Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Liver transplantation is used to treat a broad spectrum of end-stage liver diseases. Hepatitis C virus (HCV) and alcoholic and nonalcoholic fatty liver disease–induced cirrhosis are the leading indications in North America, Europe, and South America, whereas in Asia, hepatitis B virus (HBV)-induced cirrhosis still accounts for a large percentage of liver transplants. Original disease recurrence is common in adults, but less so in children, and all liver allografts are susceptible to a variety of technical complications. Therefore an entire textbook devoted solely to histopathology is needed to thoroughly address all potential issues. In addition, the field is becoming more mature, and many centers throughout the world are contributing to advancements. We have relied therefore on review articles for many subjects and have not attempted to cite primary references because the bibliography is already quite extensive.
This chapter will focus on conditions unique to allografts, such as rejection, small-for-size syndrome, preservation-reperfusion injury, and immunosuppression (IS) minimization. It will also cover unique aspects of common liver diseases that are affected by the transplant setting. Because this entire textbook is devoted to liver transplantation, many of the clinical manifestations and risk factors of various disorders are covered elsewhere and kept to a minimum in this chapter. Tabular presentations will be used for easier reference, especially those prepared by the Banff working groups on acute and chronic rejection, late allograft dysfunction, and weaning of IS. This information has also recently been presented in other chapters, in slightly different formats, but overlap exists.
Several groups have reported histopathological findings in potential living donors. Disqualification rates based on biopsy findings alone vary from 3% to 21%. Most biopsies either have normal findings or show mild steatosis, but 20% to 50% show, mostly mild, abnormalities. The most common histopathological abnormality is macrovesicular steatosis, which is present in 14% to 53% of potential living donors. Macrovesicular steatosis exceeding a certain, usually center-specific, threshold is the most common reason for donor disqualification.
Most programs try to limit the severity of macrovesicular steatosis in living donors to less than 30% because this level of steatosis does not appear to adversely impact the postsurgical course of either donor or recipient. Other programs more conservatively limit macrovesicular steatosis to less than 10% or 20%. Diet modification and other treatments to reduce hepatic steatosis can be used to requalify potential donors.
Other common findings in potential living donor biopsies include idiopathic low-grade chronic hepatitis, nonnecrotizing granulomas, and a variety other unexpected findings too numerous to catalogue here (e.g., unexpected early-stage primary biliary cirrhosis [PBC]). Mild (1 to 2+ on a scale of 0 to 4) periportal hepatocellular iron deposits are present in approximately 17% of donors, most of whom are male. This observation probably represents a “normal” finding in males and does not preclude transplantation. Unexplained portal tract eosinophilia is also occasionally seen but does not appear to adversely affect the postoperative donor or recipient clinical course.
Frozen section evaluation can assist in evaluation of nonideal or extended criteria donors (ECDs), defined by various characteristics. Included are increased age (>60 years), macrovesicular steatosis (>40%), cold ischemia time exceeding 12 hours, partial-liver allografts, donation after cardiac death (DCD), hemodynamic instability, use of vasopressors, hypernatremia (>155 mEq/L), HBV or HCV infection or anti–hepatitis B core (HBc) antibody positivity, or presence of a liver mass, fibrosis, or other focal lesions and history of cancer. Feng et al introduced the important concept of a “donor risk score” based on a study of more than 20,000 transplants. The score is based on differential weighing for each of the following parameters: old age (>60 years), anoxic and cerebrovascular causes of death, black race, short height, DCD and split/partial grafts, regional or national sharing, and cold ischemic time. The parameter scores are then summed, and higher total scores translate into greater risk and inversely correlate with 1- and 3-year survivals.
Donor frozen section requests are most often prompted because of the gross appearance, “feel,” or color of the donor liver; known preexisting donor disease (e.g., HCV); or a suspicious clinical history or circumstances surrounding donor death or harvesting procedure. Tissue for frozen section evaluation should be representative and obtained fresh, preferably in the presence of the pathologist, who should also grossly inspect the donor liver.
We request three tissue samples if the gross appearance is uniform: two 2-cm 16-gauge needle cores, one each from the right and left lobes and one 2-cm subcapsular right lobe wedge biopsy. Cores are used to stage fibrosis, if present; a wedge can be additionally helpful for evaluating arterial/arteriolar disease and providing a larger sample for an assessment of steatosis.
The fresh tissue samples should be immediately transported to the frozen section room on a paper towel moistened with preservation solution or in a plastic specimen container. Storage in “physiologic” saline, air drying, and placement of the tissue sample on an absorbent substrate should be absolutely avoided . Air drying and storage in physiologic saline can cause hepatocytes to appear shrunken/necrotic, leading to overestimation of ischemic injury. Absorbent substrates blot fat out of the tissue, resulting in false underestimation of the extent of fatty infiltration.
Difficultly cutting the frozen section should alert the pathologist to the possibility of a steatotic donor liver, which can be correlated with the gross appearance. Recognition of hepatocytes in various stages of necrosis or apoptosis can be enhanced by staining several sections for increasing lengths of time in eosin. This approach enhances contrast between viable and damaged/nonviable hepatocytes: the latter are more hypereosinophilic and often show early nuclear karyorrhexis.
Any histopathological findings are then correlated with the donor history and laboratory values. Partial or fragmented clinical histories are common and can be substantially misleading. Therefore the pathologist should request additional information and/or samples if biopsy findings do not correlate with the given history or events. Donor biopsy evaluation, however, is only one test. The pathologist is unable to predict the adequacy of organ function after transplantation based on frozen section light microscopic evaluation before the operation in the absence of significant histopathological findings.
Reasons for donor organ disqualification vary by center, but most institutions will refuse donors that are found to be positive for certain infections (e.g., human immunodeficiency virus, rabies) or have a recent or even remote history of a high-risk malignancy. Biopsy findings that disqualify organs are also center specific but often include hepatocyte coagulative-type necrosis involving greater than 25% of amply sampled parenchyma, severe macrovesicular steatosis (discussed later), advanced intrahepatic atherosclerosis, and definite evidence of bridging fibrosis. Polarization microscopy can be used to quickly, easily, and accurately assess liver fibrosis without special stains. It is helpful therefore to equip frozen section room microscopes with this capability.
A grossly fatty appearance is the most common reason for requesting frozen section evaluation of a deceased donor liver. Experienced donor surgeons are usually able to accurately estimate steatosis severity before biopsy evaluation. Gross estimates, however, can be misleading in donors with small vacuolar steatosis or when the donor liver is poorly illuminated during examination.
Large vacuolar or macrovesicular steatosis is typically defined as fat globules that are larger than the nuclear diameter and result in displacement of the hepatocyte nucleus to the periphery of the cell. Small vacuolar or microvesicular steatosis is defined as multiple tiny fat globules, usually substantially less than nuclear diameter, where the hepatocyte nucleus remains centrally placed. Macrovesicular steatosis (>30%) increases susceptibility to preservation-reperfusion injury, impairs regeneration, and is associated with decreased graft survival. Small vacuolar microvesicular steatosis is often found after a short period of warm ischemia and other insults and usually does not adversely affect outcome. One study, however, associated “high-grade” small vacuolar or microvesicular steatosis with delayed graft function. It can be difficult in some cases to decide whether the fats vacuoles are large (macro-) or small (micro-) because the fat globules vary substantially in size. Our approach in such cases is to use nuclear size as a rough gauge to make the distinction between microsteatosis and macrosteatosis.
