Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The hereditary periodic fever syndromes are a group of monogenic diseases that present with recurrent bouts of fever and associated pleural and/or peritoneal inflammation, arthritis, and various types of skin rash. A number of identifiable disorders present with recurrent episodes of inflammation, although fevers may not be common feature. Therefore the term systemic autoinflammatory diseases is used to include all diseases that present with seemingly unprovoked episodes of inflammation, without the high-titer autoantibodies or antigen-specific T cells typically seen in autoimmune diseases. Whereas the autoimmune diseases are disorders of the adaptive immune system, driven by B and T lymphocyte effector cells, autoinflammatory diseases largely represent disorders of the phylogenetically more primitive innate immune system, mediated by myeloid effector cells and germline-encoded receptors. Autoinflammatory diseases exhibit episodic or persistent inflammation characterized by an acute-phase response with elevation of the erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and serum amyloid A (AA). In some patients, untreated autoinflammatory disorders over time will lead to AA amyloidosis (see Chapter 189 ).
It is important to note that autoinflammatory disorders are rare, whereas fever in childhood caused by innocuous illness is very common. The approach to a child with fevers should include a detailed history, physical examination, and limited laboratory investigations to rule out other conditions that lead to fevers, including autoimmune disorders and malignancies ( Table 188.1 ). If there is evidence of recurrent infections with fevers, an immune deficiency could be considered and evaluated. If the workup is reassuring, the inflammatory episodes resolve, and the child is otherwise well without unusual physical findings, observance is often warranted because these episodes are likely to resolve as the child's immune system matures.
See Table 188.2 .
Infectious
Hidden infectious focus (e.g., aortoenteric fistula, lung sequestration)
Recurrent infection/reinfection (e.g., chronic meningococcemia, immune deficiency)
Specific infection (e.g., Whipple disease, malaria)
Noninfectious inflammatory disorder:
Adult-onset Still disease
Systemic-onset juvenile idiopathic arthritis
Periodic fever, aphthous stomatitis, pharyngitis, and adenitis
Schnitzler syndrome
Behçet syndrome
Crohn disease
Sarcoidosis
Neoplastic
Lymphoma (e.g., Hodgkin disease, angioimmunoblastic lymphoma)
Solid tumor (e.g., pheochromocytoma, myxoma, colon carcinoma)
Histiocytic disorders
Vascular (e.g., recurrent pulmonary embolism)
Hypothalamic
Psychogenic periodic fever
Factitious or fraudulent
Because of the rapidly expanding number of autoinflammatory disorders and their varied clinical presentation, it can be difficult to group these disorders in a meaningful manner. Some autoinflammatory disorders present with prominent fevers and are known as hereditary periodic fever syndromes . These include 2 disorders with an autosomal recessive mode of inheritance, familial Mediterranean fever ( FMF ; MIM249100) and the hyperimmunoglobulinemia D (hyper-IgD) with periodic fever syndrome ( HIDS ; MIM260920). Hereditary periodic fever syndromes with an autosomal dominant mode of inheritance include the tumor necrosis factor (TNF) receptor–associated periodic syndrome ( TRAPS ; MIM191190) and a spectrum of disorders known as the cryopyrin-associated periodic syndromes ( CAPS ), or cryopyrinopathies. From mildest to most severe, CAPS include the familial cold autoinflammatory syndrome ( FCAS1 ; MIM120100), Muckle-Wells syndrome ( MWS ; MIM191100), and neonatal-onset multisystem inflammatory disease ( NOMID ; MIM607115) (also known as chronic infantile neurologic cutaneous and articular syndrome, CINCA ) ( Table 188.2 ).
