 11 y |
| (35 infected) | ||||||
| Mason et al. | 2500 cells/mL b | 60 | — | — | 86 knees | Not mentioned |
| (36 infected) | ||||||
| Trampuz et al. | 1700 | 65 | — | — | 133 knees | > 6 mo |
| (34 infected) | ||||||
| Parvizi et al. | 1760 | 73 | — | — | 145 knees | Not mentioned |
| (78 infected); 23 hips | ||||||
| (16 infected) | ||||||
| Trampuz et al. | 1700 a | 65 a | 10 a | 30 a | 331 joints | Not mentioned |
| (207 knees, 124 hips; | ||||||
| 79 infected) | ||||||
| Della Valle et al. | 3000 cells/mL b | 65 a | 10 a | 30 a | 94 knees | Not mentioned |
| (41 infected) | ||||||
| Nilsdotter-Augustinsson et al. | 1700 a | — | 10 a | 30 a | 85 knees | Uninfected: 9 (1–22) Infected: 3 (0.2–16) |
| (25 infected) | ||||||
| Ghanem et al. | 1100 | 64 | 10 a | 30 a | 429 knees (161 infected) | 1.2 (0.1–7.8) |
| Schinsky et al. | 4200 cells/mL b | 80 | 10 a | 30 a | 201 hips | Uninfected: 8 Infected: 4.5 (including 7 < 6 wk) |
| (55 infected) | ||||||
| Parvizi et al. | 1100 | 64 | 10 a | 30 a | 296 knees (116 infected) | Not mentioned |
| Ghanem et al. | — | — | 20.5 | 31 | 479 hips | Not mentioned |
| (127 infected) |
a Values not based on independent receiver operating characteristic (ROC) analysis performed for the purpose of the study in question but rather on thresholds set by prior studies.
b Units given in these cases are as reported in source literature.
Because serologic markers of inflammation (ESR, CRP), synovial WBC count, and differential are normally elevated in the early postoperative period, conventional wisdom has been that these tests may be of debatable utility in the first few weeks after surgery. Recent work by Yi and associates has revealed in a study of more than 6000 primary THAs that these markers of inflammation can be used reliably in the diagnosis of periprosthetic infection of the hip in the early postoperative period (< 6 weeks from surgery) but at threshold levels higher than those previously reported. Specifically, the ESR was not found to be useful in the early postoperative period, but the CRP level was useful, with an optimal cutoff value of 93 mg/L. The synovial fluid WBC count was the best test for diagnosing acute postoperative infections, with a threshold of less than 12,800 WBCs/µL being the best cutoff. The differential was also helpful, with an optimal balance of sensitivity and sensitivity at greater than 89% PMNs ( Table 105.2 ). These numbers are similar to those reported by Bedair et al. for diagnosing infection in the early postoperative period following total knee arthroplasty. Based on these 2 works, our present algorithm for evaluating infection in the early postoperative period includes a low threshold for getting a serum CRP if there is any question of an infection early after surgery. If the serum CRP is near or above 100 mg/L (normal, 10 mg/L) we aspirate the hip joint. If the synovial fluid WBC count is less than 10,000 and the differential is less than 90%, infection is unlikely. If we are still unsure, antibiotics are administered and the cultures are monitored closely.
| Authors | WBCs/µL | PMNs, % | CRP, mg/L | ESR, mm/h | Population | Time From Index Surgery |
|---|---|---|---|---|---|---|
| Bedair et al. | 27,800 | 89 | 95 | — | 146 knees | < 6 wk |
| (19 infected) | ||||||
| Yi et al. | 12,800 | 89 | 93 | — | 73 hips | < 6 wk |
| (36 infected) |
The use of biomarkers, most notably alpha defensin, has demonstrated promising results in the diagnosis of prosthetic joint infection (PJI). By combining tests for elevated synovial alpha-defensin levels and an elevated synovial CRP (commercially marketed as Synovasure), the diagnostic accuracy of this combination has been reported to be as high as 97% sensitive and 100% specific for PJI in 1 report. While some use this test routinely, we have found it most useful in cases in which the results of the synovial fluid WBC count, differential, and culture are unclear or discordant. The most common scenario that we see is an elevated synovial fluid WBC count and differential with negative cultures. In this scenario, Synovasure testing is obtained at the time of repeat aspiration to confirm the diagnosis and the presence of a culture-negative infection. When we are faced with a potential culture-negative infection, we also inoculate any synovial fluid obtained directly into blood culture bottles, which may increase the yield of cultures, obtain cultures for acid-fast bacilli and fungus (which we do not normally obtain) and hold the cultures for a full 14 days (normally, we hold them for just 72 hours).