In our opinion the severity of macrovesicular steatosis can be roughly estimated on hematoxylin-eosin (H & E)–stained slides alone. Fat stains are not necessary. Automated morphometric analysis is not really practical in the frozen section setting. We have found that comparison of the microscopic findings to a standardized diagram illustrating various percentages of macrovesicular steatosis improves accuracy and reproducibility of steatosis scoring ( Fig. 84-1 ).

Most studies confirmed the reproducibility of evaluating donor macrovesicular steatosis by pretransplant frozen section evaluation, but cutoff values lower than 30% can decrease reproducibility. Other modalities used to assess steatosis include clinical and biochemical parameters and hepatic computed tomography imaging in conjunction with other noninvasive clinical data, and magnetic resonance imaging.
Use of steatotic donor livers is controversial and varies by center. Most centers disqualify donor livers when macrovesicular steatosis exceeds 50% of hepatocyte volume because it has been reliably associated with increased risk for early dysfunction and failure. This practice, however, has been questioned, particularly if other risk factors (e.g., cold ischemic time [CIT]) or complications are absent or have been mitigated.
Some studies have applied an evenly distributed range for scoring macrovesicular steatosis (mild < 30%, moderate = 30% to60%, and severe > 60%), but we and others use a scale that more closely reflects the triage algorithm for ECD at our center. Our algorithm allows for consideration of multiple factors, as follows: Mild donor macrovesicular steatosis (<10%) does not at all influence the decision-making process. Livers with moderate macrovesicular steatosis (10% to 30%) are usually still used for transplantation, but other factors are also taken into consideration in the decision-making process (e.g., ECD characteristics). Livers with greater than 30%, or severe steatosis, are used only under special circumstances (e.g., when cold ischemic time is kept to a minimum) and when there are very few or no other ECD risk factors. The outcome in such situations can be comparable to nonsteatotic donor livers.
Some nonsteatosis ECD factors can also have histopathological manifestations: advanced age (>60 years), HCV infection, and cardiovascular instability/ischemic injury. Other factors are not reliably associated with specific histopathological findings and do not justify biopsy evaluation: black race, short stature, cerebrovascular cause of death, hypernatremia (>155 mEq/L), cold ischemia time exceeding 12 hours, and partial-liver allografts. DCD status usually fits in the latter category.
American Society of Transplant Surgeons best practice guidelines for DCD suggest that warm ischemia should be limited optimally to less than 20 minutes. Even under ideal circumstances DCD donors are still more susceptible to ischemic cholangiopathy, which usually develops several weeks to months after transplantation. Frozen section evaluation, however, is rarely helpful in evaluating DCD donors because (1) organ reperfusion is required for histopathological manifestations of necrosis and inflammation to be recognized, and (2) the compartment most susceptible to injury, the biliary tree, is not practically accessible for needle biopsy evaluation.
Necrosis in donor biopsies has negatively impacted recipient outcome in some but not all studies. An algorithmic approach or reproducible method of quantifying the necrosis has not been reported and might account for the different observations. In our experience the donor liver is usually disqualified if greater than 20% (roughly estimated) of hepatocytes are necrotic and the necrosis diffusely involves both the wedge and needle cores. Immediately subcapsular necrosis with neutrophilic inflammation is common in donor organs and should not be included in the assessment. Importantly, the extent of necrosis should be correlated with serial liver injury tests in the donor. Caution is urged when necrosis on donor biopsy findings is combined with rising levels of alanine aminotransferase and aspartate aminotransferase. This suggests that the biopsy findings might not accurately represent the final extent of the necrosis.
Many centers use mildly diseased HCV-positive donors to prolong the life of a HCV-positive recipient with end-stage HCV–induced liver failure and rarely use them in HCV-negative recipients with fulminant hepatic failure. Graft and patient survival outcomes have been minimally impacted by donor HCV status, but more rapid progression of fibrosis can occur. All HCV-positive donors at our institution are subjected to frozen section biopsy analysis: those with nonbridging fibrosis (<3/6, Ishak scale) are offered to potential recipients after informed consent. Other groups report one stage lower fibrosis cutoff (<2/6). The advent of new, more effective, anti-HCV therapy will likely significantly alter this process.
Anti-HBc–positive donors can transmit HBV to naive and unvaccinated recipients ; the risk is lower in vaccinated and anti-HBc–positive recipients and can be further reduced by anti-HBV medications and passive antibodies. Donor biopsy evaluation is generally not helpful in this circumstance because the vast majority of biopsies have normal findings in the absence of other diseases.
A variety of neoplastic, infectious, and metabolic diseases have been inadvertently transferred from donors to recipients. Examples that might be detectable by pathologists include various cancers, amyloidosis, hemochromatosis, and fungal, viral, and parasitic diseases. Metabolic diseases, such as familial amyloid polyneuropathy, oxalosis, and possibly α 1 -antitrypsin deficiency can be intentionally or unintentionally transferred with the donor organ in “domino” transplants with the expectation that the latency period between transplantation and recipient disease onset is gained life years. Finally, revisiting any donor data in recipients with unexpected complications can potentially provide valuable insights.
Accurate biopsy interpretation requires familiarity with the donor and recipient operations. This is because many insults that contribute to allograft dysfunction can be traced back to agonal events in the donor and surgical complications. Donor and recipient are usually matched for size and ABO blood groups, and there are numerous surgical variations (e.g., live donors, split livers, alternate vena caval anastomoses) that are beyond the scope of this chapter but are important to understand. The most common operation is a whole deceased donor organ implanted into the recipient using end-to-end anastomoses for the portal vein, hepatic artery, bile duct, and vena cava and is described in Chapter 49, Chapter 50, Chapter 51, Chapter 52, Chapter 53, Chapter 54, Chapter 55, Chapter 56, Chapter 57, Chapter 58, Chapter 59, Chapter 60 . Living donor operations are much more common in Asia, and variable operative technical approaches impact subsequent histopathological interpretation of graft dysfunction.
In general, more complicated and technically demanding operations deviate from reconstruction of normal anatomy. This also increases the risk for both vascular and biliary tract complications. Therefore all reduced-size grafts such as split cadaveric and living donor grafts are at risk for increased biliary and vascular complications. Reduced-size grafts also substantially increase the risk for sampling issues or errors for the pathologist.
Accurate biopsy interpretation also requires an understanding of basic immunobiology, approach to IS, and the evolution of technical complications. In turn, these factors influence characteristic time periods when certain causes of allograft dysfunction occur ( Table 84-1 ). Knowledge of the original disease, time after transplantation, and liver injury test profile therefore often provides enough information to generate a reasonably accurate differential diagnosis. The primary diseases for which liver transplantation is carried out in the United States are covered in Chapter 8, Chapter 9, Chapter 10, Chapter 11, Chapter 12, Chapter 13, Chapter 14, Chapter 15, Chapter 16, Chapter 17, Chapter 18, Chapter 19, Chapter 20, Chapter 21, Chapter 22, Chapter 23, Chapter 24, Chapter 25, Chapter 26, Chapter 27, Chapter 28 . Also, more than one cause of liver injury is often present. Therefore thorough clinicopathological evaluation should be carried out before a final histopathological diagnosis is given.