| DISEASE | GENETIC DEFECT/PRESUMED PATHOGENESIS | INHERITANCE | AFFECTED CELLS | FUNCTIONAL DEFECTS | ASSOCIATED FEATURES |
|---|---|---|---|---|---|
| Familial Mediterranean fever | Mutations of MEFV (lead to gain of pyrin function, resulting in inappropriate IL-1β release) | AR | Mature granulocytes, cytokine-activated monocytes | Decreased production of pyrin permits ASC-induced IL-1 processing and inflammation following subclinical serosal injury; macrophage apoptosis decreased | Recurrent fever, serositis, and inflammation responsive to colchicine. Predisposes to vasculitis and inflammatory bowel disease |
| Mevalonate kinase deficiency (hyper IgD syndrome) | Mutations of MVK (lead to a block in the mevalonate pathway). Interleukin-1β mediates the inflammatory phenotype | AR | Affecting cholesterol synthesis; pathogenesis of disease is unclear | Periodic fever and leukocytosis with high IgD levels | |
| Muckle–Wells syndrome | Mutations of NLRP3 (also called PYPAF1 or NALP3 ) lead to constitutive activation of the NLRP3 inflammasome | AD | PMNs, monocytes | Defect in cryopyrin, involved in leukocyte apoptosis and NF-κB signaling and IL-1 processing | Urticaria, SNHL, amyloidosis |
| Familial cold autoinflammatory syndrome | Mutations of NLRP3 (see above) Mutations of NLRP12 |
AD | PMNs, monocytes | Same as above | Nonpruritic urticaria, arthritis, chills, fever, and leukocytosis after cold exposure |
| Neonatal-onset multisystem inflammatory disease (NOMID) or chronic infantile neurologic cutaneous and articular syndrome (CINCA) | Mutations of NLRP3 (see above) | PMNs, chondrocytes | Same as above | Neonatal-onset rash, chronic meningitis, and arthropathy with fever and inflammation | |
| TNF receptor–associated periodic syndrome (TRAPS) | Mutations of TNFRSF1A (resulting in increased TNF inflammatory signaling) | AD | PMNs, monocytes | Mutations of 55-kDa TNF receptor leading to intracellular receptor retention or diminished soluble cytokine receptor available to bind TNF | Recurrent fever, serositis, rash, and ocular or joint inflammation |
| Pyogenic sterile arthritis, pyoderma gangrenosum, acne (PAPA) syndrome | Mutations of PSTPIP1 (also called C2BP1 ) (affects both pyrin and protein tyrosine phosphatase to regulate innate and adaptive immune responses) | AD | Hematopoietic tissues, upregulated in activated T cells | Disordered actin reorganization leading to compromised physiologic signaling during inflammatory response | Destructive arthritis, inflammatory skin rash, myositis |
| Blau syndrome | Mutations of NOD2 (also called CARD15 ) (involved in various inflammatory processes) | AD | Monocytes | Mutations in nucleotide binding site of CARD15, possibly disrupting interactions with lipopolysaccharides and NF-κB signaling | Uveitis, granulomatous synovitis, campodactyly, rash, and cranial neuropathies, 30% develop Crohn disease |
| Chronic recurrent multifocal osteomyelitis and congenital dyserythropoietic anemia (Majeed syndrome) | Mutations of LPIN2 (increased expression of the proinflammatory genes) | AR | Neutrophils, bone marrow cells | Undefined | Chronic recurrent multifocal osteomyelitis, transfusion-dependent anemia, cutaneous inflammatory disorders |
| Early-onset inflammatory bowel disease | Mutations in IL-10 (results in increase of many proinflammatory cytokines) | AR | Monocyte/macrophage, activated T cells | IL-10 deficiency leads to increase of TNFγ and other proinflammatory cytokines | Enterocolitis, enteric fistulas, perianal abscesses, chronic folliculitis |
| Early-onset inflammatory bowel disease | Mutations in IL-10RA (see above) | AR | Monocyte/macrophage, activated T cells | Mutation in IL-10 receptor alpha leads to increase of TNFγ and other proinflammatory cytokines | Enterocolitis, enteric fistulas, perianal abscesses, chronic folliculitis |
| Early-onset inflammatory bowel disease | Mutations in IL-10RB (see above) | AR | Monocyte/macrophage, activated T cells | Mutation in IL-10 receptor beta leads to increase of TNFγ and other proinflammatory cytokines | Enterocolitis, enteric fistulas, perianal abscesses, chronic folliculitis |
A variety of mendelian autoinflammatory disorders may or may not exhibit prominent fevers and are not considered periodic fever syndromes, but do have continuous or repeated episodes of spontaneous inflammation with unique clinical characteristics. These include the syndrome of pyogenic arthritis with pyoderma gangrenosum and acne ( PAPA ; MIM604416), deficiency of the interleukin-1 (IL-1) receptor antagonist ( DIRA ; MIM612852), Blau syndrome caused by mutations in NOD2 (also known as early-onset sarcoidosis ; MIM186580), autoinflammation with phospholipase Cγ 2 -associated antibody deficiency and immune dysregulation ( APLAID ; MIM614878), and deficiency of adenosine deaminase-2 ( DADA2 ). Other disorders include congenital sideroblastic anemia with B-cell immunodeficiency, periodic fevers, and developmental delay ( SIFD ) caused by biallelic mutations of the TRNT1 gene (MIM616084), autoinflammation with infantile enterocolitis caused by mutations in NLRC4 ( AIFEC ; MIM616060), familial cold autoinflammatory syndrome type 2 caused by mutations in NLRP12 ( FCAS2 ; MIM611762), CARD14 (MIM607211), and deficiency in IL-36 receptor antagonist ( DITRA ; 614204).
In addition to the previous autoinflammatory disorders, a variety of disorders are characterized by inappropriate interferon expression , the interferonopathies . Type 1 interferons (e.g., IFN-α, IFN-β) are cytokines expressed by many cells in response to viral infections. Disorders that result in spontaneous interferon production and inflammatory manifestations include STING-associated vasculopathy of infancy ( SAVI ; MIM615934) and chronic atypical neutrophilic dermatosis with lipodystrophy and elevated temperature ( CANDLE ; MIM256040).