Nuclear medicine tests may aid in the diagnosis of periprosthetic infection. A technetium-99 radioisotope scan may have some value, with a negative test most probably ruling out infection; however, a positive test cannot differentiate septic from aseptic failure. The use of an indium-111 leukocyte scan, in which host leukocytes are tagged with the radioisotope and reintroduced into the body, has shown some utility, namely, in its negative predictive value of 95%. The differential use of technetium and indium scans may distinguish areas of enhanced metabolic activity and inflammation. When a technetium-99 sulfur colloid marrow scan is combined with an indium scan, accuracy improves to 88%. However, routine use of these studies is debatable, given their high cost and the relative accuracy of synovial fluid WBC count, differential, and culture. Newer nuclear medicine techniques, such as technetium-99 immunoglobulin scintigraphy and 18F positron emission tomography, may provide improved diagnostic accuracy. These techniques are not currently in wide use, however.
Unfortunately, there are many clinical scenarios in which preoperative testing is unable to definitively diagnose or rule out periprosthetic infection. If the clinical picture continues to cause concern for infection, analysis of fluid and tissue around the prosthesis obtained at the time of reoperation is indicated. Gram staining of periprosthetic fluid not only has poor sensitivity but may also generate false-positive results. It should not be done routinely and certainly should not be relied on as the only method of screening for periprosthetic joint infection.
Many authors have investigated the role of histologic analysis of periprosthetic tissue in the diagnosis of infection. In these studies, an attempt was made to correlate frozen sections from the time of revision surgery with the diagnosis of infection. Although specific cutoff values are debated—likely secondary to inconsistencies in sampling, technique, and pathologists—the essence of the findings is that the increased number of neutrophils observed in the tissue surrounding the prosthesis appears to correlate with infection. In an attempt to synthesize this variability, some authors have recommended using the cutoff of at least 5 neutrophils in each of three 400× high-power microscopic fields to diagnose infection.
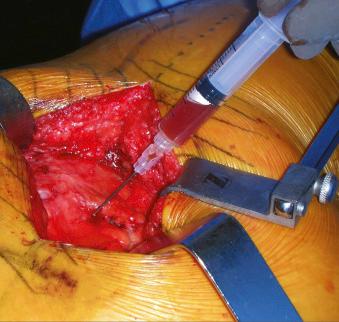
Any test for infection will have a given sensitivity and specificity that will not perfectly predict or rule out the presence of infection around a THA, even those tests considered to be gold standards. Based on Bayesian theory, the predictive values of tests are related to the prevalence of the disease in the given patient population. The burden thus lies on the treating physician to acquire the relevant data and apply them to a specific patient; the utility of any test will vary based on the specific clinical scenario. We recommend an initial thorough history and physical examination paired with ESR and CRP level in all patients undergoing revision surgery or who present with a painful or otherwise failed THA. Should the clinical suspicion be high based on the history and physical examination or if the ESR and CRP level are elevated, the hip joint should be aspirated and the fluid obtained sent for a synovial fluid WBC count with differential and culture. These results, taken together, should be helpful in establishing the diagnosis in the vast majority of patients. If reoperation is indicated, or if the diagnosis remains uncertain, infection can be ruled in or out through additional intraoperative testing, including intraoperative aspiration of the joint for synovial fluid WBC count and differential, an intraoperative frozen section, and culture. Given that operative cultures can be both falsely positive and negative, we recommend obtaining multiple cultures (typically, 3–5 sets) from several different sites (including the most suspicious-appearing areas) at the time of revision. To perform an aspiration intraoperatively, the capsule is exposed and directly visualized as per the surgeon's normal surgical approach, and an 18-gauge spinal needle is used to obtain synovial fluid through the capsule ( Fig. 105.2 ). A result for the synovial fluid WBC count is typically available within 30 minutes; the differential can take up to 45 minutes to obtain. Advantages of this test include its objective nature, low cost, and availability to surgeons worldwide.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here