| Syndrome | Clinical Associations/Observations | Peak Time Period |
|---|---|---|
| Preservation-reperfusion injury | Long cold (>12 hr) or warm (>120 min) ischemic time; older (>60 yr), hemodynamically unstable, DCD, “redo” of anastomoses; poor bile production; prolonged cholestatic phase predisposes to biliary sludge syndrome | Recognized primarily in postreperfusion biopsy specimens and biopsy specimens obtained within the first several weeks after OLT. Changes can persist for several months depending on the severity of the initial injury. |
| Antibody-mediated rejection | ABO-incompatible donor; high titer (>1:32) lymphocytotoxic crossmatch DSA; presents with persistently low platelet counts and complement levels during first several weeks after transplantation | First several weeks to months after transplantation; later onset less common and not well defined. |
| Acute cellular rejection | Younger, “healthier,” female, and inadequately immunosuppressed recipients, long cold ischemic times, and those with disorders of dysregulated immunity (e.g., PSC, AIH, PBC) | Peak dependent on IS regimen; usually between 3 and 40 days; later onset usually associated with inadequate IS. |
| Chronic rejection | Usually occurs in inadequately immunosuppressed patients (e.g., infections, tumors, PTLD) and patients have a history of moderate or severe or persistent acute rejection episodes or are noncompliant | Bimodal distribution; early peak during first year and later increase in noncompliant and inadequately immunosuppressed patients. |
| Hepatic artery thrombosis | Suboptimal anastomosis; pediatric/small caliber vessels; donor and/or recipient atherosclerosis; suboptimal or difficult arterial anastomosis; large difference in vessel caliber across anastomosis; hypercoagulopathy; suboptimal arterial flow (vasospasm from small-for-size syndrome | Bimodal distribution; early peak between 0 and 4 wk and later peak between 18 and 36 mo (see text). |
| Biliary tract obstruction or stricturing | Arterial insufficiency or thrombosis; long cold ischemia; DCD; difficult biliary anastomosis; AMR; original disease of PSC | Variable, but timing can be used to determine cause: < 6 mo usually mechanical, preservation-reperfusion injury (ischemic cholangiopathy) or AMR; > 6 mo recurrent disease, mechanical |
| Venous outflow obstruction | Difficult “piggyback “ hepatic vein reconstruction; cardiac failure | Usually during the first several months. |
| “Opportunistic “ viral (CMV, EBV, adenovirus, etc.) and fungal infections (see text) | Seropositive donors to seronegative recipients (often pediatric); overimmunosuppression | 0-8 wk, much less common thereafter except for EBV-related PTLDs and other EBV-related tumors (see text). |
| Recurrent or new onset of viral hepatitis (e.g., HBV, HCV, HEV). | Original disease HBV, HCV, or acquired HEV-induced hepatitis in patients with contact with animals or culinary inconsistencies | Usually first becomes apparent 4-6 wk after transplantation and persists thereafter; earlier onset (within 2 wk) in aggressive cases. |
| Recurrent AIH, PBC, and PSC | Original disease of AIH, PBC or PSC (see text for risk factors) | Usually more than 6 mo after transplantation; incidence of recurrence increases with time after transplantation. |
| Alcohol abuse | Psychiatric comorbidity/social instability; noncompliance with treatment protocols; GGTP:ALP ratio > 1.4 (see text for risk factors) | Usually > 6 mo |
| Nonalcoholic steatohepatitis | Original disease nonalcoholic steatohepatitis or cryptogenic cirrhosis; persistent or worsening risk factors for NASH in general population | Usually > 3-4 weeks and increases with time if risk factors persist. |
Allograft biopsy monitoring is used (1) to determine the cause of dysfunction or cause of the liver lesion, if present; (2) to assess the effect of therapy and/or progression of disease; and (3) to document the immunological and/or architectural status to help guide chronic IS therapy. Tissue triage therefore depends on the reason for biopsy, clinical differential diagnosis, and time after transplantation. For native livers, the American Association for the Study of Liver Disease has recommended two passes with a 16-gauge needle for adequate assessment of fibrosis because staging is subject to increasing latitude of sampling error for small biopsies (<20 mm long). Needle biopsies containing less than 11 portal tracts might not be representative. Similar guidelines should be followed for liver allograft biopsies, particularly those obtained late after transplantation where assessment of fibrosis and architectural integrity are important.
Most diagnostically important histopathological stains can be completed on routinely processed, formalin-fixed, paraffin-embedded sections. A clinical differential that includes antibody-mediated rejection (AMR) optimally should include fresh frozen tissue for immunofluorescent staining for immunoglobulin and complement, but less sensitive formalin-fixed paraffin-embedded samples can also be used for C4d staining. For each biopsy we routinely prepare two H & E stained slides each containing two to four step sections. Additional special stains are ordered only on indication after review of the H & E slides. Commonly requested stains include trichrome, iron, and copper to detect chronic cholestasis. Cytokeratin 7 or 19 can be used to help localize bile ducts and/or ductular metaplasia of periportal hepatocytes in cases with suspected ductopenia and chronic rejection. Sign-out stations should have access to the electronic medical records and serial laboratory results, IS drug levels, and donor-specific antibodies (DSA) data.
Optimal information needed for interpretation of posttransplant allograft biopsies includes the original disease, ABO compatibility, DSA status and related alloantibody information (e.g., C1q activity), time after transplantation, and type of transplant (e.g., standard whole organ deceased, DCD livers, reduced-size deceased, living related). These variables influence the susceptibility to specific complications and consequently affect the histopathological differential diagnosis. A clinical differential diagnosis can also be very helpful in directing the histopathological workup. Complete clinical information, however, can also bias biopsy interpretation. It is best therefore to first complete the slide review and then correlate findings with the clinical history and laboratory results to generate a differential diagnosis. Final interpretation, however, should be based on a complete clinicopathological correlation. We also routinely compare current findings to findings in previous biopsies. Doing so greatly assists with the interpretation and assessment of therapeutic intervention and disease progression. Re-review of all liver allograft biopsies at a weekly clinicopathological conference is an essential quality assessment tool that provides feedback to clinical physicians and pathologists.
Gross examination of failed allografts should follow a protocol-directed approach similar to that used for native hepatectomy specimens, described elsewhere. Special attention should be given to dissection and inspection of anastomotic sites: biliary, hepatic artery, and portal and hepatic vein. This might require assistance of the operative surgeon to explain the surgical anatomy. Routine tissue sampling for microscopy should include (1) anastomoses, if present; (2) superficial and deep sections of the right and left lobe; (3) at least one deep hilar section with cross sections of the medium-sized bile ducts and arteries; and (4) any grossly obvious defects.
The most common causes of allograft failure vary according to the time after transplantation. Preservation-reperfusion injury or primary nonfunction, the incidence of which is decreasing, vascular thrombosis, and patient death are the leading causes of allograft failure within the first several weeks after transplantation. Very few allografts fail because of acute cellular rejection (ACR) or AMR, but the latter can be missed if the management team is not aware of its manifestations. Recurrent disease, delayed manifestations of technical complications, such as vascular thrombosis or biliary sludge syndrome, and patient death are most commonly responsible for late graft failures occurring more than 1 year after transplantation. Chronic rejection is relatively uncommon as the cause of graft failure, and the incidence is decreasing. Recurrent HCV-induced cirrhosis is a leading cause for allograft failure that challenges organ allocation algorithms, but this is likely to change substantially over the next several years.
Normal liver structure and function is dependent on optimal portal venous and hepatic artery inflow and adequate venous outflow and biliary drainage. Transplanting only a portion of the liver (e.g., living donors or splits) necessarily compromises at least one of these vascular or biliary conduits, especially near the cut edge of the residual liver fragment. It is important therefore for the pathologist to be aware of the technical details of the operation because sampling errors can be quite misleading in reduced size/living donor allografts .
