Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The parvoviruses are a family of single-stranded DNA viruses that have a wide cellular tropism and broad host range, causing infection in invertebrate species and vertebrates, from insects to mammals. Although many parvoviruses are important veterinary pathogens, there are only two human pathogens in the family: human parvovirus B19 and the more recently described human bocavirus. Human bocavirus seems to be primarily a respiratory pathogen of young children and is not discussed further here. Human parvovirus B19 is most commonly referred to as parvovirus B19 or simply B19. B19 is the prototype of the genus Erythrovirus in the Parvoviridae family, and a new genus and name have been proposed for this virus, Erythrovirus B19, based on its cellular tropism for erythroid lineage cells and to distinguish it from the other mammalian parvoviruses. Compared with most other common human viruses, B19 is a relatively new pathogen, but since its initial description, B19 has come to be associated with a variety of seemingly diverse clinical syndromes in many different patient populations ( Table 28-1 ). Although the list of clinical manifestations caused by B19 infection is probably not yet complete, some proposed relationships, such as to rheumatologic disease and neurologic disorders, remain controversial.
| Diseases | Primary Patient Groups |
|---|---|
| Diseases Associated With Acute Infection | |
| Erythema infectiosum (fifth disease) | Normal children |
| Polyarthropathy | Normal adolescents and adults |
| Transient aplastic crisis | Patients with hemolytic anemia or accelerated erythropoiesis or both |
| Papular-purpuric “gloves and socks” syndrome | Normal adolescents and adults |
| Diseases Associated With Chronic Infection | |
| Persistent anemia (red blood cell aplasia) | Immunodeficient or immunocompromised children and adults |
| Nonimmune fetal hydrops | Intrauterine infection |
| Congenital anemia | Intrauterine infection |
| Chronic arthropathy | Rare patients with parvovirus B19–induced joint disease |
| Infection-associated hemophagocytosis | Normal or immunocompromised patients |
| Vasculitis or purpura | Normal adults and children |
| Myocarditis | Intrauterine infection, normal infants and children, immunocompromised patients |
Parvovirus B19 was accidentally discovered by Cossart and associates in 1975 as an anomalous band of precipitation while screening blood donor serum for hepatitis B antigen by counterimmunoelectrophoresis. The name B19 refers to the donor unit from which it was originally isolated. Initial analysis of the new virus revealed it had physical features characteristic of the known parvoviruses, allowing classification in this family. Because the donors from whom it was originally isolated were asymptomatic, B19 infection was not initially associated with any illness, and for the next several years after its description, it was a virus in search of a disease.
In 1981, Pattison and colleagues noted a high prevalence of antibodies to this virus in the serum of children hospitalized with transient aplastic crisis of sickle cell disease and proposed B19 as the viral cause of this clinically well-described event. Serjeant and colleagues later confirmed this association in population studies of sickle cell patients in Jamaica. In 1983, 8 years after its initial description, Anderson and coworkers proposed B19 as the cause of the common childhood exanthem erythema infectiosum (EI), or fifth disease. The name fifth disease derives from the 19th century practice of numbering the common exanthems of childhood—EI was the fifth rash designated in this scheme, and it is the only one for which this numeric designation has persisted in clinical practice. The others in the series included measles, scarlet fever, rubella, and Filatov-Dukes disease (a mild variant of scarlet fever that is no longer recognized).
The possibility of fetal disease associated with EI was considered long before the viral etiology was known primarily because of comparison with rubella and the incidence of congenital rubella syndrome after community epidemics. Advances in knowledge of the virology of other animal parvoviruses and their known propensity to cause disease in the fetus and newborn animal further fueled this concern. This suspicion was confirmed in 1984 when two reports of B19 infection in pregnant women associated with adverse fetal outcomes appeared and were later followed by a larger report of a series of cases of nonimmune hydrops fetalis caused by intrauterine infections with B19. Over the ensuing decade, various clinical manifestations associated with acute and chronic infection have since been attributed to this virus in different patient groups (see Table 28-1 ).
Since the initial reports of fetal infection, knowledge of the epidemiology, pathophysiology, and short-term outcome of fetal and neonatal infection with B19 has increased immensely based on numerous large population-based studies. B19 infection during pregnancy has probably been the subject of more such studies than any of the other manifestations with the possible exception of transient aplastic crisis of sickle cell disease. There is still much to be learned, however, regarding the long-term outcome of fetal infection, the clinical spectrum of infection in neonates, and the immunologic response to infection. Last, the potential for prevention through vaccine development is a topic of current interest and ongoing research.
