Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
A 13-year-old boy presents with frequent tripping. He enjoys playing baseball but has had several falls this season, one resulting in a fractured ankle. Born at term, his early developmental milestones were normal and he walked at 13 months (normal). On examination, he has mild wasting of the distal lower extremities without contractures. He walks well on his toes but cannot walk on his heels. There is mild weakness of ankle dorsiflexion and eversion. The ankle reflexes are absent but other reflexes are preserved. Sensation is intact. There is no family history of neuromuscular disorders. His father, who accompanies him to the appointment, agrees to be examined and is found to have high-arched feet and generalized areflexia. Neurophysiologic testing of the patient and his father reveals marked slowing of ulnar nerve motor conduction (18 m/s), with absent sensory responses. Genetic testing is positive for a duplication of the PMP22 gene on chromosome 17, confirming the clinical diagnosis of Charcot-Marie-Tooth disease type 1A.
The hereditary sensory and motor neuropathies (HSMNs) are responsible for about one-third of chronic neuropathies. Also known by the eponym Charcot-Marie-Tooth disease (CMT), they affect 1 in 2500 people and are the most common inherited neurologic disorder. CMT1A, the most common form described in the foregoing vignette, is a prototypic hereditary neuropathy responsible for almost half of all CMT cases. CMT is a genetically heterogeneous disease; over 70 genes have been identified and this number is expected to rise.
Historically, CMTs were divided based on their mode of inheritance and electrophysiologic findings. The majority are autosomal-dominant; whereas X-linked and recessive cases are less common. About two-thirds of CMTs are demyelinating; the rest are primarily axonal. CMT1 refers to dominantly inherited demyelinating neuropathies, whereas type 2 (CMT2) represents dominantly inherited axonal types. Recessively inherited neuropathies are designated as type 4 (CMT4) and can be either demyelinating or axonal. The X-linked form (CMT-X) can be demyelinating or “intermediate.”
In CMT disease, both motor and sensory fibers are affected. This contrasts with purely motor or purely sensory forms of hereditary polyneuropathies—distal hereditary motor neuropathies (dHMNs) and hereditary sensory and autonomic neuropathies (HSANs) with prominent autonomic features. Less common inherited polyneuropathies are those associated with systemic genetic degenerative disorders and inborn errors of metabolism ( Table 66.1 ). Of these, peripheral nerve involvement in familial amyloid polyneuropathies (FAPs) can be an early and defining clinical feature. Unique phenotypic features of hereditary neuropathy with liability to pressure palsies (HNPP) and hereditary neuralgic amyotrophy (HNA) place them in separate categories of inherited polyneuropathies.