For example, infarcted parenchyma or high-grade biliary or venous outflow obstruction changes can be seen in biopsy specimens obtained near the cut surface in otherwise well recipients with normal or near-normal liver injury tests. In this circumstance the histopathological changes are attributable to only localized defects of blood or bile flow and are not representative. Also, if the patient has more than one biliary anastomosis, biopsy from one lobe might show obstructive cholangiopathic changes, whereas the other lobe might be normal. It is especially important therefore to review the biopsy results in clinical and laboratory context before rendering a final diagnosis.
Early after transplantation reduced-size/living donor allografts are usually undergoing rapid growth and might be more susceptible to damage from needle biopsies and AMR. Late after transplantation, portal venopathy, low-grade ductular reactions, and nodular regenerative hyperplasia changes are also fairly common in reduced-size and living donor liver allografts.
Preservation-reperfusion injury refers to culmination of donor organ damage that occurs during agonal events in the donor, cold preservation, warm sanguineous reperfusion in the recipient, and various other perioperative events. Cold ischemia refers to damage that occurs when the donor organ is stored in preservation fluid and immersed in an ice bath; it preferentially damages sinusoidal endothelial cells, causing them to lift from the underlying matrix. Warm ischemia refers to the time the organ is exposed to blood, but suboptimally perfused. Pathophysiological mechanisms of warm and cold ischemia and preservation-reperfusion have been reviewed elsewhere and in Chapter 44, Chapter 105 . Steatosis increases susceptibility to both warm and cold ischemic injury.
Donor and recipient hypotension, warm ischemia, metabolic abnormalities, cold ischemia during organ preservation, and reperfusion injury all contribute to the syndrome of preservation-reperfusion injury. DCD donors merit special attention because these organs all suffer a warm ischemic insult, by definition, which is substantially more severe than for the organs of brain-dead donors. Even when optimally managed, considerable platelet and leukocyte sludging and subsequent damage occurs in the peribiliary plexus, which in turn results in ischemic cholangiopathy that usually becomes manifest within several weeks to months after transplantation.
Postreperfusion needle biopsies, obtained within several hours of complete revascularization, can reliably gauge the extent of preservation-reperfusion injury. Mild damage is the most common manifestation present in many liver allografts and includes microvesicular steatosis, which is usually attributable to warm ischemia, hepatocellular cytoaggregation (i.e., detachment of individual hepatocytes from each other and “rounding up” of the cytoplasm), and hepatocellular swelling. Severe injury is characterized by zonal or confluent coagulative necrosis, often periportal or midzonal, combined with severe neutrophilic inflammation. A caveat is that the pathologist should not overinterpret either “surgical hepatitis” or manipulation injury as preservation-reperfusion injury. These are characterized by perivenular sinusoidal neutrophilia without necrosis or necrosis and neutrophilia in immediate subcapsular parenchyma.
Preservation-reperfusion injury of donor livers with greater than 20% macrovesicular steatosis causes death of some fat-containing hepatocytes. This releases large lipid droplets into the sinusoids that coalesce into larger globules, which in turn trigger local sinusoidal fibrin deposition, and neutrophilia, which leads to focal obstruction of sinusoidal blood flow and sinusoidal congestion ( Fig. 84-2 ). If the liver eventually recovers, the large fat globules become surrounded by macrophages and resolve over a period of several weeks.
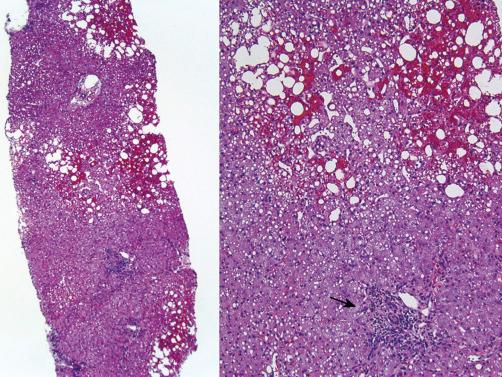
Because normal hepatocytes require only 4 to 6 hours to undergo the entire apoptotic cycle, recognition of apoptotic hepatocytes or coagulative necrosis in a biopsy specimen obtained more than several days after transplantation should arouse the suspicion of another, usually ischemic, insult.
Regenerative repair responses usually begin in 1 to 2 days after injury and are directly proportional to the severity of the insult. Mild injury triggers hepatocellular mitosis and thickening of the plates. These changes are often accompanied by mild centrilobular hepatocellular swelling and hepatocanalicular cholestasis that persist for one to several weeks. Severe preservation-reperfusion injury manifests as marked centrilobular hepatocellular swelling and hepatocanalicular and cholangiolar cholestasis that often persists for 1 or 2 months. Cholangiolar proliferation seems to be triggered by periportal and confluent bridging necrosis with collapse the reticulin framework ( Fig. 84-3 ). Coexistent sepsis, which is also commonly present during this time, can also contribute to this injury pattern. A normal architecture can be eventually restored if the graft recovers, but such patients are at risk for ischemic cholangiopathy.
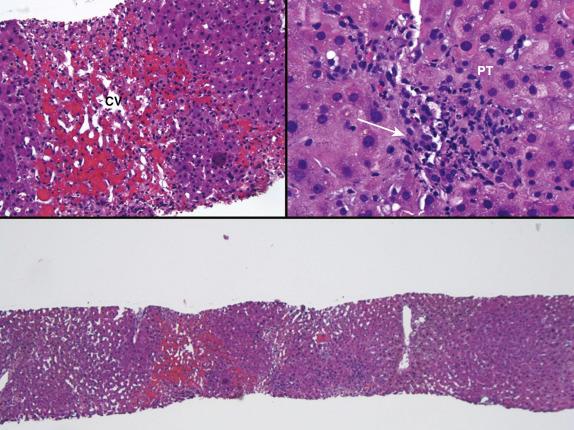
Biliary obstruction/pancreatitis, sepsis, AMR, and cholestatic hepatitis can produce histopathological changes that resemble preservation-reperfusion injury. Detailed donor information, including age and type (e.g., ECD, DCD), cold and warm ischemic times, operative difficulties, recipient clinical profile, and blood culture and crossmatch and DSA profile, and C4d staining results help to determine the likely source of injury. Preservation-reperfusion injury and operative technical difficulties most often prove to be the cause of liver injury early after transplantation.
Examination of the true bile ducts (not cholangioles) contained within the original portal tract stroma and cholangioles at the interface zone provides useful clues in distinguishing between preservation-reperfusion injury and biliary tract obstruction/stricturing ( Fig. 84-4 ). The latter is usually associated with at least some periductal lamellar edema surrounding the true bile ducts or produces stellate-shaped septal bile duct lumens. These changes are often accompanied by neutrophils within the lumen or infiltrating between biliary epithelial cells. Preservation-reperfusion injury, in contrast, does not usually show “true” bile duct changes. Instead, neutrophils surround interface zone cholangioles. Centrilobular hepatocanalicular, cholangiolar cholestasis, and intralobular neutrophil clusters are common to both disorders. The clinical context, history, and laboratory results are often needed to distinguish between sepsis and preservation-reperfusion injury.
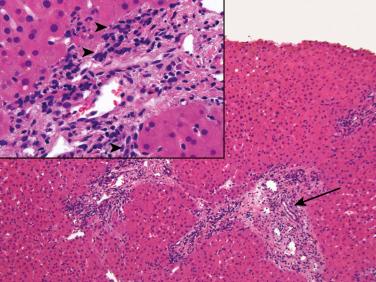
Acute cellular rejection superimposed on preservation injury is recognized by typical “rejection-type” infiltrates in portal and perivenular regions: blastic and smaller lymphocytes and especially eosinophils, which are an excellent marker of an early emerging rejection reaction, as is convincing lymphocytic cholangitis and lymphocytic central perivenulitis.