There are also a number of autoinflammatory disorders with a complex mode of inheritance. These include the syndrome of periodic fever with aphthous stomatitis, pharyngitis, and adenitis ( PFAPA ) and chronic recurrent multifocal osteomyelitis ( CRMO ; MIM259680). Other genetically complex disorders that are sometimes considered autoinflammatory include systemic-onset juvenile idiopathic arthritis (see Chapter 180 ), Behçet disease (see Chapter 186 ), and Crohn disease (see Chapter 362.2 ).
Distinguishing autoinflammatory disorders from one another can be difficult because their presentations can vary, and many display similarities. Some disorders have characteristic fever patterns ( Fig. 188.1 ), whereas others have characteristic skin findings that can aid in a diagnosis ( Table 188.3 ). Others can have characteristic physical features or organ involvement. Some of these disorders have bone involvement ( Table 188.4 ). Other clinical features can also be helpful, such as ethnicity, age of onset, triggers, laboratory testing, and response to therapies ( Table 188.5 ). Genetic panels are increasingly being used to screen for most if not all of these defects in a single test, rather than individual genetic assessment based on clinical findings.
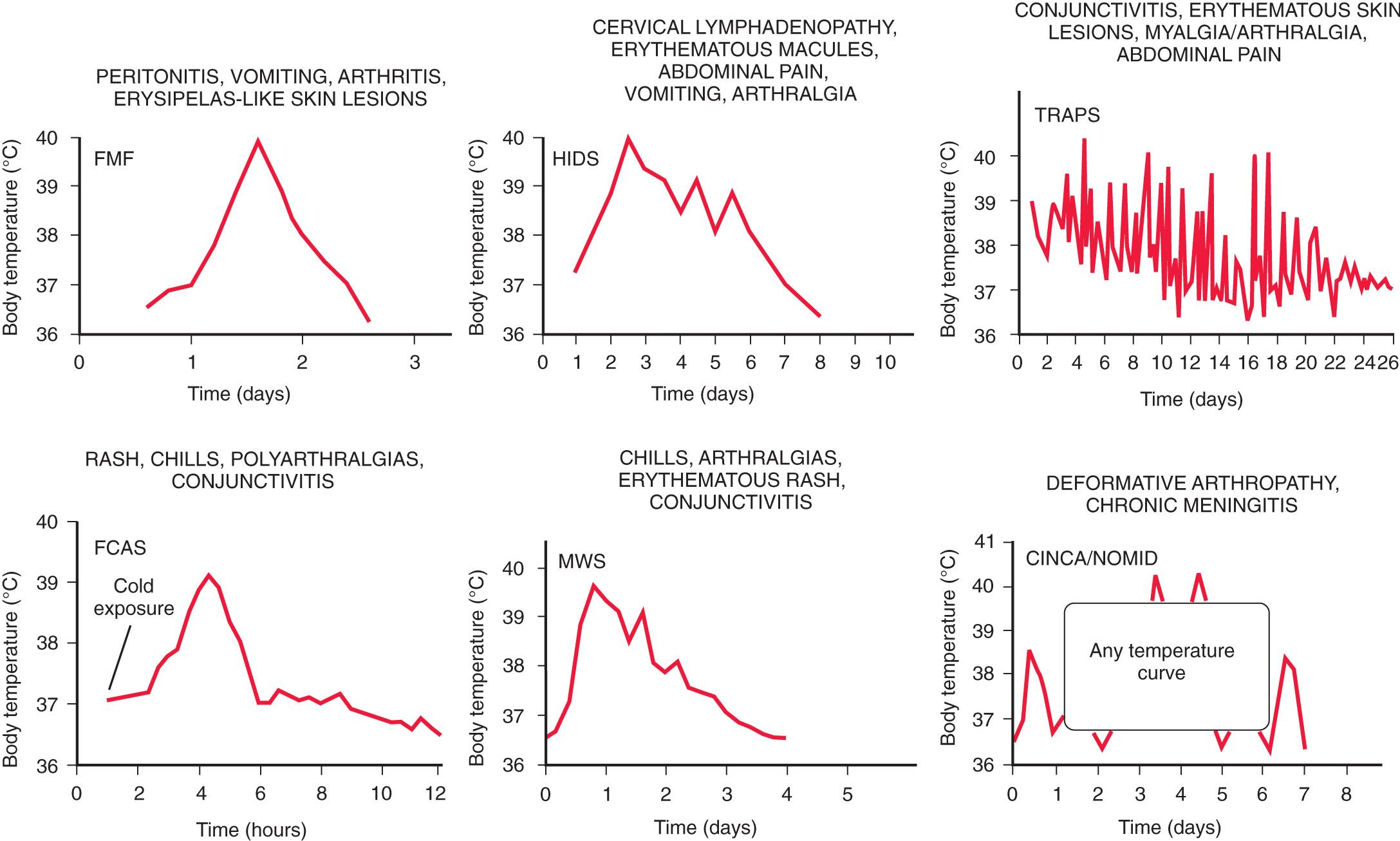
Neutrophilic urticaria (the cryopyrinopathies)
Recurrent fever attacks of short duration (typically <24 hr)
CAPS/FCAS: familial cold autoinflammatory syndrome
CAPS/MWS: Muckle-Wells syndrome
FCAS2/NLRP12
Continuous low-grade fever
CAPS/NOMID: neonatal-onset multisystem inflammatory disease (NOMID)/chronic infantile neurologic cutaneous and articular syndrome (CINCA)
Granulomatous skin lesions and minimal or low-grade fever attacks
Blau syndrome/early-onset sarcoidosis (pediatric granulomatous arthritis)
Pustular skin rashes and fever
With inflammatory bone disease
DIRA: deficiency of interleukin-1 receptor agonist
Majeed syndrome
With pyogenic arthritis
PAPA: pyogenic arthritis, pyoderma gangrenosum, and acne syndrome
Without other organ involvement
DITRA: deficiency of interleukin-36 receptor antagonist
CAMPS: CARD14-mediated psoriasis
Atypical neutrophilic dermatosis with histiocytic-like infiltrate
CANDLE: proteasome associated autoinflammatory syndromes
Livedo reticularis, vasculopathy with ulcerations
SAVI; STING associated vasculopathy, infantile onset
Livedo racemosa, vasculitis with ulcerations
ADA2; adenosine deaminase-2 deficiency
CAPS, Cryopyrin-associated periodic syndromes.
| CRMO | MAJEED SYNDROME | DIRA | CHERUBISM | CMO AND LUPO MICE | |
|---|---|---|---|---|---|
| Ethnicity | Worldwide, but mostly European | Arabic | European, Puerto Rican, Arabic | Worldwide | Occurs in various backgrounds |