Similar to other members of the family Parvoviridae, parvovirus B19 is a small, nonenveloped, single-stranded DNA virus. The taxonomy for this family has been revised to include two subfamilies, the Densovirinae, which are insect viruses, and the Parvovirinae, which infect vertebrates. The Parvovirinae subfamily is composed of five genera, four of which are capable of infecting humans: Dependoviru s, Parvovirus , Erythrovirus, and Bocavirus. The dependoviruses require coinfection with another unrelated helper virus (adenovirus or herpesvirus) to complete their life cycle. Some Dependovirus strains infect humans (e.g., adeno-associated viruses), but the infection is asymptomatic and without clinical consequence. In contrast to Dependovirus strains, members of the genera Parvovirus, Erythrovirus, and Bocavirus are able to replicate autonomously. Previously included in the genus Parvovirus, B19 is now classified as an Erythrovirus . At present, the genus Erythrovirus consists of only two members: B19 and a simian parvovirus that has a genomic organization similar to B19 and several related viruses identified in cynomolgus monkeys and macaques that have a similar genomic organization and a similar tropism for erythroid cells. Although many parvoviruses are pathogenic to other mammals (e.g., canine parvovirus, feline panleukopenia virus), B19 and human bocavirus are the only parvoviruses proven to cause disease in humans. A fourth human parvovirus, termed Parv4, has been recently described. Originally isolated from the blood of an intravenous drug abuser with hepatitis C, it has subsequently been found in plasma pools and asymptomatic blood donors. To date, no disease has been associated with Parv4 infection, and its classification into a parvovirus genus awaits further evaluation of its genomic organization.
There is only one recognized serotype of B19. Minor variations in the nucleotide sequence occur among different B19 viral isolates from different geographic areas, but these have not been definitely shown to affect clinical patterns of infection or pathogenicity. Two isolates of human parvovirus, V9 and V6, whose nucleotide sequence differs significantly (>10%) from B19, have been described. Both were isolated from patients with transient red blood cell aplasia indistinguishable clinically from typical B19-induced aplastic crisis. The clinical significance of these variants and whether they represent different genotypes or merely geographic variants of B19 remain topics of debate.
The B19 genome is very small (approximately 5.6 kb) and contained within an icosahedral protein capsid. The capsid structure and lack of an envelope make the virus very resistant to heat and detergent inactivation, features that seem to be important in transmission. The genome seems to encode only three proteins. Two are capsid proteins, designated VP1 and VP2. VP2 is smaller but more abundant and makes up approximately 96% of the capsid protein. VP1 is larger and makes up about 4% of the capsid but contains a unique region that extends out from the capsid surface and serves as the attachment site for the cellular receptor. VP2 has the unique ability to self-assemble into capsids that are morphologically and antigenically similar to B19 viruses when expressed in cell culture systems in vitro. When present with VP1, the capsids incorporate proteins, but VP1 alone does not self-assemble.
The third gene product is a nonstructural protein designated NS1. The function of this protein is unclear, but it has been shown to be involved in regulation of the viral promoter and seems to have a role in DNA replication. Studies of NS1 have been hampered by the observation that it seems to be toxic to cells by an unknown mechanism. More recent studies have further suggested that production of NS1 can lead to programmed cell death (apoptosis) mediated by stimulation of cytokine production.
Because of its limited genomic complement, B19 requires a mitotically active host cell for replication. It can replicate only in certain erythroid lineage cells stimulated by erythropoietin, such as erythroid precursors found in bone marrow, fetal liver, umbilical cord blood, and a few erythroleukemic cell lines. B19 cannot be propagated in standard cell cultures, a fact that had previously limited the availability of viral products for development of diagnostic assays. Much of this limitation has been overcome by the development of molecular methods for the detection of viral nucleic acid and commercial assays for detection of both antibodies and viral DNA are available.
The cellular receptor for the virus has been identified as globoside, a neutral glycosphingolipid that is present on erythrocytes, where it represents the P blood group antigen. The presence of this lipid is necessary for viral infection to occur, and individuals who lack this antigen (p phenotype) are naturally immune to B19 infection. The P antigen is also present on other cells, such as endothelial cells, fetal myocardial cells, placenta, and megakaryocytes. The tissue distribution of this receptor may explain some of the clinical manifestations of infection with this virus (see “Clinical Manifestations Other Than Intrauterine Infection” ).
Although the P antigen is necessary for B19 viral infection, it is insufficient because some cells, particularly nonerythroid tissues, that express the receptor are incapable of productive viral infection. More recently, a co-receptor has been described on human cells that are permissive for B19 infection. The hypothesis is that the globoside is necessary for viral attachment, but the co-receptors somehow allow viral entry into the cell where viral replication can occur. If confirmed, this hypothesis may provide an alternative explanation of the pathogenesis of infection in nonerythroid tissues that express globoside without a co-receptor.
Parvovirus B19 requires a mitotically active host cell to complete its full replicative life cycle. The primary target for B19 infection seems to be erythroid progenitor cells that are near the pronormoblast stage of development. The virus can be propagated only in human erythroid progenitor cells from bone marrow, umbilical cord blood, fetal liver, peripheral blood, and a few erythroid leukemic cell lines. B19 lytically infects these cells with progressive loss of targeted cells as infection proceeds. In vitro hematopoietic assays show that B19 suppresses formation of erythroid colony-forming units, and this effect can be reversed by addition of serum containing anti-B19 immunoglobulin G (IgG) antibodies.
The virus has little to no effect on the myeloid cell line in vitro but causes inhibition of megakaryocytopoiesis in vitro without viral replication or cell lysis. Clinically, this is best illustrated in transient aplastic crisis of sickle cell disease. Patients have fever, weakness, and pallor on presentation, with a sudden and severe decrease in their reticulocyte count. This cessation of red blood cell production coupled with the shortened red blood cell survival because of chronic hemolysis produces a profound anemia. Examination of the bone marrow typically reveals hypoplasia of the erythroid cell line and a maturational arrest; giant pronormoblasts are often seen with intranuclear viral inclusions. With development of specific antibodies, viral infection is controlled, and reticulocyte counts begin to increase.
