Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Drug-induced liver injury: a penalty for progress. Hans Popper, MD
Drug-induced liver injury (DILI) represents a spectrum of acute and chronic forms of hepatic injury that can mimic virtually any liver disease. The diagnosis of hepatic drug injury is challenging mainly because it is a diagnosis of exclusion, layered on top of pre-existing illness. No specific biomarker exists for DILI, so the diagnosis rests on clinical suspicion, knowledge of the clinical-pathological patterns of injury associated with the suspected agent(s) and a careful exclusion of other causes. More than a half-century ago, Professor Hans Popper, the father of modern hepatology, referred to the adverse reactions to drugs as a ‘penalty for progress’. In the intervening decades, medical progress has been matched by an increase in penalties from adverse reactions to the many new medications and their expanded use by the general population. Indeed, it is estimated that more than 1000 medicinal, chemical and herbal agents can produce hepatic injury. ,
Drugs are important and not uncommon causes of liver injury. Indeed, drugs are the major cause of acute liver failure in the Western world and DILI is one of the most frequent causes of withdrawal, restrictions on use and failure to move forward in the development of a new drug products. , DILI accounts for 1–5% of hospital admissions for jaundice and approximately 10% of cases of unexplained marked serum enzyme elevations. Importantly, cases of DILI are more likely to undergo liver biopsy in attempts to make a secure diagnosis than routine cases of acute viral hepatitis or choledochal lithiasis in which noninvasive diagnostic tests are reliable. Drugs are also an important cause of chronic liver disease and can resemble autoimmune hepatitis (AIH), alcoholic liver disease and nonalcoholic steatohepatitis (NASH). Drugs and toxins are the primary and perhaps sole cause of sinusoidal obstruction syndrome (SOS, formerly referred to as veno-occlusive disease [VOD]) and acute fatty liver with microvesicular steatosis. Drugs are also likely causes of nodular regenerative hyperplasia (NRH) and peliosis hepatis. Finally, drugs are a well-proven cause, although rare, of benign and malignant tumours of the liver, including hepatocellular adenoma (HCA), angiosarcoma and hepatocellular carcinoma (HCC).
The incidence of clinically apparent DILI is not well defined. In population-based surveys from France and Iceland, the incidence was estimated to be 13 to 19 per 100,000. A recent epidemiological study attempted to capture all DILI cases in the state of Delaware and recorded an annual incidence of 2.7 cases per 100,000 persons. The causes of DILI vary geographically, reflecting what drugs are available and the relative roles of traditional versus conventional (Western) medical practice. In the United States and Western Europe, the most common causes of clinically apparent liver injury from medications have been antibiotics, particularly amoxicillin-clavulanic acid, isoniazid (or isonicotinic acid hydrazide, INH), nitrofurantoin and various macrolide and fluoroquinolone antibiotics ( Table 12.1 ). In Asian countries, herbal medications and traditional Chinese or Ayurvedic medications tend to be more frequent causes of liver injury, although drugs for tuberculosis (TB) and antibiotics are also frequent causes. In recent years, herbal and dietary supplements (HDSs) have become more common causes of clinically apparent liver injury in the United States and Latin America. ,
| Andrade (Spain) | Bjornsson (Sweden) | DILIN (United States) | Devarbhavi (India) | |
|---|---|---|---|---|
| No. of patients | 461 | 784 | 899 | 313 |
| Time period | 1994–2004 | 1970–2004 | 2003–2013 | 1997–2008 |
| Most common drugs | ||||
| Anti-infectives | 32% | 27% | 45.4% | 70% |
| Amoxicillin-clavulanate Isoniazid, pyrazinamide, rifampicin Trovafloxacin |
Flucloxacillin Erythrocycin Isoniazid Trimethoprim-sulfamethoxazole Ciprofloxacin Dicloxacillin |
Amoxicillin-clavulanate Isoniazid Nitrofurantoin Trimethoprim-sulfamethoxazole Minocycline Cefazolin |
Anti-TB drugs Dapsone Trimethoprim-sulfamethoxazole Amoxicillin-clavulanate |
|
| CNS agents | 17% | 13% | 9% | 16% |
| Carbamazepine Paroxetine Valproate |
Carbamazepine Chlorpromazine |
Phenytoin Lamotrigine Duloxetine Valproate |
Phenytoin Olanzapine Carbamazepine Valproate |
|
| Musculoskeletal | 17% | 5% | 10% | 5% |
| Ibuprofen Diclofenac Nimesulide |
Diclofenac Naproxen Sulindac |
Diclofenac Allopurinol |
Nimesulide Diclofenac Ibuprofen |
|
| Herbals and dietary supplements | 2% | 16% | 1.3% | |
| Death or liver transplantation | 5.2% | 9.2% | 6.2% | 17% |
| Chronicity * | 10.3% | 17% | 4.2% |
Risk factors are often mentioned in discussing DILI, but they probably play a relatively minor role. Thus, liver injury from medications is claimed to be more common in elderly persons and women. , However, these groups are more likely to take medications, and in population-based studies, age and sex have little effect. Black Americans may incur greater injury than White Americans although the spectrum of drugs taken differs. Risk factors may be important for certain drugs. Thus, alcohol use appears to predispose to acute liver injury from antituberculosis medications and methotrexate, and excessive alcohol use is a well-established risk factor for acetaminophen (paracetamol) toxicity. Pre-existing hepatic disease is generally not regarded as increasing the risk of idiosyncratic DILI, but there may be exceptions. Importantly, some medications may exacerbate the underlying liver disease, such as antiretroviral therapy that can cause acute exacerbations of chronic hepatitis B or C. Drugs that cause weight gain (such as corticosteroids or atypical antipsychotic agents) can worsen an underlying NASH. Oestrogens and androgens may cause accelerated growth of HCAs or HCCs. Furthermore, the underlying liver disease may alter the clinical expression and pattern of injury as well as complicating the determination of a drug-induced injury. In general, risk factors may be important for specific agents but are not very predictive of clinically apparent DILI.
Agents that can produce hepatic injury have been classically divided into two main categories: intrinsic (predictable or true) hepatotoxins and idiosyncratic (unpredictable) hepatotoxins that produce liver injury only in a small proportion of individuals who are unusually susceptible ( Table 12.2 ). The pathophysiological mechanisms by which drugs injure the liver generally involve their interacting with or disrupting cellular targets ( Table 12.3 ). , The concepts defining idiosyncrasy, dose dependency and latency periods have undergone incremental changes over the last two decades, and our understanding of direct and indirect hepatotoxicity has been modified by advances in hepatotoxicity mechanisms at both the subcellular and cytokine release levels, as will be discussed later in the chapter. Identification of toxic intermediaries producing oxidative and other forms of intracellular stress continues and offers new insights into the expanding spectrum of DILI ( Fig. 12.1 ). ,
| Category | Incidence on exposure | Experimental reproducibility | Dose dependent | Mechanism | Histological lesion | Examples |
|---|---|---|---|---|---|---|
| Direct, intrinsic | Direct destruction by peroxidation in: | |||||
| Cytotoxic | High | Easy | Yes | Hepatocytes | Necrosis and/or steatosis | Carbon tetrachloride, phosphorus |
| Cholestatic | High | Easy | Yes | Ductal cells | Cholestasis Duct destruction |
Paraquat |
| Indirect, intrinsic | Interference with specific pathways or production of selective lesions in: | |||||
| Cytotoxic | High | Easy | Yes | Hepatocytes | Necrosis and/or steatosis | Acetaminophen |
| Cholestatic | High | Easy | Yes | Ductal cells | Cholestasis Duct destruction |
Methylene-dianiline |
| Host idiosyncrasy | ||||||
| Immunological | Low | Difficult | No | Drug allergy | Necrosis or cholestasis | Phenytoin, sulphonamides, chlorpromazine, amoxicillin-clavulanate |
| Metabolic | Low | Difficult | No | Hepatotoxic metabolites | Necrosis or cholestasis | Isoniazid, valproic acid, diclofenac, troglitazone |
| Target | Hepatocyte injury | Mechanism |
|---|---|---|
| Hepatocyte membrane | Blebbing, rupture, cell lysis | Disassembly of actin fibrils |
| Canaliculus | Loss of villous processes | Interruption of transport pumps |
| Endoplasmic reticulum | Cytochrome P-450 system | Creation of nonfunctioning adducts |
| Vesicles | Attack by T cells | Immune response by cytolytic T cells and cytokines |
| Nuclear chromatin | Apoptosis | Activation of TNF or FAS triggers a cascade of intercellular caspases |
| Mitochondria | Impaired oxidation and respiratory chain enzymes | Lactic acidosis, reactive oxygen species |
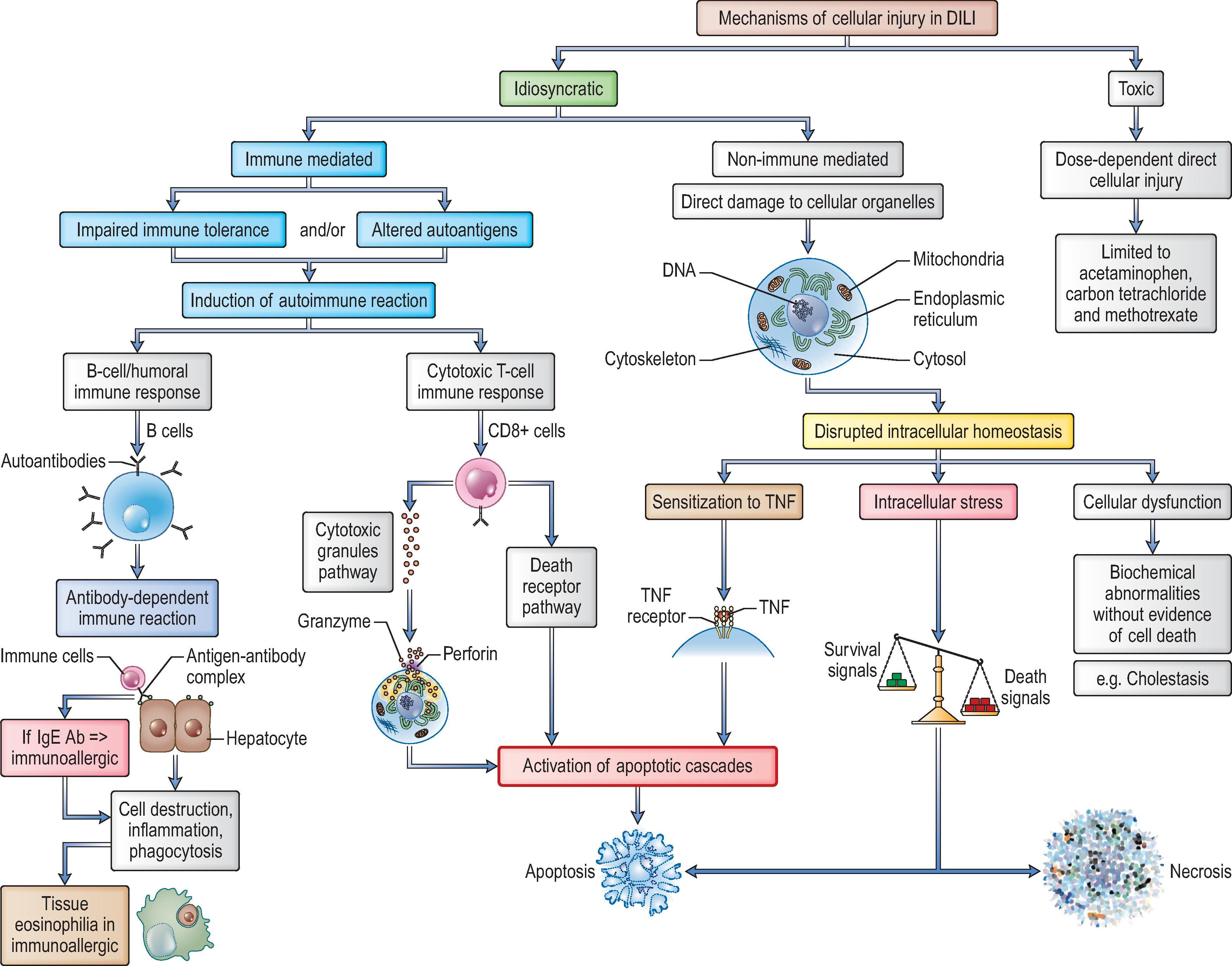
Intrinsic hepatotoxins are generally defined as agents which cause hepatic injury in a dose-dependent, reproducible manner. They produce injury as a result of their chemical composition rather than depending on variation in metabolic processing or immune response to a metabolite that may be present in one individual but not another. Historically they have been divided into direct and indirect intrinsic hepatotoxins although the division in these categories is not always clear. Direct hepatotoxins include chemicals like ferrous salts and phosphorus compounds that cause direct injury to cell membranes, organelles and macromolecules as well as chemicals like carbon tetrachloride that require some enzymatic processing to produce the highly reactive intermediate. Carbon tetrachloride is processed by the cytochrome P450 system to the trichloromethyl radical. This radical is highly reactive and can freely interact with macromolecules in the cell. The degree of injury is dependent on dose and small doses can be well tolerated but larger doses are lethal. No pharmaceutical agents fall into the category of direct hepatotoxins.
Indirect hepatotoxins are metabolically processed to reactive intermediates as well but affect specific metabolic pathways. Acetaminophen is the most common example. This drug is also processed by the cytochrome P450 system to a reactive intermediate: N -acetyl- p -benzoquinone. This intermediate reacts with glutathione, depleting this protective molecule and making the cell more susceptible to oxidative stress. Patients are variably sensitive to injury from acetaminophen depending on their nutritional status (which affects glutathione stores) and chronic alcoholism (which affects both the levels of the cytochrome P450 enzymes and the nutritional status). Because intrinsic hepatotoxins cause reproducible liver injury, they are used extensively in animal models of hepatotoxicity.
Nearly all drugs that have been described as causing hepatic injury do so rarely and seemingly unpredictably. Very few agents are recognized as dose-dependent intrinsic hepatotoxins (e.g. acetaminophen, aspirin and some chemotherapeutic agents). Drugs that injure the liver unpredictably in susceptible individuals may be divided into two categories. In immunological idiosyncrasy, the mechanism appears to be autoimmune (discussed in the section on chronic hepatitis-like drug injury) or immunoallergic, while in toxic-metabolic idiosyncrasy the mechanism appears to be a metabolic aberration of the host that permits production or accumulation of damaging amounts of hepatotoxic metabolites. A third type of idiosyncratic hepatotoxicity is represented by the checkpoint inhibitors and other biologicals, for which the mechanism of toxicity is a function of the drug’s mechanism of action.
Immunoallergic drug injury (also called drug allergy or hypersensitivity drug injury) develops after a relatively short sensitization period of 1–8 weeks, recurs promptly on readministration of the agent (sometimes within a day or two) and tends to be accompanied by other clinical findings of hypersensitivity. These may include fever, rash, eosinophilia, lymphadenopathy and atypical lymphocytosis. The rash may be severe and toxic epidermal necrolysis or Stevens–Johnson syndrome can develop. Jaundice develops later in the injury. The liver biopsy tends to show a mixed injury pattern with both cholestasis and hepatitis (see later section on cholestatic hepatitis). There may be an eosinophil-rich or granulomatous inflammatory infiltrate. A subset of cases of immunoallergic liver injury may result in the DRESS (drug reaction with eosinophilia and systemic symptoms) syndrome. DRESS has been reported with a variety of drugs, including phenytoin, carbamazepine, allopurinol and nevirapine sulphonamides, dapsone and a number of other drugs. It should be noted that these systemic symptoms, particularly fever and rash, are relatively nonspecific, so additional evidence of hypersensitivity should be present before classifying a drug reaction as immunoallergic.
Convincing evidence for the role of immunological factors in some forms of drug-induced injury can be found in the case of halothane-induced DILI, where injury was found to be accompanied by the development of antibodies and sensitized T cells that react with rabbit hepatocytes modified by an oxidative metabolite of halothane. Mouse models of halothane hepatitis have demonstrated both the involvement of eosinophils as well as the development of an immune response directed against trifluoroacetylated protein adducts. Injury could be increased by specific depletion of myeloid-derived suppressor cells. Ticrynafen (tienilic acid) has been found to have antibodies that react with ticrynafen-altered hepatocyte proteins. One such ticrynafen antibody reacts with microsomes. This anti-liver–kidney microsomal antibody type 2 (anti-LKM2) appears to be an antibody to the cytochrome P450 isoform involved in the metabolism of ticrynafen. These studies provide suggestive evidence that the pathogenesis of the injury is the consequence of conversion of hepatocyte proteins to neoantigens by reaction with a metabolite of the drug and that the ensuing immunological reaction leads to hepatic injury. Cell-mediated immunity has also been implicated in immunoallergic injury and this has led some investigators to try to use the stimulation of peripheral blood mononuclear cells by a drug as causal evidence of DILI. However, when this idea was tested in a series of patients with DILI, less than 10% of cases showed a positive stimulation of lymphocytes by the injurious agent.
Many drugs have been shown to have injury associated with specific human leukocyte antigen (HLA) subtypes ( Table 12.4 ). The combination drug amoxicillin-clavulanate has been associated with increased prevalence of specific HLA-DR subtypes, suggesting that the injury may be mediated by HLA class II antigen presentation. Flucloxacillin injury showed a strong association with HLA-B*5701 in a genome-wide association study. Individuals carrying this allele are 80 to 100 times more likely to experience flucloxacillin-related liver injury. It should be noted that the idea that drug hypersensitivity alone will lead to hepatic injury is an oversimplification. Generalized hypersensitivity per se does not always lead to liver injury. Penicillin is a frequent cause of drug hypersensitivity, yet almost never causes liver injury. On the other hand, anticonvulsant-related hypersensitivity almost always includes a component of liver injury. The mere presence of circulating antidrug antibodies is insufficient to induce liver disease. For example, despite the presence of serum antibodies to halothane and other anaesthetics in 158 paediatric anaesthesiologists, only a single individual developed liver injury.
| Hepatotoxic agent | Predisposing metabolic/genetic factor |
|---|---|
| Acetaminophen | Deficiency of glutathione synthetase |
| Amineptine | Postulated familial susceptibility; extensive dextromethorphan oxidation capacity; genetic deficiency of a yet to be identified cell defence mechanism |
| Atrium (meprobamate) | Poor metabolizer of S -mephenytoin from CYP2C19 deficiency |
| Chlorpromazine | Sulphoxidation deficiency; poor metabolizer of S -carboxy- l -methyl-cysteine and/or extensive metabolizer of sparteine |
| Halothane | Postulated familial susceptibility of epoxide hydrolase deficiency |
| Isoniazid | Deficiency of glutathione- S -transferase due to mutations at GST M1 and GST T1 loci; and N -acetyltransferase-2 (NAT2) polymorphisms |
| Perhexiline | Poor metabolizer of debrisoquine and/or dextromethorphan (seen in 5–10% of Europeans) from CYP2D6 deficiency |
| Sulphonamides, dihydralazine | Slow acetylation due to deficiency of N -acetyltransferase-2 activity |
| Human leukocyte antigen (HLA) associations | |
| Amoxicillin-clavulanate | HLA-A*02:01, HLA-B*18:01, HLA-DRB1*15:01, HLA-DQB1*06:02; inverse association with HLA-DRB1*07 |
| Antimycobacterials | HLA-DQB1*02:01; inverse association with HLA-DQA1*01:02 |
| Clometacin | HLA-B*08 |
| Fenofibrate | HLA-A*33:01 |
| Flucloxacillin | HLA-B*57:01 |
| Flupirtine | HLA-DRB1*16:01-DQB1*05:02 |
| Lapatinib | HLA-DRB1*07:01-DQA1*02:01 |
| Lumiracoxib | HLA-DRB1*1501-HLA-DQB1*0602-HLA-DRB5*0101-HLA-DQA1*1102 |
| Minocycline | HLA-B*35:02 |
| Nevirapine | HLA-DRB1*01:02 |
| Terbinafine | HLA-A*33:01 |
| Ticlopidine | HLA-A*33:01 |
| Tiopronine | HLA-A*33 |
| Ximelagatran | HLA-DRB1*07-DQA1*02 |
| Non-HLA associations | |
| Efavirenz | ERN1 |
| Flucloxacillin | ST6GAL1 |
| Rifampicin | FAM65B |
Hypersensitivity-mediated reactions may also result from a metabolic defect. , In one study, patients who had sustained hepatic injury in a hypersensitivity-type reaction to phenytoin had an apparent defect in converting the active metabolite (arene oxide) to the inactive dihydriol. The active metabolite presumably could serve as a hapten or could be cytotoxic. Other potential genetic predispositions to developing DILI are given in Table 12.4 . , With the burgeoning field of pharmacogenomics, it is expected that the ability to identify susceptible individuals will permit a greater understanding of the mechanisms involved as well as a means to avoid serious toxicity in such patients. ,
Idiosyncratic hepatic injury that is not accompanied by clinical hallmarks of hypersensitivity, is not promptly reproduced by readministration of the drug and which may appear after widely varying periods of exposure to the drug (between 2 weeks and several months) is thought to result from metabolic rather than immunological mechanisms. Identification of reactive metabolites that are thought to be responsible for acute DILI has been made for some agents, such as perhexiline maleate, valproic acid, INH and iproniazid. For the majority of other agents, however, the exact cause remains undefined. Interindividual differences in susceptibility may account for this type of DILI.