Distinguishing preservation injury from AMR is discussed later in the AMR section.
Preservation-reperfusion injury can be difficult to distinguish from cholestatic hepatitis without the clinical context and history. Cholestatic hepatitis, however, has been reported only in patients infected with HBV or HCV or those with drug-induced liver injury, all of which are rare earlier than 3 to 4 weeks after transplantation, when preservation-reperfusion injury is most common. In addition, cholestatic hepatitis usually worsens with time unless the patient is specifically treated with decreased IS and/or antiviral therapy, whereas the onset of the insult is obvious for preservation-reperfusion injury and the trend is toward gradual improvement.
Adequate liver structure, function, and growth are dependent on finely balanced portal and hepatic artery inflow and hepatic venous outflow and biliary tract drainage. It is difficult, however, for surgeons to precisely reduce the size of a donor liver (e.g., living donor, split) such that all of these preconditions are satisfied in the donor and recipient. This is especially true when a reduced-size/living donor allograft (<30% of expected recipient liver volume or <0.8% recipient body weight) is placed into the hyperdynamic and hypertensive portal circulation of a cirrhotic recipient: the donor liver fragment might be unable to accommodate the significantly increased portal inflow, especially if the hepatic venous drainage is also compromised. Too much portal venous inflow can injure portal venous and periportal sinusoidal endothelium and contributes to allograft dysfunction referred to as the “portal hyperperfusion (PHP)” or “small-for-size syndrome (PHP/SFSS).”
The arterial buffer response, or the reciprocal regulation of portal venous and hepatic artery hepatic blood flow inflow, is an important contributor to the PHP/SFSS, the pathophysiology of which are discussed elsewhere in this text. Conversely, liver regeneration is highly dependent on portal venous blood flow: too little portal venous inflow can impair regeneration and cause graft steatosis. Moreover, the degree of portal pressure elevation after partial hepatectomy is dependent on the size of the graft, and the regeneration rate is directly proportional to the level of increased portal pressure and flow and inversely correlated with residual volume. Portal venous inflow has to be “just right” to prevent hyperperfusion-related injury, discussed earlier, high enough to promote liver regeneration. Failure of liver regeneration is generally not a major PHP/SFSS clinical problem unless also impeded by suboptimal hepatic venous drainage.
Dahm et al defined PHP/SFSS as at least two of the following complications and on 3 consecutive days within the first several weeks after transplantation: elevated bilirubin level (>100 μmol/L), international normalized ratio or greater than 2, and grade 3 or 4 encephalopathy after exclusion of technical, immunological, or infectious complications. Exclusion of technical complications, however, is not trivial for these complicated operations. Conversely, PHP/SFSS can also contribute to so-called technical complications, which are also found with increased frequency in reduced-size allografts(e.g., arterial vasospasm predisposing to thrombosis ).
Initial events in the PHP/SFSS are observed reliably in experimental animal models of PHP/SFSS, and occasionally in postreperfusion or early posttransplant allograft biopsy specimens obtained within the first several days in humans. Widespread recognition of this phenomenon, however, results in maneuvers to avoid this complication. Consequently, histopathological manifestations are becoming less prevalent.
Denudation of portal vein and periportal sinusoidal endothelium occur as early as 5 minutes following transplantation. ∗
∗ References .
In severe cases microvascular rupture at portal vein–sinusoidal junction can result in hemorrhage into the portal and periportal connective tissue that can dissect into the hepatic parenchyma.
Reparative changes begin to occur if the allograft survives the initial crisis: endothelial cell hypertrophy and subendothelial edema accompanied by an ingrowth of myofibroblasts and endothelial cells into the subendothelial space. Eventually this leads, most commonly, to fibrointimal hyperplasia/intimal thickening and, rarely, luminal obliteration or thrombosis with recanalization.
Venous findings typical of PHP/SFSS are uncommonly seen in peripheral core needle biopsy specimens. Instead, one often observes a constellation of nonspecific findings that usually include centrilobular hepatocanalicular cholestasis, centrilobular hepatocyte steatosis or hepatocyte atrophy with sinusoidal dilatation, and/or centrilobular hepatocyte necrosis and a low-grade ductular reaction. Review of the operative and radiographic reports and clinical history and discussion with the operative surgeon are useful in determining whether histopathological changes and graft dysfunction in a peripheral needle biopsy specimen can be attributed to PHP/SFSS.
Hilar sections of allografts that fail from PHP/SFSS frequently show changes of traumatic injury to larger portal vein endothelium, focal fibrointimal hyperplasia of the vein branches, evidence of arterial vasospasm, and in some cases, ischemic cholangitis, particularly if the hepatic artery has thrombosed. If the graft eventually recovers, portal hypertension and ascites resolve over a period of several weeks. This is accompanied by restoration of the normal architecture, except that some grafts eventually develop significant nodular regenerative hyperplasia. This probably occurs as a result of portal venopathy either from initial injury or from initial segmental suboptimal perfusion and localized collapse.
Suboptimal arterial flow because of arterial thrombosis or stricturing, sepsis, hypotension, and biliary tract obstruction/structuring and ischemic cholangitis can cause histopathological changes similar to PHP/SFSS. Preservation injury might also be a differential diagnostic consideration, but living donor grafts are usually not significantly affected by this complication. Because PHP can lead to diminished arterial flow and thrombosis, more than one complication might be present. Biliary tract obstruction/stricturing alone, however, is usually not accompanied by either portal tract connective tissue hemorrhage or centrilobular hepatocyte ischemic changes. Therefore, when these other changes are present, PHP/SFSS should be considered.
Arterial vasospasm, manifest as medial thickening and mural myocyte vacuolization along with luminal narrowing and wrinkling of the internal elastic lamina, is usually detectable microscopically only in failed allografts and only when it is severe. The vessels most commonly affected are medium-sized perihilar arteries.
Most vascular complications occur during the first several months after transplantation and are related to anastomotic imperfections or iatrogenically induced vascular abnormalities (e.g., narrowing, flaps, dramatic caliber reductions), preexisting atherosclerotic disease, manipulation of the vascular tree, creation of “kinks” or abnormal tortuosity, metabolic or physiological abnormalities that predispose to thrombosis, or a combination of these factors. Vascular problems are often associated with factors that increase the technical difficulty of the operation and vascular anastomoses (e.g., small-caliber vessels and/or abnormal anatomy) and with physiological or metabolic abnormalities that decrease hepatic blood flow and/or promote coagulation (e.g., cardiac failure, clotting abnormalities). Altogether they increase the risk for thrombosis, stimulate perianastomotic atherogenesis, and/or serve as nidi for infection.
Hepatic artery thrombosis (HAT) is the most frequent major vascular complication and an important cause of allograft damage and failure that occurs in 2% to 20% of transplants usually within 30 days of transplantation. The allograft parenchyma and biliary tree are more susceptible to arterial ischemia than similar structures in native livers, at least early after transplantation. This is because allografts are devoid of a collateral arterial circulation during the first several months. Risk factors for early HAT include pediatric age-group, retransplantation, arterial conduit use, long operation time, variant arterial anatomy, and low-volume transplantation centers. Early thromboses usually occur at or near the suture line.