Evaluation of infection in normal volunteers has shown similar hematologic changes, but because of the longer life of red blood cells, these changes are clinically insignificant. Adult volunteers inoculated intranasally with B19 developed viremia after 5 to 6 days with a mild illness. Their reticulocyte counts decreased to undetectable levels, and this was accompanied by a modest decline in hemoglobin and hematocrit. Platelets and granulocyte counts also declined. Specific antibody production with IgM followed by IgG developed, and viremia was cleared rapidly. A second phase of illness developed at 17 to 18 days, with rash and arthralgias but without fever, and hematologic indices had returned to normal.
The tissue distribution of the cellular receptor for the virus (P antigen) may explain the predominance of hematologic findings associated with B19 infection. Its presence on other tissues may help to explain other clinical manifestations, such as myocardial disease, congenital infection, and vasculitis syndromes. Although the cellular receptor is present and the virus can attach, in contrast to the erythroid cell, these cells are nonpermissive for viral replication; that is, the virus is unable to undergo a complete life cycle with the resultant lysis of the host cells, as described previously. Instead, interaction in these tissues leads to accumulation of the nonstructural protein NS1. This protein is essential for viral replication and has various proposed functions, but it seems to be toxic to most mammalian cell lines when present in excess. NS1 has been associated with apoptosis and programmed cell death. NS1 has also been linked to production of tumor necrosis factor-α and interleukin-6, a potent proinflammatory cytokine. Cellular injury may occur through cytokine pathways and provide another mechanism aside from lytic infection for some of the clinical manifestations.
Chronic infections in immunocompromised patients develop when patients are unable to mount an adequate neutralizing antibody response. These infections are characterized by viral persistence in serum or bone marrow and lack of detectable circulating antibody. Clinical manifestations include chronic anemia or red blood cell aplasia and may include granulocytopenia and thrombocytopenia. The mechanism for leukopenia and thrombocytopenia is unknown, although it has been shown that B19 causes disturbances in megakaryocytic replication when infected in vitro.
Parvovirus B19 is a highly contagious and common infection worldwide. In the United States, 60% or greater of white adults are seropositive (have IgG antibodies to B19 in their sera). This seropositivity indicates a previous infection usually acquired in childhood. Among African Americans, the rate of seropositivity is about 30%. Transmission of B19 from person to person is probably by droplets from oral or nasal secretions. This mode of transmission is suggested by the rapid transmission among individuals in close physical contact, such as schoolmates or family members, and from a study of healthy adult volunteers experimentally infected with B19, in whom virus was found in blood and nasopharyngeal secretions for several days beginning 1 or 2 days before symptoms appeared. In the volunteer study, no virus was detected in urine or stool.
Given the highly contagious nature of B19 infections, it is not surprising that most outbreaks occur in elementary schools and occasionally child care centers. Susceptible seronegative adult school personnel are at high risk for acquiring the infection from students. Some outbreaks in schools may be seasonal, often late winter and spring, and epidemic, with many children and staff acquiring the infection and developing symptoms of EI. At other times, the infection is endemic, with transmission occurring slowly and with only a few manifesting symptoms.
Parvovirus B19 infections occur worldwide. Serologic evidence of B19 infection has been found everywhere studied, including developed countries, undeveloped countries, urban and rural areas, and isolated island populations. The diseases and associated signs and symptoms are the same worldwide. No clinically or epidemiologically important strain or antigenic differences have been detected, and serologic assays are independent of the source or location of patient serum. Disease resulting from B19 seems to be unrelated to specific viral genotypes, although analysis of the antigenic variation or nucleotide sequences of widely dispersed B19 isolates shows some heterogeneity of unknown significance.
Transmission of parvovirus B19 continues throughout the year; however, there are seasonal variations in transmission rates. Outbreaks of EI most often occur in winter and spring in temperate climates and less frequently in fall and summer. In schools or daycare centers, outbreaks of EI may persist for months, usually starting in late winter or early spring and ending with summer vacation. Figure 28-1 highlights multiyear outbreaks of B19 exposure among pregnant women and the associated seasonal variation in Pittsburgh, Pennsylvania. Yearly, most cases occurred in late spring and summer.
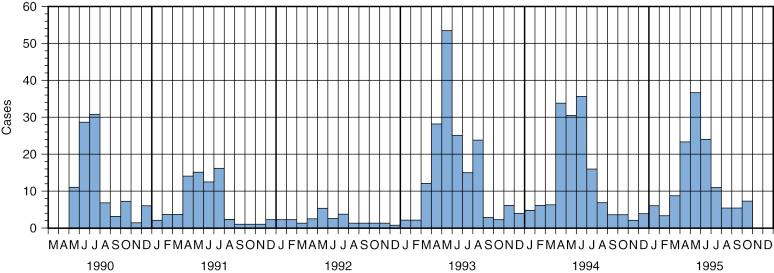
In the island nation of Jamaica, careful studies of people with sickle cell disease showed that epidemics of transient aplastic crises occurred about every 5 years, with little disease inside this interval. Epidemics of B19 infections at 5-year intervals were also observed in Rio de Janeiro, Brazil. In Japan, age-related serologic evaluation of stored serum samples showed no evidence for B19 epidemics over a 10-year period. The prevalence of IgG antibodies to B19 among three tribes of Amerindians living in remote regions of Brazil was very low (<11%), and in one tribe, it was zero for people younger than 30 years. School nursing records in Iowa over 14 years identified cases of EI in all but one year.