There appears to be some degree of dose dependency in idiosyncratic liver injury , despite its characterization as a dose-independent form of liver injury. A study of a U.S. pharmaceutical database and the large Spanish DILI registry found that drugs given in a daily dose of 50 mg or greater were far more likely to cause DILI compared to drugs given in daily doses of 10 mg or less. In the Spanish registry, 77% of agents were given in daily doses of more than 50 mg, and such agents were more likely to result in acute liver failure, liver transplantation and death. The degree to which a drug undergoes hepatic metabolism also relates to its risk of causing DILI. Compounds with greater than 50% hepatic metabolism were associated with higher rates of elevated alanine aminotransferase (ALT) (more than three times the upper limit of normal [ULN]), liver failure and death when compared to drugs undergoing lesser degrees of hepatic metabolism.
The diagnosis of drug and toxic hepatic injury begins with a careful morphologic evaluation of the liver for the types of cellular injury as well as the overall pattern , ( Table 12.5 ). While it is true that hepatoxicity can replicate essentially every other type of liver disease, individual hepatotoxins display a more limited range of injury. Thus, identification of the pattern of injury can be very helpful in both including and excluding hepatotoxicity as the cause of liver dysfunction. Hepatotoxins may injure any or all of the cell types found in the liver, but particular attention should be paid to hepatocytes, bile duct cells and endothelial cells. Ultimately the type of cellular injury will depend on the particular agent involved. Cell injury may result in necrosis, apoptosis, cholestasis, steatosis, cytoplasmic inclusions or pigment accumulation depending on the degree of injury and the cellular elements involved. Cellular injury may vary across the hepatic lobule and may affect the different cell types in different ways. Severe injury may lead to fulminant hepatic failure by several pathways, while subacute and chronic injury can lead to bile duct loss or advanced fibrosis. The overall outcome is a pattern of injury that is characteristic of the agent involved, and this pattern may be modified by the genetic and biochemical background of the host as well as the response of the host’s immune system to the injury.
| Type of injury | Examples of agents |
|---|---|
| Necroinflammatory | |
| Acute coagulative (zonal) necrosis * | |
| Perivenular (zone 3) | CCl 4 , acetaminophen, halothane |
| Periportal (zone 1) | Phosphorus, allylformate, albitocin |
| Acute hepatitis-like | Isoniazid, sulphonamides, halothane, diclofenac |
| Mononucleosis-like | Phenytoin, para-aminosalicylate, dapsone |
| Chronic hepatitis-like | Oxyphenisatin, methyldopa, nitrofurantoin, dantrolene, clometacine, papaverine, sulphonamides |
| Granulomatous | |
| Cholestatic | |
| Acute intrahepatic cholestasis (‘bland cholestasis’) | C-17 alkylated anabolic and contraceptive steroids |
| Chronic intrahepatic cholestasis (ductopenic, PBC-like) | CPZ, haloperidol, imipramine, organic arsenicals, thiabendazole, tolbutamide |
| Biliary sclerosis (PSC-like) | Floxuridine by hepatic artery perfusion |
| Mixed hepatocellular-cholestatic injury (cholestatic hepatitis) | Amoxicillin-clavulanate, statins |
| Steatosis | |
| Microvesicular | Ethionine, tetracycline, phosphorus, ethanol |
| Macrovesicular | Ethanol, MTX |
| Steatohepatitis-like | Perhexiline maleate, amiodarone |
| Phospholipidosis | Amiodarone |
| Vascular | |
| Sinusoidal dilatation | OCs |
| Peliosis hepatis | Anabolic steroids, vinyl chloride, arsenicals |
| Sinusoidal obstruction syndrome/veno-occlusive disease | Pyrrolizidine alkaloids, antineoplastic agents |
| Hepatic vein thrombosis (Budd–Chiari syndrome) | Contraceptive steroids |
| Hepato-portal sclerosis | Arsenicals, copper sulphate, antineoplastic agents |
| Nodular regenerative hyperplasia | 6-Thioguanine, azathioprine |
| Fibrosis/cirrhosis | Methotrexate, drugs which cause chronic injury |
| Neoplasms | |
| Hepatocellular adenoma | OCs, anabolic steroids |
| Hepatocellular carcinoma | Anabolic steroids, TPN, OCs, Th-O, vinyl chloride |
| Cholangiocarcinoma | |
| Angiosarcoma | Th-O, vinyl chloride, As, CuSO 4 , anabolic steroids |
The number of major morphological patterns of drug and toxin injury is limited, and these are listed in Table 12.5 . Definition of the forms of injury produced by the intrinsic hepatotoxins has been relatively simple, since the hepatic damage can be reproduced in experimental animals. Characterization of the forms of idiosyncratic injury, however, has been more difficult and has depended on collation of material from reports of individuals and groups of cases. It is important to remember that when reporting instances of DILI, pathologic descriptive words like hepatitis, necrosis, steatosis and cholestasis should be used only if a biopsy or other tissue is available for examination. Biochemical injury is typically described as ‘hepatocellular’ when the aminotransferases are elevated, and ‘cholestatic’, when alkaline phosphatase (ALP) is significantly elevated. Elevated serum bilirubin levels may be observed in either biochemically hepatocellular or cholestatic injuries.
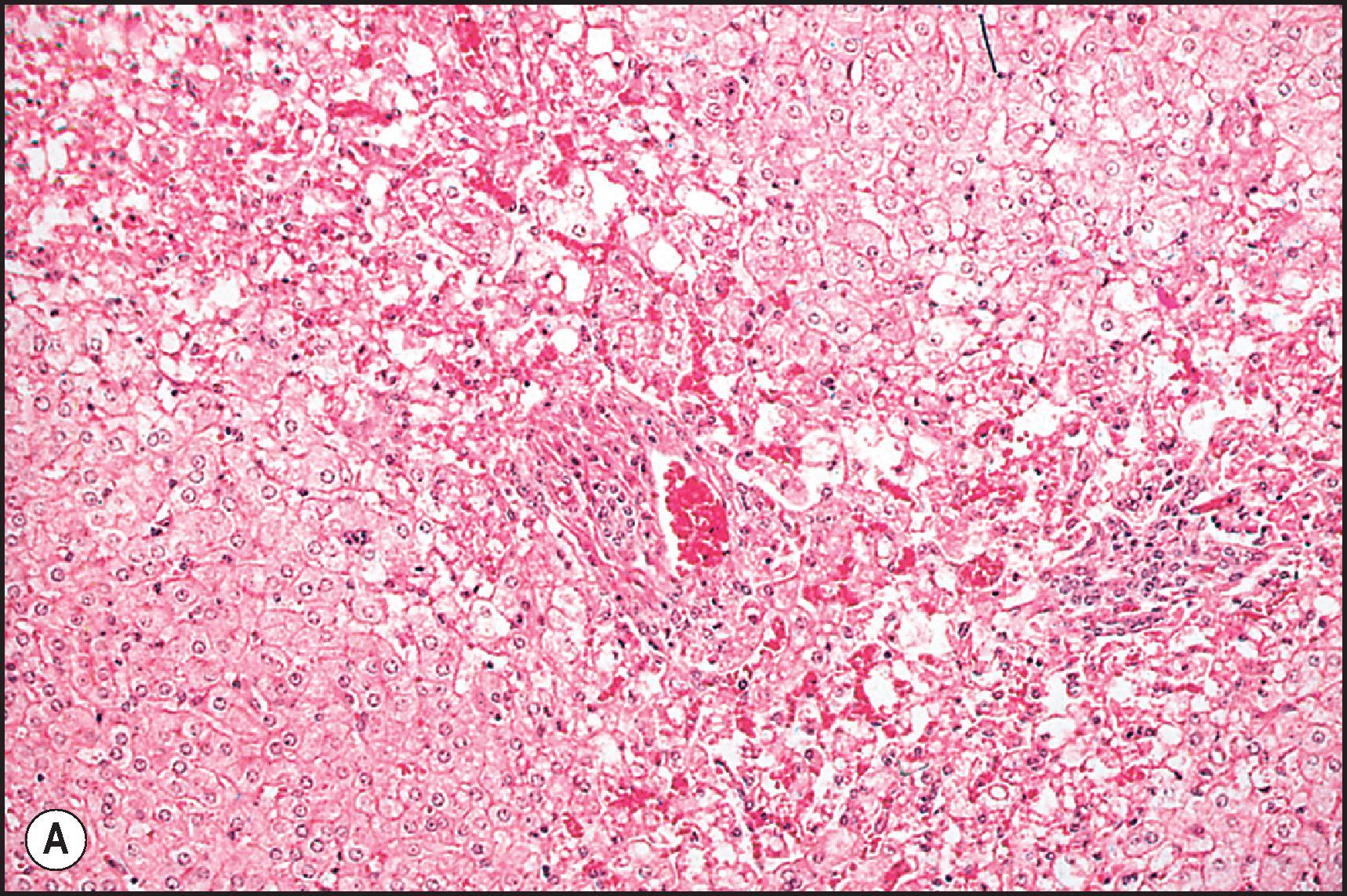
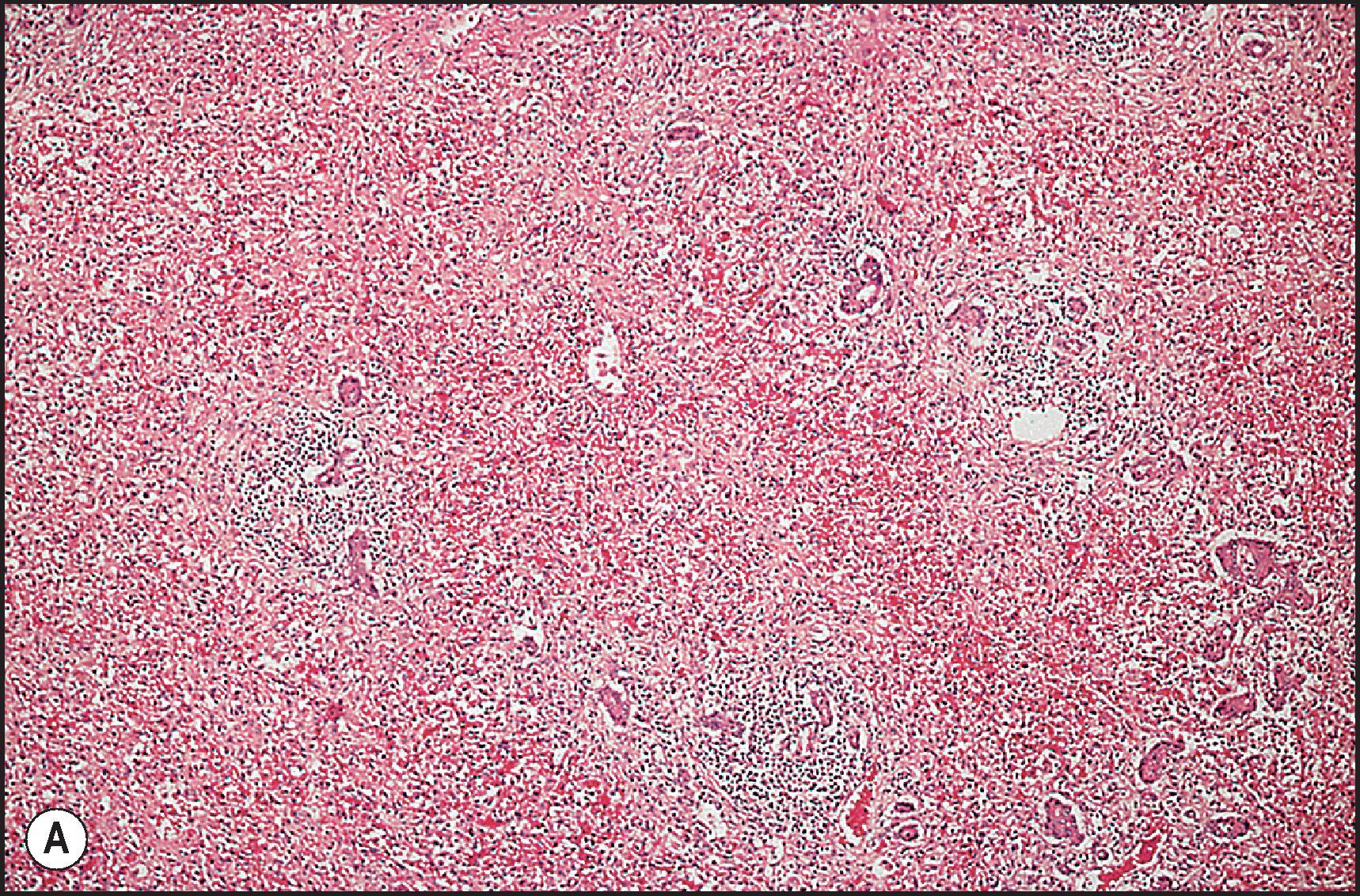
Toxic injury which comes to clinical attention frequently takes the form of either necroinflammatory injury or cholestatic injury. In toxicity that is mainly necroinflammatory, the relative degree of cell death and inflammation may vary from lesions that are almost entirely necrotic with little inflammation to those that may mimic chronic viral hepatitis or AIH. Most intrinsic toxins produce predominantly cytotoxic rather than cholestatic injury. Idiosyncratic injury may be cholestatic or cytotoxic. Some drugs characteristically produce a mixed pattern of injury, with both hepatocellular and cholestatic injury. The necrosis observed in necroinflammatory injury may be nonzonal, zonal or massive. As a general rule, the necrosis produced by intrinsic hepatotoxins is zonal ( Figs. 12.2 12.3 ), while that produced by idiosyncratic injury is usually inflammatory and nonzonal ( Figs. 12.4 12.5 ). Extreme degrees of both zonal and nonzonal necrosis can result in massive necrosis ( Fig. 12.4 ).
![Figure 12.2, Paracetamol (acetaminophen) toxicity. (A) Confluent coagulative necrosis involving the perivenular and mid-zones. (Haematoxylin & eosin [H&E].) (B) Necrotic hepatocytes are shrunken, eosinophilic, rounded and have lost their nuclei. (H&E.) Figure 12.2, Paracetamol (acetaminophen) toxicity. (A) Confluent coagulative necrosis involving the perivenular and mid-zones. (Haematoxylin & eosin [H&E].) (B) Necrotic hepatocytes are shrunken, eosinophilic, rounded and have lost their nuclei. (H&E.)](https://storage.googleapis.com/dl.dentistrykey.com/clinical/HepaticInjuryduetoDrugsDietaryandHerbalSupplementsChemicalsandToxins/1_3s20B9780702082283000120.jpg)
Individual cells involved in necroinflammatory injury can undergo a variety of changes. Cell death can occur by two processes: necrosis and apoptosis. , Necrosis refers to destructive disintegration of the cell, apparently initiated by plasma membrane injury and yielding only debris. Apoptosis (programmed cell death) is an energy-dependent event leading to cell shrinkage, nuclear fragmentation, condensation of chromatin and the production of cytoplasmic blebs yielding distinct fragments of the cells or entire condensed shrunken cells. The condensed cells are referred to as apoptotic or free acidophilic bodies. The mechanisms for the two are different. Activation of apoptotic pathways involves tumour necrosis factor alpha receptor or Fas-mediated release of intercellular caspases that lead to cell death. Necrosis results when there is massive injury to the cells. Frequently this is due to massive injury to the cell membrane and organelles. Apoptosis may occur when the cell suffers similar direct injury, but in lesser degrees. Thus, apoptotic hepatocytes are sometimes seen adjacent to zones of confluent necrosis. Apoptosis may also result from immune-mediated injury, in which cytotoxic T cells, antibodies or cytokines initiate the apoptotic pathway. Ballooning hepatocellular changes, steatosis and Mallory body formation may also be seen as components of necroinflammatory injury, depending on the agent. The presence of cholestasis (either canalicular or hepatocellular) should lead the pathologist to consider agents that cause mixed injury (discussed later).
Pure zonal necrosis may involve the perivenular (zone 3), periportal (zone 1) or rarely the midzone (zone 2) depending on the agent and is usually the result of intrinsic toxins ( Table 12.6 ). However, there are exceptions. In the idiosyncratic injury caused by halothane and other halogenated anaesthetics, the necrosis is often perivenular and is strikingly similar to that of carbon tetrachloride, an intrinsic toxin. In addition, drug-induced acute hepatitis may have zone 3 necrosis and a number of the drugs listed in Table 12.6 show this pattern of injury. In pure acute zonal necrosis there is confluent coagulative necrosis that involves the liver evenly, striking the same distribution in each acinus. Cells of the innate immune system may be present within and around the zones of necrosis, but there is little lymphocytic infiltrate in the pure forms of this injury. As the injury becomes more severe it will extend to involve other zones with bridging necrosis. Massive necrosis of the zonal type tends to retain its zonal character. For example a rim of hepatocytes might remain adjacent to the portal areas in severe toxicity from an agent that affects zone 3 first.
| Chemicals | Drugs |
|---|---|
| 2-Chloropropane (Z3) 2-Nitropropane (D) Aflatoxins (Z1, Z3) Albitocin (Z1) Alloxan (Z1) Allyl compounds (Z1) Amanitin (Z3) Aniline (D) Arsenic compounds (Z3, D) Bacillus cereus toxin (Z2) Beryllium (Z2) Bromobenzene (Z3) Carbon tetrachloride (Z3) Chlorinated benzenes (Z3, D) Chlorinated diphenyls (D) Chlorinated naphthalene (D) Chloroprene (Z3) Cocaine (Z1, Z3) Copper sulphate (Z3) DDT (Z3) Dichloropropane (Z3) Dimethylnitrosamine (Z3) Dinitrobenzone (Z3, D) Dinitrotoluene (Z3, D) Dioxane (Z2, D) Diphtheria toxin (Z3) Ethylene dibromide (Z3) Ethylene dichloride (Z3) Galactosamine (D) Halogenated hydrocarbons (Z3, D) Luteoskyrin (Z3) Manganese compounds (Z1) Mushrooms (Z3, D) Naphthalene (Z3) Ngaione (Z2) Proteus vulgaris endotoxin (Z1) Paraquat (Z3) Phalloidin (Z3) Phosphorus (Z1) Rubratoxin (Z3) Selenium (D) Sporidesmin (Z1) Tetrachloroethane (Z3, D) Tetrachloroethylene (Z3) |
Acetaminophen (paracetamol) (Z3) Amodiaquine (Z3) Anastrozole (Z3) Benorylate (Z3) Chloroform (Z3) Chlorzoxazone (Z3) Ciprofloxacin (Z3) Citalopram (Z3) Cyclophosphamide (Z3) Diclofenac (Z3) Disulfiram (D) Duloxetine (Z3) Ebrotidine (Z3) Enflurane (Z3) Ephedrine (Z3) Ethionamide (Z3) Ferrous sulphate (Z1) Fimasartan (Z3) Fluroxene (Z3) Halothane (Z3) Hycanthone (D) Hydroxychloroquine (Z3) Imatinib mesylate (Z3) Imipramine (Z3) Indomethacin (Z3) Infliximab (Z3) Iodoform (Z3) Iproniazid (Z3) Isoflurane (Z3) Isoniazid (Z3) Ketoconazole (Z3) Labetalol (Z3) Lamotrigine (Z1, Z3) Levetiracetam (Z3) Levofloxacin (Z3) Lisinopril (Z3) Lovastatin (Z3) Mesalamine (Z1) Methoxyflurane (Z3) Methyldopa (Z3) Nefazodone (Z3) Nevirapine (Z3) Norfloxacin (Z3) Nortriptyline (Z3) |
| Thioacetamide (Z3) Trichloroethylene (Z3) Trinitrotoluene (Z3, D) |
Nortriptyline (Z3) Olanzapine (Z3) Oxaprozin (Z3) Pantoprazole (Z3) Phenytoin (Z3) Piroxicam (D) Plicamycin (Z3) Propylthiouracil (Z3) Quetiapine (Z3) Quinidine (Z3) Rifampin (Z3) Rivaroxaban (Z3) Roxithromycin (Z3) Sulfasalazine (Z3) Sulindac (Z3) Synthaline (Z1) Tacrine (Z3) Tacrolimus (Z3) Telithromycin (Z3) Ticrynafen (Z3) Tocilizumab (Z3) Tolazamide (Z3) Toloxatone (Z3) Troglitazone (Z3) Trovafloxacin (Z3) Urethane (ethyl carbamate) (Z3) Valproic acid (Z3) Venlafaxine (Z3) |
| Herbals/natural products | |
| Black cohosh (Z3) Chaparral (Z3) Germander (Teucrium) (Z3) Greater celandine (Z3) Green tea extract (Z3) Impila (D) Kava kava (D) Ma huang (Z3) Oil of cloves (Z3) Pennyroyal oil (Z3) Syo-saiko-to (Z3) Tannic acid (Z3) Turmeric (D) Usnic acid |
Perivenular (zone 3) necrosis is the most common type of zonal necrosis and is the characteristic lesion produced by several intrinsic toxins. The toxins in this category that are relevant to human disease include carbon tetrachloride (CCl 4 ), chloroform (CHCl 3 ), copper salts, pyrrolizidine alkaloids (PAs), tannic acid and the toxins of Amanita phalloides , among others. Acetaminophen in high doses may cause perivenular necrosis ( Fig. 12.2 ) often accompanied by steatosis of the remaining viable hepatocytes. Perivenular necrosis induced by PAs and stem cell transplant preparative regimens is accompanied by occlusive injury to the hepatic veins, adding a haemorrhagic component to the necrosis. , Periportal (zone 1) necrosis ( Fig. 12.3 ) is usually due to direct toxins such as phosphorus, , poisonous doses of ferrous sulphate, ingestion of concentrated (90%) acetic acid and allyl alcohol and its esters.