| Inheritance | Primary Neurophysiology | |
|---|---|---|
| Charcot-Marie-Tooth Disease and Related Neuropathies | ||
| CMT1 | AD | Demyelinating |
| CMT2 | AD or AR | Axonal |
| CMT3 | AD or AR | Demyelinating |
| CMT4 | AR | Demyelinating or axonal |
| Intermediate CMT | AD or AR | Mixed |
| CMT-X | X-linked | Mixed |
| Hereditary sensory and autonomic neuropathies (HSAN) | AD or AR | Axonal |
| Hereditary motor neuropathies (HMN) | AD, AR, or X-linked | Axonal |
| Hereditary neuropathy with liability to pressure palsies (HNPP) | AD | Demyelinating |
| Neuropathies Associated With Inborn Errors of Metabolism | ||
| Lipid Disorders | ||
| Cerebrotendinous xanthomatosis | AR | Mixed |
| Abetalipoproteinemia | AR | Mixed |
| Ataxia with vitamin E deficiency | AR | Mixed |
| Tangier disease | AR | Demyelinating, mixed |
| Refsum disease | AR | Demyelinating |
| Adrenomyeloneuropathy (AMN) | X-linked | Axonal |
| Mitochondrial Cytopathies | ||
| NARP | Mitochondrial | Mixed |
| MNGIE | AR | Mixed |
| Leigh disease | AR, mitochondrial or X-linked | Mixed |
| Lysosomal Storage Diseases | ||
| Globoid cell leukodystrophy | AR | Demyelinating |
| Metachromatic leukodystrophy | AR | Demyelinating |
| Fabry disease | X-linked | Axonal |
| Sphingomyelin Lipidoses | ||
| Niemann–Pick disease type C | AR | Demyelinating |
| Farber disease (lipogranulomatosis) | AR | Demyelinating |
| Porphyrias | ||
| Acute intermittent porphyria | AD | Axonal |
| Hereditary coproporphyria | AD | Axonal |
| Variegate porphyria | AD | Axonal |
| Familial Amyloid Polyneuropathies (FAPs) | ||
| FAP I and II (transthyretin-related) | AD | Axonal or demyelinating |
| FAP III (apolipoprotein A1-related) | AD | Axonal |
| FAP IV (gelsolin-related) | AD | Axonal |
| Disorders With Defective DNA Synthesis or Repair | ||
| Ataxia telangiectasia | AR | Axonal |
| Cockayne syndrome | AR | Demyelinating |
| Neuropathies Associated With Spinocerebellar Ataxias | ||
| Friedreich ataxia, other SCAs | AR | Axonal |
| Neuroacanthocytosis | X-linked | Axonal |
| Other Inherited Neuropathies | ||
| Hereditary neuralgic amyotrophy | AD | Axonal |
| Giant axonal neuropathy | AD | Axonal |
| Infantile neuroaxonal dystrophy | AR | Axonal |
| Andermann syndrome | AR | Axonal |
A typical patient with CMT presents in his or her first two decades with foot deformities and abnormal gait ( Fig. 66.1 ). This has been termed the “classical” phenotype. Less frequent presentations include hypotonia and delayed motor milestones in infancy (“infantile-onset” phenotype) or leg weakness in adulthood (“adult-onset” phenotype). Symptom onset is characteristically insidious and cannot be dated with certainty. Patients may recall being called “clumsy” while playing sports as children. Often reported is the history of inability to ice or roller