Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Malformation of the heart and arterial trunks is the most common form of congenital anomaly found in humans. They occur in approximately 6 of every 1000 live births and in 8 to 10 of every 1000 pregnancies. Fetal echocardiography, or the use of ultrasonic technologies to evaluate the fetal cardiovascular system, enables the diagnosis of structural heart defects and offers a way to observe complex physiologic processes prior to birth. The primary benefits of fetal echocardiography include the ability to counsel parents prior to birth as to the expectations for a child born with a congenitally malformed heart and the ability to implement appropriate postnatal management strategies in an anticipatory fashion so as to maximize outcome and to identify and treat cardiovascular diseases prior to birth.
As imaging technologies and the skills of operators continue to improve, a higher percentage of congenital cardiac malformations can be detected accurately before birth. Analysis of large registries has shown that rates of detection can vary from 15% to 25%. Many studies have documented the utility of the four-chamber view of the heart in identifying the malformations during fetal life. In these studies, rates of detection using this view alone ranged from 4.5% to 81%. Malformations of the outflow tracts, however, and discordant ventriculoarterial connections, may frequently be missed when this view is used in isolation. When views showing the right and left ventricular outflow tracts are included in the obstetric screen, the reported detection rate increases to between 43.8% and 85.5%. In contrast, if fetal echocardiography—defined as a detailed, focused assessment of the fetal cardiovascular system—is performed, rates of detection are significantly higher and diagnostic accuracy rates may exceed 85% to 90%. Given the disparity in rates of detection between those specializing in fetal echocardiography and routine obstetric screening, the question of who should be referred for fetal echocardiography remains an important consideration.
Currently the American Society of Echocardiography recommends fetal echocardiography for fetal, maternal, and familial indications ( Box 8.1 ). In the past, many women were referred for fetal echocardiography due to a family history of congenital cardiac disease, an umbilical cord containing two vessels, maternal diabetes, or maternal exposure to teratogens. Patterns of referral, however, have changed because of improved techniques for imaging have become available. Referrals for fetal echocardiography producing a high yield now include an abnormal obstetric ultrasound evaluation, in which up to two-thirds of referrals have congenital cardiac malformations, and a chromosomal anomaly, with half of such referrals proving to have congenital cardiac lesions. Referrals with a low yield include the presence of a single umbilical artery or exposure to teratogens, with congenital cardiac lesions detected infrequently in mothers referred with these indications. A family history of congenital cardiac disease accounts for between one-quarter and one-third of all referrals for fetal echocardiography but less than one-twentieth of cases with detected malformations. Increased nuchal translucency noted on screening during the first trimester, from 10 to 13 weeks of gestation, has now emerged as an important indication for fetal echocardiography. Indeed, increased nuchal translucency of greater than 3 mm as seen during scanning when the fetus is from 10 to 14 weeks of age is a published marker for aneuploidy. Even in the absence of a chromosomal anomaly, fetuses found to have increased nuchal translucency on early scanning have been shown to be at increased risk for congenital cardiac disease. Fetuses conceived via artificial fertilization, particularly when intracytoplasmic injection of sperm is used, have a twofold increased risk of major birth defects, including congenital cardiac disease, compared with infants conceived naturally. Therefore fetal echocardiography has emerged as a benefit to this group of patients.
Family history of congenital cardiac disease
Metabolic disorders, such as phenylketonuria or diabetes
Exposure to teratogens
Exposure to inhibitors of prostaglandin synthetase, such as ibuprofen, salicylic acid, or indomethacin
Infection with rubella
Autoimmune disease, such as systemic lupus erythematosus, or Sjögren syndrome
Familial inherited disorders, such as Ellis van Creveld syndrome, Marfan syndrome, or Noonan syndrome
Artificial fertilization
Abnormal result following obstetric ultrasonic screening
Extracardiac abnormality
Chromosomal abnormality
Arrhythmia
Hydrops
Increased nuchal translucency in first trimester
Multiple gestation and twin-twin transfusion
Fetal echocardiography is best performed between 18 and 22 weeks’ gestation. At this gestational age there is adequate amniotic fluid to allow good visualization of the cardiac structures and vasculature. After 30 weeks’ gestation, increase in the fetal body mass and the shadowing effects of the fetal ribs may make the acquisition of images more difficult. Maternal transabdominal fetal imaging may be performed between 15 and 18 weeks’ gestation, although visualization may be suboptimal. At some centers, first-trimester screening for congenital heart disease may occur as early as 10 to 14 weeks’ gestation, especially in populations known to be at high risk, such as those noted to have increased nuchal translucency during scanning at 10 to 14 weeks, those with suspected aneuploidy, or those with a family history of congenital cardiac disease. Feasibility studies have demonstrated adequate image acquisition at these gestational ages for both maternal transabdominal and transvaginal imaging. At some centers with experienced practitioners, the prenatal detection rate for major congenital heart disease exceeds 86% to 90% within the first trimester.
