Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Instability is one of the most common indications for revision total hip arthroplasty in the United States.
The cost burden for treating dislocation is substantial.
Multiple risk factors for instability are known, including patient-specific (female gender, increasing age, lumbar spine disease, underlying diagnosis of avascular necrosis [AVN] or femoral neck fracture) and surgeon-specific factors (surgical approach, component orientation, head size, proper restoration of leg lengths and offset, and surgical volume).
Proper treatment of instability requires an understanding of the timing and cause of the dislocation coupled with a thorough physical examination and radiographic evaluation.
Treatment options include closed reduction, component revision, modular component exchange, increasing femoral head size, soft tissue augmentation, trochanteric advancement, conversion to a dual-mobility or tripolar arthroplasty, and use of a constrained liner.
Total hip arthroplasty (THA) is an exceptionally cost-effective and successful medical intervention. Other than implant loosening, dislocation, infection, and osteolysis secondary to wear of the bearing surface are the 3 most common complications affecting the long-term success of THA. Dislocation rates ranging from 0.3% to 10% of primary THAs and up to 28% for revision THAs have been reported. The incidence of dislocation appears to be highest within the first year after the index procedure but rises cumulatively with time. For Charnley THA with a 22-mm diameter femoral head and a conventional polyethylene bearing, the cumulative instability rate was found to rise about 1% per 5 years to 7% at 25 years postoperatively. Population-based estimates have predicted a substantial increase in the future number of primary and revision THA procedures performed in the United States over the next few decades. Despite advances in surgical technique and implant technology, the revision rate for THA has not changed substantially with time. In addition, a recent study evaluating a national database demonstrated that instability/dislocation was one of the most common diagnoses resulting in revision THA in the United States, representing 17.3% of revision THA cases.
The cost burden for treating dislocations is substantial. Estimates place the hospital costs of treating dislocations following THA at $74,000,000 per year. Investigators have reported that dislocations treated by closed methods represent a hospital cost of 19% of the overall cost of an uncomplicated primary THA, and revision surgery results in hospital costs of 148% of an uncomplicated primary THA. Operative treatment of dislocations has been reported to increase the hospital cost 342% after primary THA and 352% after revision THA. These direct costs do not account for patient morbidity or lost work production. This cost burden is expected to increase as the numbers of primary THA and subsequent revision cases for instability increase over the next few decades.
Multiple factors are correlated with the risk of instability after THA. Patient-specific risk factors include female gender, increasing age, underlying diagnosis (including avascular necrosis [AVN] and femoral neck fracture), combined preoperative hip range of motion (ROM) greater than 115 degrees, and associated comorbidities. Surgeon- and prosthesis-specific variables include surgical approach, component orientation, femoral head size and prosthetic neck geometry, offset, soft tissue integrity, leg length, and impingement and surgeon volume, all of which contribute to the risk of dislocation.
Various authors have investigated how gender influences hip stability. Berry and associates, in a study of more than 6000 THAs, reported a 25-year cumulative risk of dislocation of 8.9% for females compared with 4.5% for males, resulting in a relative risk of dislocation of 2.1 for women. It is hypothesized that greater tissue laxity among females contributes to this reported risk. However, other authors have reported that gender does not play a role in instability risk. In a recent multicenter study, Fessy et al. did not find that gender was a significant risk for dislocation following primary THA. Furthermore, Meek and colleagues reviewed more than 14,000 THAs from the Scottish National Arthroplasty Registry, Conroy and coworkers evaluated more than 65,000 THAs from the Australian Orthopaedic Association National Joint Replacement Registry, and Khatod and associates reported on approximately 2000 THAs from a US community registry (Kaiser Permanente Total Joint Registry). None of these authors found gender to be an independent risk factor for dislocation.