The zonality of necrosis may be related to the mechanism of injury. The perivenular location of the lesion induced by CCl 4 , bromobenzene and acetaminophen reflects zonal variation in enzyme systems responsible for the conversion of these agents to hepatotoxic metabolites as well as sensitivity of those hepatocytes to oxidative stress and mitochondrial injury. The periportal necrosis of ferrous sulphate toxicity in children is presumed to be a direct cytopathic effect of the high concentration of iron reaching the liver via the portal vein. Phosphorus is absorbed very quickly in the stomach and intestine; it reaches the liver soon after ingestion. The specific mechanism of phosphorus toxicity is unclear, but it appears to interfere with multiple intracellular functions.
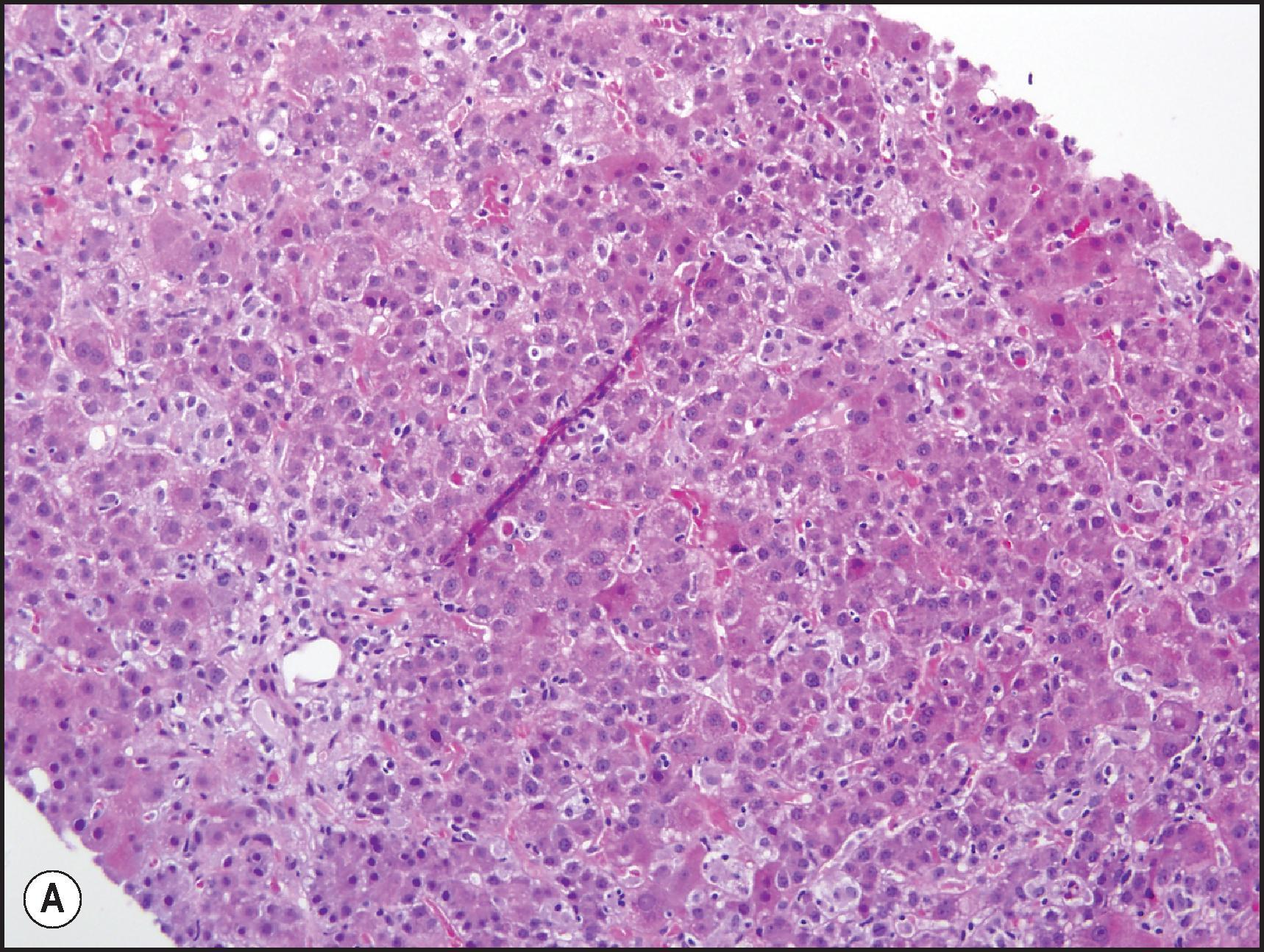
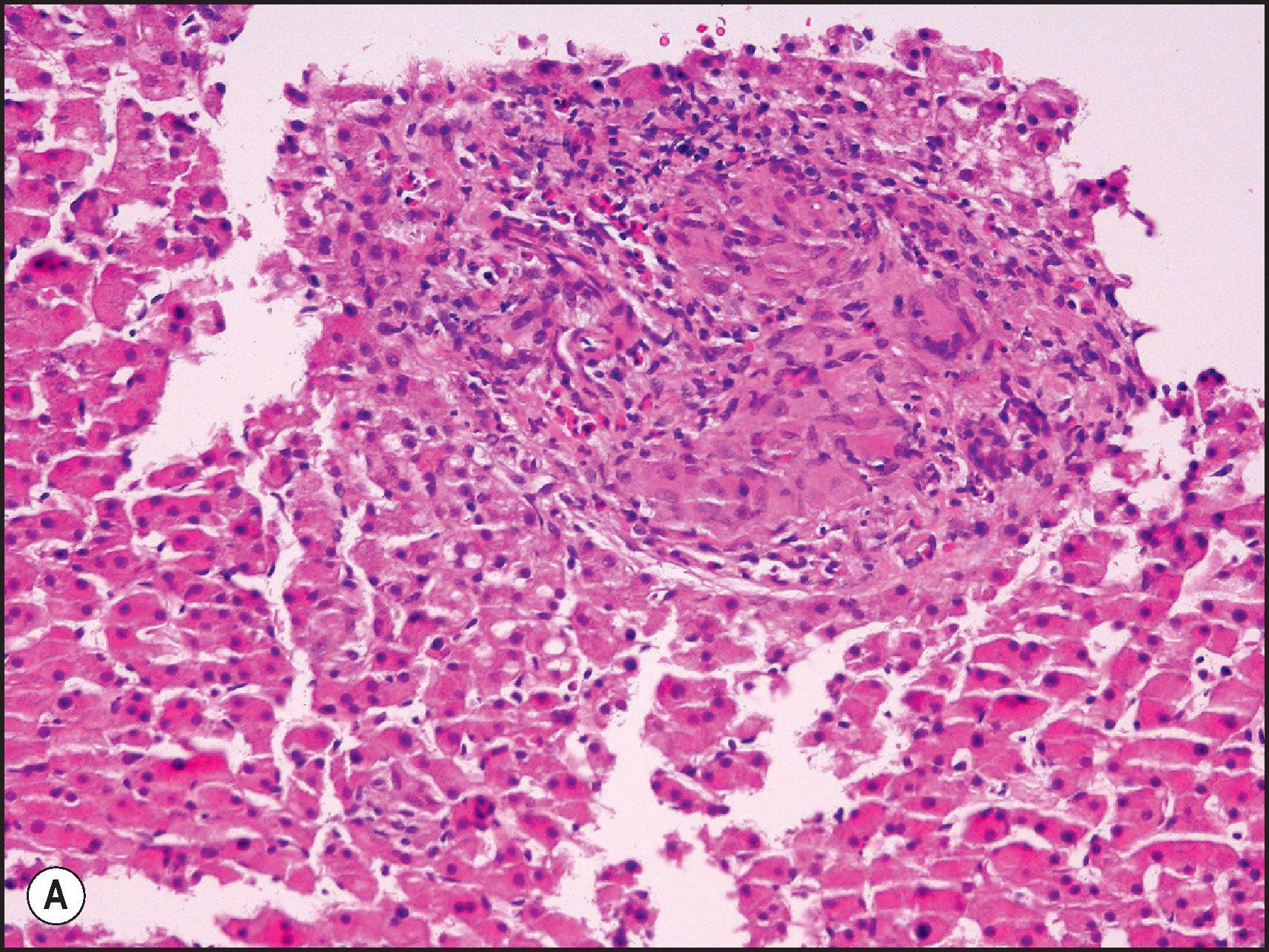
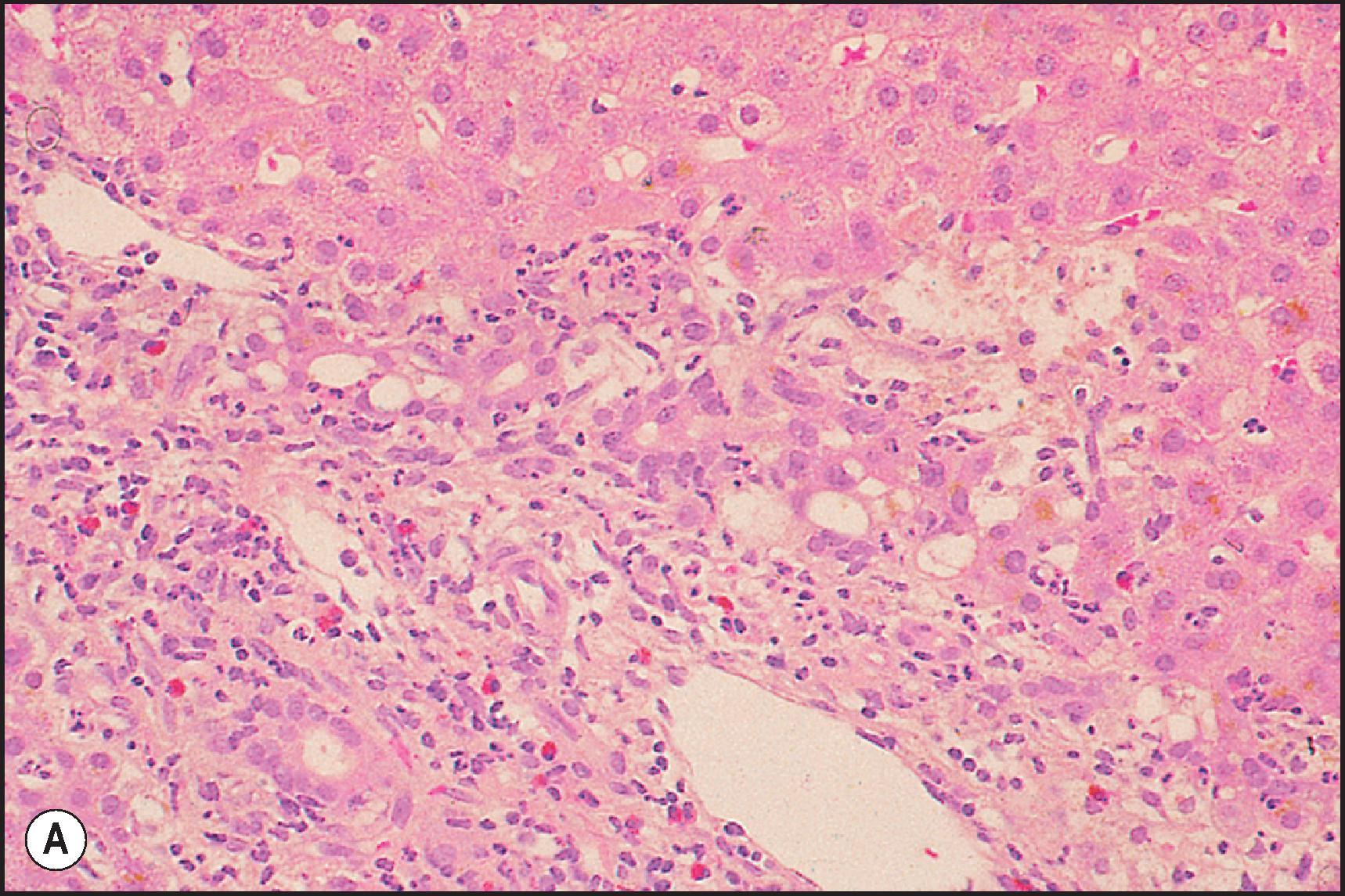
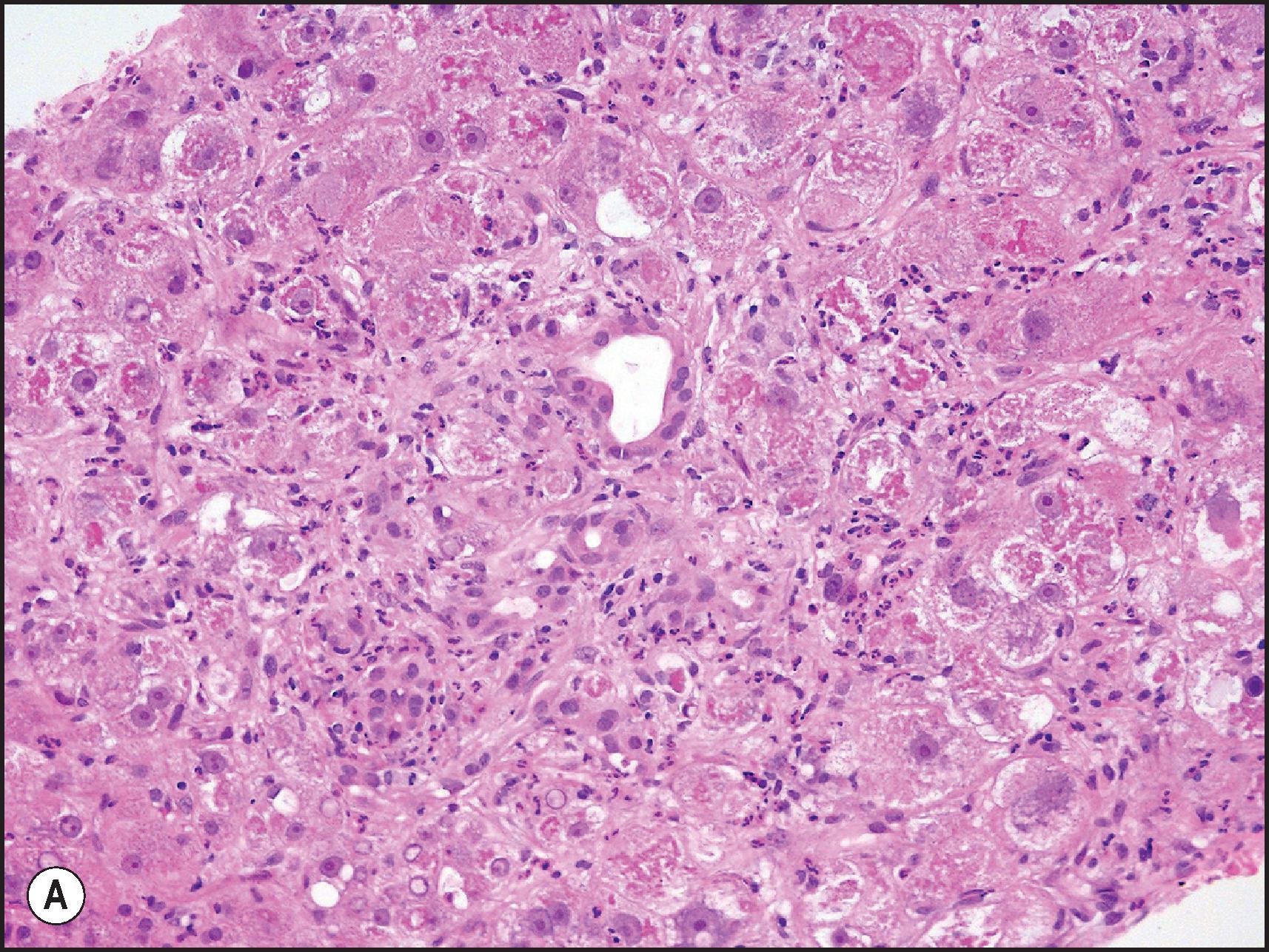
Another common pattern of acute hepatoxicity is that of multiple small areas of degeneration or ‘spotty’ necrosis, similar to acute viral hepatitis ( Table 12.7 ). Acute hepatitis without concomitant cholestasis accounted for 20% of a large series of idiosyncratic DILI cases. It is characterized by a predominantly lobular, lymphocytic infiltrate with scattered apoptotic hepatocytes. Increased numbers of eosinophils can be seen, which may help suggest the diagnosis of DILI. There may be other evidence of hepatocellular injury, such as cytoplasmic clearing, cellular swelling or steatosis, and regenerative changes, such as mitoses, hepatocyte rosette formation and the proliferation of ductular hepatocytes. In severe cases there is often lobular disarray ( Fig. 12.5 ), in which the normal sinusoidal architecture is lost amid a sea of inflammation, hepatocyte rosettes and apoptosis. This may lead to areas of confluent necrosis from extensive apoptotic cell death ( Fig 12.6 ). The massive necrosis that results from drugs in this category of injury may be most prominent in zone 3, mimicking the zonal necrosis pattern ( Fig. 12.4 ). In the DILI Network (DILIN) series, 18% of the acute hepatitis cases showed zone 3 necrosis. Portal inflammation and interface hepatitis are often present, as is bile duct injury, but they do not dominate the overall inflammatory pattern. Reticulin stains will show the zones of collapse and periportal fibrosis may be present. More extensive fibrosis may develop as a result of extensive necrosis and subsequent scarring if the patient survives. In case reports of DILI, necrosis has been one histologic feature clearly associated with a fatal outcome, although the degree of necrosis is undoubtedly important. ,
| Acute (lobular-predominant) hepatitis | Chronic (portal-predominant) hepatitis | |
|---|---|---|
| Drugs | Drugs | |
| Acarbose Acetylsalicylic acid Acitretin Adalimumab Doxorubicin Amiodarone Amitriptyline Amlodipine Amodiaquine Amoxapine Anakinra Aprindine Asparaginase Atomoxetine Benzarone Bromfenac Bupropion Captopril Carbamazepine Carbenicillin Cetirizine Chlorotetracycline Chlorpromazine Chlorzoxazone Cimetidine Cinchophen Ciprofloxacin Cisplatin Citalopram Corticosteroids Clarithromycin Clindamycin Clometacin Cromolyn Cyclofenil Dantrolene Dapsone Darunavir Desflurane Dichloromethotrexate Diclofenac Diflunisal Dihydralazine Disulfiram |
Lapatinib Leflunomide Lergotrile Levofloxacin Losartan Mepacrine Mercaptopurine Metformin Methimazole Methotrexate Methoxyflurane Methyldopa Minocycline Mirtazapine Mitomycin C Naproxen Nevirapine Nicotinic acid Nimesulide Nitrofurantoin Norfloxacin Olanzapine Oxacillin Oxaprozin Oxcarbazepine Oxyphenisatin Papaverine Paroxetine Pemoline Perhexiline maleate Phenazopyridine Phenindione Phenobarbital Phenprocoumon Phenylbutazone Phenytoin Pirprofen Pomalidomide Pregabalin Propylthiouracil Pyrazinamide Quetiapine Ranitidine |
Acetaminophen (paracetamol) * Acetylsalicylic acid Adalimumab Doxorubicin Amlodipine Atomoxetine Atorvastatin Azapropazone Benzarone Chlorpromazine Clometacin Cyclofenil Dantrolene Doxidan Doxorubicin Erlotinib Erythromycin Etanercept Etretinate Everolimus Ezetimibe Fenofibrate Gliclazide Haloperidol Halothane Infliximab Ipilimumab Irbesartan Isoniazid Labetalol Levofloxacin Lisinopril Losartan Methyldopa Naproxen Nevirapine Nicotinic acid Nimesulide Nitrofurantoin Oxacillin Oxyphenisatin Pemoline Pentamidine |
| Doxepin Duloxetine Ecstasy Eletriptan Enflurane Erlotinib Etanercept Etodolac Etoposide (VP-16) Etretinate Fenofibrate Fluroxene Gatifloxacin Gliclazide Glibenclamide Gold Haloperidol Halothane Hydralazine Hydrazines Hydrochlorothiazide Hydroxychloroquine Ibuprofen Imatinib mesylate Infliximab Interleukin-2 Ipilimumab Isoniazid Ivermectin Ketoconazole Lamotrigine |
Rifampin Riluzole Rosuvastatin Sertraline Sevoflurane Sibutramine Simvastatin Sorafenib Spironolactone Streptozotocin Sulfadiazine Sulfadoxine-pyrimethamine Sulfamethizole Sulfamethoxazole Sulfasalazine Sulindac Suloctidil Telithromycin Teniposide Thalidomide Thiotepa Ticrynafen Tolazamide Tolvaptan Trazodone Trastuzumab Troglitazone Trovafloxacin Vancomycin Verapamil Ximelagatran |
Perhexiline maleate Phenprocoumon Phenytoin Propylthiouracil Pyrazinamide Rosuvastatin Simvastatin Sulfamethizole Thalidomide Ticrynafen Tolazamide Trazodone Ximelagatran |
| Herbals/Natural Products | Herbals/Natural Products | |
| Black cohosh Chaparral Chaso/onshido Germander (Teucrium) Greater celandine Green tea extract Jin bu huan Skullcap Syo-saiko-to |
Baracol Germander (Teucrium) Turmeric |
|
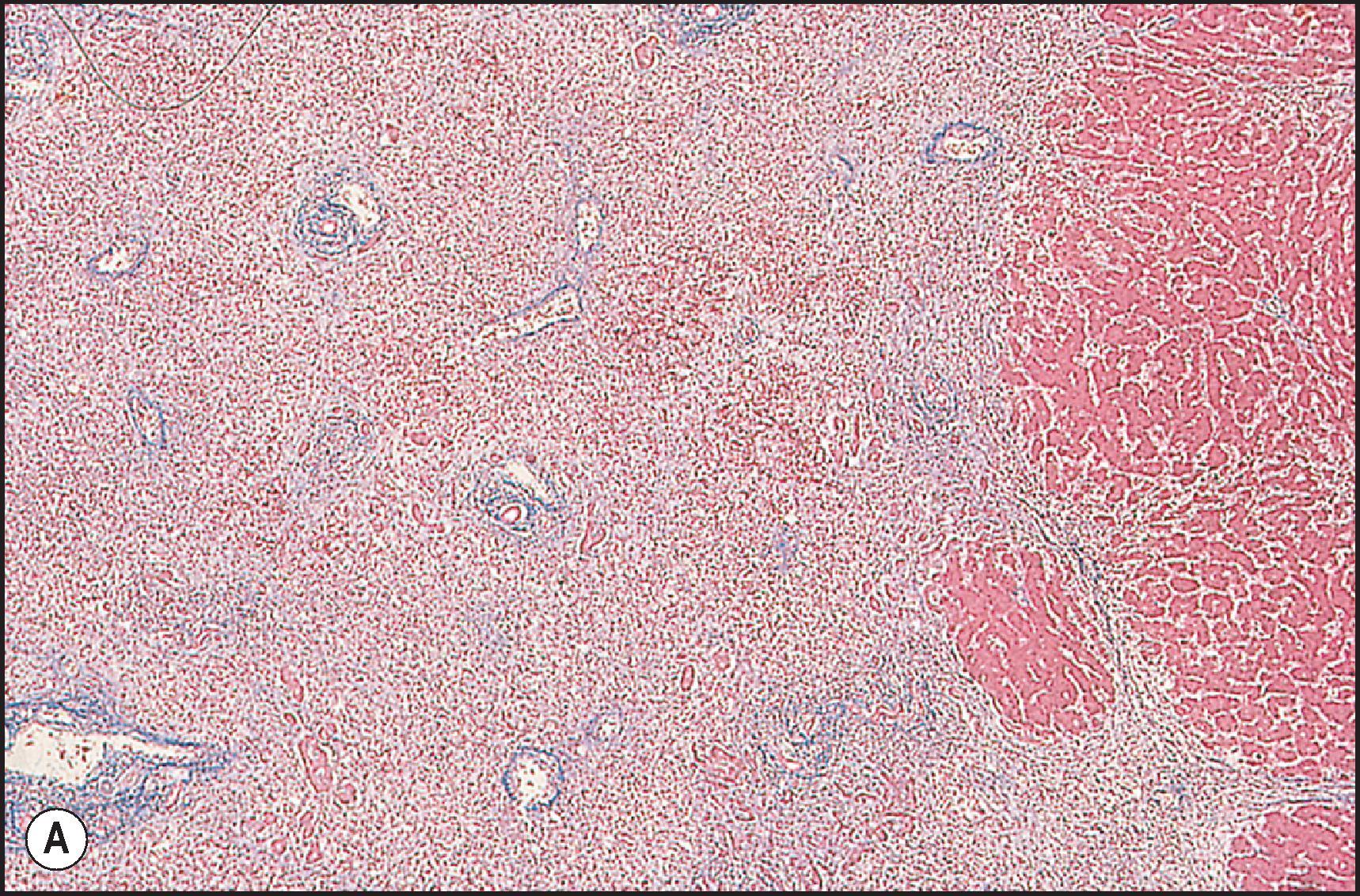
There is an alternate acute hepatitis pattern that most resembles the hepatitis of Epstein–Barr virus (EBV) infection. In this pattern there is sinusoidal beading of lymphocytes like the mononucleosis hepatitis of EBV; granulomas and eosinophils can be seen. The archetypical drug that causes this pattern of injury is phenytoin ( Fig. 12.7 ), although it can also cause cholestatic and mixed injury patterns. , Other drugs reported to show this pattern include para-amino salicylate, dapsone and the sulphonamides.