The allograft hepatic artery supplies primarily the extrahepatic and intrahepatic bile ducts, hilar and portal tract connective tissue, and hilar lymph nodes. Therefore these structures are preferentially damaged by inadequate arterial flow or after arterial thrombosis, often resulting in an all-encompassing phrase “ischemic cholangitis” or “ischemic cholangiopathy.” These phrases refer to ischemic damage to the biliary tree manifest as frank necrosis, poor wound/anastomotic healing, biliary leaks, and cholangitic abscesses, all of which eventually result in the biliary sludge syndrome ( Fig. 84-5 ).
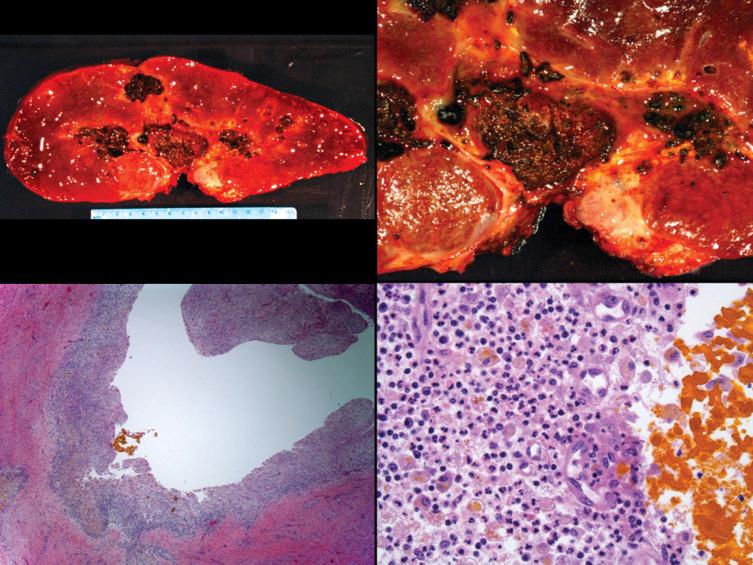
Perihilar tissue and large bile ducts are the structures most susceptible to ischemic injury from hepatic artery compromise but are not routinely sampled in peripheral core needle biopsies. Therefore peripheral core needle biopsy evaluation is not an entirely reliable method of establishing the diagnosis. For example, the biopsy results might be unremarkable, because either the patient has developed vascular collaterals and the thrombosis is inconsequential or there has been necrosis of the hilar bile ducts and changes secondary to biliary sludging have not yet developed in the periphery. Occasionally, however, histopathological changes that suggest suboptimal arterial flow can be detected. Included are centrilobular coagulative necrosis or marked centrilobular hepatocyte swelling and biliary tract complications secondary to ischemic cholangiopathy, such as cholangiolar proliferation, acute cholangiolitis, and obstruction or stricturing. HAT can also occasionally present as spotty acidophilic necrosis of hepatocytes or so-called ischemic hepatitis and mimic the acute lobular phase of viral hepatitis (see later). Chronic suboptimal arterial flow can cause centrilobular hepatocellular atrophy and sinusoidal widening.
Failed allografts with HAT often show necrosis of hilar/perihilar bile ducts with leakage of bile into the surrounding connective tissue, biliary sludge, biliary abscesses seeded with fungi and bacteria, infarction of hepatic hilar lymph nodes, and patchy parenchymal infarction.
Because HAT or narrowing can mimic almost every liver allograft syndrome, a high index of suspicion should be maintained because uncommon presentations are easily misdiagnosed. Ischemic necrosis, often centrilobular, most reliably points toward HAT, but suboptimal flow can also result in centrilobular hepatocyte swelling and/or biliary tract obstruction/cholangitis. Chronic suboptimal arterial flow can cause biliary epithelial cell senescence changes that resemble chronic rejection. “Ischemic hepatitis” (spotty hepatocyte apoptosis) can be virtually indistinguishable from the lobular phase of acute viral hepatitis. In one study, however, the former was shown to have a higher compensatory mitotic rate than the latter. The relationship between arterial thrombosis and biliary tract complications is so common that examination of hepatic arterial patency should be routinely considered when biliary tract complications are encountered .
Portal vein complications are much less frequent than those involving the hepatic artery, involving approximately 1% to 2% of recipients. Included are thrombosis, strictures, and poor flow because of persistent collateral circulation or hypotension. Risk factors include reduced-size and living donor transplants, small portal vein diameter, pediatric recipients, previous portal vein thrombosis, surgical shunting before transplantation, splenectomy, and use of cryopreserved venous interposition grafts.
The histopathological findings depend on the severity of portal vein flow compromise, time after transplantation, and the condition/structure of the allograft. Suboptimal flow because of strictures, kinks, or persistent collateral circulation can result in periportal to midzonal hepatocyte atrophy and/or necrosis and unexplained zonal or panlobular macrovesicular steatosis and nodular regenerative hyperplasia. Complete portal venous obstruction early after transplantation in a noncirrhotic allograft often causes massive coagulative necrosis. Bacterial or fungal seeding of a partial portal vein thrombus can result in miliary seeding of the liver with small abscesses. Long-surviving liver allografts with recurrent disease/cirrhosis are also are susceptible to portal vein thrombosis, as with any cirrhotic liver.
Suboptimal portal vein blood flow can be difficult to distinguish from suboptimal hepatic venous drainage. Linear zones of ischemic necrosis and/or hepatocellular atrophy favor the former, whereas red blood cell congestion within central veins and centrilobular sinusoids and obliterative central venopathy favor the latter. Ultrasonography and/or angiography are needed to more accurately characterize the cause of the vascular abnormality. Cases of suboptimal portal vein flow presenting with intrahepatic steatosis need to be distinguished from recurrent or de novo steatosis/steatohepatitis. The former usually appears within several weeks after transplantation, whereas the latter usually requires 6 months or more.
Suboptimal hepatic venous drainage is relatively uncommon. Risk factors include reduced-size/living donor allografts without inclusion of the middle hepatic vein and difficulties reconstructing the venous outflow tract or alternative anastomoses such as the “piggyback” approach. Significant stenosis or thrombosis of the outflow tract is mostly always associated with either significant clinical or histopathological manifestations.
Congestion and hemorrhage involving the hepatic venules and surrounding perivenular sinusoids are the most reliable histopathological findings that point toward suboptimal hepatic venous drainage ( Fig. 84-6 ). Bland centrilobular hepatocyte necrosis and dropout are also usually present. Chronic changes can include perivenular fibrosis, focal central vein occlusion, and nodular regenerative hyperplasia. Substantial perivenular fibrosis can also be accompanied by a prominent ductular reaction at the interface zone and in perivenular areas. This can make it difficult to recognize architectural landmarks and to exclude biliary obstruction. Severe and/or long-standing suboptimal venous outflow eventually results in venocentric cirrhosis.
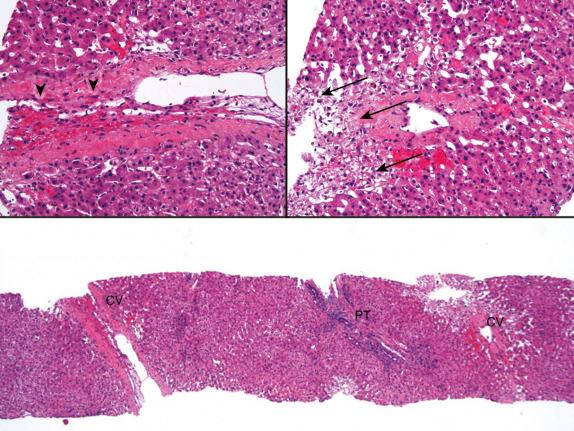
If any of the previously mentioned ischemic and/or congestive changes are not accompanied by significant inflammation, a mechanical/outflow obstruction cause should be suspected. Conversely, if centrilobular changes are accompanied by significant lymphocytic, histiocytic, and/or lymphoplasmacytic inflammation, an immunologically mediated cause of injury, such as acute and chronic rejection, plasma cell hepatitis (PCH)/autoimmune hepatitis (AIH), and drug-induced liver injury (less likely) should be suspected. However, there are two caveats. First, relatively bland perivenular and perivenular subsinusoidal fibrosis has recently associated with chronic AMR; correlation with serum DSA and C4d stains can provide useful information (see Acute and Chronic Antibody Mediated Rejection). Second, perivenular inflammation associated with cellular rejection can wax and wane but nonetheless be associated with significant perivenular fibrosis. If the perivenular fibrosis is first detected during the “hypocellular” phase, the histopathological changes can be indistinguishable from mechanical causes of venous outflow obstruction. Review of the clinical history and previous biopsy results is often helpful in making the distinction.