| Fever | Uncommon | Common | Uncommon | No | Not assessed |
| Sites of osseous involvement | Metaphyses of long bones > vertebrae, clavicle, sternum, pelvis, others | Similar to CRMO | Anterior rib ends, metaphyses of long bones, vertebrae, others | Mandible > maxilla Rarely ribs |
Vertebrae hind > forefeet |
| Extraosseous manifestations | PPP, psoriasis, IBD, others | Dyserythropoietic anemia, Sweet syndrome, HSM, growth failure | Generalized pustulosis, nail changes, lung disease, vasculitis | Cervical lymphadenopathy | Dermatitis, extramedullary hematopoiesis, splenomegaly |
| Family history of inflammatory disorders | Psoriasis, PPP, arthritis, IBD, others | Psoriasis in some obligate carriers | No known associations | No known associations | Heterozygotes normal |
| Inheritance | Not clear | Autosomal recessive | Autosomal recessive | Autosomal dominant; incomplete penetrance | Autosomal recessive |
| Gene defect | Unknown | LPIN2 | IL1RN | SH3BP2 >> PTPN11 | Pstpip2 |
| Protein name | ? | Lipin2 | IL-1Ra | SH3BP2 | PSTPIP2 (MAYP) |
| Protein function | ? | Fat metabolism: (PAP enzyme activity), ↑ message to oxidative stress, ? role in mitosis | Antagonist of IL-1 receptor | ↑ Myeloid cell response to M-CSF and RANKL, ↑ TNF-α expression in macrophages | Macrophage proliferation, macrophage recruitment to sites of inflammation, cytoskeletal function |
| Cytokine abnormalities | ↑ serum TNF-α | Not tested | ↑ IL-1α, IL-1β, MIP-1α, TNF-α, IL-8, IL-6 ex vivo monocyte assay; skin reveals ↑ IL-17 staining | ↑ serum TNF-α in mouse model | cmo: ↑ serum IL-6, MIP-1α, TNF-α, CSF-1, IP-10 Lupo: ↑ serum MIP-1α, IL-4, RANTES, TGF-β |
| AGE OF ONSET | |
| At birth | NOMID, DIRA, MWS |
| Infancy and 1st yr of life | HIDS, FCAS, NLRP12 |
| Toddler | PFAPA |
| Late childhood | PAPA |
| Most common of autoinflammatory syndromes to have onset in adulthood | TRAPS, DITRA |
| Variable (mostly in childhood) | All others |
| ETHNICITY AND GEOGRAPHY | |
| Armenians, Turks, Italian, Sephardic Jews | FMF |
| Arabs | FMF, DITRA (Arab Tunisian) |
| Dutch, French, German, Western Europe | HIDS, MWS, NLRP12 |
| United States | FCAS |
| Can occur in blacks (West Africa origin) | TRAPS |
| Eastern Canada, Puerto Rico | DIRA |
| Worldwide | All others |
| TRIGGERS | |
| Vaccines | HIDS |
| Cold exposure | FCAS, NLRP12 |
| Stress, menses | FMF, TRAPS, MWS, PAPA, DITRA |
| Minor trauma | PAPA, MWS, TRAPS, HIDS |
| Exercise | FMF, TRAPS |
| Pregnancy | DITRA |
| Infections | All, especially DITRA |
| ATTACK DURATION | |
| <24 hr | FCAS, FMF |
| 1-3 days | FMF, MWS, DITRA (fever) |
| 3-7 days | HIDS, PFAPA |
| >7 days | TRAPS, PAPA |