Patient age has also been investigated in relation to hip stability. Berry and colleagues demonstrated a relative risk of dislocation of 1.3 for patients older than 70 years. Additional authors have demonstrated a 2-fold to 3-fold increased risk of dislocation in the elderly, specifically patients older than 80 years. Levy and coworkers explained that predisposing factors in this elderly population that potentially increased their risk of dislocation included cognitive decline, muscle weakness, impaired proprioception, and laxity of the capsular structures. Cognitive decline has been shown to have an odds ratio of 3.9 (95% CI, 2.15–7.1) for dislocation after primary THA. In contrast, other authors could not find a correlation between increasing age and dislocation rate in 2 large registry databases. In addition, a 3.5% dislocation rate was reported by Lachiewicz and associates in a single surgeon review of patients older than 75 years—a dislocation rate that is relatively on par with other reports in the literature, irrespective of age.
AVN of the femoral head, acute fracture or nonunion of the proximal part of the femur, inflammatory arthritis, and previous hip surgery are generally agreed upon risk factors for dislocation. Berry and associates reported that AVN, acute fracture or nonunion of the proximal femur, and inflammatory arthritis had relative rates of dislocation of 1.9, 1.8, and 1.5, respectively, compared with an underlying diagnosis of osteoarthritis, in a study of more than 6000 THAs. In the same report, Berry and colleagues did not find a significantly higher dislocation rate with developmental dysplasia of the hip or posttraumatic arthritis when compared with osteoarthritis. Khatod and coworkers reported a 6-fold increased dislocation risk with the underlying diagnosis of rheumatoid arthritis, and Meek and associates reported it as a strong independent risk factor for instability. Zwartele and colleagues described rheumatoid arthritis as an independent risk factor and hypothesized that it resulted from overall poorer soft tissue quality. Lewinnek and coworkers noted a significantly increased dislocation risk in patients with previous hip surgery and suggested the inferior soft tissue envelope as a potential causative factor. The soft tissue insult could also explain the higher dislocation rates reported by various authors for acute femoral neck fracture after total hip arthroplasty. In addition, the higher combined preoperative ROM in the nonarthritic hip fracture cohort could contribute to the higher rate of dislocation.
Body mass index (BMI) and medical comorbidities have been studied to evaluate their relationship to instability following THA. Jolles and associates reported a 10-fold increased risk of dislocation with an American Society of Anesthesiologists (ASA) score of 3 or 4. Khatod and colleagues found a 2.3-fold dislocation risk for patients who had ASA scores of 3 or 4 compared with patients with scores of 1 or 2. Khatod and coworkers did not find BMI to be a risk factor for dislocation ; this finding is in accordance with most reports in the literature. However, a recent study of 2100 primary THAs in male patients from a Swedish registry determined that BMI was a risk factor for dislocation. In addition, Kim and associates reported a six times higher dislocation risk in the obese patient population in a study of revision THA.
Recently, the impact of lumbar spine disease on outcomes after THA has been a topic of interest. Degenerative lumbar spine conditions and lumbar fusions are known to decrease lumbar lordosis and limit lumbar flexion. This leads to an alteration in pelvic mechanics with increased pelvic tilt, acetabular anteversion and retroversion of the pelvis. These alterations lead to a positive sagittal balance, which requires hip hyperextension to stand upright. Furthermore, patients with spinal disease or spinal fusion have limited ability to increase pelvic retroversion in a seated position, leading to decreased posterior femoral head coverage in a seated position and increased risk for posterior dislocation. Lumbar disease has been shown to have a relative risk of 1.76 to 2.0 (95% CI, 1.63–2.20) for dislocation after THA. In patients with previous lumbar fusion, Perfetti et al. found a 7.19 times increased risk for dislocation after THA. In a Medicare database analysis of over 800,000 patients without spinal fusion and over 14,000 patients with spinal fusion, Buckland et al. found a dislocation rate of 1.55% in the control group while patients with 1- to 2-level fusions had a dislocation rate of 2.96% and those with 3 to 7 levels of fusion had a dislocation rate of 4.12%.
