Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Herpes simplex encephalitis (HSE) is the most common cause of fatal sporadic encephalitis worldwide. In adults and older children, most cases of HSE are caused by the herpes simplex virus 1 (HSV-1) virus. Patients initially present with nonspecific neurologic signs, including altered mental status, focal cranial nerve defects, hemiparesis, dysphagia, aphasia, ataxia, and seizures, usually with accompanying fevers. Symptoms of encephalopathy then progress with devastating results; the mortality rate exceeds 70% without treatment, with only 11% of survivors returning to baseline function. Acyclovir has been the mainstay of treatment for HSE, and the estimated mortality reduction is 70% to 28%. The percent of survivors who return to normal function is also higher among acyclovir-treated cohorts. Delayed initiation of acyclovir therapy is directly associated with poorer outcome. Thus early recognition of HSE and aggressive empiric treatment with acyclovir is vital to improve morbidity and mortality.
The HSV enters the brain via the cranial nerves. Clinical presentation is usually with an acute onset of encephalopathy, with fever being present in almost all cases. Recruitment of local inflammatory responses leads to cytotoxic edema, presenting as restricted diffusion, which is the most apparent imaging finding in the early stage. The initial site of HSE involvement is usually the medial temporal lobes, either unilaterally or bilaterally (in which case it is often asymmetric; Fig. 7.1 ). HSE has a strong predilection for the limbic system and spreads along these pathways to involve the contralateral medial temporal lobe, anterior temporal lobe, parahippocampal gyrus, amygdala, orbitofrontal gyri, mammillary bodies, insula, and fornix. The thalamus, parietal lobes, and occipital lobes may occasionally also be affected. The basal ganglia and brainstem are characteristically spared.
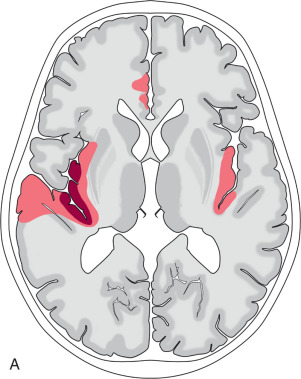
As the disease progresses the restricted diffusion normalizes and T2/FLAIR signal increases. Hemorrhage and enhancement can be variably present on imaging, but generally do not impact prognosis. However, fulminant hemorrhagic necrosis of the affected areas leads to a high mortality rate. Survivors often have long-term sequela, manifesting as regional parenchymal volume loss and encephalomalacia on follow-up imaging. A small subset of patients has recurrence of symptoms that is felt to be immune mediated.
It is unclear whether HSE is caused by reactivation of a latent HSV-1 infection or if it represents a primary infection. Latent HSV-1 remains in the trigeminal ganglia of asymptomatic patients after the acute illness subsides, and it is postulated that the encephalitis is a result of reactivation of the latent virus. An alternative theory is that HSE is a primary infection in which the virus travels to the brain either through the trigeminal nerves or olfactory tracts. The latter is supported by studies demonstrating that in at least half of cases of HSE the strain of the virus identified is different from the one responsible for herpes labialis in that individual, as well as studies demonstrating that the percent of HSE patients that provide a history of herpes labialis is not higher than in the general population.
Regardless of the timing of inoculation, the most widely accepted theory is that the virus gains access to the central nervous system via the cranial nerves, either through the olfactory or trigeminal nerves. Once the virus breaches the brain parenchyma, it triggers an immune reaction that may itself contribute to cell death and tissue destruction.
Clinically, HSE presents with nonspecific neurologic findings of an acute (<1 week) duration. Neurologic findings include hemiparesis, dysphagia, aphasia, ataxia, or focal seizures. Fever is the most discriminating finding, present in 90% of patients. Patients often have a prodromal syndrome of upper respiratory tract infection.
Although head CT examination is often obtained in the setting of encephalopathy, in most patients, no abnormality is detected in the first 4 to 6 days of disease. MR is much more sensitive than CT, especially early in the disease course. HSE leads to early development of cytotoxicity, and diffusion-weighted imaging is therefore sensitive in the acute and subacute phase of the disease. In the early disease phase, DWI increases the conspicuity of lesions identified on CT and demonstrates many more areas of signal abnormality in regions that appear normal on CT.
DWI remains the most sensitive sequence for detection of HSE until approximately 2 weeks of disease duration, at which point FLAIR shows more pronounced signal abnormality ( Figs. 7.2–7.4 ). Hemorrhage, both of the large intraparenchymal and petechial variety, is present in less than half of cases. Contrast-enhancement can be seen on CT and MR but is less frequent than FLAIR hyperintensity or restricted diffusion. Of all MR imaging findings in the acute stage, the only one that is predictive of disease morbidity is restricted diffusion. The presence of hemorrhage or contrast enhancement does not portend a worse prognosis.
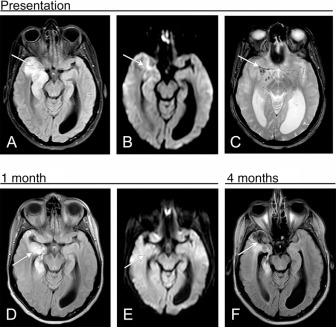
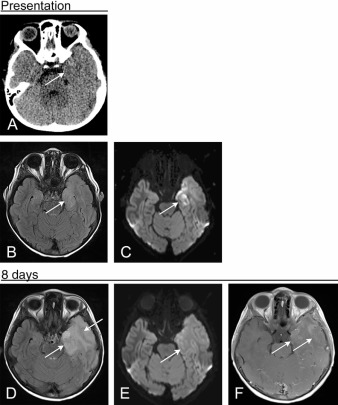
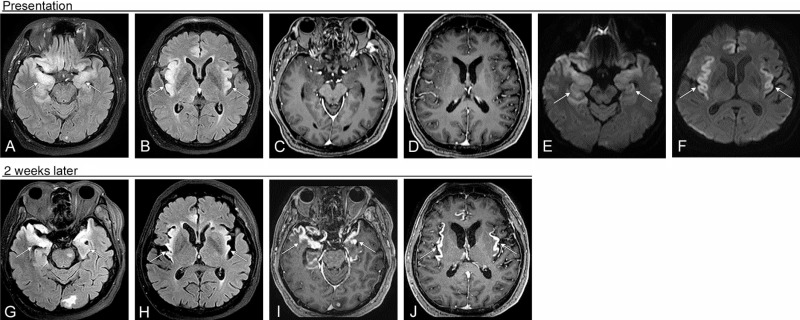
Several months after primary infection there is encephalomalacia in the affected brain, with parenchymal volume loss, frequent cystic changes, and ex vacuo dilatation of the ventricles. FDG-PET imaging performed up to months after presentation shows FDG avidity of the affected cortex. Progressive diffuse brain atrophy is a frequent finding of HSE, seen even in patients with resolving neurologic deficits.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here