| Almost always “in attack” | NOMID, DIRA |
| INTERVAL BETWEEN ATTACKS | |
| 3-6 wk | PFAPA, HIDS |
| >6 wk | TRAPS |
| Mostly unpredictable | All others |
| Truly periodic | PFAPA, cyclic neutropenia |
| USEFUL LABORATORY TESTS | |
| Acute-phase reactants must be normal between attacks | PFAPA |
| Urine mevalonic acid in attack | HIDS |
| IgD > 100 mg/dL | HIDS |
| Proteinuria (amyloidosis) | FMF, TRAPS, MWS, NOMID |
| RESPONSE TO THERAPY | |
| Corticosteroid dramatic | PFAPA |
| Corticosteroid partial | TRAPS, FCAS, MWS, NOMID, PAPA * |
| Colchicine | FMF, PFAPA (30% effective) |
| Cimetidine | PFAPA (30% effective) |
| Etanercept | TRAPS, FMF arthritis |
| Anti–IL-1 dramatic | DIRA (anakinra), FCAS, MWS, NOMID, PFAPA |
| Anti–IL-1 mostly | TRAPS, FMF |
| Anti–IL-1 partial | HIDS, PAPA |
The first descriptions of autoinflammatory disorders focused on genetic diseases that presented with prominent fevers, the periodic fever syndromes. As new autoinflammatory diseases were discovered, it was clear that a variety of inflammatory disorders can occur in the absence of fever.
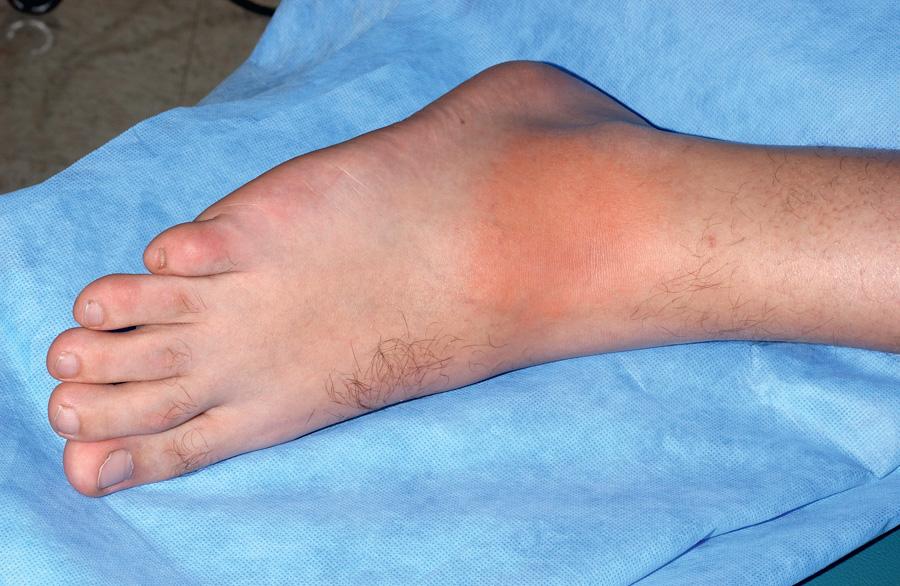
FMF is a recessively inherited autoinflammatory disease usually characterized by recurrent, short-lived (1-3 days), self-limited episodes of fever, serositis, mono- or pauciarticular arthritis, or an erysipeloid rash, sometimes complicated by AA amyloidosis. Most patients with FMF present with symptoms in childhood, with 90% presenting before age 20. Clinical features of FMF may include fever, serositis presenting as pleuritic chest pain or severe abdominal pain, arthritis, and rash. The pleural pain is typically unilateral, whereas the abdominal pain (sterile peritonitis) can be generalized or localized to 1 quadrant, similar to other forms of peritonitis. FMF-associated arthritis occurs primarily in the large joints, may be accompanied by large, neutrophil-rich effusions, and is usually nonerosive and nondestructive. The hallmark cutaneous finding is an erysipeloid erythematous rash that overlies the ankle or dorsum of the foot ( Fig. 188.2 ). Other clinical findings include scrotal pain caused by inflammation of the tunica vaginalis testis, febrile myalgia, exercise-induced myalgia (particularly common in children), and an association with various forms of vasculitis, including Henoch-Schönlein purpura, in as many as 5% of pediatric patients. FMF episodes may be triggered by stress, menses, or infections. Between flares, patients are generally symptom free but may have persistent elevation of their inflammatory markers. The attack frequency can vary from weekly to 1-2 flares per year. Table 188.6 lists diagnostic criteria for FMF.