The biliary tree continues to be the Achilles' heel of liver transplantation with complications occurring in approximately 20% of recipients—an even higher incidence is observed in reduced-size allografts. Most biliary tract complications are ultimately attributable to ischemic and/or traumatic or immunological injury or a surgically introduced abnormal anatomy. These factors predispose to poor biliary tree wound healing, inadequate drainage, or inordinate reflux, or a combination of these three.
The distal-most portion of the extrahepatic donor bile duct is particularly vulnerable to ischemic-injury: it is supplied by only one of the three terminal branchings of the hepatic artery, the peribiliary plexus, and arterial flow originates more proximal to the resection margin. The peribiliary plexus is particularly vulnerable to preservation-reperfusion injury and damage during operative manipulation. For example, in DCD donors, sludging of blood or use of the more viscous University of Wisconsin solution can prevent adequate reperfusion of the peribiliary plexus in the recipient. This can be lessened by arterial, and consequently peribiliary plexus, perfusion by thrombolytic agents and/or less viscous preservation solutions (e.g., histidine-tryptophan-ketoglutarate) before reperfusion in the recipient. Other causes of biliary tract ischemia include (1) small-for-size syndrome and severe arterial vasospasm, (2) prolonged cold ischemia, (3) older donors with atherosclerotic disease, and (4) preformed or de novo antidonor antibodies (e.g., ABO isoagglutinin or anti–human leukocyte antigen (HLA) antibodies ).
Complications include anastomotic dehiscence, transmural necrosis, bile leakage, cholangitic abscesses, ascending cholangitis, bile casts, strictures, obstruction, ampullary dysfunction, and biliary-vascular fistulas. These occur in approximately 15% of whole cadaveric allografts and up to 30% to 40% of reduced-size/living donor allografts.
Strictures are the most common complication; they are categorized according to time after transplantation and location: into those occurring early and late, and into anastomotic and nonanastomotic or intrahepatic. Intrahepatic strictures are further categorized into hilar versus peripheral.
Anastomotic strictures usually appear within the first several months after transplantation but continue to occur, at a reduced rate, for many years. Multiple risk factors for anastomotic strictures include postoperative bile leaks, female donor–male recipient sex combination, and more recent year of transplantation. Compared to nonanastomotic strictures, anastomotic strictures are generally more amenable to radiographically directed and/or surgical intervention and have less of a negative impact on long-term graft and patient survival.
Conversely, nonanastomotic strictures usually occur later after transplantation, are generally untreatable and progressive, and negatively impact graft and patient survival. Risk factors for nonanastomotic strictures include use of high-viscosity preservation solution, primary sclerosing cholangitis (PSC) original disease, Roux-en-Y biliary anastomoses, and cytomegalovirus [CMV] infection.
Early (<1 year) nonanastomotic strictures are often associated with preservation-related injury, including DCD and extended criteria donors and are usually located in perihilar bile ducts. As expected, risk factors include long cold and warm ischemic times, high-viscosity preservation solution, older recipient age, a duct-to-duct biliary anastomosis, and bile leaks. Late (>1 year) nonanastomotic strictures are usually peripheral and associated with immunological risk factors, such as PSC as the original disease.
Allograft biliary tract complications are histopathologically identical to those seen in native livers ( Fig. 84-7 ). Typical findings of stricturing/obstructive cholangiopathy include portal and periductal edema, predominantly neutrophilic portal inflammation, intraepithelial and intraluminal neutrophils within true bile ducts, an interface ductular reaction of varying severity, centrilobular hepatocanalicular cholestasis, bile infarcts, and small clusters of neutrophils throughout the lobules. Over time chronic biliary tract strictures, or intermittent obstruction, are often associated with mixed or predominantly chronic portal inflammation, biliary epithelial cell senescence changes, and low-grade ductopenia involving the small bile ducts. More than 1 year after transplantation, in addition to the classic features described earlier, biliary strictures are a relatively common cause of portal eosinophilia. A frequent situation is recurrent placement of biliary stents to facilitate drainage and intermittent stricturing that leads to a combination of the findings mentioned previously.
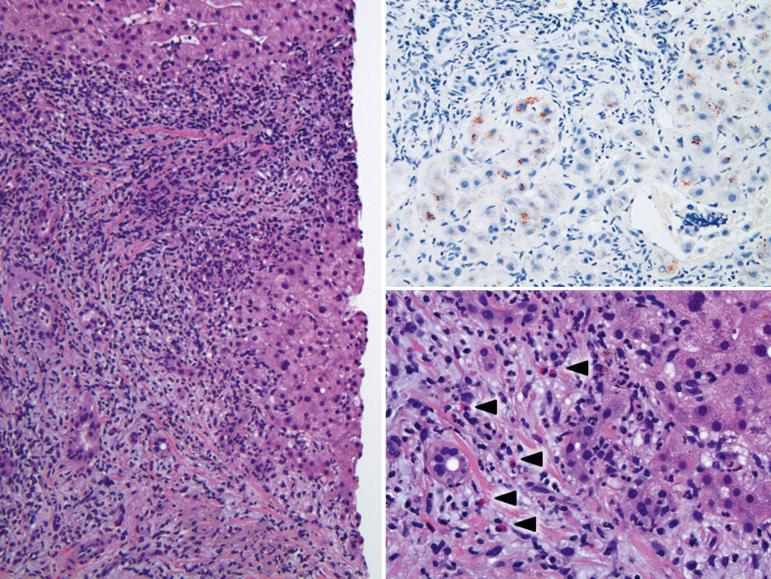
Biliary-vascular fistulas are recognized by red blood cells in lumen of bile ducts or bile concretions in blood vessels. Occasionally, inordinate elevations of the serum bilirubin level, beyond the normal pathophysiological range, can also be seen with a fistula. In contrast, periductal hemorrhage surrounding small interlobular bile ducts is an inconsequential finding in asymptomatic patients when a biopsy is obtained within a day or so after transhepatic cholangiography.
The time after transplantation and original disease are important considerations in the differential diagnosis of obstructive cholangiopathy. Within the first several weeks after transplantation biliary obstruction/cholangitis can be difficult to distinguish from preservation-reperfusion injury (discussed earlier) and ACR, particularly if the patient was treated with increased IS before the biopsy was obtained. Features that favor biliary tract obstruction/stricturing over ACR include neutrophilic-predominant portal infiltrate, periductal edema, retention of the normal nuclear-cytoplasmic ratio and biliary epithelial cells, and an absence of perivenular mononuclear inflammation. Acute rejection is favored when mixed portal infiltrate composed of blastic and small lymphocytes, plasma cells, and eosinophils; lymphocytic cholangitis; increased nuclear-cytoplasmic ratio in biliary epithelial cells; and perivenular inflammation are seen. Portal eosinophilia in early acute rejection can be quite striking, especially in patients treated with “steroid-sparing” immunosuppressive regimens.