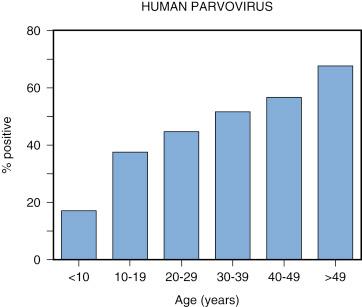
In numerous studies of parvovirus B19 infection based on serologic testing, the seroprevalence of B19 infection increases with age. Figure 28-2 shows the age-dependent increase in seroprevalence from Richmond, Virginia. Transplacentally acquired maternal antibodies are undetectable by 1 year of age. In children younger than 5 years, the prevalence of IgG antibodies to B19 is usually less than 5%. The greatest increase in seroprevalence and B19 infection occurs between 5 and 20 years of age. By age 20 years, the seroprevalence of B19 infection increases from about 5% at 5 years of age to nearly 40%. Afterward, without regard to risk factors, B19 seroprevalence increases slowly. In adult blood donors, the seroprevalence of IgG antibodies to B19 ranges from 29% to 79% (median, 45%). By age 50 years, the seroprevalence may be greater than 75%. Similar results on age-related seroprevalence of B19 infections were observed in India.
In most studies, the prevalence of antibodies to parvovirus B19 in sera obtained from men and women is similar. At least six studies have reported that women have a higher rate of B19 infection than men, however. ∗
∗ References .
In one study of adult blood donors, the proportion of women who were seropositive, 47.5%, was 1.5 times higher than in men. The prevalence of IgG antibodies averaged 51% for women of all ages compared with 38% for men in one of two family studies in Richmond, Virginia, and 64% for women and 50% for men in the other study. In Taiwan, the prevalence of IgG antibodies to B19 among women was significantly higher than among men (36.4% vs. 29.4%; P < .001). The most likely explanation for the higher rates of B19 infection among women compared with men is that women are likely to have more frequent contact with children, especially school-age children who, because of school attendance, are the major sources of B19 transmission. For adults, contact with school-age children is the major risk factor for B19 infection.
In the United States, there are significant differences in the seroprevalence to parvovirus B19 between African Americans and whites. In Richmond, Virginia, approximately 60% of whites are seropositive compared with 45% of African Americans. The reasons for the lower rate of infection among African Americans are unknown but likely reflect the racial segregation of children in schools.
In tests of serum from random blood donors for evidence of a recent parvovirus B19 infection, the rate of infection using antigen detection is 0 to 2.6 per 10,000 individuals tested, with a median of 1 per 10,000, whereas using DNA detection, the rate is 0 to 14.5 per 10,000, with a median of 2 per 10,000. By contrast, when IgM antibodies to B19 were used to detect recent infection, the rate was zero, but all studies included fewer than 1000 patients. As for seroprevalence, women may have a greater risk for infection during outbreaks of EI. During an epidemic of EI in Port Angeles, Washington, the attack rate for women was 15.6%, more than twice the rate of 7.4% for men.
In Spain and Chile, children have the highest rates of B19 infection, which is true for children ages 0 to 4 years and 5 to 9 years. A study of 633 children with sickle cell disease, followed at Children’s Hospital in Philadelphia from 1996 to 2001, found that 70% were seronegative (susceptible), and during this period, 110 of these patients developed B19 infections, for an incidence of 11.3 per 100 patients per year. Of the 110 patients infected, there were 68 episodes of transient aplastic crisis, characterized not only by an acute exacerbation of anemia but also by acute chest syndrome, pain, and fever. The high incidence of disease among children with sickle cell disease emphasizes the need for a vaccine against B19.
Parvovirus B19 is efficiently transmitted among persons residing in the same home, with attack rates, based on the development of signs and symptoms of EI, of 17% to 30%. Using serologic testing to identify asymptomatic infection and to exclude immune individuals, the secondary attack rate for susceptible household contacts is 50%. Most secondary cases of EI or aplastic crisis in the home occur 6 to 12 days after symptoms develop in the index case, A serologic study of pregnant Danish women indicated that seropositivity was significantly correlated with increasing number of siblings, having a sibling of the same age, number of own children, and occupational exposure to children.
During epidemics, B19 transmission is widespread among school-age children. Studies of school or classroom outbreaks of EI with at least one serologically confirmed case of acute B19 infection revealed student infection rates of 1% to 62% based on the occurrence of a rash illness. The median infection rate for all studies was 23%. Because asymptomatic infections are common, and other signs and symptoms of EI may be mild and overlooked, these studies undoubtedly underestimate the true incidence of infection. Studies of students using serologic assays to identify B19 infection during outbreaks report infection rates of 34% to 72%, with most not associated with a rash illness. Higher rates of infection occur in elementary schools and daycare centers compared with secondary schools, and in students in boarding schools compared with students who live at home.