Surgical approach has been described as an additional factor affecting stability and risk of dislocation following THA. Traditionally, the posterior approach has been associated with the highest risk of dislocation compared with transtrochanteric, anterolateral, and anterior-based approaches. In a study of more than 21,000 primary THAs, Berry and colleagues reported dislocation rates at 10-year follow-up of 3.1%, 3.4%, and 6.9% for anterolateral, transtrochanteric, and posterolateral approaches, respectively. Masonis and Bourne performed a meta-analysis of the literature and found the posterior approach to have a 6-fold higher dislocation rate compared with a direct lateral approach. However, multiple authors have reported that dislocation rates with the posterior approach can be reduced by repair of the capsule and short external rotators. Furthermore, Kim and coworkers advocated preserving the external rotators during the posterior approach—a technique that resulted in zero dislocation in one study. The direct anterior approach has become increasingly popular with purported benefits of faster recovery, more precise acetabular orientation, accurate assessment of limb length, and lower dislocation rates. In a national registry analysis, Sheth et al. found an adjusted hazard ratio of 0.44 (95% CI, 0.22–0.87) for dislocation with the direct anterior approach compared with the posterior approach. However, other studies have shown similar dislocation rates between the direct anterior and posterior approaches.
Implant positioning is paramount in optimizing the result of THA and minimizing dislocation risk. Lewinnek and associates defined the “safe zone” of acetabular cup placement as anteversion of 15 ± 10 degrees and abduction of 40 ± 10 degrees, which resulted in a dislocation rate using the posterior approach of 1.5%. Comparatively, when the cup was placed outside of this safe zone, the dislocation rate was 6.1%. McCollum and Gray demonstrated similar dislocation rates using a posterior approach when cup position was maintained within a safe zone of 30 to 50 degrees abduction and 20 to 40 degrees anteversion. However, the accuracy of intraoperative assessment of cup positioning has been shown to be poorer than expected, with 21 of 50 cups unexpectedly falling outside the safe zone when examined radiographically in the postoperative period in one study. In contrast, multiple authors have described intraoperative techniques that focus on specific landmarks to accurately reproduce acetabular cup position and minimize dislocation risk. Archbold and associates described using the transverse acetabular ligament to determine cup anteversion, which enabled them to achieve a dislocation rate of 0.6% in 1000 hips treated with a posterior approach. Sotereanos and colleagues advocated using the bony palpable landmarks of the ischium, superior pubic ramus, and superior acetabulum for cup placement and demonstrated both reproducible cup placement and a dislocation rate of less than 1%. Finally, some have advocated for computer-assisted navigation to more accurately reproduce the desired acetabular cup position.
The effect of head size on the stability of THA has been investigated by numerous authors. “Jump distance” increases as the femoral head diameter increases. Berry and coworkers demonstrated a reduction in dislocation rates for anterolateral, posterolateral, and transtrochanteric hip approaches as the femoral head diameter increased from 22 mm to 28 mm to 32 mm. Smith and associates reported zero dislocations after large-head (38-mm) metal-on-metal (MOM) THA. Dislocation rates for 38-mm versus 28-mm MOM THAs were zero and 2.5%, respectively, at 3-month follow-up in a study conducted by Cuckler and associates. Peters and colleagues also demonstrated zero dislocations with 38-mm MOM THA with 52-month follow-up compared with a 2.5% rate of dislocation with 28 mm (mostly metal-on-polyethylene) despite the posterior approach for the large-head bearings and the direct lateral for the 28-mm bearings. Peters and coworkers reported a 0.4% dislocation rate with 38- to 56-mm-diameter MOM heads in THA with 36-month follow-up. Smit studied anatomically sized—or “jumbo”—femoral heads in primary THA and had zero dislocations at 1-year follow-up in a cohort of 235 patients. However, Lachiewicz and associates found no change in dislocation risk using 36- and 40-mm metal femoral heads on polyethylene liners compared with historical controls. In addition, Amstutz and colleagues reported a dislocation rate of 3.5% in 57 patients with primary THA using femoral heads with diameters of at least 36 mm, which is not dissimilar to historical reports. Scifert and coworkers demonstrated that with larger head sizes, bony impingement becomes much more of a prominent problem than traditional component neck-cup impingement as the levering force in hip dislocations. Large head diameters associated with polyethylene bearings have the drawbacks of potentially creating more volumetric polyethylene wear (which may be mitigated with cross-linked polyethylene) and requiring thinner polyethylene, which may be at greater risk for fracture. Additionally, soft tissue impingement and iliopsoas tendinitis may occur with larger-diameter femoral heads and acetabular components owing to the prominence of the implants changing direction and tension on the tendon.