* Requirements for diagnosis of FMF are ≥1 major criteria or ≥2 minor criteria.
Typical attacks †
† Typical attacks are defined as recurrent (≥3 of the same type), febrile (≥38°C), and short (lasting between 12 hr and 3 days).
with peritonitis (generalized)
Typical attacks with pleuritis (unilateral) or pericarditis
Typical attacks with monoarthritis (hip, knee, ankle)
Typical attacks with fever alone
Incomplete abdominal attack
Incomplete attacks ‡
‡ Incomplete attacks are defined as painful and recurrent attacks not fulfilling the criteria for a typical attack.
involving chest pain
Incomplete attacks involving monoarthritis
Exertional leg pain
Favorable response to colchicine
FMF is caused by autosomal recessive mutations in MEFV , a gene encoding a 781 amino acid protein denoted pyrin (Greek for “fever”). Pyrin is expressed in granulocytes, monocytes, and dendritic cells (DCs) and in peritoneal, synovial, and dermal fibroblasts. The N-terminal approximately 90 amino acids of pyrin are the prototype for a motif (the PYRIN domain) that mediates protein-protein interactions and is found in >20 different human proteins that regulate inflammation and apoptosis. Many of the FMF-associated mutations in pyrin are found at the C-terminal B30.2 domain of pyrin, encoded by exon 10 of MEFV. More than 50 such FMF mutations are listed in an online database ( http://fmf.igh.cnrs.fr/ISSAID/infevers/ ), almost all of which are missense substitutions. Homozygosity for the M694V mutation may be associated with an earlier age of onset, arthritis, and an increased risk of amyloidosis. The substitution of glutamine for glutamic acid at residue 148 (E148Q) is considered either a mild mutation or a functional polymorphism in the pyrin protein. The carrier frequency of FMF mutations among several Mediterranean populations is very high, suggesting the possibility of a heterozygote advantage.
FMF occurs primarily among ethnic groups of Mediterranean ancestry, most frequently Jews, Turks, Armenians, Arabs, and Italians. Because of a higher frequency of the M694V mutation, FMF is more severe and more readily recognized in the Sephardic (North African) than the Ashkenazi (East European) Jewish population. With the advent of genetic testing, mutation-positive FMF has been documented worldwide, although at lower frequency than in the Mediterranean basin and Middle East.
Through PYRIN-domain interactions, pyrin can activate caspase-1 , the enzyme that converts the 31 kDa pro–IL-1β molecule into the biologically active 17 kDa IL-1β, which is a major mediator of fever and inflammation. FMF mutations lead to a gain-of-function activation of caspase-1 and IL-1β–dependent inflammation, with a gene-dosage effect. These results may explain why as many as 30% of heterozygous carriers of FMF mutations have biochemical evidence of inflammation.
Prophylactic daily oral colchicine decreases the frequency, duration, and intensity of FMF flares. This regimen also prevents the development of systemic AA amyloidosis. Colchicine is generally well tolerated and safe in children, with the most common side effects being diarrhea and other gastrointestinal (GI) complaints. Some patients develop lactose intolerance while taking colchicine. GI side effects can be minimized by initiating therapy at a low dose (for young children, 0.3 mg/day) and slowly titrating upward. A dose-related transaminitis may also be observed; bone marrow suppression is rarely seen at the dosages prescribed for FMF. Pediatric patients may require doses of colchicine similar to those needed in adults (1-2 mg/day), reflecting that children metabolize the drug more rapidly than adults. It is not always possible to find a tolerated dose of colchicine at which all symptoms are suppressed, but approximately 90% of patients have a marked improvement in disease-related symptoms. A small percentage of FMF patients are either unresponsive to or intolerant of therapeutic doses of colchicine. Based on the role of pyrin in IL-1β activation, a trial demonstrated the safety and effectiveness of rilonacept, an IL-1 inhibitor, in FMF; there are case reports of the effectiveness of anakinra, a recombinant interleukin-1 receptor (IL-1R) antagonist.