During school epidemics, employees in contact with children have the highest rates of infection compared with community control subjects. The attack rate based on detection of rash illness or arthropathy may be relatively low—12% to 25%. The seroprevalence of B19 IgG antibodies to B19 in school employees is greater than in adult community control subjects, however—50% to 75%. When serologic testing is used to identify employees with asymptomatic infection and to exclude immune employees, the attack rate among the susceptible individuals is usually very high. In four school outbreaks where serologic testing was used, the attack rate among teachers ranged from 19% to 84%, and the frequency of asymptomatic infection was greater than 50% in all but one outbreak. The highest infection rates occurred among susceptible elementary school teachers compared with middle and high school teachers; this may reflect either exposure to more infected children or a greater likelihood of contact with respiratory secretions in younger children. During a community-wide outbreak of EI in Connecticut in 1988, the infection rate among susceptible women was 16% for school teachers, 9% for daycare workers and homemakers, but only 4% for other women working outside the home.
The risk of infection may be increased for school employees even in the absence of recognized epidemics of EI. In a study of 927 susceptible school employees conducted during a 3.5-year period when no community outbreaks were detected, the annual incidence of specific IgG seroconversion was 2.9% compared with 0.4% for a control population of 198 hospital employees. The rate was higher, 3.4%, for school employees with jobs involving direct contact with children compared with only 0.6% observed for persons with jobs that did not involve contact with children. Most (>50%) of the individuals who seroconverted did not recall an illness characterized by rash or arthropathy.
Salivary antibodies can be used to detect IgG and IgM antibodies to B19 because serum antibodies passively diffuse into saliva. Testing saliva for antibodies to B19 was useful in documenting outbreaks in schools and households. In an outbreak in England, school attack rates ranged from 8% to 50%, including an attack rate of 45% among the teaching staff. The household transmission attack rate was 45% for 11 susceptible individuals. These rates are similar to what has been previously observed. Crowding and low socioeconomic status are not proven risk factors for B19 infection. These risk factors are suggested, however, by the observation that in Rio de Janeiro, the seroprevalence of IgG antibodies to B19 is 35% in children aged 5 years or younger, and in Niger, it was 90% by 2 years of age.
Parvovirus B19 can be transmitted from infected patients to hospital workers. Most, but not all, investigations reveal that hospital transmission of B19 to laboratory personnel is common and includes direct patient-to-patient transmission and indirect transmission from materials or specimens known to contain B19. One patient with sickle cell anemia became ill with aplastic crisis 9 to 11 days after contact in the hospital with a patient with hereditary spherocytosis hospitalized for aplastic crisis; B19 infection was confirmed in both patients. An outbreak of EI occurred on a pediatric ward where 13 (26%) of 50 children developed a rash illness. B19 seroconversion occurred in 5 (71%) of 7 children with rash illness and in 9 (35%) of 26 children who were asymptomatic.
Transmission from patient to health care worker occurred twice in one hospital after admission of patients with aplastic crisis. In the first case, 4 (36%) of 11 susceptible employees with close contact had IgM antibodies to B19, indicating recent infection; in the second case, 10 (48%) of 21 employees either had specific IgM antibodies to B19 or seroconverted from IgG negative to positive. Eleven (79%) of 14 employees were symptomatic with rash or arthropathy. Another study of an outbreak of EI among health care workers on a pediatric ward found that 10 (33%) of 30 susceptible health care workers had serologic evidence of acute B19 infection, along with 2 (17%) of 12 immunocompromised patients being cared for on the ward. The two infected patients were not symptomatic, but analysis of preexisting sera showed they acquired B19 while hospitalized. Onset of symptoms among the employees was temporally clustered, indicating a chronic source, such as an immunocompromised patient or person-to-person transmission.
Studies in Hong Kong identified three immunocompromised patients who seemed to transmit genetically identical strains of B19 from patient to patient. At least one of these three patients seemed to be able to transmit the virus over many months. Immunocompromised patients often have chronic infections and may be infectious for long periods. DNA sequence analysis was also used in Japan to document B19 transmission between hospital staff members, including nursing staff, office workers, and a physiotherapist.
Other investigations have observed little, if any, risk for hospital transmission. No evidence of patient-to-employee transmission was found among 10 susceptible health care workers with frequent contact with a chronically infected patient hospitalized for 24 days before institution of isolation precautions. Transmission to hospital employees did not occur after exposure to a B19-infected mother, her infected stillborn fetus, and contaminated objects in the hospital room. During a community outbreak of B19, none of 17 susceptible pregnant health care workers with possible exposure had serologic evidence (IgM antibodies to B19) of a recent infection. In a case-control study of hospital transmission, serologic testing was used to determine the infection rates among personnel exposed to patients with sickle cell disease and transient aplastic crisis before the patients were placed in isolation. Only 1 of 32 susceptible exposed hospital workers acquired a B19 infection compared with 3 of 37 susceptible workers not exposed. This study suggested that hospital workers who cared for patients with aplastic crisis were not at an increased risk for B19 acquisition.
Two prospective studies from one institution determined the incidence of infection in health care workers during endemic (nonepidemic) periods. The first study found the annual seroconversion rate to be 1.4% for 124 susceptible female health care workers followed for an average of 1.7 years. In a subsequent study of 198 susceptible hospital employees, the annual rate was 0.4% compared with 2.9% for school employees. Taken as a whole, the evidence indicates that one must assume that B19 may be highly contagious in the hospital, although perhaps not in every circumstance. Many potential variables may affect rates of transmission from patients to staff, including the type of patient, immunocompromised status or nonimmunocompromised status, the duration of B19 infection at the time of hospitalization, and potentially the viral load of the infected patient. Patients with erythrocyte aplasia or others with suspected EI or B19 infection should be presumed to have a B19 infection until proven otherwise. These patients should receive respiratory and contact isolation while hospitalized.