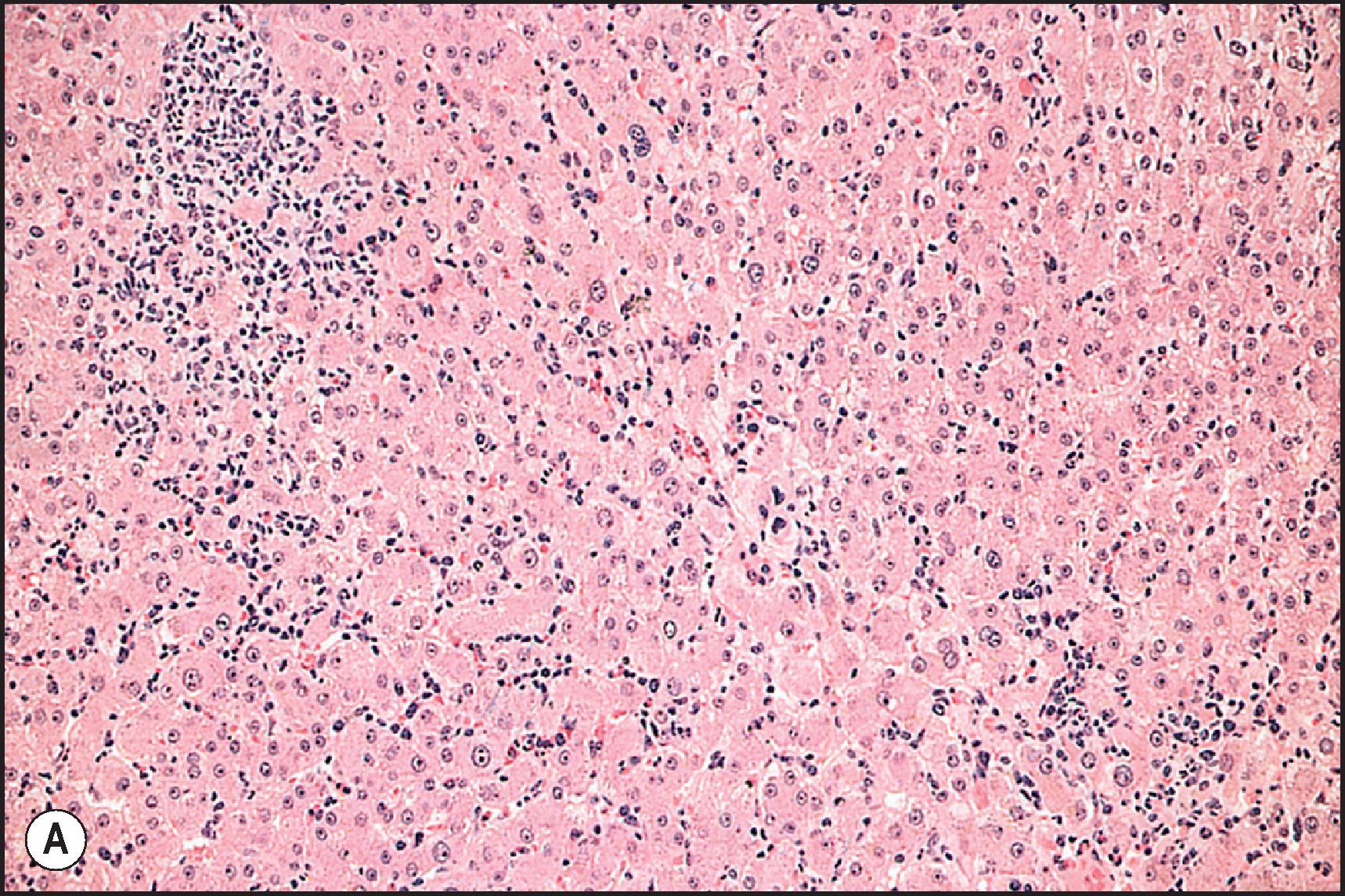
Subacute hepatic necrosis is an insidious form of necroinflammatory disease that was a dreaded occupational disease in the first half of the twentieth century. It results from prolonged exposure to benzene derivatives. , Subacute hepatic necrosis has also been associated with long-term administration of INH or methyldopa , and was seen in recipients of cinchophen. The histological features include varying degrees of necrosis, fibrosis and regeneration ( Fig. 12.6 ).
Many of the drugs that are known to cause an acute hepatitis-like pattern are also responsible for a pattern of injury that resembles chronic viral hepatitis or AIH ( Table 12.7 ). About 14% of cases from the U.S. DILIN series showed a chronic hepatitic pattern of injury. Although the pattern resembles chronic hepatitis, this does not imply that the injury is long-term or that it will persist once the agent is withdrawn. Like chronic viral hepatitis, this pattern is characterized by a necroinflammatory infiltrate that is mainly portal and periportal in distribution. The portal inflammation is mixed, but composed predominantly of CD8-(+) lymphocytes and macrophages. The lymphocytes are cytotoxic, as demonstrated by colocalization with the cytotoxic protein TIA-1. Plasma cells and eosinophils may be present. There is interface hepatitis associated with variable periportal fibrosis ( Fig. 12.8 ). Lobular inflammation is present in the form of spotty necrosis along with scattered apoptotic hepatocytes, but the injury is typically not severe enough to cause lobular disarray. Some agents may cause injury leading to significant fibrosis or cirrhosis ( Box 12.1 ) ( Fig. 12.9 ).
Acetohexamide
Amiodarone
Chlorpromazine
Cinchophen
Cyanamide
Dantrolene
Diclofenac
Ebrotidine
Etretinate
Fenofibrate
Ferrous fumarate
Halothane
Isoniazid
Lisinopril
Mercaptopurine
Methotrexate
Methyldopa
Methyltestosterone
Nimesulide
Nitrofurantoin
Oxyphenisatin
Papaverine
Perhexiline maleate
Phenylbutazone
Propylthiouracil
Tamoxifen
Thiabendazole
Ticrynafen
TPN
Valproic acid
Bush tea
Comfrey
Germander (Teucrium)
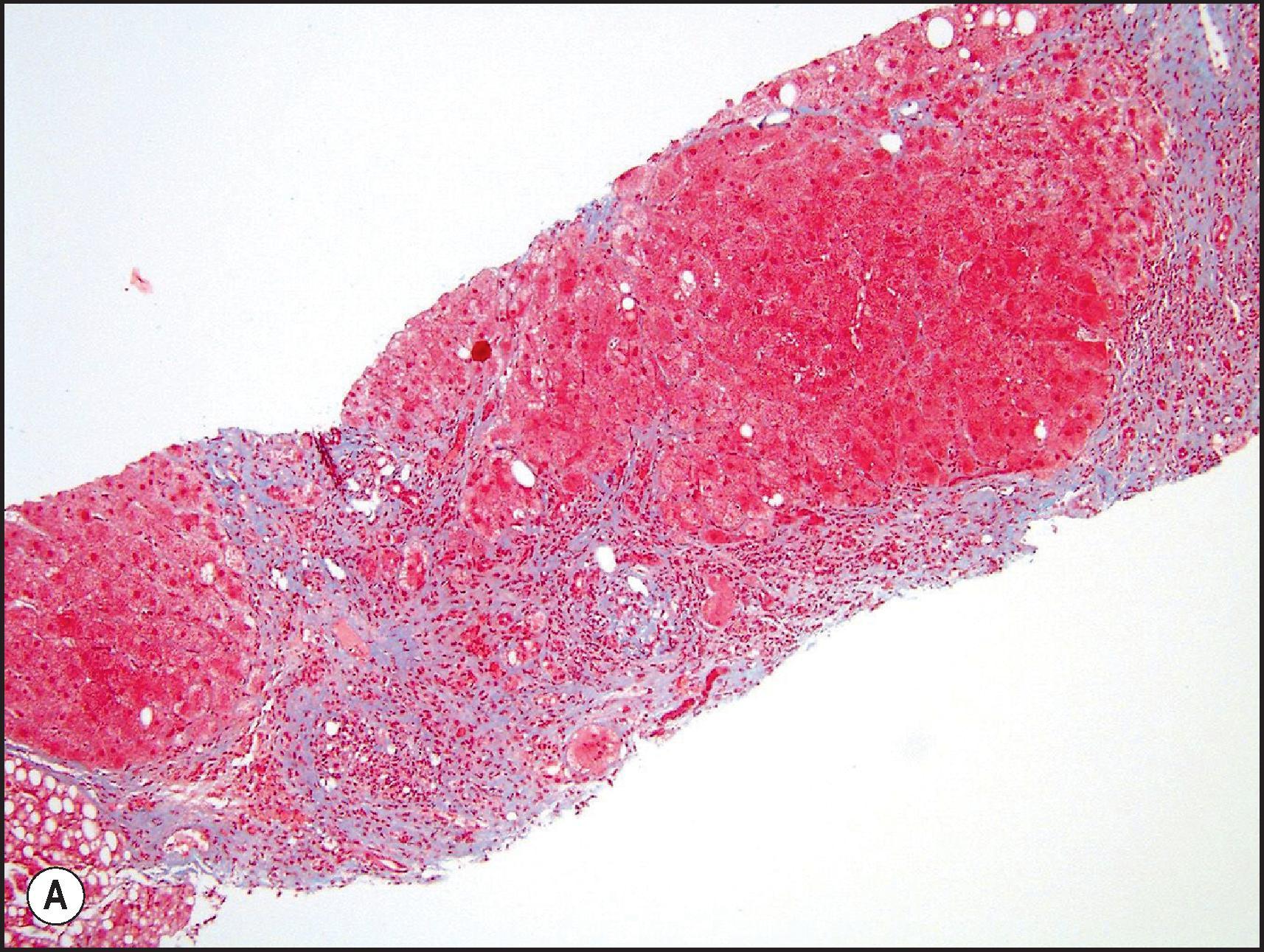
In the chronic hepatitis-like pattern, hepatocytes may show degenerative changes or steatosis as in acute hepatitis. Since cholestasis is not a feature of chronic viral hepatitis until late in the disease, its presence should raise the possibility of drug injury (or a second process). Mixed cholestatic-hepatitic injury is discussed further later. Duct injury can be seen in the chronic hepatitis pattern, even in the absence of bile stasis, and may mimic primary biliary cirrhosis. Pseudoxanthomatous changes (cholatestasis) or periportal copper accumulation should cause one to consider a chronic cholestatic injury.
The frequency with which acute DILI leads to chronic liver disease is thought to be low but clearly depends upon the definition of ‘chronicity’. Acute or subacute injury that becomes chronic from continued use of certain medications may lead to fibrosis and even cirrhosis ( Box 12.1 ). Aithal and Day published a series of cases that confirmed the chronicity of what were mostly cholestatic hepatotoxins. Chronic injury that persists after the alleged agent is discontinued appears to be far less common. In the large registry from Spain ( Table 12.1 ), chronicity was seen in 10–12% of patients, depending on the biochemical presentation. Similar figures were described among patients in the DILIN registry, with 17% of cases showing persistence of abnormal liver-associated enzymes (LAEs) after 6 months of enrollment. In a more recent study with a longer follow-up, the proportion of patients with persistent enzyme abnormalities fell from 21% at 6 months to 12% at 12 months. Biopsies available for review from cases with chronic injury mainly showed chronic cholestasis and duct loss rather than chronic hepatitis. Most cases also showed some degree of fibrosis progression. It is well described that recovery from acute DILI generally takes longer if the injury is cholestatic (16 weeks or longer) compared to acute hepatocellular necrosis (4–12 weeks). , In most cases of acute DILI, it is thought that clinical, biochemical and histological recovery is complete where the offending agent is discontinued. Chronic viral hepatitis, steatohepatitis and other non-DILI chronic liver diseases should always be excluded when evaluating patients with persistent liver enzyme abnormalities following DILI.
Drug-induced chronic hepatitis may be associated with a syndrome similar to AIH. The clinical syndrome mimics idiopathic AIH very closely. The onset of symptoms is insidious, often developing more than 2 months after the first dose of drug. The serum transaminases are elevated and there are elevated titres of serum antinuclear or anti-smooth muscle actin antibodies along with hyperglobulinaemia. Other manifestations of autoimmunity, including skin rashes and joint pains, may be observed. Recognition that drug-induced injury can mimic AIH came some 55 years ago with reports of the lesion in patients taking oxyphenisatin , an ingredient of laxative preparations no longer in use in the United States. Thereafter, the syndrome was reported in those treated with nitrofurantoin, methyldopa, , minocycline, hydralazine, halothane and statins, among others ( Table 12.8 ). Not all recipients of the drugs implicated in causing an autoimmune type of hepatitis show the expected autoantibodies. Almost 20% of patients with nitrofurantoin-associated chronic hepatitis and at least 40% of patients with methyldopa-induced chronic hepatitis are seronegative. Some agents, including hydralazine and tienilic acid, are associated with antibodies directed against microsomal components. These are CYP2C9 (also called LKM2) in tienilic acid-induced disease and the isoform CYP1A2 (a liver microsomal protein (LM)) in hydralazine-induced hepatitis. The isoforms are involved in the metabolism of the respective drug and it may be presumed that, in the process of biotransformation of the drug, an effect of the drug metabolite on the enzyme protein renders it a neoantigen. Autoimmune markers may be associated with drug reactions other than chronic hepatitis. Antinuclear antibodies may accompany chronic cholestatic injury induced by flucloxacillin and the reaction to aniline-contaminated rapeseed oil in the ‘toxic oil syndrome’ reported from Spain. They also may mark drug reactions without hepatic disease, as in the drug-induced systemic lupus erythematosus syndrome induced by hydralazine.
| Drug | Serology |
|---|---|
| Agents associated with ANA and ASMA | |
| Multiple cases | |
| Adalimumab | ANA, ASMA |
| Clometacine | ASMA, anti-DNA |
| Etanercept | ANA |
| Hydralazine | ANA, ASMA |
| Infliximab | ANA, ASMA |
| Methyldopa | ANA (16%), ASMA 35% |
| Minocycline | ANA, anti-DNA |
| Nitrofurantoin | ANA (80%), ASMA (72%) |
| Oxyphenisatin | ANA (67%), ASMA (67%) |
| Statins | ANA (80–90%), ASMA (25%) |
| Few cases (<5) | |
| Benzarone | ASMA |
| Diclofenac | ANA |
| Ecstasy (3,4-methylenedioxymethamphetamine) | ANA |
| Fenofibrate | ANA |
| Germander | ANA, ASMA |
| Papaverine | ANA, ASMA |
| Pemoline | ANA, antimicrosomal |
| Propylthiouracil | ANA |
| Agents associated with other autoantibodies | |
| Dihydralazine | Anti-CYP1A2 |
| Ticrynafen | Anti-CYP2C9 (LKM2) |
| Halothane | Anticarboxylesterase, antiprotein disulphide isomerase |
| Iproniazid | Antimicrosomal Ab 6 |
Histologically, there may be little to distinguish idiopathic AIH from drug-induced AIH. In both cases there may be a plasma-cell rich portal inflammatory infiltrate with aggressive interface hepatitis and bridging necrosis. Bridging fibrosis may be present but cirrhosis is unusual at presentation. Drug-induced AIH is more often associated with hepatocellular or canalicular cholestasis, while hepatocyte rosette formation and emperipolesis of inflammatory cells are more common in idiopathic AIH, but these findings are not absolutely specific. In follow-up, patients with drug-induced AIH are more often able to discontinue steroid therapy than patients with idiopathic AIH. Differences in nonclassical autoantibodies may exist between these two diseases that may ultimate help distinguish them.