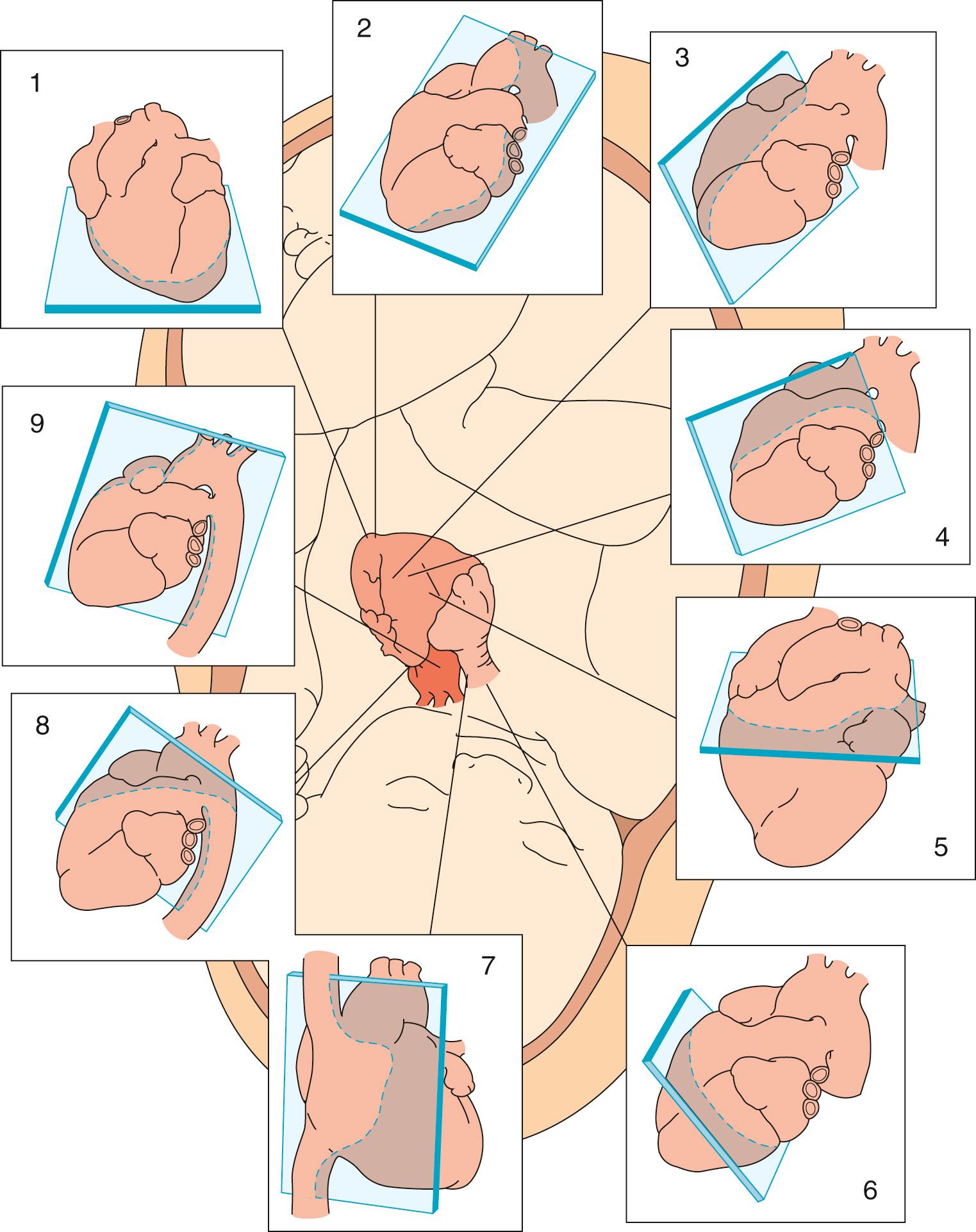
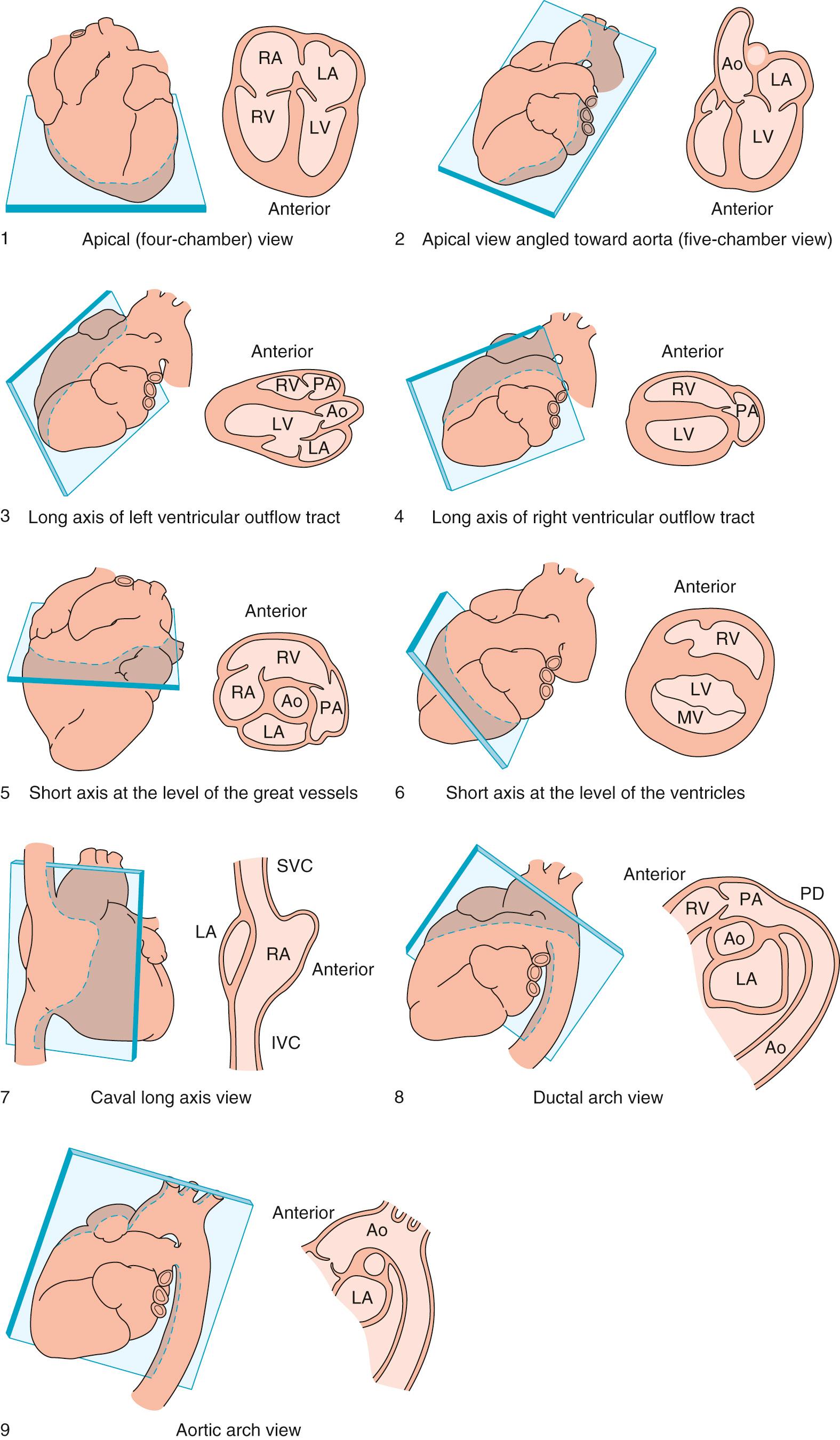
Cross-sectional imaging remains the gold standard for the diagnosis of structural cardiac disease during fetal life. The Pediatric Council of the American Society of Echocardiography recommends obtaining multiple cross-sectional tomographic views of the heart in order to make an accurate diagnosis. Fetal echocardiography should include an apical four-chamber view of the heart, an apical five-chamber view, a long-axis view of the left ventricular outflow tract, a long-axis view of the right ventricular outflow tract, a short-axis view at the level of the arterial trunks, a short-axis view at the level of the ventricles, a long-axis view of the caval veins, a view of the ductal arch, and a view of the aortic arch ( Table 8.1 ; Figs. 8.1 and 8.2 ). The American Institute of Ultrasound in Medicine also recommends the three-vessel tracheal view, which has proven useful in the assessment of critical congenital heart disease as well as aortic arch anomalies. The fetal heart rate should be documented and any arrhythmia confirmed with M-mode imaging. The diameters of the orifices of all valves should be measured in systole at right angles to the plane of flow. Reference ranges for the diameters of all valves over the course of gestation have been published. Cardiac dysfunction may be assessed by cross-sectional interrogation by the presence of ascites, pleural or pericardial effusions, skin edema, and cardiomegaly as defined by a ratio of cardiothoracic areas greater than 0.36. A ratio of cardiothoracic areas greater than 0.6 is associated with an extremely poor outcome.
| Feature | Essential Component |
|---|---|
| Anatomic overview |
|
| Biometric examination |
|
| Cardiac imaging views/sweeps |
|
| Doppler examination |
|
| Measurements |
|
| Examination of rhythm and rate |
|
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here