skate, attributed to “weak ankles.” Patients rarely report sensory symptoms, and if they do, negative symptoms (i.e., numbness) prevail over positive (e.g., tingling, prickling). On examination, one finds distal weakness and wasting, absent reflexes, and impaired distal sensation. Foot deformities are common; high arches (pes cavus) and curling of the toes (hammertoes) are detected in most cases. Imbalance between weaker anterior muscles (tibialis anterior and peroneus brevis) and stronger posterior muscle group (tibialis posterior, peroneus longus, and extensor digitorum longus) is hypothesized to be responsible for the typical foot appearance in CMT. Some CMT forms come accompanied by skeletal abnormalities such as scoliosis or hip dysplasia, optic atrophy (CMT2A), or tremor (CMT1). An uncommon but interesting presentation is one in which symptoms follow exposure to neurotoxic drugs in a patient with mild or yet undiagnosed CMT; chemotherapy agents paclitaxel and vinca alkaloids are examples.
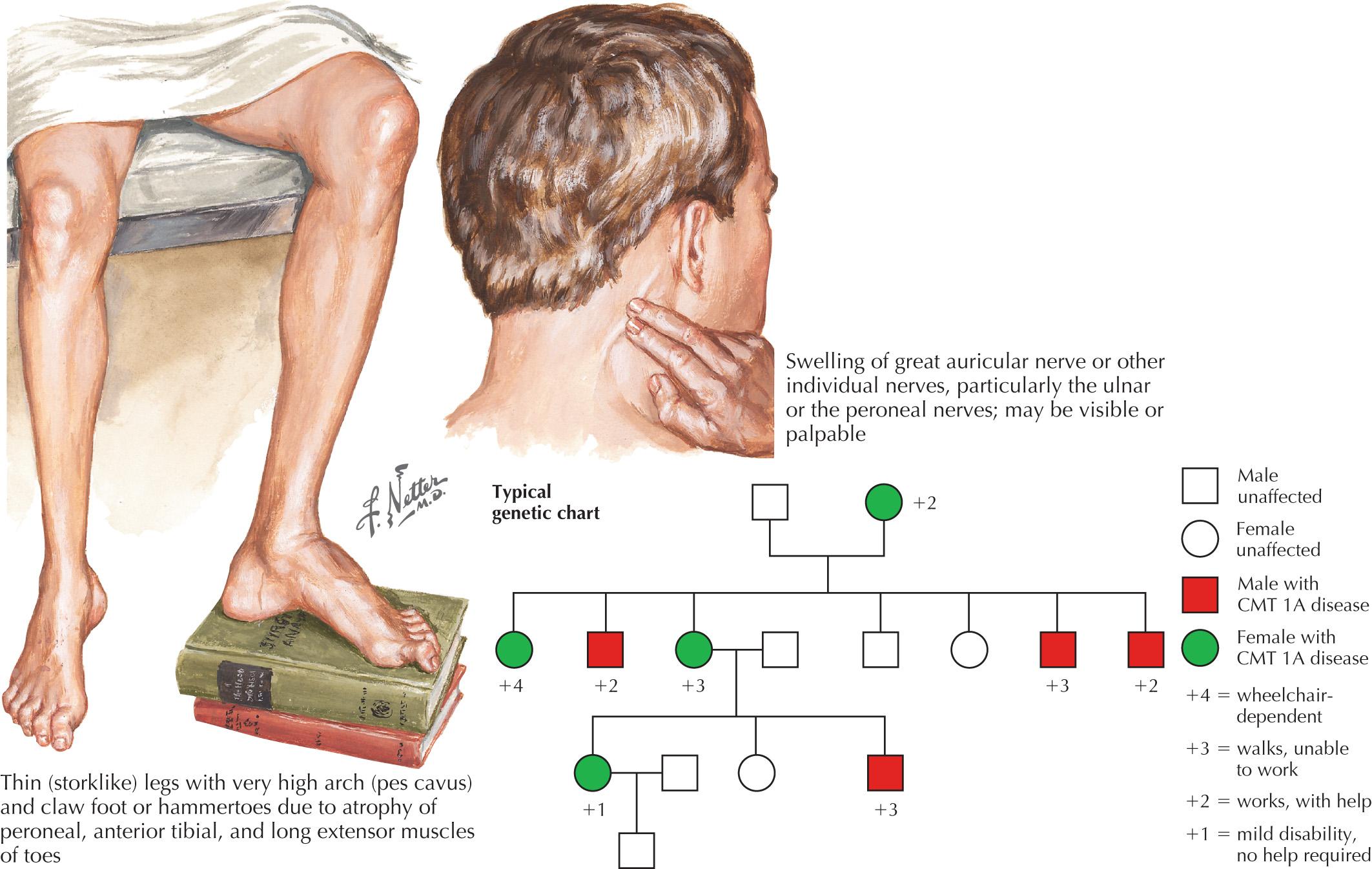
CMT1—autosomal dominant demyelinating CMT—is the most common form of CMT ( Table 66.2 ). CMT1A—the most common subtype responsible for about 70% of CMT1 and 40%–50% of all CMT cases—is considered a prototypic hereditary neuropathy. Almost all CMT1A patients become symptomatic in their first two decades with characteristic findings of pes cavus and steppage gait due to footdrop. Although sensory loss is not a common presenting complaint, reduced sensation to all modalities is often noted on exam. Nerve conduction velocity is uniformly slowed, most often in 15–35 m/s range. Nerve biopsy is rarely needed; if performed, it shows a hypertrophic “dysmyelinating” neuropathy with “onion bulb” formation. Negative family history is not uncommon; about 10% of CMT1A cases are due to sporadic mutations. In some cases that appear sporadic, one of the parents accompanying the child may have pes cavus or slow motor conduction, indicating an undiagnosed CMT. The term Roussy-Levy syndrome is applied to those CMT1A patients who have postural tremor. A small minority of CMT1A cases present as Dejerine-Sottas syndrome (DSS), a severe “infantile” phenotype with extreme conduction slowing (MCV < 15 m/s). CMT1A is caused by duplication of the peripheral protein 22 gene (PMP22) on chromosome 17. Interestingly, a heterozygous deletion of the same region causes HNPP, further discussed later. A fraction of CMT1 cases—CMT1E—is caused by point mutations in the PMP22 gene and can have a severe DSS phenotype. Of the genes associated with CMT1, myelin protein zero (MPZ) is the second most common (10%–15% of CMT1s). MPZ mutations are associated with a range of phenotypes, from the most severe infantile type (DSS), to classical CMT and milder adult-onset cases with borderline conduction slowing. The remaining CMT1 subtypes are exceedingly rare.