Person-to-person spread of parvovirus B19 probably occurs through contact with respiratory secretions. Viral DNA is present in saliva at levels similar to those in blood, and in a volunteer study infection was initiated by intranasal inoculation of B19. B19 cannot be detected in columnar epithelial cells of the large airways. Indirect evidence suggests B19 is not transmitted by aerosols. Viruses transmitted by aerosols, such as measles and influenza, are rapidly spread during outbreaks, but new cases of EI are spread out over many months during school outbreaks, suggesting that B19 transmission is inefficient. B19 DNA may be found in the urine, but it is unlikely that this is associated with infectious virus.
The only well-documented route of spread for B19 is vertically from mother to fetus and from parenteral transfusion with contaminated blood products or needles. Vertical transmission is discussed later. Although transmission of B19 by transfusion occurs, it is rare because of the low prevalence of B19 viremia among donors of blood and blood products; however, the risk increases for pooled blood products. B19 DNA is frequently found in clotting factor concentrates, including products treated with solvents and detergents, steam, or monoclonal antibodies, and even treated products may be infectious. Seroprevalence of IgG antibodies to B19 is high among hemophiliacs compared with age-matched controls and is higher for individuals who received frequent infusions of clotting factors prepared from large donor pools, compared with infusions prepared from small donor pools.
Parvoviruses are resistant to chemical inactivation. In one hospital, B19 transmission occurred without recognized direct patient contact, suggesting possible transmission via fomites or environmental contamination. That B19 is transmitted by fomites has not been directly established, but, considering the stability of related animal parvoviruses, this possibility exists. B19 DNA, not infectious virus, was found in a study of a suspected nosocomial outbreak in a maternity ward. B19 DNA was detected by polymerase chain reaction (PCR) assay on the hands of the mother of a stillborn fetus infected with B19 and on the sink handles in her hospital room. Samples from countertops, an intravenous pump, and telephone were also positive by a sensitive nested PCR DNA technique. PCR assay is so sensitive that minute quantities of DNA can be detected via this technique, and the presence of B19 DNA on surfaces does not imply that these surfaces are sources of infection. Infected fetal tissues and placental or amniotic fluids are more likely sources of infection for health care workers than fomites.
We completed a large epidemiologic study to determine the relative risk of parvovirus B19 acquisition for women of childbearing age in daily contact with children, including nurses, daycare employees, and teachers at all levels. We identified risk factors for B19 infections for hospital and school employees during an endemic period. Serologic testing was used to monitor 2730 employees of 135 schools in three school systems and 751 employees of a hospital, all in Richmond, Virginia. Of participants, 60% were initially seropositive. After adjusting for age, race, and gender, risk factors for seropositivity were contact with children 5 to 18 years of age at home or at work and employment in elementary schools. Over 42 months, only 1 of 198 susceptible hospital employees seroconverted (0.42% annual rate), compared with 62 of 927 (2.93% annual rate) school employees (relative risk, 6.9). Four factors associated with seroconversion were employment at elementary schools, contact with children 5 to 11 years of age at home, contact with children 5 to 18 years of age at work, and age younger than 30 years. Women in daily contact with school-age children had a fivefold increased annual occupational risk for B19 infection.
Several observations indicate that B19 infections were endemic but not epidemic or pandemic in the Richmond area during the 42-month prospective evaluation. First, few cases of B19 infection were reported by the school nurses, and no cluster of cases was observed at any single school or group of schools. Second, the seroconversion rates during each of three consecutive study periods were the same for all groups or subgroups. Third, for employees, B19 infections were not clustered at individual schools or groups of schools. Fourth, the infection rates that we observed in employees, even for elementary school teachers, were less than those observed for the 1988 Connecticut epidemic, where 46 infections occurred among 236 susceptible individuals exposed in the schools, for a minimum annual infection rate of 19%. Also, in a study of secondary B19 infections among exposed household members, rates ranged from 30% to 50%.
B19 infections are often asymptomatic or without a rash, and low-level endemics go unnoticed. We observed that 28 of 60 infected employees were asymptomatic, and only 20 knew of a specific exposure. In a study of 52 household contacts of patients with B19 infections during an Ohio epidemic, infections without a rash occurred in 15 of 16 (94%) African Americans and 17 of 35 (47%) whites, and completely asymptomatic infections occurred in 11 of 16 (69%) African Americans and 6 of 30 (20%) whites. During the Connecticut outbreak, 5 of 65 (8%) teachers who were never exposed to a child with a rash became infected. The observations of high secondary attack rates during epidemics and the high rates of infections without a rash or asymptomatic infections provide strong evidence that even during periods when EI is inapparent in the community, school or hospital personnel in contact with children have a significant occupational risk for B19 infections.