Granulomatous inflammation is observed in a large number of drug-associated injuries ( Table 12.9 ). Non-necrotizing epithelioid granulomas were seen in about 5% of cases from the DILIN series. Granulomas may be seen alone or associated with other types of hepatic injury ( Fig. 12.10 ), including acute and chronic hepatitis, cholestasis and steatosis. More than 60 drugs have been implicated, although most are isolated case reports. A few agents appear to be well documented as typically causing granulomatous hepatitis, usually as part of a hypersensitivity syndrome (e.g. phenylbutazone, sulphonamides, allopurinol, phenothiazines and penicillins). Drug-induced granulomas have been reported with a frequency of up to 29% of all causes of hepatic granulomas, although the prevalence has usually been far lower. In the McMaster case series, the drugs that were listed as probably or possibly related were predominantly from antihypertensive, antirheumatic, anticonvulsant and antimicrobial drug classes and included methyldopa, hydralazine, phenytoin, INH, cephalexin, penicillin, sulphonamides and procainamide, among others. In other series, 6–9.5% of granulomas were due to many of the same drug classes and included chlorpropamide, allopurinol, PBZ, synthetic penicillins, quinidine, carbamazepine and phenothiazines, among others.
| Drugs | |
|---|---|
| Acetylsalicylic acid Acitretin Albendazole Allopurinol Amoxicillin-clavulanate Aprindine Atenolol Azapropazone Bacille Calmette–Guérin (BCG) Carbamazepine Carbutamide Cefalexin Chlorpromazine Chlorpropamide Clavulanic acid Clometacin Cyclofenil Dapsone Detajmium tartrate Didanosine Diflunisal Diltiazem Disopyramide Etanercept Feprazone Glibenclamide Gold Halothane Hydralazine Imipramine Interferon alpha Isoniazid Mebendazole Mestranol Methimazole Methotrexate Methyldopa Metolazone Nitrofurantoin Nomifensine Norethindrone (norethisterone) Norethynodrel Norfloxacin |
Norgestrel Oral contraceptives Oxacillin Oxyphenbutazone Oxyphenisatin Papaverine Penicillin Phenazone Phenprocoumon Phenylbutazone Phenytoin Prajmalium Procainamide Procarbazine Quinidine Quinine Ranitidine Succinylsulfathiazole Sulfadiazine Sulfadimethoxine Sulfadoxine-pyrimethamine Sulfasalazine Sulfonylurea Tacrine Thorotrast Ticarcillin-clavulanate Tocainide Tolbutamide Trichlormethiazide Trimethoprim-sulfamethoxazole Troglitazone Vemurafenib Verapamil Herbals/natural products Green-lipped mussel (Seatone) Mineral oil Polyvinyl pyrrolidone |
Microgranulomas are small foci of macrophages one to three hepatocytes in size. They are a common finding in DILI (over half the cases in the DILIN series) and other forms of liver disease and should not, by themselves, lead to a diagnosis of granulomatous hepatitis. Drug-induced hepatic granulomas have no specific features that distinguish them from those of sarcoidosis or other causes of noncaseating epithelioid granulomas, although the presence of eosinophils should suggest the possibility of a drug injury. Fibrin ring granulomas have been attributed to allopurinol and most recently to combination therapy with ipilimumab and nivolumab. , Granulomas due to Bacillus Calmette–Guerin, whether resulting from vaccination or from treatment of bladder carcinoma, may contain acid-fast bacilli. Systemic granulomatous disease does not preclude the possibility of drug-induced granulomas, since sarcoid-like granulomas have been reported in the liver, lungs and other organs as a result of chronic exposure to metals such as beryllium, copper and aluminium as well as interferon alfa.
Drug-induced granulomatous hepatitis may be related to immunoallergic DILI, particularly when associated with tissue eosinophilia. The prominence of eosinophils in the sinusoids, however, is usually a reflection of peripheral eosinophilia. , Patients with immunoallergic DILI tend to present with an acute febrile illness, rash and peripheral eosinophilia, along with jaundice and elevated LAEs. A latency of 1–16 weeks is described by McMaster et al., which is consistent with the timeframe for other immunoallergic reactions from drugs (usually within 8 weeks). Histologic features suggesting a drug-induced cause include the relatively uniform age of the granulomas and the presence of eosinophils, apoptotic bodies, acute cholangitis and/or vasculitis. In contrast, tissue eosinophilia is not seen in sarcoidosis or TB. The prognosis is generally good with recovery after the offending agent is withdrawn. Healing without sequelae is the rule. ,
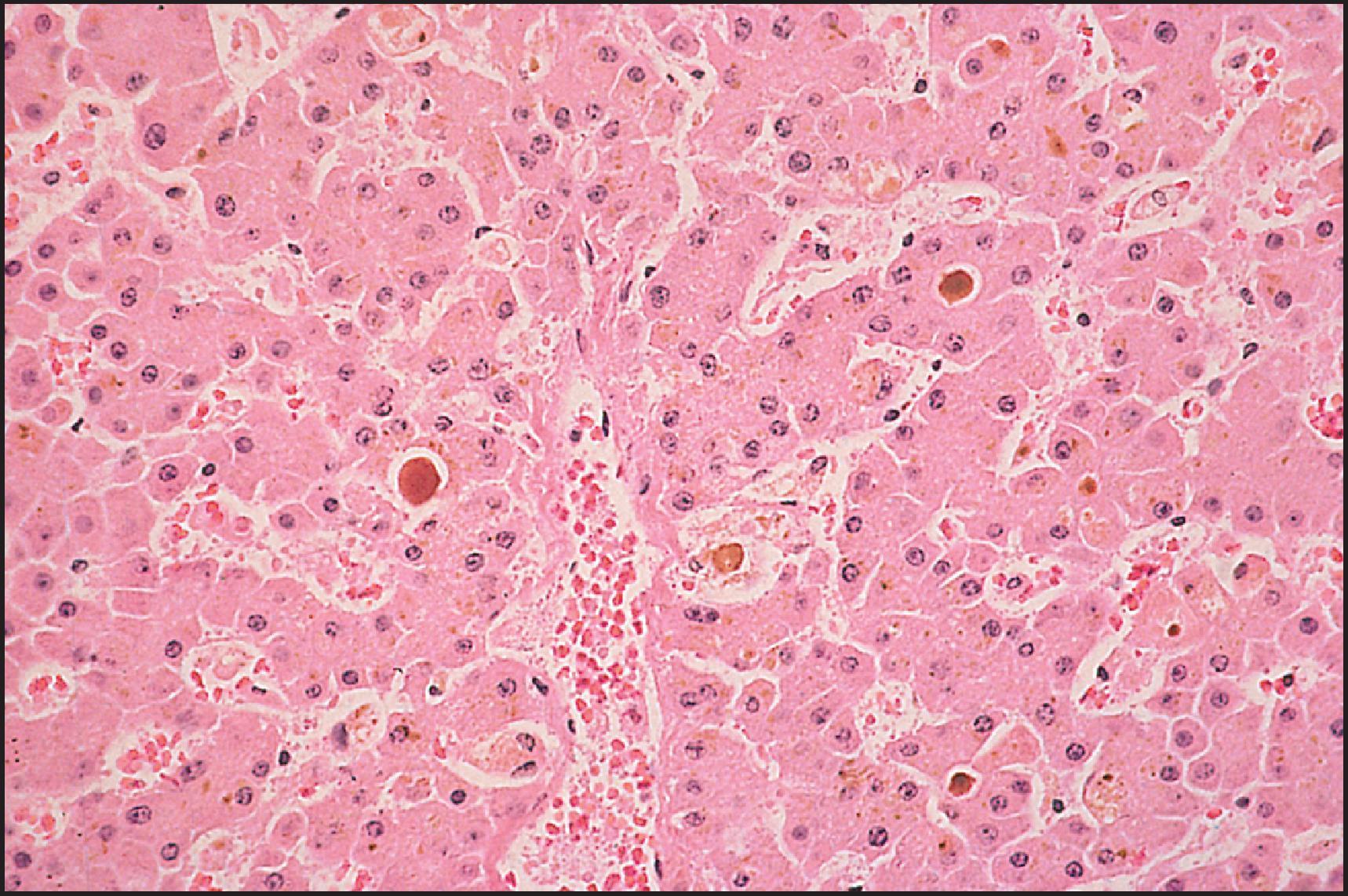
Some agents lead to injury that spares the parenchyma and instead causes arrested bile flow , ( Table 12.10 ). Intrahepatic cholestasis accompanied by significant portal or lobular inflammation should be classified as a mixed hepatocellular and cholestatic injury (cholestatic hepatitis), while the cholestasis occurring with little or no portal or lobular inflammation has been termed ‘acute’ or ‘bland’ intrahepatic cholestasis. Acute intrahepatic cholestasis accounted for 9% of cases in the DILIN series. This mild form of cholestatic injury consists mainly of bile accumulation in the cytoplasm of liver cells (hepatocellular cholestasis) and in canaliculi (canalicular cholestasis). Hepatocellular cholestasis is frequently marked by cell swelling, and the bile itself may be difficult to identify without the use of special stains. Iron and copper stains may both be more useful in this regard than a haematoxylin and eosin-stained section because of their light counterstains that allow the bile pigment to be more easily seen. An iron stain is doubly useful in that it can be used to distinguish iron (blue) from lipofuscin (granular and dirty brown) from bile (pale green to greenish brown). The cholestasis is typically most prominent in zone 3, and other processes that result in zone 3 cholestasis need to be excluded. Macrophages, sometimes bile stained, accumulate in the sinusoids of zone 3. A mild degree of parenchymal injury and inflammation (particularly portal inflammation) may be observed. This occurs with, for example, the cholestatic injury associated with chlorpromazine and erythromycin estolate ( Fig. 12.11 ) but rarely in the cholestasis associated with steroids ( Fig. 12.12 ). Different drugs may also lead to different distributions of bile accumulation. Chlorpromazine is associated with both hepatocellular and canalicular cholestasis while steroids are typically associated with pure canalicular cholestasis. Cholangiolar cholestasis like that seen in sepsis may be caused by benoxaprofen. Sometimes, the acute intrahepatic cholestasis is accompanied by acute cholangitis, which in turn may lead to chronic duct injury and loss. Temporally acute forms of vanishing bile duct syndrome (VBDS) may show acute intrahepatic cholestasis, minimal inflammation and ductopenia.
| Acute (intrahepatic) cholestasis) | Cholestatic hepatitis (combined hepatocellular–cholestatic injury) | |
|---|---|---|
| Drugs | Drugs | |
| Allopurinol Amsacrine Amitriptyline Anastrozole Androgens Aprindine Azapropazone Azathioprine Azithromycin Benoxaprofen Captopril Carbamazepine Celecoxib Chloramphenicol Chlordiazepoxide Chloropurine Chlorozotocin Chlorpromazine Chlorpropamide Chlortetracycline Cimetidine Cinnarizine Ciprofloxacin Citalopram Cloxacillin Contraceptive steroids Cyanamide Cyclosporin Danazol Diazepam Disopyramide Erythromycin Ethambutol Febuxostat Flucloxacillin Fluoxymesterone Flurazepam Gabapentin Gatifloxacin Glibenclamide Gold Haloperidol Idoxuridine Infliximab Interleukin-2 Interleukin-6 Iprindole Lansoprazole Letrozole Medroxyprogesterone |
Acetohexamide Acitretin Adalimumab Alfuzosin Allopurinol Aminoglutethimide Aminosalicylate Amoxicillin-clavulinate Ampicillin-sulbactam Aprindine Atomoxetine Atorvastatin Azathioprine Bortezomib Bromfenac Bupropion Busulfan Captopril Carbamazepine Carbarsone Carbimazole Cefadroxil Cefazolin Cefdinir Celecoxib Cetirizine Chlorambucil Chloramphenicol Chlordiazepoxide Chlorpromazine Chlorpropamide Chlortalidone Chlortetracycline Chlorzoxazone Cimetidine Cisplatin Clarithromycin Clavulanic acid Clometacin Clopidogrel Clorazepate ClozapineContraceptive steroids Cyclosporin Cyproterone Dacarbazine Dantrolene DapsoneDextropropoxyphene |
Meglumine antimoniate Meprobamate Mercaptopurine Mesalamine Metformin Metoclopramide Methyldopa Mycophenolate Naproxen Nevirapine Nicotinic acid Nifedipine Nimesulide Nitrofurantoin Nomifensine Oxacillin Oxyphenisatin Papaverine Paroxetine Pazopanib Penicillamine Phenelzine Phenindione Phenobarbital Phenothiazines (class) Phenylbutazone Phenytoin Pioglitazone Piperacillin Piperacillin-tazobactam Piroxicam Pizotifen Prajmalium Prasugrel Procainamide Prochlorperazine Propafenone Pyrazinamide Quetiapine Quinethazone Quinidine Ramipril Ranitidine Repaglinide Rifampin Risperidone Rivaroxaban Rofecoxib Sertraline Sildenafil |
| Meloxicam Mercaptopurine Meropenem Mestranol Metformin Methandrostenolone Methimazole Methyldopa Methyltestosterone Methylthiouracil Montelukast Nimesulide Nitrofurantoin Norethindrone Norethynodrel Norgestrel Oestrogens, synthetic Oxymethalone Penicillamine Phenobarbital Phenylbutazone Phenytoin Pioglitazone Piroxicam Pravastatin Prochlorperazine Ritonavir Rosiglitazone Stavudine Sulfadiazine Sulfamethoxazole Tacrolimus Tamoxifen Terfenadine Thioguanine Thioridazine Tolbutamide Valacyclovir Warfarin |
Diclofenac
Disopyramide |
Sirolimus Sulfadiazine Sulfamethoxazole Sulfasalazine Sulindac Tamoxifen Telithromycin Temozolomide Terbinafine Thiabendazole Thioguanine Thiopental sodium Ticarcillin-clavulanate Ticlopidine Tocilizumab Tolazamide Tolbutamide Tolvaptan TPN Tranylcypromine Trazodone Triazolam Trifluoperazine Trimethobenzamide Trimethoprim Trimethoprim-sulfamethoxazole Tripelennamine Troglitazone Troleandomycin Valdecoxib Valproic acid Vancomycin Vemurafenib Venlafaxine Verapamil Zimelidine Zonisamide |
| Herbals/natural products | ||
| Cascara sagrada Chaparral Greater celandine Green tea extract Kava kava Senna Syo-saiko-to |
||
When hepatocellular or canalicular cholestasis is associated with a hepatitic necroinflammatory injury, the result is a mixed form of hepatic injury that may be termed ‘cholestatic hepatitis’. Cholestatic hepatitis was the most common pattern of histological injury in the DILIN series, accounting for almost 30% of cases. Many agents are associated with this pattern of injury and it should strongly suggest a drug aetiology when present ( Table 12.10 ). , By clinical presentation and histological evaluation, cases of cholestatic hepatitis fall into two general patterns. On one end are cases with an acute hepatitis-like pattern of inflammation associated with generally mild cholestasis. Patients have high levels of serum transaminases and jaundice. Zonal necrosis may be present. Agents that appear both under acute hepatitis ( Table 12.7 ) and cholestatic hepatitis ( Table 12.10 ), such as methyldopa, have this type of presentation. The histological differential diagnosis is usually with acute viral hepatitis and sometimes with acute AIH. In the other subtype of cholestatic hepatitis, the inflammation is mild to moderate, sometimes with the low-power appearance of chronic hepatitis, with prominent portal inflammation. The cholestasis is usually apparent from low to medium magnification. This pattern blends into the acute intrahepatic cholestatic pattern as the inflammation becomes less severe. Patients present with modest elevations of transaminases and ALP as well as jaundice. Drugs that have cholestatic hepatitis as their main pattern of injury, such as amoxicillin-clavulanate, tend to have this subtype of cholestatic hepatitis. Bile duct injury may be prominent in either form and some cases that present with cholestatic hepatitis will evolve into ductopenic chronic cholestasis. ,
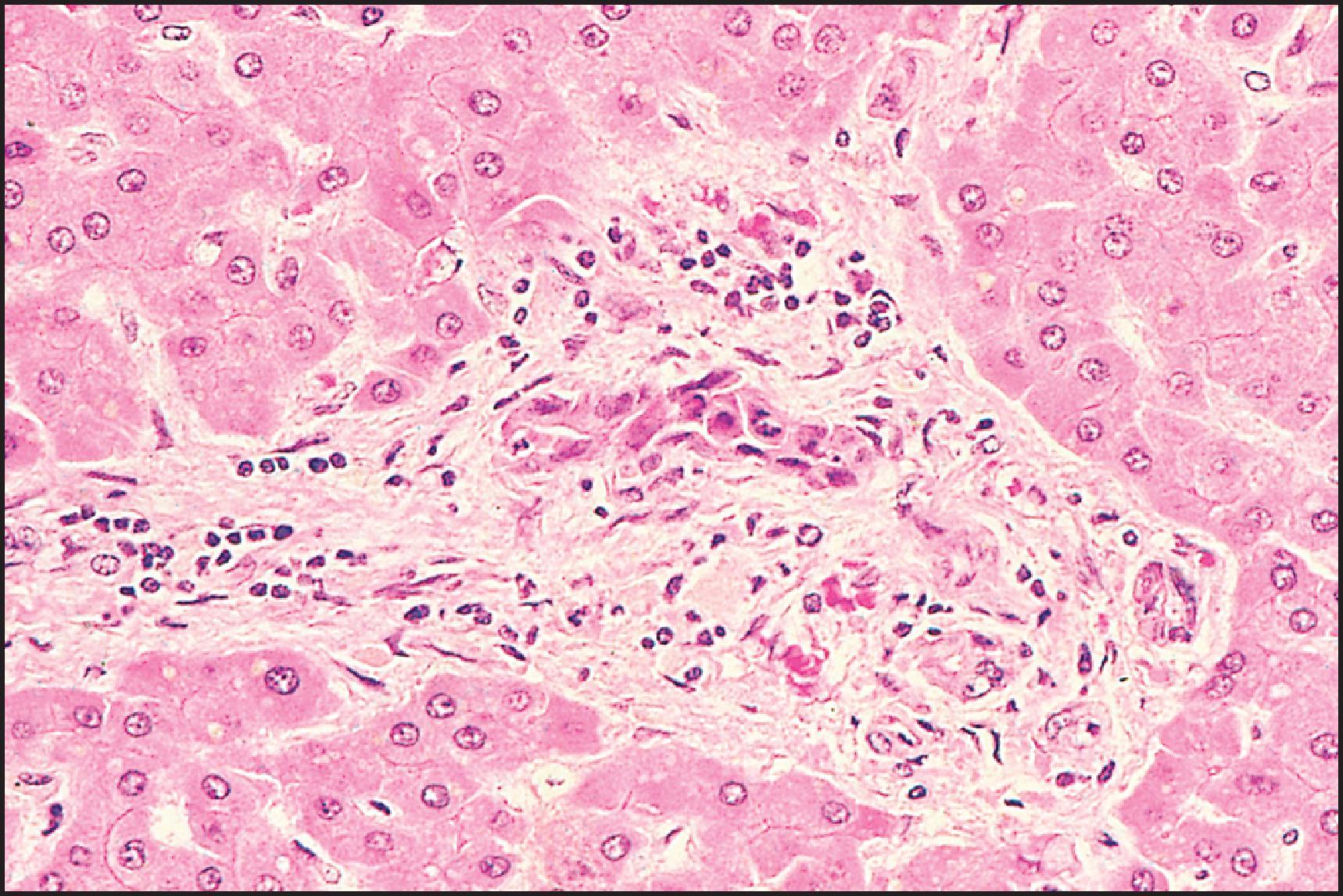
Drug-induced chronic cholestatic injury may take a variety of forms, but the central feature is chronic injury to the intrahepatic ducts. While this duct injury often results in ductopenia, chronic cholestatic changes can be observed in biopsies with apparently preserved ducts. Over 5% of the cases of acute DILI in the DILIN biopsy study had evidence of ductopenia at initial biopsy and about 10% of cases showed evidence of chronic cholestatic injury. , The injury may mimic primary biliary cholangitis (PBC) or primary sclerosing cholangitis (PSC) or the ducts may be destroyed by direct injury as exemplified by the changes ( Fig. 12.13 ) of paraquat poisoning. , Chronic cholestasis with duct damage may occur with a number of therapeutic drugs, e.g. chlorpromazine, chlorpropamide, sulphamethoxazole-trimethoprim, amoxicillin-clavulanate and others ( Table 12.11 ), and PSC-like changes may be seen on imaging. Indeed, ductal injury may accompany cholestatic hepatitis and can lead to drug-induced VBDS. , , A syndrome that resembles primary biliary cirrhosis has followed acute cholestasis due to chlorpromazine, prochlorperazine, amitryptiline, imipramine, organic arsenicals, tolbutamide and ajmaline, among others. , The degree of duct destruction and portal inflammation tends to be less prominent in the drug-induced syndrome than in PBC ( Fig. 12.14 ). Chronic cholestatic injury may lead to fibrosis and cirrhosis, and even if the drug is withdrawn, recovery may be very slow. Chronic cholestasis as demonstrated by persistently elevated ALP was the most common clinical phenotype for chronic DILI, and of those who had biopsies, over half had evidence of ductopenia. Histological features may include other changes seen in non-DILI chronic cholestasis, including periportal cholate-stasis (pseudoxanthomatous change) of hepatocytes, copper accumulation and ductular reaction. Acute cholestatic and mixed hepatocellular/cholestatic injuries may progress to chronic cholestasis as the injury evolves.