Amyloidosis is the most serious complication of FMF, and in its absence FMF patients may live a normal life span. Amyloidosis may develop when serum AA, an acute-phase reactant found at extremely high levels in the blood during FMF attacks, is cleaved to produce a 76–amino acid fragment that misfolds and deposits ectopically, usually in the kidneys, GI tract, spleen, lungs, testes, thyroid, and adrenals. Rarely, cardiac amyloidosis may develop; macroglossia and amyloid neuropathy are generally not seen with the amyloidosis of FMF. The most common presenting sign of AA amyloidosis is proteinuria. The diagnosis is then usually confirmed by rectal or renal biopsy. In a small number of case reports, mostly from the Middle East, amyloidosis may actually precede overt FMF attacks, presumably because of subclinical inflammation. Risk factors for the development of amyloidosis in FMF include homozygosity for the M694V MEFV mutation, polymorphisms of the serum AA gene (encoding AA), noncompliance with colchicine treatment, male gender, and a positive family history of AA amyloid. For unclear reasons, country of origin is also a major risk factor for amyloidosis in FMF, with patients raised in the Middle East having a much higher risk than genotypically identical patients raised in the West. Aggressive lifelong suppression of the acute-phase reactants should be the goal in patients with FMF amyloidosis, and documented cases show this may result in resorption of amyloid deposits. The natural history of untreated amyloidosis in FMF is the inexorable progression to renal failure, often within 3-5 yr.
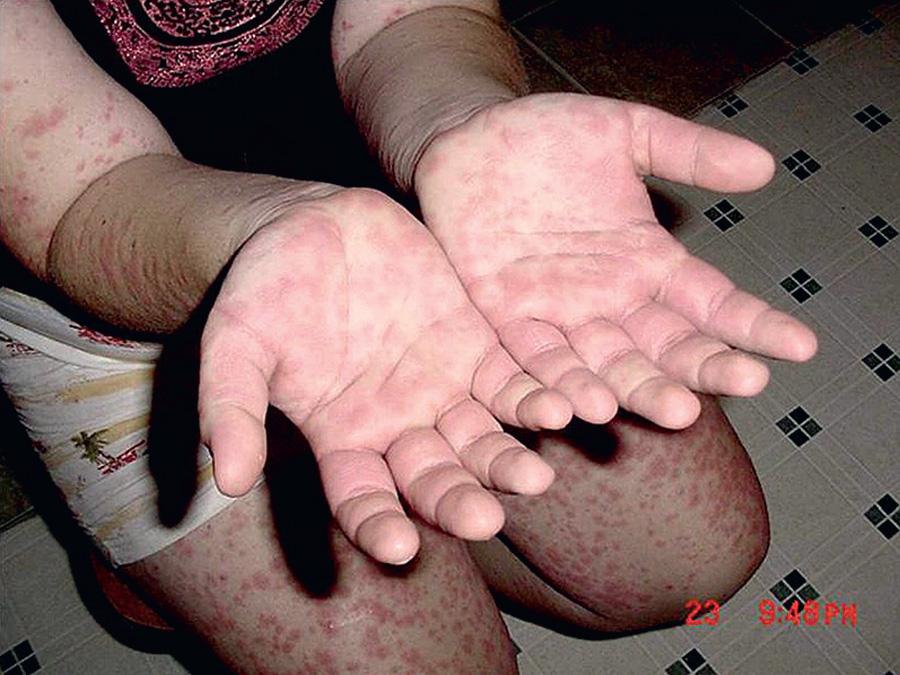
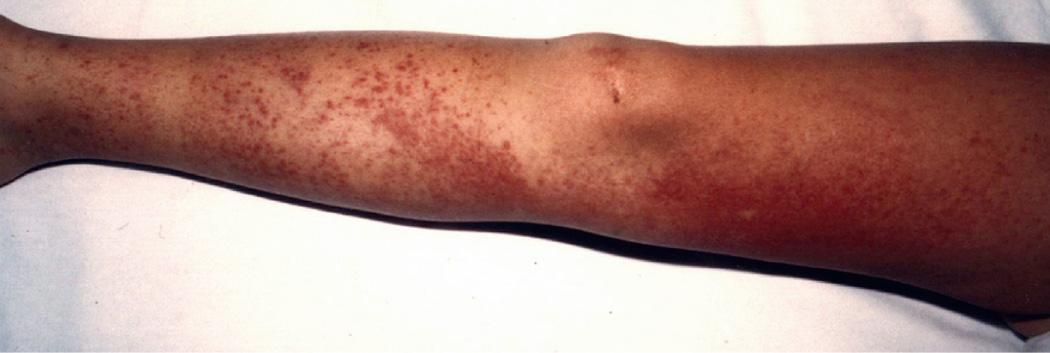
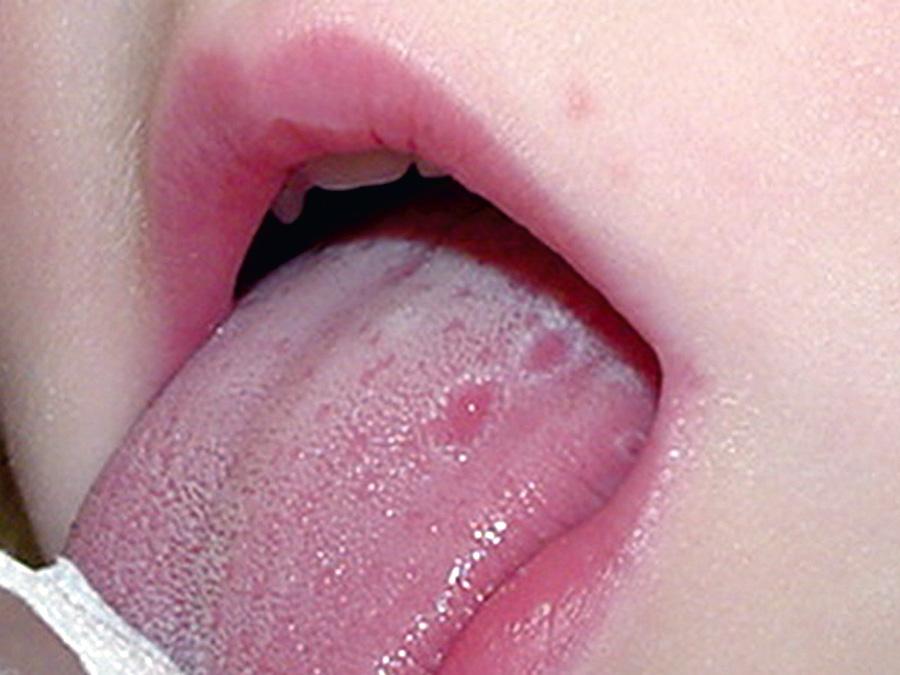
HIDS, also known as mevalonate kinase deficiency , was initially described in a cohort of Dutch patients and occurs primarily in patients of Northern European descent. HIDS is recessively inherited and caused by mutations of MVK , a gene that encodes mevalonate kinase (MK). The clinical features of HIDS generally appear within the 1st 6 mo of life. Febrile attacks last 3-7 days, with abdominal pain often accompanied by diarrhea, nausea, and vomiting. Other clinical manifestations include cervical lymphadenopathy, diffuse macular rash, aphthous ulcers, headaches, and occasional splenomegaly ( Figs. 188.3 to 188.5 ). Arthritis or arthralgia can be present in an oligoarticular or polyarticular pattern. Inflammatory disease–like illness and Kawasaki disease–like presentation have also been reported. Attacks are often precipitated by intercurrent illness, immunizations, and surgery. Families