Contact with elementary school–age children, whether at home or at work, may be the most important risk factor for B19 acquisition. When seropositivity for persons with children at home was stratified by the child’s age, the association between seropositivity and children at home was significant ( P < .05) when all children 5 to 18 years of age were included; for seroconversion, the significant association was with elementary school–age children at home. The low seroprevalence and seroconversion rate among hospital employees without known contact with children indicates that this group has a low occupational risk for acquiring B19 infections. We and others have not observed an increased rate of seroprevalence among daycare workers, but this has been observed in other studies.
The major conclusions from these studies were that when EI is inapparent in the community, school or hospital personnel in contact with children still have a significant occupational risk for B19 infections and that school employees have an approximately twofold greater risk of acquiring B19 from children at work than from elementary school–age children at home. We also found that hospital employees without contact with children have a low risk for acquiring B19.
Using the Richmond data and assuming that on average 50% of pregnant women are immune, we estimate that, in endemic periods, 1% to 4% of susceptible women become infected during pregnancy. If the rate of fetal death after maternal infection is 5% to 10% (see “Intrauterine Transmission Rates, Clinical Manifestations, and Fetal Outcomes” ), the occupational risk of fetal death for a pregnant woman with unknown serologic status is between 1 in 500 and 1 in 4000. These rates are so low that during endemic periods, they do not justify intervention, such as serologic testing for pregnant women or furloughing or temporary transfer of pregnant seronegative employees to administrative or other positions without child contact (see “Prevention” ).
Knowing B19 infection rates during endemic periods may be more important than knowing rates during epidemic periods. In the United States, B19 infections are endemic most of the time. Because greater than 75% of B19 infections are inapparent, most women who acquire B19 infection during pregnancy do so during endemic periods, not during epidemics. For establishing public health policy and assessing the potential importance of immunizing against B19, knowing that for seronegative women the endemic infection rate is 1% to 4% is more important than knowing epidemic rates, which vary widely depending on the frequency of susceptible individuals in a given population at a particular time.
The most common clinical manifestation of infection with parvovirus B19 is EI, or fifth disease, a well-known rash illness of children. EI begins with a mild prodromal illness consisting of low-grade fever, headache, malaise, and upper respiratory tract symptoms. This prodrome may be so mild as to go unnoticed. The hallmark of the illness is the characteristic exanthem. The rash usually occurs in three phases, but these are not always distinguishable. The initial stage consists of an erythematous facial flushing described as a “slapped-cheek” appearance. In the second stage, the rash spreads quickly to the trunk and proximal extremities as a diffuse macular erythema. The third stage is central clearing of macular lesions, which occurs promptly, giving the rash a lacy, reticulated appearance. Palms and soles are usually spared, and the rash tends to be more prominent on the extensor surfaces. Affected children at this point are afebrile and feel well. Adolescents and adults often complain of pruritus or arthralgias concurrent with the rash. The rash resolves spontaneously but typically may recur over 1 to 3 weeks in response to various environmental stimuli, such as sunlight, heat, exercise, and stress.
Lymphadenopathy is not a consistent feature but has been reported in association with EI 101 and as a sole manifestation of infection. A mononucleosis-like illness associated with confirmed B19 infections has occasionally been reported, but B19 does not typically cause a mononucleosis-like illness. Atypical rashes not recognizable as classic EI have also been associated with acute B19 infections; these include morbilliform, vesiculopustular, desquamative, petechial, and purpuric rashes.
Asymptomatic infection with B19 also occurs commonly in children and adults. In studies of large outbreaks, asymptomatic infection is reported in approximately 20% to 30% of serologically proven cases.
As noted earlier, transient aplastic crisis was the first clinical illness to be definitively linked to infection with parvovirus B19. An infectious etiology had been suspected for this condition because it usually occurred only once in a given patient, had a well-defined course and duration of illness, and tended to occur in clusters within families and communities. Attempts to link it to infection with any particular agent had repeatedly failed until 1981, when Pattison and colleagues reported six positive tests for B19 (seroconversion or antigenemia) among 600 admissions to a London hospital; all six positive tests were in children with sickle cell anemia admitted with aplastic crisis. This association was confirmed by studies of an outbreak of aplastic crisis in the population with sickle cell disease in Jamaica.
Although such transient aplastic crises are most commonly associated with sickle cell anemia, any patient with a condition of increased red blood cell turnover and accelerated erythropoiesis can experience a similar transient red blood cell aplasia with B19 infection. B19-induced aplastic crises have been described in many hematologic disorders, including other hemoglobinopathies (e.g., thalassemia, sickle-C hemoglobin), red blood cell membrane defects (e.g., hereditary spherocytosis, stomatocytosis), enzyme deficiencies (e.g., pyruvate kinase deficiency, glucose-6-phosphate dehydrogenase deficiency), antibody-mediated red blood cell destruction (autoimmune hemolytic anemia), and decreased red blood cell production (e.g., iron deficiency, blood loss). B19 is not a significant cause of transient erythroblastopenia of childhood, another condition of transient red blood cell hypoplasia that usually occurs in younger, hematologically normal children and follows a more indolent course.
In contrast to EI, patients with a transient aplastic crisis are ill at presentation with fever, malaise, and signs and symptoms of profound anemia (e.g., pallor, tachypnea, tachycardia). Rash is rarely present in these patients. The acute infection causes a transient arrest of erythropoiesis (see “General Aspects of Pathogenesis” ) with a profound reticulocytopenia. Given the short half-life of red blood cells in these patients and their dependence on active erythropoiesis to counterbalance their increased red blood cell turnover, this arrest of erythropoiesis leads to a sudden and potentially life-threatening decrease in serum hemoglobin. Children with sickle hemoglobinopathies may also develop a concurrent vasoocclusive pain crisis, which may complicate the clinical picture further.