| Chronic cholestasis | Duct loss (vanishing bile duct syndrome) |
|---|---|
| Drugs | Drugs |
| Ajmaline Amineptine Amitriptyline Amoxicillin-clavulinate (PBC-like) Ampicillin Azathioprine Bortezomib Candesartan Carbamazepine Carbutamide Cefuroxime Celecoxib Chlorpromazine (PBC-like) Chlorpropamide Cimetidine Cromolyn (PBC-like) Cyamemazine Cyproheptadine Detajmium tartrate Dicloxacillin Doxycycline Enalapril Erythromycin Ezetimibe Fenofibrate Flucloxacillin Floxuridine (PSC-like) Gabapentin Gatifloxacin Gold Haloperidol Ibuprofen Imipramine Irbesartan Itraconazole Leflunomide |
Aceprometazine Ajmaline Allopurinol Amineptine Amitriptyline Amoxicillin/clavulanic acid Ampicillin Androgenic anabolic steroids Artemisinin Atorvastatin Atovaquone-proguanil Azathioprine Azithromycin Barbiturates Carbamazepine Carbutamide Cefalexin Cefazolin Cefdinir Celecoxib Chlorothiazide Chlorpromazine Cimetidine Ciprofloxacin Clindamycin Cotrimoxazole Cromolyn sodium Cyamemazine Cyclohexyl propionate Cyproheptadine Diazepam Efavirenz Enalapril Erythromycin Estradiol Fenofibrate |
| Lenalidomide Losartan Meropenem Mesalamine Methyltestosterone Montelukast Moxifloxacin Naproxen Norandrostenolone Phenothiazines (class) Phenylbutazone Phenytoin Practolol (PBC-like) Prochlorperazine Ramipril Scolicides (PSC-like) Sertraline Sulfonylurea Sulpiride Temozolomide Terbinafine Tetracycline Thalidomide Thiabendazole (PBC-like) Tiopronin Tolazamide Tolbutamide (PBC-like) TPN Troleandomycin Xenalamine Zonisamide |
Flucloxacillin Gabapentin Glibenclamide Glycyrrhizin Gold Haloperidol Hydrochlorothiazide Ibuprofen Infliximab Imipramine Itraconazole Levofloxacin Lamotrigine Lansoprazole Lenalidomide Meropenem Mesalamine Metoclopramide Methyltestosterone Montelukast Moxifloxacin Naproxen Nevirapine Norandrostenolone Olanzapine Omeprazole Oxcarbazepine Pazopanib Pelubiprofen Pexadartinib Phenobarbital Phenylbutazone Phenytoin Prochlorperazine Sertraline Temozolomide |
| Terbinafine Tetracyclines Thalidomide Thiabendazole Tibolone Tiopronin Tolbutamide Trifluoperazine Tolbutamide Trimethoprim-sulfamethoxazole |
|
| Troleandomycin Valproic acid Xenalamine Zonisamide |
|
| Herbals | |
| Glycyrrhizin | Artemisia annua |
| Prostata | |
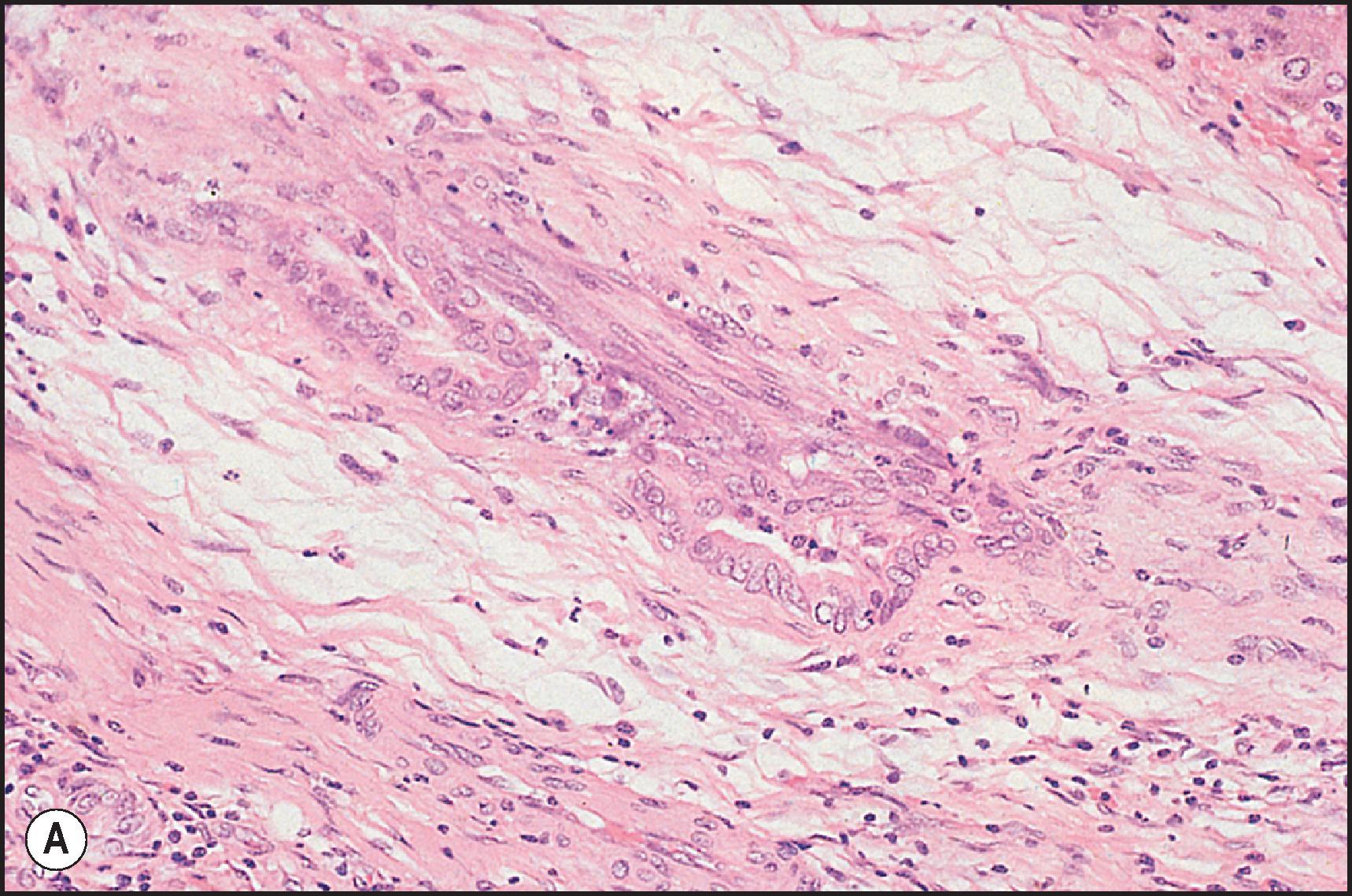
‘Biliary sclerosis’ is the term that has been applied to the biliary tree injury produced by hepatic arterial infusion therapy with floxuridine (FUDR) for metastatic carcinoma , and from the use of scolicides for treatment of echinococcal cysts. The incidence with FUDR appears to be high, and the lesion consists of blebs and oedema in the duct epithelial surface and compression and distortion of the duct lumen ( Fig. 12.15 ). Ludwig et al. have attributed the ductal injury to occlusive arterial injury and have labelled it an ‘ischaemic cholangiopathy’. On cholangiography, the lesion resembles PSC.
Nonalcoholic fatty liver disease (NAFLD) is a rubric currently used to encompass all forms of liver disease that have some degree of steatosis and in which alcohol and other known liver diseases do not play a role. NAFLD is most often associated with insulin resistance; the estimated prevalence of NAFLD is very high, and up to 5% of the general population may have some form of NAFLD. Consequently, the presence of steatosis in the liver biopsy is not a very specific finding, particularly when present in only a mild to moderate degree. Nevertheless, steatosis has been associated with a large number of agents ( Table 12.12 ) and may be seen combined with other forms of liver injury, such as zonal necrosis.
| Steatosis | Steatohepatitis | |
|---|---|---|
| Chemicals and toxins | Drugs | Drugs |
| 2-Chloropropane Aflatoxins (micro) Amanitin (macro) Antimony Arsenic compounds Azaserine Bacillus cereus toxin Barium salts Borates (both) Bromobenzene Cadmium (macro) Carbon disulphide Carbon tetrachloride (macro) Chlorinated diphenyls Chlorinated naphthalene Chloroprene Chromates (macro) Cocaine (both) DDT Dichloroethylene (macro) Dichloropropane Dimethylformamide (both) Dimethylhydrazine Dimethylnitrosamine Dinitrobenzone Dinitrotoluene Ethanol (both) Ethionine (both) Ethyl bromide (macro) Ethyl chloride (macro) Ethylene dibromide Ethylene dichloride Flectol H Galactosamine Halogenated hydrocarbons (macro) Hydrazine Hypoglycin A (micro) Luteoskyrin Mercury Methyl bromide (macro) Methyl chloride (macro) Methyl chlorobromide Methyl dichloride (macro) Methylchloroform Mushrooms (both) Naphthalene Organic solvents (macro) Orotic acid (macro) Phalloidin (micro) Phosphorus (both) Rare earths (low atomic #) Safrole Tetrachloroethane (macro) Tetrachloroethylene (macro) Trichloroethylene (macro) Trinitrotoluene Uranium compounds (macro) |
Acetaminophen (Paracetamol) (macro) Acetylsalicylic acid (both) Amineptine (micro) Amiodarone (micro) Amsacrine Asparaginase (macro) Azacytidine Azauridine Calcium hopantenate Camphor (micro) Chloroform (macro) Chlortetracycline (micro) Cisplatin (macro) Clometacin (macro) Corticosteroids (macro) Cortisone (macro) Cyanamide (macro) Demeclocycline (micro) Desferoxamine (micro) Dichloromethotrexate Didanosine (micro) Oestrogens, synthetic Etretinate (macro) Fialuridine (both) Gold (macro) Hydralazine (macro) Hydrazines Indinivir Indomethacin (both) Interleukin-2 Iodoform Irinotecan (macro) Isoniazid (macro) Ketoprofen (micro) Lomitapide Linezolid (micro) Mefloquine Methimazole (macro) Methotrexate (macro) Methyl salicylate (micro) Methyldopa (macro) Minocycline (macro) Mitomycin C (macro) Nifedipine Nitrofurantoin (macro) Oxytetracycline (micro) Pegaspargase Perhexiline maleate (macro) Piroxicam Pirprofen (micro) Puromycin Rifampin (macro) Riluzole (micro) Risperidone Ritonavir Rolitetracycline (micro) Spironolactone Stavudine (both) Streptozotocin |
Amiodarone Corticosteroids Oestrogens, synthetic Irinotecan Methotrexate Perhexiline maleate Raloxifene Risperidone Spironolactone Sulfasalazine Tamoxifen |
| Sulfasalazine (macro) Sulindac (macro) Synthaline |
||
| Tamoxifen (macro) Tetracycline (micro) Thiotepa Tolmetin (micro) TPN (macro) Valproic acid (both) Vitamin A (micro) Zidovudine (macro) |
||
| Herbals/natural products | Drugs associated with phospholipidosis | |
| Jin bu huan (micro) Margosa oil (micro) Pennyroyal oil (micro) Pentanoic acid (micro) Syo-saiko-to (micro) Tannic acid (macro) |
Amiodarone Amitryptyline Chloramphenicol Chloroquine Chlorpromazine Oestrogens, synthetic Gentamycin Ketoconazole Perhexiline maleate Promethazine Thioridazine TPN Trimethoprim-sulfamethoxazole |
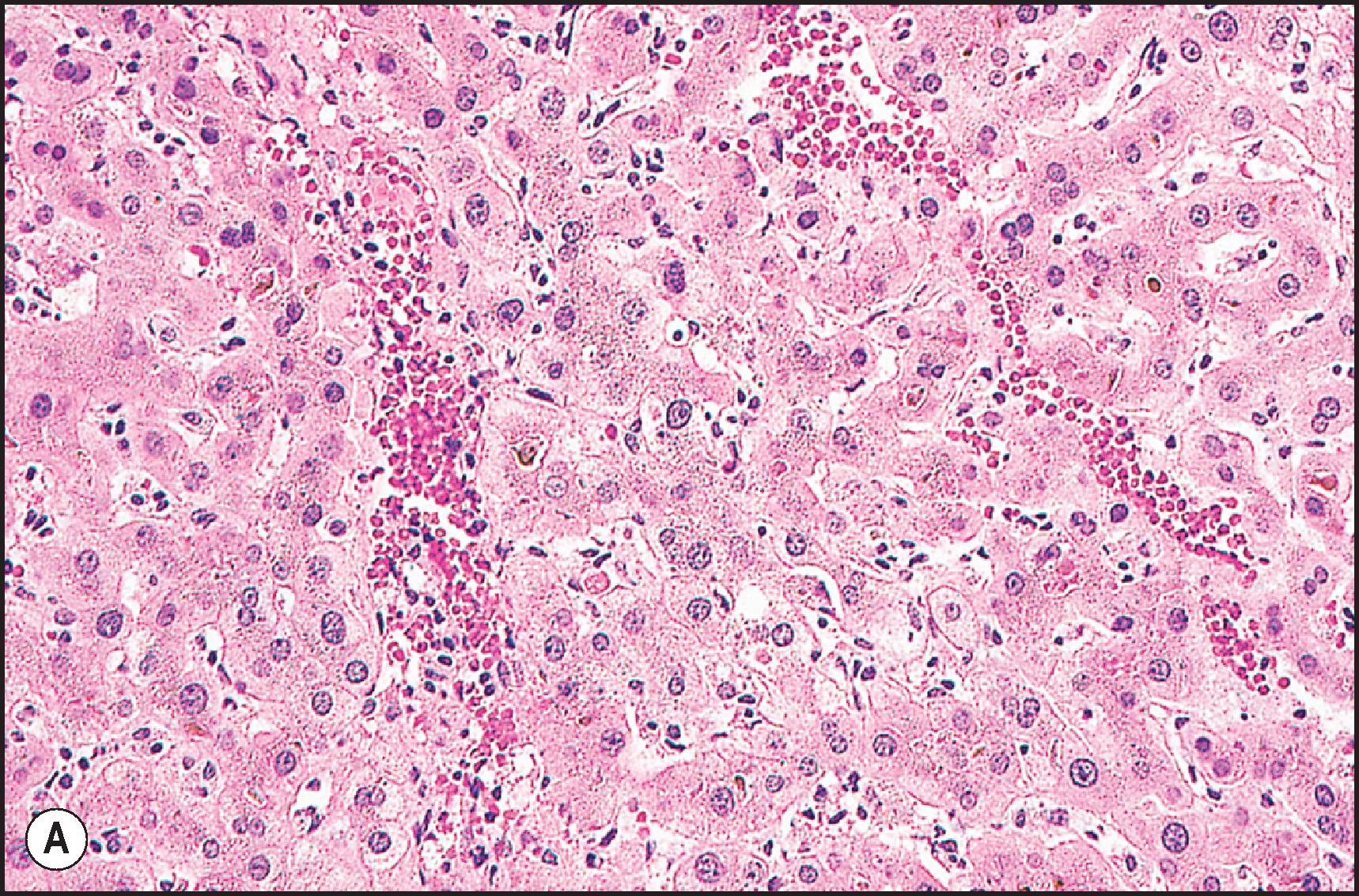
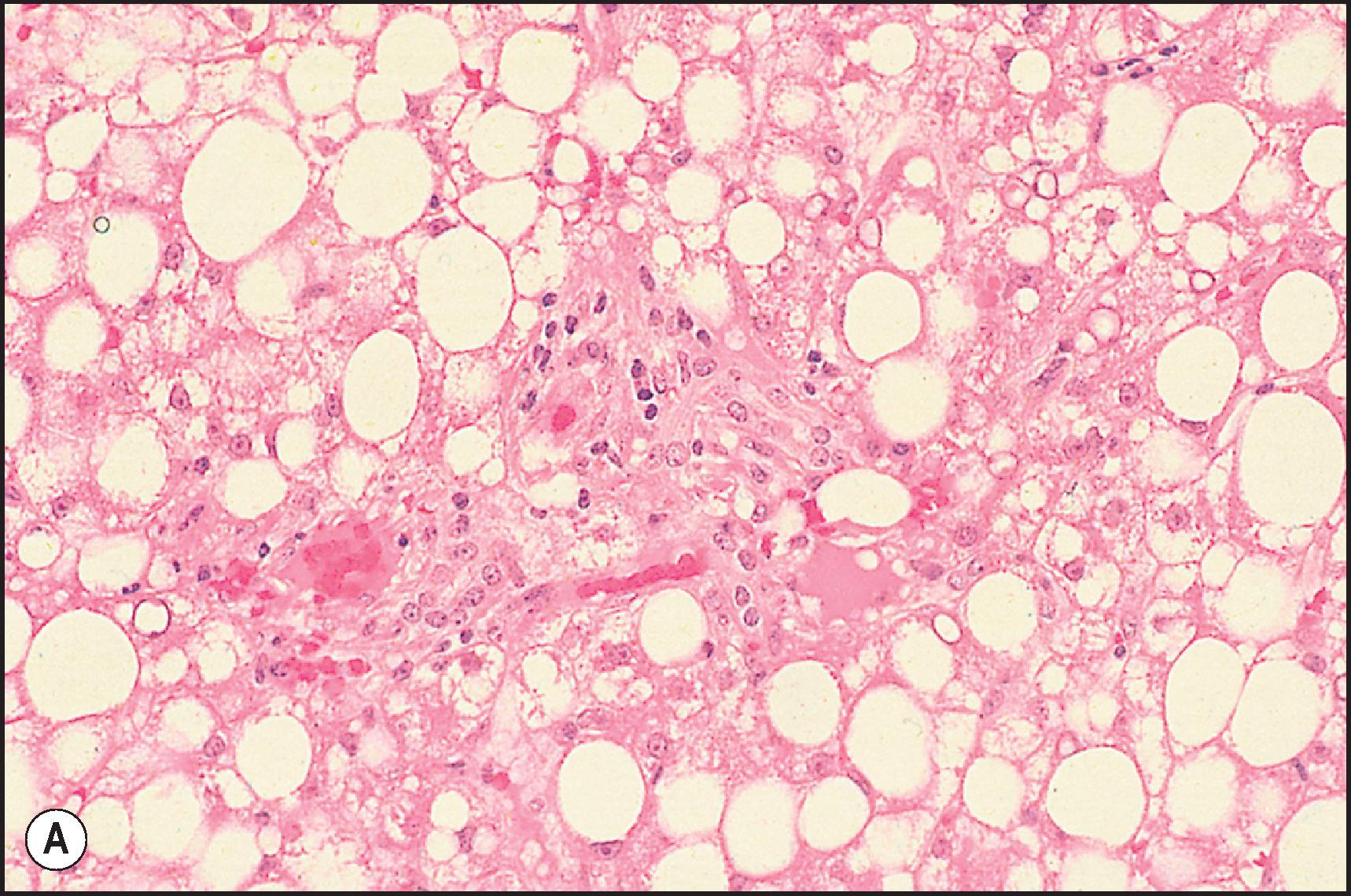
Two main types of steatosis occur—macrovesicular and microvesicular—and both may be present simultaneously in various combinations. Microvesicular steatosis is generally associated with acute presentations with jaundice and lactic acidosis and is characterized by a foamy change in the hepatocyte cytoplasm composed of tiny fat droplets that do not displace the nucleus ( Fig. 12.16 ). Ideally the droplets are so tiny that they are truly innumerable. Drug-induced microvesicular steatosis has been observed with tetracycline, valproic acid, , some nucleoside analogues and salicylates (Reye syndrome). Nontherapeutic toxins associated with microvesicular steatosis include ackee poisoning in children from hypoglycin A (Jamaican vomiting sickness), occupational toxicity from dimethylformamide and cocaine abuse. In at least some of these toxicities, mitochondrial injury is a common theme, but the actual mechanism may vary. Mitochondrial injury has been linked with uncoupling of oxidative phosphorylation, inhibition of fatty acid oxidation, inhibition of the mitochondrial respiratory chain, opening of mitochondrial permeability transition pores and damage to mitochondrial DNA. In the DILIN biopsy series, microvesicular steatosis was associated with serious outcomes, including clinical hepatic failure, liver transplantation and death, even when present only focally.
In macrovesicular steatosis , the hepatocytes contain a medium or large-size cytoplasmic fat droplet that displaces the nucleus to the periphery. Frequently multiple vacuoles of varying sizes are observed in a single cell. This mixed form of steatosis should still be considered macrovesicular steatosis. Macrovesicular steatosis is characteristic of alcohol abuse, methotrexate injury and a number of other agents. Steatosis may be secondary to another drug effect, like weight gain or lipodystrophy. The steatosis may be the sole finding or may be associated with varying degrees of inflammation and necrosis and, as with microvesicular steatosis, may be associated with mitochondrial injury. The combination of zonal necrosis and steatosis is observed with a number of toxic agents, including acetaminophen, halogenated hydrocarbons, tannic acid and arsenicals. Glucocorticoid steatosis is frequently bland, while methotrexate-related steatosis is associated with inflammation and fibrosis. There are differences in the zonal distribution of fat of different toxic aetiologies. Phosphorus leads mainly or initially to accumulation of fat in the periportal zone, while tetracycline and alcohol lead predominantly or initially to perivenular steatosis.