Kelley and associates reported that 22-mm heads coupled with acetabular components measuring 56 mm and larger, and 28-mm heads coupled with acetabular components measuring 62 mm and larger, are associated with markedly increased risk of dislocation. Kelley and colleagues proposed that head-cup diameter mismatch led to 3 events that potentially increased dislocation rates: (1) prosthetic impingement due to the relatively small arc of motion of the smaller head coupled with a wider-diameter acetabular shell; (2) increased laxity due to movement of the pseudocapsule farther from the head as the result of the use of larger shells; and (3) anatomic femoral head mismatch, which might cause greater laxity.
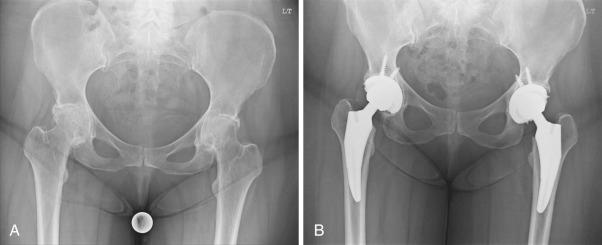
Another option to gain a larger effective head size is the use of dual-mobility constructs. The dual-mobility components typically have a metal acetabular liner with a large polyethylene bipolar femoral head ( Fig. 106.1 ), which is in contrast to an unconstrained tripolar component that uses a polyethylene acetabular liner and a metal bipolar femoral head with an inner polyethylene liner between the head components. Dual mobility has been advocated for use in primary THA to reduce the risk of instability. Harwin et al. reported on 249 patients at high risk for dislocation and had no cases of dislocation at a mean of 3.3 years follow-up. In a systematic review of 59 articles, De Martino reported that the mean rate of dislocation with the use of dual mobility in primary THA was 0.9%.

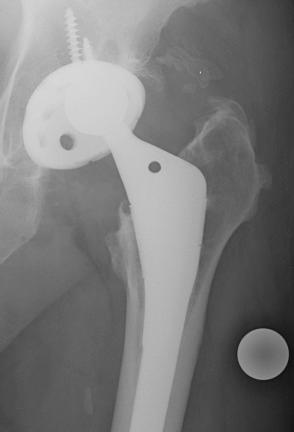
Proper balancing of the soft tissues achieved by restoration of offset and leg length is of critical technical importance to optimize outcomes and minimize complications such as dislocation and limp ( Fig. 106.2 ). Inadequate offset restoration increases the risk of dislocation by increasing the laxity of the soft tissue envelope. Limb length inequality is often associated with component malposition that can be mistakenly addressed by increasing neck length and increasing offset in an attempt to restore tension on the soft tissues to prevent instability. Recognition of these factors is critical in understanding the stability of THA.
Use of high-wall, elevated-rim, or lipped liners has been advocated by some authors to prevent dislocation following primary THA. Cobb and coworkers reported a significant decrease in dislocation rate at 2-year follow-up using 10-degree lipped liners compared with standard liners in a series of 5167 THAs. Concern about nonarticular wear if the prosthetic neck impinges on the elevated rim and impingement causing instability in the opposite direction (e.g., an elevated rim placed posteriorly may decrease anterior stability) have decreased enthusiasm for routine use of elevated liners.
Surgeon procedural volumes have been correlated with outcomes of THA, including dislocation. Battaglia and associates demonstrated that increasing surgical volume is associated with lower dislocation rates and showed that the correlation is more clearly related to individual surgeon volumes than to hospital volumes. Katz and colleagues identified lower dislocation rates in patients treated by surgeons who performed more than 50 THAs per year compared with surgeons who performed 5 THAs or fewer.