Late-onset and/or chronic biliary tract complications can occasionally present with predominantly mononuclear portal inflammation and biliary epithelial cell senescence changes, and low-grade ductopenia can develop ; they can also cause isolated portal eosinophilia and therefore mimic acute and chronic rejection, viral hepatitis, and recurrent autoimmune disorders. Clinicopathological correlation can also be quite helpful in distinguishing late-onset biliary tract complications from late-onset acute rejection. Adequate immunosuppressive drug levels with preferential elevation of the γ-glutamyl transpeptidase (GGTP) and alkaline phosphatase levels favors obstructive cholangiopathy because late-onset acute rejection is unusual in this circumstance.
Chronic rejection and biliary strictures, especially recurrent PSC , occur in similar risk populations, they cause “cholestatic” elevation of liver injury test results, and biopsy findings can show hepatocanalicular cholestasis, biliary epithelial cell senescence changes, and ductopenia. Careful examination of the clinical history, evaluation of serial biopsy findings, and histopathological evaluation are needed to distinguish with confidence between biliary strictures/recurrent PSC and chronic rejection. Features that favor biliary strictures/recurrent PSC over chronic rejection in a needle biopsy include a history of biliary tract complications and/or PSC original disease, periductal lamellar edema involving true bile ducts, stellate portal expansion, stellate-shaped bile duct lumens, portal neutrophilia, and a ductular reaction affecting at least some portal tracts and copper or copper-associated protein deposition in periportal hepatocytes. Features that favor acute and/or chronic rejection include a previous history of rejection and/or inadequate IS, lymphoplasmacytic portal inflammation, small portal tracts, absence of a ductular reaction, absence of copper deposits, and active central perivenulitis and/or perivenular fibrosis.
Additional findings in failed allografts can also be used to distinguish between biliary strictures/recurrent PSC and chronic rejection. Chronically rejected livers are usually of normal or slightly increased weight, whereas those with obstructive cholangiopathy are usually significantly enlarged. In the hilar lymph nodes, obstructive cholangiopathy usually causes bile-pigmented sinus histiocytosis, whereas chronically rejected nodes are usually atrophic and/or fibrotic. The perihilar arteries are either normal or show mild and focal eccentric fibrointimal hyperplasia, whereas foam cell arteriopathy and significant concentric fibrointimal hyperplasia are typical of chronic rejection. In obstructive cholangiopathy, extrahepatic/large intrahepatic bile ducts often show focal ulceration, periductal lymphoplasmacytic inflammation, and fibrosis, whereas in chronic rejection ulceration is unusual. When the infiltrate in either a periphery needle biopsy or explant perihilar section of a large bile duct shows plasma cell–rich infiltrates, the possibility of recurrent or de novo IgG4 sclerosing disease should be considered.
Angiography and cholangiography are also useful in making the distinction between obstructive cholangiopathy and chronic rejection. “Pruning” of the peripheral arterial and biliary trees and poor peripheral filling are seen in chronic rejection, whereas some intrahepatic duct dilation is observed with obstructive cholangiopathy and arterial changes are either not present or not significant.
It is not yet possible to reliably distinguish recurrent PSC from other causes of biliary tract obstruction or stricturing on the basis of needle biopsy evaluation alone.
Biliary strictures can be difficult to distinguish from chronic hepatitis in some cases. Cholangitis favors a biliary tract complication, whereas cholangiolitis and lobular disarray favor chronic hepatitis. The cholestatic liver injury tests profile favors obstructive cholangiopathy, unless the patient has cholestatic variant of viral hepatitis. Review of HBV and/or HCV nucleic acid levels in the blood helps to distinguish between the two because cholestatic viral hepatitis is variably associated with very high levels of viral replication (e.g., >30 million International Units/mL HCV RNA).
As in other organs, liver allograft rejection is broadly categorized into acute and chronic AMR, ACR, and chronic rejection. Relatively isolated AMR usually occurs within the first several weeks after transplantation in ABO-incompatible allografts and in recipients with a strongly positive lymphocytotoxic crossmatch harboring DSA. DSA also often appear in association with uncontrolled acute cellular and chronic rejection, leading to a mixed pattern of acute cellular and antibody-mediated rejection. Late-onset or chronic AMR has not been adequately defined, but some potential features are discussed later. Effective IS has made otherwise typical acute and chronic rejection uncommon-to-rare causes of allograft failure.
AMR can be caused by preexisting or preformed antibodies that are present in the recipient before transplantation or new-onset (de novo) antibodies that develop after transplantation of the donor organ. Natural or isoagglutinins antibodies directed at ABO blood group antigens and anti- HLA antibodies are the most important and best-characterized antibody-antigen systems in solid organ transplantation. It is of critical importance therefore to know the distribution of ABO/ABH and HLA antigens within the liver ( Table 84-2 ). ABH and HLA class I molecules are relatively stable and ubiquitously expressed on the vascular endothelial cells, biliary epithelium, and hepatocytes. In contrast, HLA class II molecules are more restricted in distribution, and their expression is linked to the presence of inflammatory mediators (especially-interferon-γ) within the organ (see Table 84-2 ). For example, class II HLA antigens are normally restricted to antigen-presenting cells within the allograft, but both structural and inflammatory changes can upregulate HLA class II on portal capillary endothelial cells, biliary epithelium, and parenchymal cells. More work, however, is needed in this area, especially with the advent of interest in AMR.
| Antigen | HC | BEC | SEC | KC | SC | Artery/Vein Endothelia | DC | Portal Microvascular Endothelia |
|---|---|---|---|---|---|---|---|---|
| AB | − | + | + | − | − | +++ | − | ++ |
| H | − | ++ | + | − | − | +++ | − | ++ |
| HLA A,B | +/−/+ | +++ | ++ | ++ | +/++ | ++ | ++ | ++ |
| HLA DR | −/+ | −/++ | −/++ | +/++ | +/++ | −/++ | +++ | Tr/vr/++ |
| HLA DP | −/+ | −/+ | −/+ | +/++ | −/− | −/++ | +++ | −/+ |
| HLA DQ | −/− | −/+/− | −/+/− | +/+ | −/− | −/+/− | +++ | −/+ |
Crossing ABO blood group barriers causes predictable and severe liver allograft injury and is generally avoided in most North American programs because doing so leads to a high incidence (approximately 60%) of significant AMR and/or graft failure. However, ABO-incompatible liver allografts are still used in Asian programs, where the donor pool is more limited. Plasmapheresis and vigorous antirejection and microcirculatory protective therapy are needed to achieve reasonable results under these circumstances. Even so, ABO-I recipients are still at risk for late biliary tract complications, such as ischemic cholangitis and strictures.
Liver allografts are demonstrably less susceptible than other solid organ allografts to damage from AMR because of anti-HLA class I and II antibodies but are not completely spared from injury by these or other antibodies. Regardless, DSA status does not routinely influence organ triage/recipient selection at most centers, even though it is associated with increased rejection rates and decreased graft survival in some studies. Cases of late-onset, combined AMR and cell-mediated rejection have been reported, and several Japanese studies show an association between perivenular C4d staining, DSA, and perivenular and perivenular subsinusoidal fibrosis, which improved after increasing IS (see Immunosuppression Optimization Protocols, later). However, criteria for establishing the diagnosis of chronic isolated AMR have not been clearly defined, and this area is in need of further study.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here