Steatohepatitis caused by drugs and toxins has essentially the same histologic features as other forms of steatohepatitis. The basic pattern of injury is a zone 3 hepatocellular injury characterized by hepatocellular ballooning and Mallory–Denk body formation with variable lobular inflammation. Portal inflammation may also be present. The associated fibrosis is typically perisinusoidal and perivenular at first but can progress to bridging fibrosis and cirrhosis. In drug-induced steatohepatitis, the basic pattern of zone 3 injury may be altered, but the components of inflammation, steatosis and Mallory–Denk body formation remain. For example, in amiodarone injury ( Fig. 12.17 ), periportal Mallory–Denk bodies may be observed. Many of the drugs that have been associated with steatohepatitis and Mallory–Denk body formation are also associated with phospholipidosis ( Fig. 12.17C, D ). The drugs in current use chiefly responsible for the entity are perhexiline maleate and amiodarone. Steatohepatitis or Mallory–Denk bodies (without phospholipidoses) have also been produced by diethylstilboesterol, prednisolone, nifedipine, tamoxifen and didanosine ( Table 12.12 ). Some of these drugs, notably tamoxifen, methotrexate, corticosteroids, diethylstilboestrol and oestrogens, seem to have their effect by exacerbating underlying steatohepatitis. Some of the antiretroviral drugs have been associated with a syndrome of dyslipidaemia, fat maldistribution and insulin resistance that has been termed ‘human immunodeficiency virus (HIV)-associated lipodystrophy syndrome’. The steatosis and steatohepatitis that have been observed in patients with HIV infection may be more related to lipodystrophy than to a direct toxic effect on the liver. The implicated drugs include ritonavir, amprenavir, nelfinavir, indinavir, saquinavir, atazanavir and the combination drug lopinavir/ritonavir. Because the nondrug causes of steatosis and steatohepatitis are common, caution is advised in making new associations between drug injury and fatty liver disease.
Phospholipidosis is an additional form of drug-induced lipid accumulation. The lesion was first recognized in Japan in 1969 as a complication of the early coronary vasodilator diethylaminoethoxyhexoestrol. Phospholipidosis is characterized by enlarged, foamy or granular hepatocytes or Kupffer cells by light microscopy ( Fig. 12.17C ); lamellated or crystalloid inclusions can be seen by electron microscopy ( Fig. 12.17D ). The lesion resembles the changes seen in inborn disorders of phospholipid metabolism. It has since been seen in patients taking perhexiline maleate , or amiodarone , and has been reproduced in experimental animals by amphiphilic compounds. , Indeed, the amphiphilic character of the drugs appears to account for the accumulation of phospholipids in lysosomes of hepatocytes and other cells. The drugs bind to phospholipids and inhibit their hydrolysis by lysosomal phospholipase A. The development of phospholipidosis may represent a form of lysosomal defence in which the agent is sequestered in the stacks of phospholipid lamellae, which may ultimately be extruded into the extracellular environment. Although most drugs associated with phospholipidosis are also associated with steatohepatitis, there are some only associated with phospholipidosis ( Table 12.12 ).
Fibrosis and cirrhosis may result from a variety of drug- and toxin-induced injuries ( Table 12.8 ). Agents which cause chronic hepatitis, chronic cholestasis and steatohepatitis may all have cirrhosis as an end point. , Acute hepatic injury does not appear to lead to cirrhosis after biochemical and clinical resolution of the acute injury following drug discontinuation. This is similar, for example, to the expected course seen after acute viral hepatitis A. It is important in such instances that underlying hepatic diseases be excluded, including chronic viral hepatitis, chronic cholestatic diseases and fatty liver (both alcoholic and nonalcoholic), that may be responsible for the chronic disease that is found. Fibrosis may follow massive or submassive hepatic necrosis if the patient survives without transplantation.
Periportal fibrosis may be seen in drug-induced acute and chronic hepatitis and chronic cholestasis. In the acute injury phase, the periportal fibrosis probably resolves quickly after the agent is withdrawn. Methotrexate can lead to periportal fibrosis when used in the treatment of leukaemia and to both periportal and bridging fibrosis (and even cirrhosis) when used in the long-term therapy of psoriasis and, rarely, of rheumatoid arthritis ( Fig. 12.18 ). Hypervitaminosis A, discussed later, can lead to perivenular fibrosis. This is accompanied by marked hypertrophy of the hepatic stellate cells, atrophy of hepatocytes, veno-occlusive lesions and sinusoidal dilatation.
Macronodular and micronodular cirrhosis, , congestive hepatopathy resembling cardiac cirrhosis and a biliary cirrhosis-like lesion can all result from toxin- or drug-induced liver damage. Macronodular or micronodular cirrhosis may be a sequel to continued or repetitive injury or the result of subacute necrosis, or chronic necroinflammatory disease. A single episode of necrosis may leave architectural distortion, but cirrhosis does not occur. A single episode of zonal necrosis in experimental animals (e.g. CC1 4 poisoning), even when extensive, is followed by complete histological restitution in surviving animals.
The most important path to drug-induced cirrhosis occurs via the lesions of chronic hepatitis ( Fig. 12.9 ), subacute necrosis ( Fig. 12.6 ) and phospholipidosis-steatohepatitic injury ( Fig. 12.17 ). Biliary cirrhosis may occur as a sequel to chronic cholestatic drug injury, and as noted earlier, an obstructive biliary cirrhosis type of injury can be a consequence of the biliary sclerosis produced by hepatic artery infusion of floxuridine ( Fig. 12.15 ). Fibrosis progression has been found in a biopsy series of patients with chronic DILI and in patients with HIV on long-term highly active antiretroviral therapy (HAART).
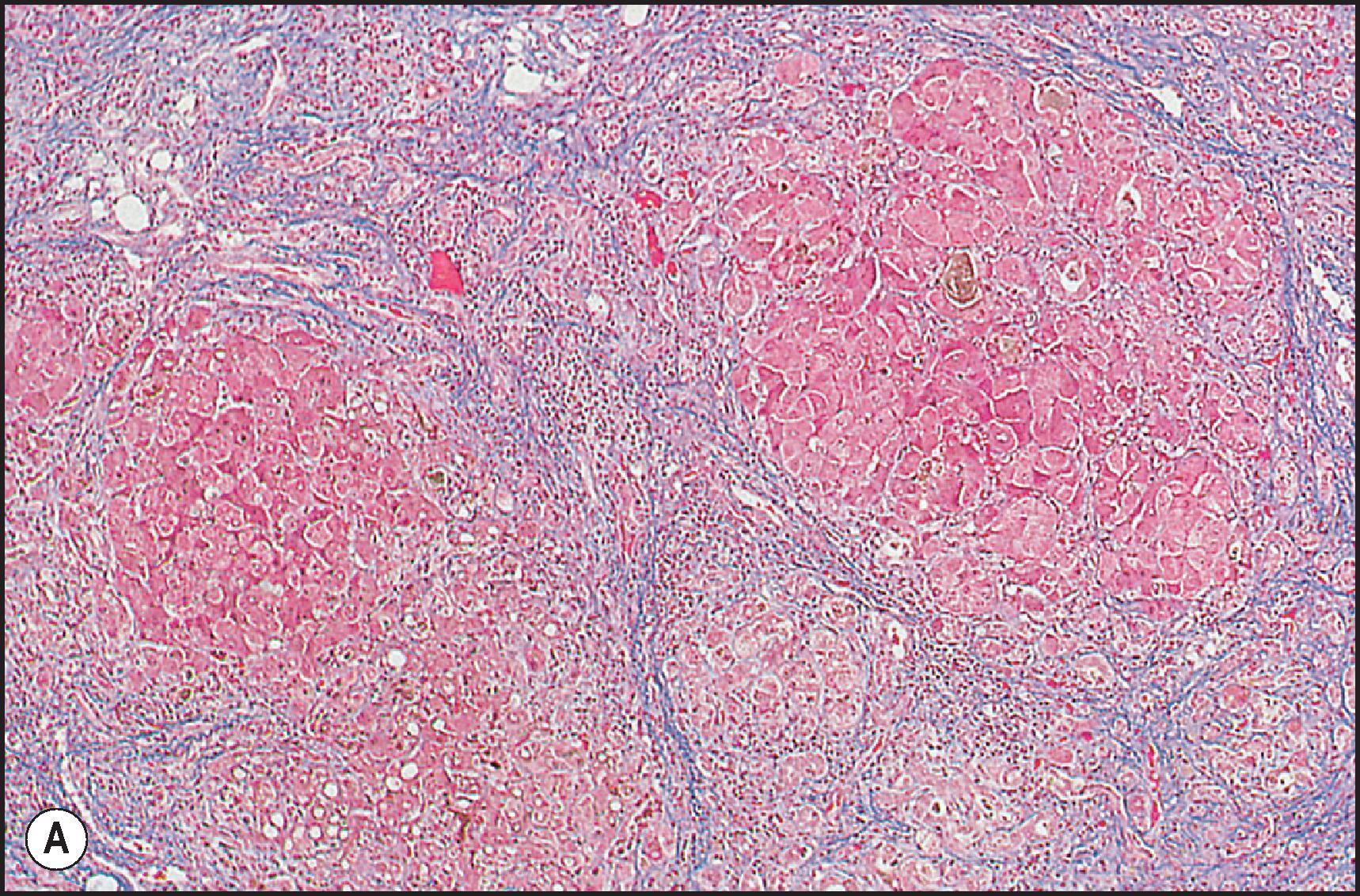
A number of important vascular lesions can be produced by drugs ( Table 12.13 ). , Two involve blockade of efferent blood flow with congestive hepatopathy, leading to perivenular necrosis; when severe, midzone necrosis also occurs. Prolonged occlusion leads to fibrotic bridging between adjacent hepatic vein branches and a picture resembling cardiac cirrhosis. The two occlusive lesions are thrombosis of the hepatic veins ( Fig. 12.19 ) and fibrotic occlusion of the hepatic venules and sinusoids ( Fig. 12.20 ). Additional lesions include peliosis hepatis ( Fig. 12.21 ), sinusoidal dilatation ( Fig. 12.22 ), perisinusoidal fibrosis and hepatoportal sclerosis ( Fig. 12.23 ). Abnormal blood flow that results from a number of these lesions may cause irregular regeneration, resulting in NRH ( Fig. 12.24 ). It has been suggested that these lesions are linked by a common pathway of endothelial cell injury.
| VOD/SOS | Peliosis | Sinusoidal Dilation |
|---|---|---|
| Drugs | Drugs | Drugs |
| Azathioprine Busulfan Carboplatin Carmustine (BCNU) Chlormethine (Mechlorethamine) Cisplatin Contraceptive steroids Cyclophosphamide Cysteamine Cytarabine Dacarbazine Dactinomycin Danazol Daunorubicin Dimethylbusulfan Doxoribicin Floxuridine (FUDR) Gemtuzumab Indicine Mercaptopurine Mitomycin C Oxaliplatin Tacrolimus Tamoxifen Thioguanine (tioguanine) Urethane (ethyl carbamate) Vinblastine Vincristine |
Androgens Azathioprine Busulfan Contraceptive steroids Corticosteroids Danazol Diethylstilboestrol Oestrogens, synthetic Estrone sulphate Fluoxymesterone Glucocorticoids Hydroxyprogesterone Hydroxyurea (hydroxycarbamide) Medroxyprogesterone Mercaptopurine Methandrostenolone Methyltestosterone Oxaliplatin Tamoxifen Testosterone Thioguanine (tioguanine) Thorotrast Vitamin A |
Androgens Azathioprine Carmustine (BCNU) Contraceptive steroids Dacarbazine Daunorubicin Metoclopramide Mitomycin C Oxaliplatin Thioguanine (tioguanine) Valproic acid Vinblastine Vitamin A |
| Herbals/natural products | Chemicals and toxins | |
| Bush tea Comfrey Mate tea Pyrrolizidine alkaloids |
Vinyl chloride | |
| Nodular regenerative hyperplasia | Budd–Chiari syndrome | Vasculitis |
| Drugs | Drugs | Drugs |
| Ado-trastuzumab emtansine Androgens Azathioprine Contraceptive steroids Corticosteroids Cytarabine Daunorubicin Interleukin-2 Mercaptopurine Oxaliplatin Thioguanine (tioguanine) Thorotrast |
Contraceptive steroids Cyclophosphamide Dacarbazine Vincristine |
Allopurinol Chlorpropamide Phenylbutazone Phenytoin |
| Hepatoportal sclerosis | ||
| Chemicals and toxins | Agents | |
| Copper sulphate Toxic oil (rapeseed) Vinyl chloride |
Arsenic compounds Contraceptive steroids Vinyl chloride |
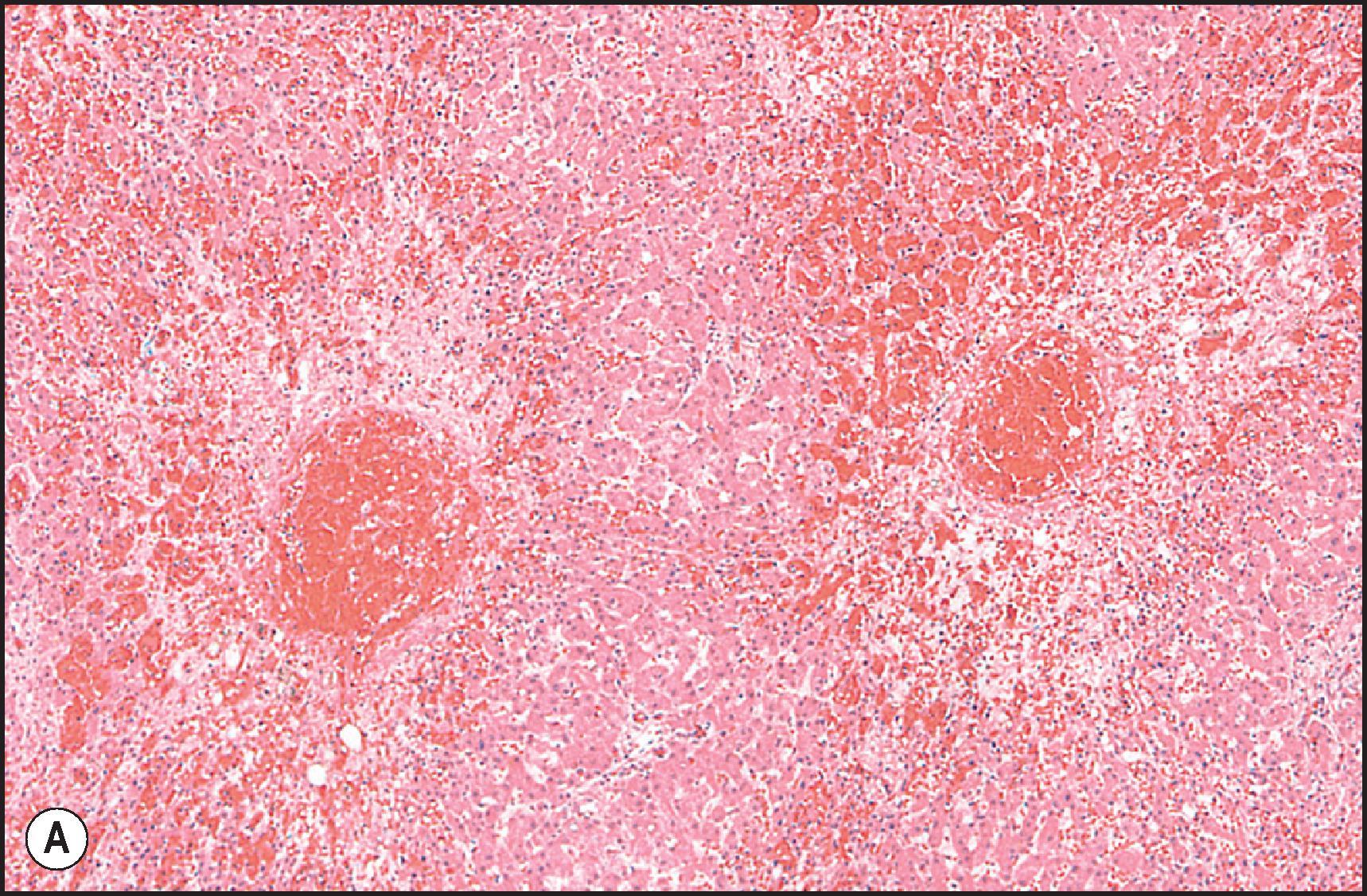
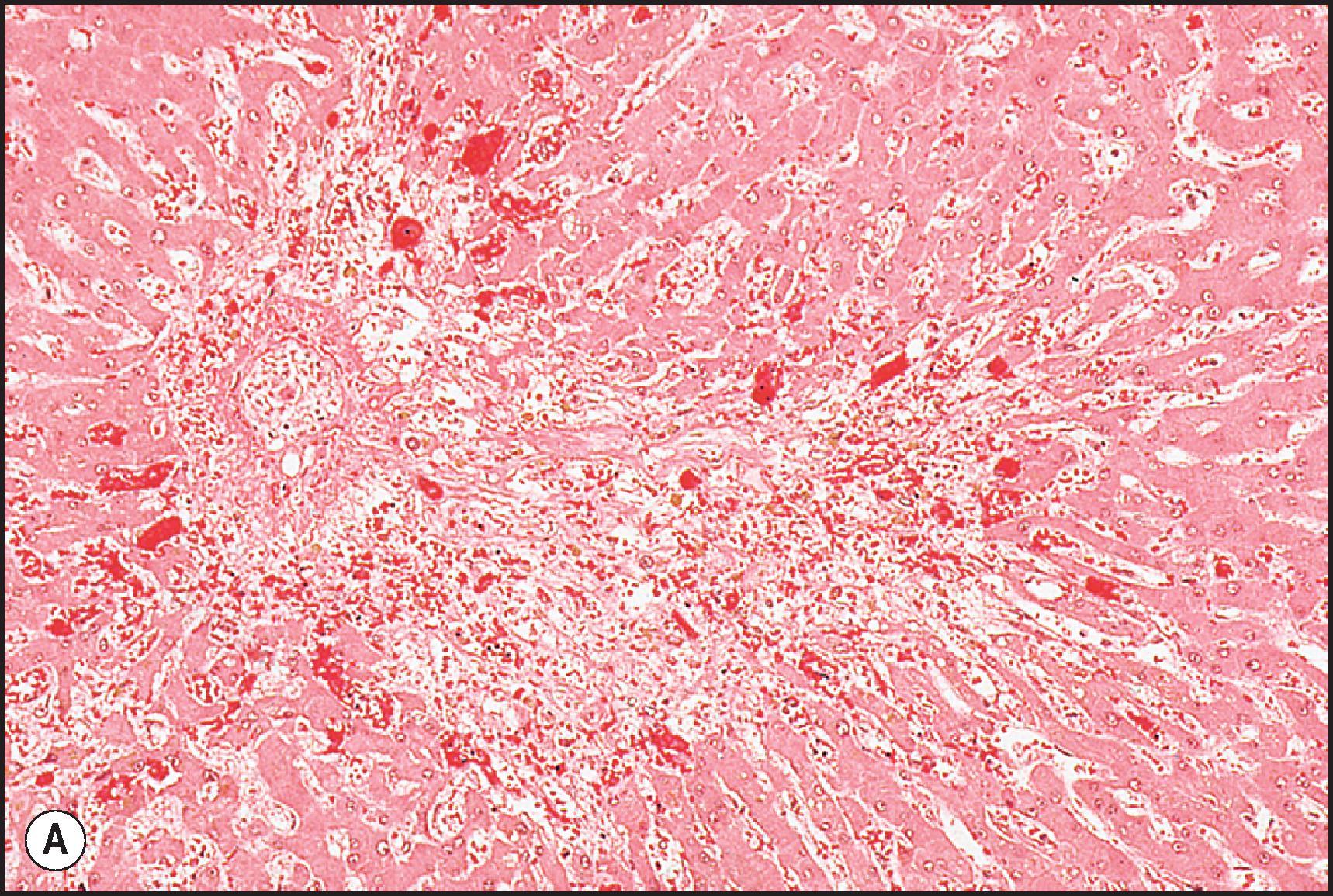