| % of CMT1 | Locus | Gene | Phenotype | |
|---|---|---|---|---|
| CMT1A | 60%–70% | 17p12 | PMP22 (duplication) | Classical CMT |
| CMT1B | 10%–15% | 1q23.3 | MPZ | Classical CMT Infantile-onset/DSD Adult-onset |
| CMT1C | 1%–2% | 16p13.13 | LITAF/SIMPLE | Classical CMT |
| CMT1D | <1% | 10q21.3 | EGR2 | Classical CMT Infantile-onset/DSD |
| CMT1E | 1%–2% | 17p12 | PMP22 (point mutations) | Associated with deafness Infantile-onset/DSD |
| CMT1F | <1% | 8p21 | NEFL | Infantile onset/DSD Allelic with CMT2E (axonal, adult-onset) |
| Roussy-Levy | MPZ PMP22 |
CMT1A plus tremor |
CMT2—the “axonal” dominant form of CMT—is about half as common as CMT1 ( Table 66.3 ). Electrodiagnostic testing is necessary to distinguish CMT2 from CMT1 because the clinical presentation is often similar. In general, however, CMT2 patients are less likely than those with CMT1 to be globally areflexic and they tend to present later in life. In CMT2, nerve conduction studies show normal or near normal motor velocities (>38 m/s) with reduced sensory and motor amplitudes. Unlike CMT1, the number of disease genes for CMT2 is large, all of which are very rare. CMT2A—the most common subtype responsible for 20%–30% of CMT2 cases—is caused by mutation in mitochondrial fusion protein, mitofusin-2 (MFN2). Mutations in MFN2 can be associated with severe early-life weakness, optic atrophy, and/or hearing loss, but it can cause the classical CMT phenotype in other patients. Some of the other CMT2 forms can be associated with vocal cord paralysis (TRPV4), motor deficits in the hands, GARS, or severe sensory loss with mutilating arthropathy RAB7.
| Frequency | Gene | Phenotype | |
|---|---|---|---|
| CMT2A | 20%–30% of CMT2 | MFN2 | Prominent distal weakness Optic atrophy |
| CMT2B | Multiple families | RAB7 | Ulcero-mutilating |
| CMT2C | Multiple families | TRPV4 | Vocal cord paralysis Diaphragmatic involvement |
| CMT2D | Multiple families | GARS | Hand wasting Allelic with dHMN |
| CMT2E | Multiple families | NEFL | Hearing loss Allelic with CMT1F |
| CMT2F | Multiple families | HSPB1 | Motor-predominant/dHMN |
| CMT2I CMT2J |
Multiple families | MPZ | Hearing loss/pupillary abnormalities Allelic with CMT1B |
| CMT2K | Multiple families | GDAP1 | AD or AR Allelic with CMT4A (vocal cord paralysis) |
| CMT2L | Multiple families | HSPB8 | Motor-predominant Allelic with dHMN |
| CMT2M | Multiple families | DNM2 | Tremor, ophthalmoplegia |
| CMT2N | Multiple families | AARS | Typical CMT2 Allelic with dHMN |
| CMT2O | Multiple families | DYNC1H1 | Learning disability |
| CMT2P | Multiple families | LRSAM1 | Mild; sensory-predominant |
| CMT2Q | One family | DHTKD1 | Typical CMT2 |
| CMT2U | One family One sporadic |
MARS | Late-onset |
| CMT2V | One family | NAGLU | Late-onset; painful; sensory-predominant |
| CMT2W | Multiple families | HARS | Typical CMT2 Allelic with dHMN |
| CMT2Y | One family One sporadic |
VCP | Typical CMT1 Allelic with ALS14, IBMPFD1 |
| CMT2Z | Multiple families | MORC2 | Pyramidal signs |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here