When a dislocation is encountered following THA, it is imperative to elucidate the direction and cause of the instability, if possible, to guide treatment and optimize results. The spectrum of treatment options includes closed reduction with or without bracing, component revision, exchange of the modular femoral head and liner, use of a larger femoral head, conversion to an unconstrained tripolar construct, conversion to a dual-mobility construct, use of a constrained liner, removal of the acetabular component and conversion to a bipolar femoral head that fits the acetabular defect, greater trochanteric advancement, and soft tissue augmentation of deficient capsule or abductors. A thorough understanding of risk factors, causes, and management options should enable the surgeon to effectively minimize the incidence of dislocation while providing a solid armamentarium for use in successfully managing an unstable THA.
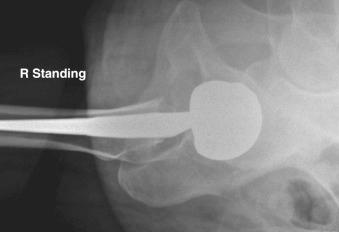
Treatment indications are dictated by the timing and cause of the dislocation. Early dislocations often occur within the first 3 months following surgery, and single episodes usually can be managed with closed reduction. Functional outcomes of patients with a single dislocation successfully treated with closed reduction have been shown to be similar nondislocators at a minimum of 1-year follow-up. Late dislocations are defined as those occurring after 5 years; they have multiple causes, including polyethylene wear, infection, trauma, neurologic decline, increased soft tissue laxity, and cup malposition. First-time late dislocations are at high risk for recurrent instability. Caution is advised when evaluating a late dislocation associated with marked polyethylene wear because an associated liner dissociation can occur; in this instance, closed reduction is not possible, and unnecessary and futile attempts at closed reduction should be avoided ( Fig. 106.3 ). Intermediate-term dislocations following THA occur between 6 months and 5 years. First-time dislocations in this group usually can be managed closed, but when instability recurs, operative management is usually indicated based on the underlying cause. Recurrent dislocations, irrespective of timing, are usually managed most effectively with an operative intervention, which will be the focus of the remainder of this chapter.
The history is critical to understanding the cause of the instability. Obtaining previous operative records to identify the prior operative approach and the make, model, and size of previously implanted components is critical when planning further surgery to ensure that appropriate modular replacements are available. The direction of dislocation and the position that results in instability are important factors that help in understanding the underlying cause. Leg lengths and neurologic function, focusing on the abductors, should be assessed and documented. Checking abductor function by having the patient lie on the side and actively abduct the leg against resistance and testing for a Trendelenburg sign during ipsilateral single leg standing are crucial examination principles. Absent abductor function narrows revision treatment options because it has been associated with failure of large-head reconstructions. In these situations, a constrained liner is often necessary, although some recently have advocated for dual-mobility constructs as an alternative. It is also imperative to evaluate for sepsis as a concomitant complication (using history, wound status, and results of erythrocyte sedimentation rate and C-reactive protein assessment followed by selective preoperative aspiration if clinical suspicion is high).
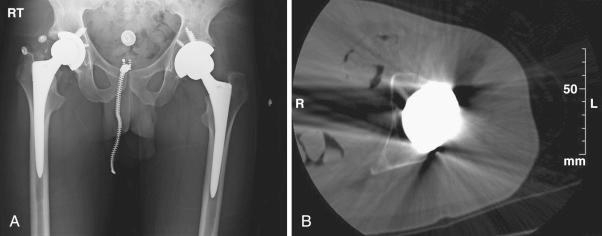
Close scrutiny of radiographs is paramount in the evaluation of instability. Anteroposterior (AP) and true lateral radiographs of the hip and an AP radiograph of the pelvis should be obtained. A shoot-through lateral can aid in providing some idea of cup anteversion ( Fig. 106.4 ). Measurement of leg lengths and offset, as well as assessment of the status of the greater trochanter and component orientation, is important ( Fig. 106.5A ). A preoperative computed tomography (CT) scan can provide precise information regarding acetabular version ( Fig. 106.5B ). A CT scan can also be used to determine femoral component anteversion provided that it includes the epicondylar axis of the knee to define the position of the femoral component in space.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here