frequently recount flares around the time of birthdays, holidays, and family vacations. The symptoms of HIDS may persist for years but tend to become less prominent in adulthood. Patients with HIDS usually have a normal life span. Unlike FMF and TRAPS, the incidence of AA amyloidosis is quite low. Complete MK deficiency results in mevalonic aciduria that presents with severe mental retardation, ataxia, myopathy, cataracts, and failure to thrive (see Chapter 103 ).
MK is expressed in multiple tissues and catalyzes the conversion of mevalonic acid to 5-phosphomevalonic acid in the biosynthesis of cholesterol and nonsterol isoprenoids. Patients with HIDS-associated mutations have greatly reduced, but not absent, MK enzymatic activity. HIDS patients usually have low-normal serum cholesterol levels, but the deficiency of isoprenoids may cause increased IL-1β production by aberrant activation of the small guanosine triphosphatase Rac1. Temperature elevation may further exacerbate this process by more complete inhibition of MK activity, leading to a possible positive feedback loop.
The diagnosis of HIDS may be confirmed either by 2 mutations in MVK (approximately 10% of patients with seemingly typical disease have only a single identifiable mutation) or by elevated levels of mevalonate in the urine during acute attacks. HIDS-associated mutations are distributed throughout the MK protein, but the 2 most common mutations are the substitution of isoleucine for valine at residue 377 (V377I), a variant that is quite common in the Dutch population, and the substitution of threonine for isoleucine at residue 268 (I268T). The eponymous elevation in serum IgD levels is not universally present, especially in young children; IgA levels can also be elevated. Conversely, serum IgD levels may be increased in other autoinflammatory disorders as well as in some chronic infections. During attacks, leukocytosis and increased serum levels of acute-phase reactants and proinflammatory cytokines are frequently present. Table 188.7 lists diagnostic criteria for HIDS.
Elevated erythrocyte sedimentation rate and leukocytosis
Abrupt onset of fever (≥38.5°C)
Recurrent attacks
Lymphadenopathy (especially cervical)
Abdominal distress (e.g., vomiting, diarrhea, pain)
Skin manifestations (e.g., erythematous macules and papules)
Arthralgias and arthritis
Splenomegaly
Elevated IgD (above upper limit of normal) measured on 2 occasions at least 1 mo apart *
* Extremely high serum concentrations of IgD are characteristic but not obligatory.
Elevated IgA (≥2.6 g/L)
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here