Leukopenia and thrombocytopenia may also occur during a transient aplastic crisis, but the incidence varies with the underlying condition. In a French study of 24 episodes of aplastic crisis (mostly in individuals with hereditary spherocytosis), 35% to 40% of patients were either leukopenic or thrombocytopenic, compared with 10% to 15% reported in a large U.S. study of mostly sickle cell patients. These transient declines in leukocyte count or platelets follow a time course similar to reticulocytopenia, although they are not as severe, and recovery occurs without clinical sequelae. The relative preservation of leukocyte and platelet counts in sickle cell anemia, compared with other hereditary hemolytic anemias, is presumably due to the functional asplenia associated with sickle cell disease.
As noted in experimental infection in human volunteers, B19 infection in normal subjects results in a decrease in the reticulocyte count, but because of the normal red blood cell half-life, this is not clinically significant or noticeable. Varying degrees of leukopenia and thrombocytopenia also occur after natural B19 infection in hematologically normal patients. Some cases of idiopathic thrombocytopenic purpura and cases of childhood neutropenia have been reported in association with acute B19 infection. Aside from these few anecdotal reports, larger studies have not confirmed B19 as a common cause of either idiopathic thrombocytopenic purpura or chronic neutropenia in children.
Joint symptoms are reported by 80% of adolescents and adults with parvovirus B19 infection, whereas joint symptoms are uncommon in children. Arthritis or arthralgia may occur in association with the symptoms of typical EI or may be the only manifestation of infection. Women are more frequently affected with joint symptoms than men.
The joint symptoms of B19 infection usually manifest as the sudden onset of a symmetrical peripheral polyarthropathy. The joints most often affected are the hands, wrists, knees, and ankles, but the larger joints can also be involved. The joint symptoms have a wide range of severity, from mild morning stiffness to frank arthritis with the classic combination of erythema, warmth, tenderness, and swelling. Similar to the rash of EI, the arthropathy has been presumed to be immunologically mediated because the onset of joint symptoms occurs after the peak of viremia and coincides with the development of specific IgM and IgG antibodies. Rheumatoid factor may also be transiently positive, leading to some diagnostic confusion with rheumatoid arthritis in adult patients. There is no joint destruction, and, in most patients, joint symptoms resolve within 2 to 4 weeks. For some patients, joint discomfort may last for months or, in rare individuals, years. The role of B19 in these more chronic arthropathies is unclear.
The arthritis associated with B19 infection may persist long enough to satisfy clinical diagnostic criteria for rheumatoid arthritis or juvenile idiopathic (formerly juvenile) arthritis. This finding has led some authors to suggest that B19 may be the etiologic agent of these conditions. This speculation has been supported by the detection of B19 DNA in synovial tissue from patients with rheumatoid arthritis and reports of increased seropositivity among patients with these conditions. The more recent findings of DNA from other viruses, in addition to B19 in synovial tissue from patients with arthritis and B19 DNA in synovium from persons without arthritis, suggest that this may be a nonspecific effect of inflammation. A review of the accumulated evidence on this topic has concluded that B19 is unlikely to be a primary etiology in these rheumatic diseases, but it may be one of several viral triggers capable of initiating joint disease in genetically predisposed individuals.
Patients with impaired humoral immunity are at risk for developing chronic and recurrent infections with parvovirus B19. Persistent anemia, sometimes profound, with reticulocytopenia is the most common manifestation of such infections, which may also be accompanied by neutropenia, thrombocytopenia, or complete marrow suppression. Chronic infections with B19 occur in children with cancer who receive cytotoxic chemotherapy, children with congenital immunodeficiency states, children and adults with acquired immunodeficiency syndrome (AIDS), and transplant recipients. Chronic infections may even occur in patients with more subtle defects in immunoglobulin production, who are able to produce measurable antibodies to B19 but are unable to generate adequate neutralizing antibodies.
B19 has also been linked to viral-associated hemophagocytic syndrome, more generally referred to as infection-associated hemophagocytic syndrome. This condition of histiocytic infiltration of bone marrow and associated cytopenias usually occurs in immunocompromised patients. B19 is only one of several viruses that have been implicated as causing viral-associated hemophagocytic syndrome. Infection-associated hemophagocytic syndrome is generally considered to be a nonspecific response to various viral and bacterial insults, rather than a specific manifestation of a single pathogen.
Infections in immunocompromised hosts can lead to chronic infection; this is most often manifested as chronic anemia (red blood cell aplasia), but varying degrees of cytopenia have been described, ranging from thrombocytopenia or neutropenia to complete bone marrow failure. Patients with an inability to produce neutralizing antibodies are at greatest risk, and this complication of B19 infection has been described in children with congenital immunodeficiency syndromes, patients receiving cytoreductive chemotherapy, transplant patients receiving immunosuppressive therapy, and adults and children with AIDS. Increased recognition of B19 infection in solid-organ transplant patients has led to many more recent reports. Although most such infections are manifested as the typical persistent anemia, an association of B19 viremia with acute graft rejection has been described.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here