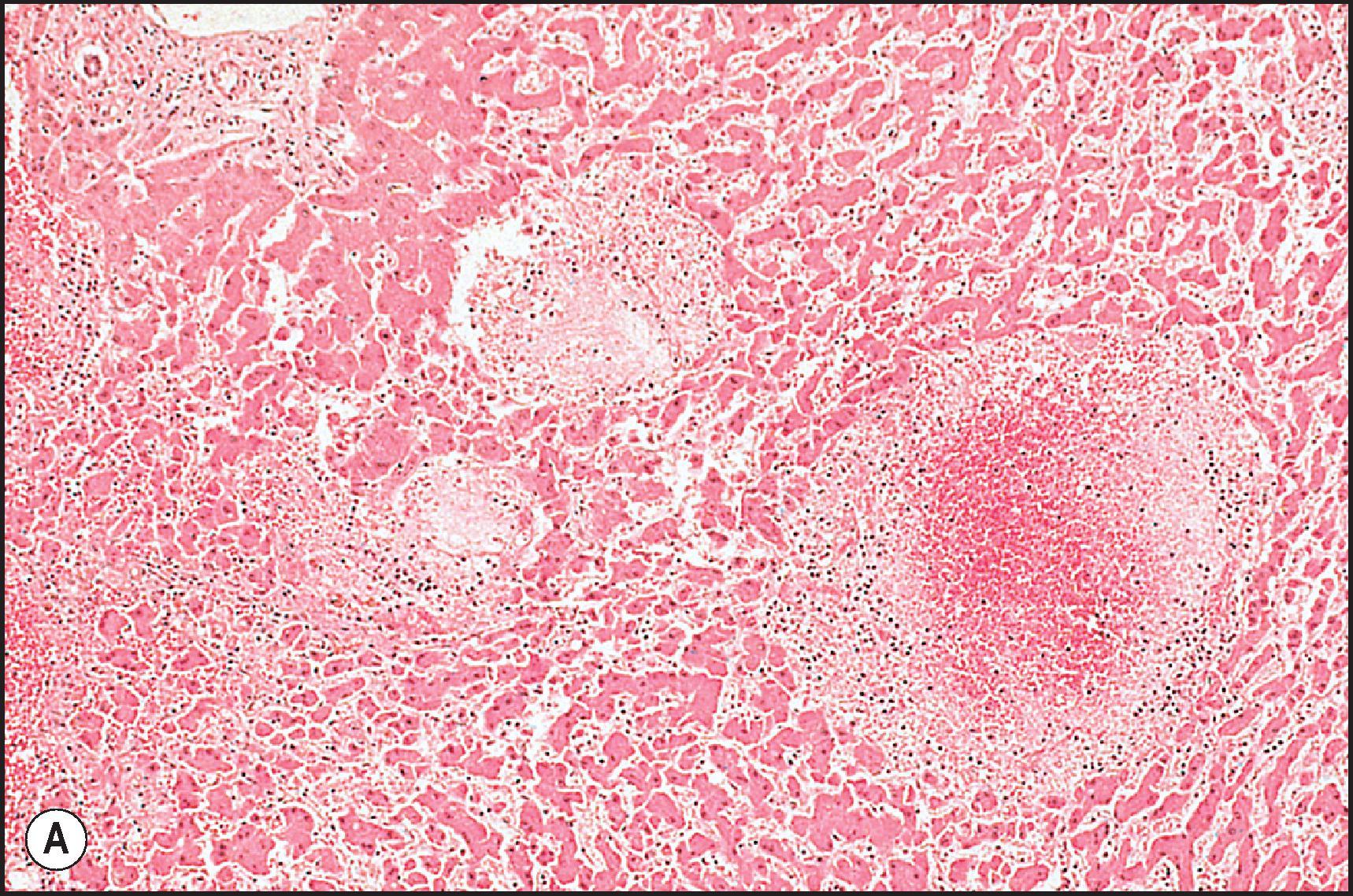
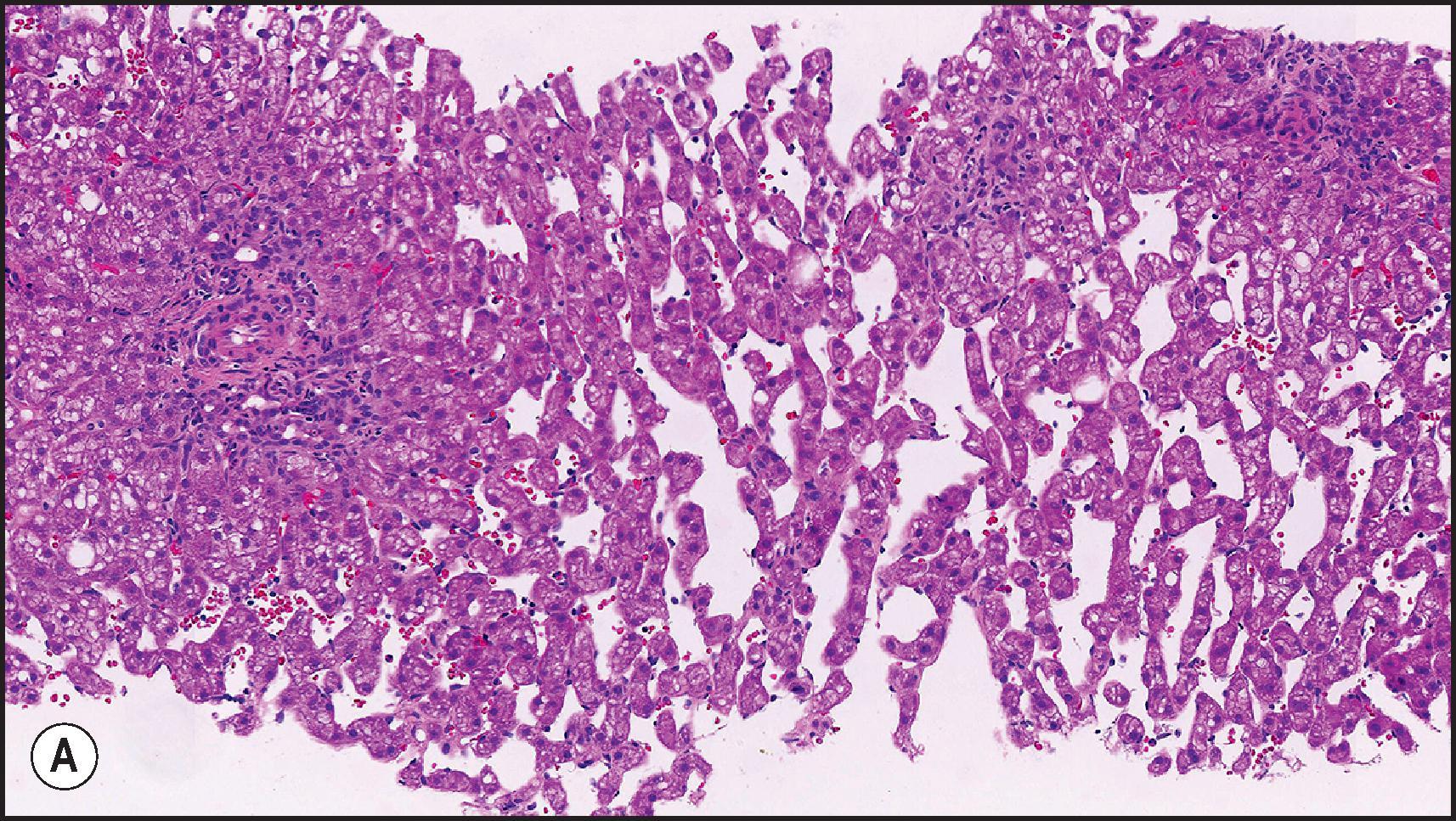
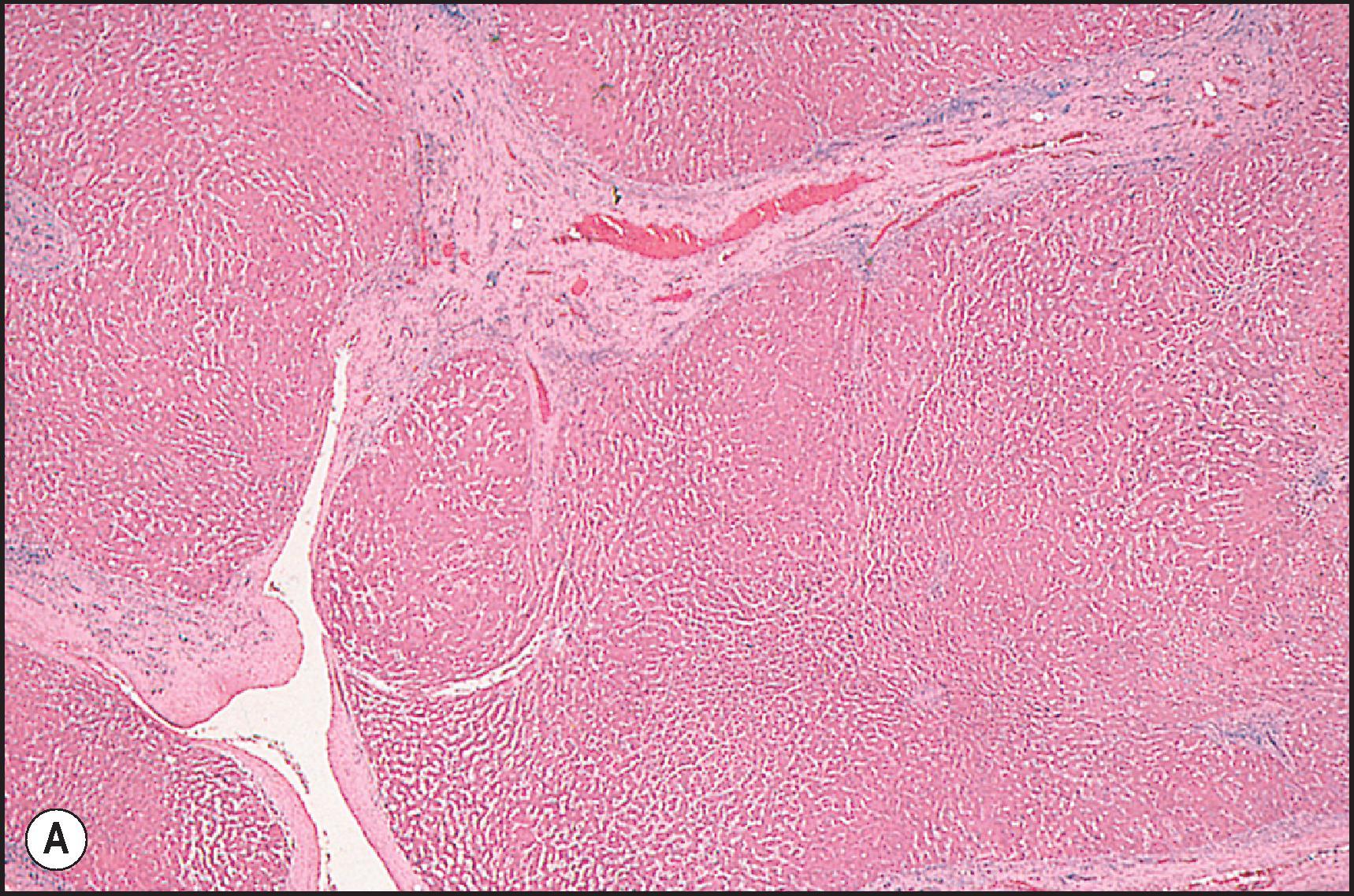
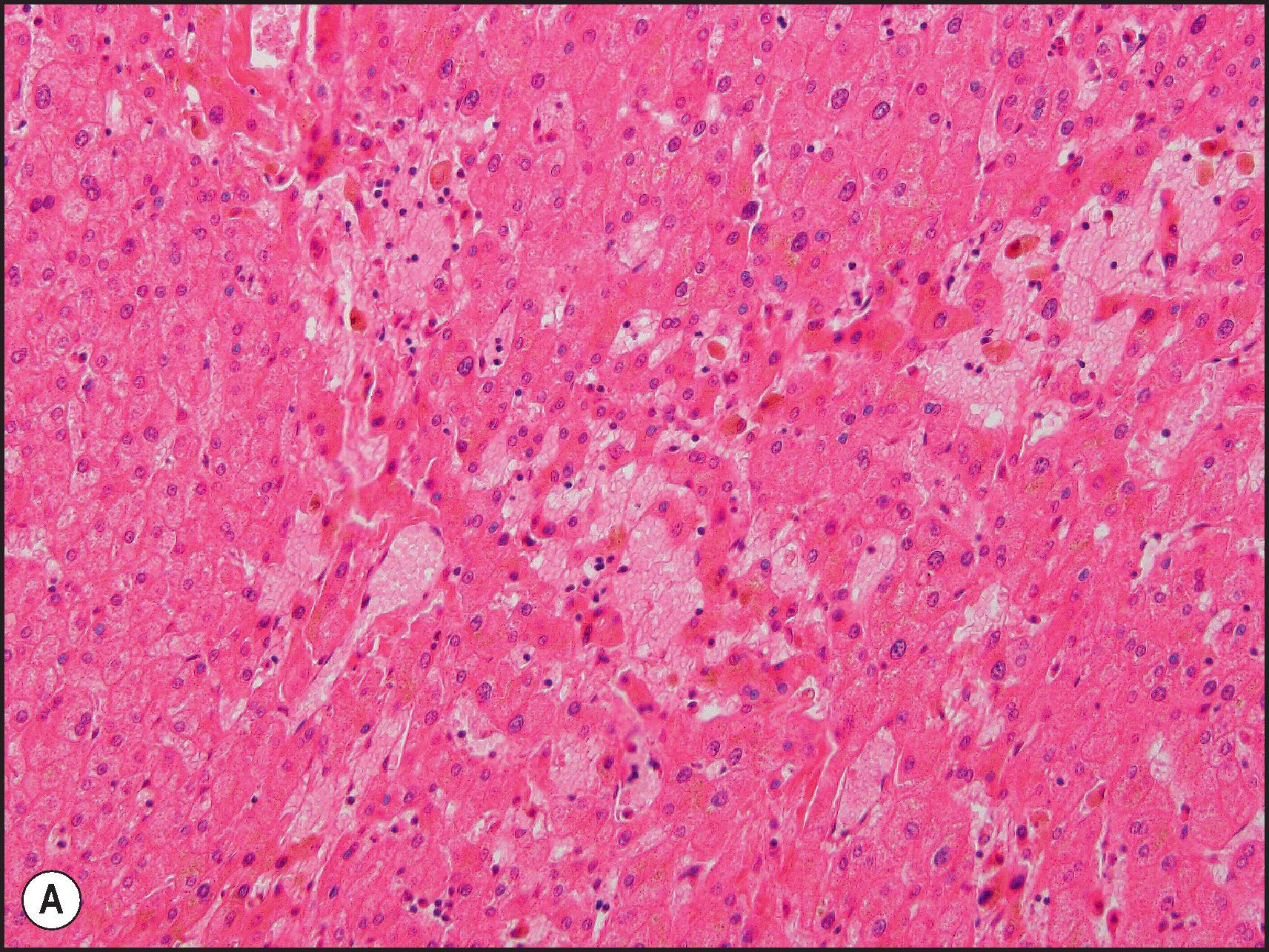
Deposition of collagen in the periportal area and space of Disse, accompanied by reduction of portal vein calibre or obliteration of the portal vein, can lead to portal hypertension ( Fig. 12.23 ). This form of noncirrhotic portal hypertension has been termed ‘hepatoportal sclerosis’. Instances of noncirrhotic portal hypertension have been attributed to chronic exposure to inorganic arsenicals, vinyl chloride and copper sulphate. Noncirrhotic portal hypertension can result from vitamin A intoxication. Several protocols employed to prepare patients for bone marrow transplantations or to treat neoplastic disease have led to noncirrhotic portal hypertension by producing hepatoportal sclerosis or NRH. NRH has also been associated with the use of azathioprine in transplantation, mercaptopurine and 6-thioguanine for Crohn disease , and oxaliplatin-containing regimens for colon cancer ( Fig. 12.24 ). , This lesion can be difficult to recognize in routinely stained sections. However, careful examination of reticulin preparations will demonstrate nodules of enlarged hepatocytes arranged in cords that are more than two cells thick and which are not organized into parallel plates. Between regenerating nodules, the hepatocytes are atrophic and compressed; there may be no fibrosis or only a small amount of sinusoidal fibrosis in the areas of hepatocyte atrophy. Sinusoidal endothelial cells may express CD34 and the normal distribution of glutamine synthetase may be altered. Sinusoidal dilation may also be seen.
Certain chemotherapeutic agents and oral contraceptives have been implicated in causing hepatic vein thrombosis ( Fig. 12.19 ). At least 100 cases have been reported in patients taking oral contraceptives and have been attributed to the thrombogenic effects of contraceptive steroids. Many patients appear to have other risk factors, so the effect of contraceptive steroids may be to increase an underlying risk. The risk of developing hepatic vein thrombosis seems to be increased at least twofold in women taking oral contraceptives.
VOD is a pattern of liver injury in which there is obliteration of small hepatic veins by loose, oedematous connective tissue that appears between the endothelium and the underlying normal collagen matrix, narrowing the lumen. This is associated with endothelial injury in the sinusoids, particularly in zone 3 with the accumulation of cell debris, trapped red cells and extracellular matrix in the sinusoids; there is frequently necrosis of the perivenular hepatocytes. Because of the key role played by sinusoidal obstruction in the pathophysiology, the term ‘sinusoidal obstruction syndrome’ has been suggested as an alternative name for this process. Depletion of glutathione and nitric oxide have been suggested as important mechansims. , Injury and occlusion of the efferent hepatic venules have long been known to be produced by PAs. , The initial lesion is sinusoidal and venular endothelial injury is accompanied and followed by progressive fibrotic decrease in venule calibre and perivenular necrosis ( Fig. 12.20 ). It can lead to portal hypertension with ascites, varices and hepatomegaly. Severe cases can develop jaundice and hepatic failure. Chronic presentations are more subtle and may result in significant fibrosis. ,
In addition to the alkaloids, causal agents include oxaliplatin, thioguanine, azathioprine and a number of other oncotherapeutic agents, as well as irradiation. , The use of multiple chemotherapeutic agents or combination chemotherapy and radiotherapy seems to be more toxic than the use of any single agent. The most important cause today is myeloablative conditioning treatment associated with bone marrow transplantation, but the incidence of SOS/VOD has fallen due to changes in bone marrow transplant protocols and the advent of nonmyeloblative regimens.
Peliosis hepatis consists of large blood-filled cavities not lined by endothelium ( Fig. 12.21 ). It has been of recognized clinical significance only since the middle of the 20th century. The lesion has been produced in experimental animals by administration of phalloidin. Phalloidin has a special proclivity for injury to membranes; this is consistent with the theory that the lesion reflects damage to sinusoidal supporting membranes.
Agents implicated as a cause of the lesion include anabolic steroids, diethylstilboestrol, contraceptive steroids, tamoxifen and azathioprine, , vitamin A and thorotrast ( Table 12.13 ). Marked sinusoidal dilation has often been observed in livers that show peliosis hepatis, even in sites remote from the cavitary lesions. Furthermore, anabolic and contraceptive steroids can lead to sinusoidal dilatation, even when no peliosis has developed. Indeed, a characteristic lesion of prominent dilatation of periportal sinusoids ( Fig. 12.22 ) has been described in patients taking contraceptive steroids. Ultrastructurally, the lesion is characterized by striking perisinusoidal fibrosis and proliferation and ‘enhanced activity’ of sinusoidal endothelial cells and perisinusoidal stellate cells. As noted previously, sinusoidal dilation may be associated with NRH, and a reticulin stain should be performed to exclude that possibility.
Subcapsular haematomas , which may rupture, have been reported in a number of patients taking oral anticoagulants (warfarin, streptokinase) , and in one patient who abused anabolic steroids.
The histopathology of drug-induced hepatic neoplasia is indistinguishable from that of non-drug-induced tumours. Both benign and malignant tumours of the liver have been associated with drugs and toxins (see Chapter 13 and Table 12.14 ).
| Hepatocellular adenoma | Hepatocellular carcinoma | Cholangiocarcinoma | Angiosarcoma |
|---|---|---|---|
| Androgens Contraceptive steroids Danazol Methyltestosterone Oestrogens, synthetic Oxymethalone Toxic oil (rapeseed) |
Aflatoxins Androgens Arsenic compounds Contraceptive steroids Ethanol Methotrexate Methyltestosterone Oestrogens, synthetic Oxymethalone Thorotrast |
Androgens Contraceptive steroids Isoniazid Methyldopa Oestrogens, synthetic Thorotrast |
Androgens Arsenic compounds Contraceptive steroids Copper sulphate Cyclophosphamide Diethylstilboestrol Phenelzine Thorotrast Vinyl chloride |
HCA, a lesion almost entirely restricted to females in the child-bearing years, was an extremely uncommon tumour prior to the era of oral contraceptives. , Convincing evidence for a causal effect is the reported regression of this tumour following withdrawal of oral contraceptives and the large case-control epidemiological study of Rooks et al. There is no relationship between focal nodular hyperplasia and contraceptive steroids.
Thorotrast has caused hepatocellular and cholangiocellular carcinomas as well as angiosarcoma, lesions that may be due to the radioactivity rather than the chemical toxicity of the agent. Epidemiological studies suggest that mycotoxins as well as synthetic chemicals encountered occupationally may also be carcinogenic in man. Several drugs have come under suspicion as possible hepatocarcinogens. The development of HCC in at least 40 long-term recipients of anabolic steroids has made these agents suspect. Contraceptive steroids have also been incriminated in the development of HCC. Tamoxifen and danazol have been implicated in hepatic carcinogenesis.
Angiosarcoma has been reported in individuals with occupational exposure to vinyl chloride. This rare tumour also developed in vintners with long exposure to inorganic arsenic, in patients with psoriasis treated with arsenicals, in individuals who had ingested arsenic-contaminated water or had other environmental exposures and in patients who had been injected with thorotrast. Other drugs incriminated rarely as causes of this tumour include androgenic-anabolic steroids, , diethylstilboestrol and phenelzine.
Ground-glass hepatocytes may develop in patients on long-term therapy with some drugs ( Fig. 12.25 ). This change was first reported by Klinge and Bannasch in patients on long-term therapy with chlorpromazine and barbiturates. Other drugs that have been implicated include azathioprine, steroids, phenytoin, resorcin and some analgesics. The change was attributed to a marked, diffuse hypertrophy of the smooth endoplasmic reticulum (SER) by Klinge and Bannasch, and this has been confirmed by ultrastructural studies. It is associated with increased activity of microsomal nicotinamide adenine dinucleotide phosphate (NADPH) cytochrome c reductase. Drug-associated ground-glass cell change may be differentiated from the ground-glass cells of chronic hepatitis B both by special stains (aldehyde fuchsin, Victoria blue and orcein) and by immunohistochemistry.
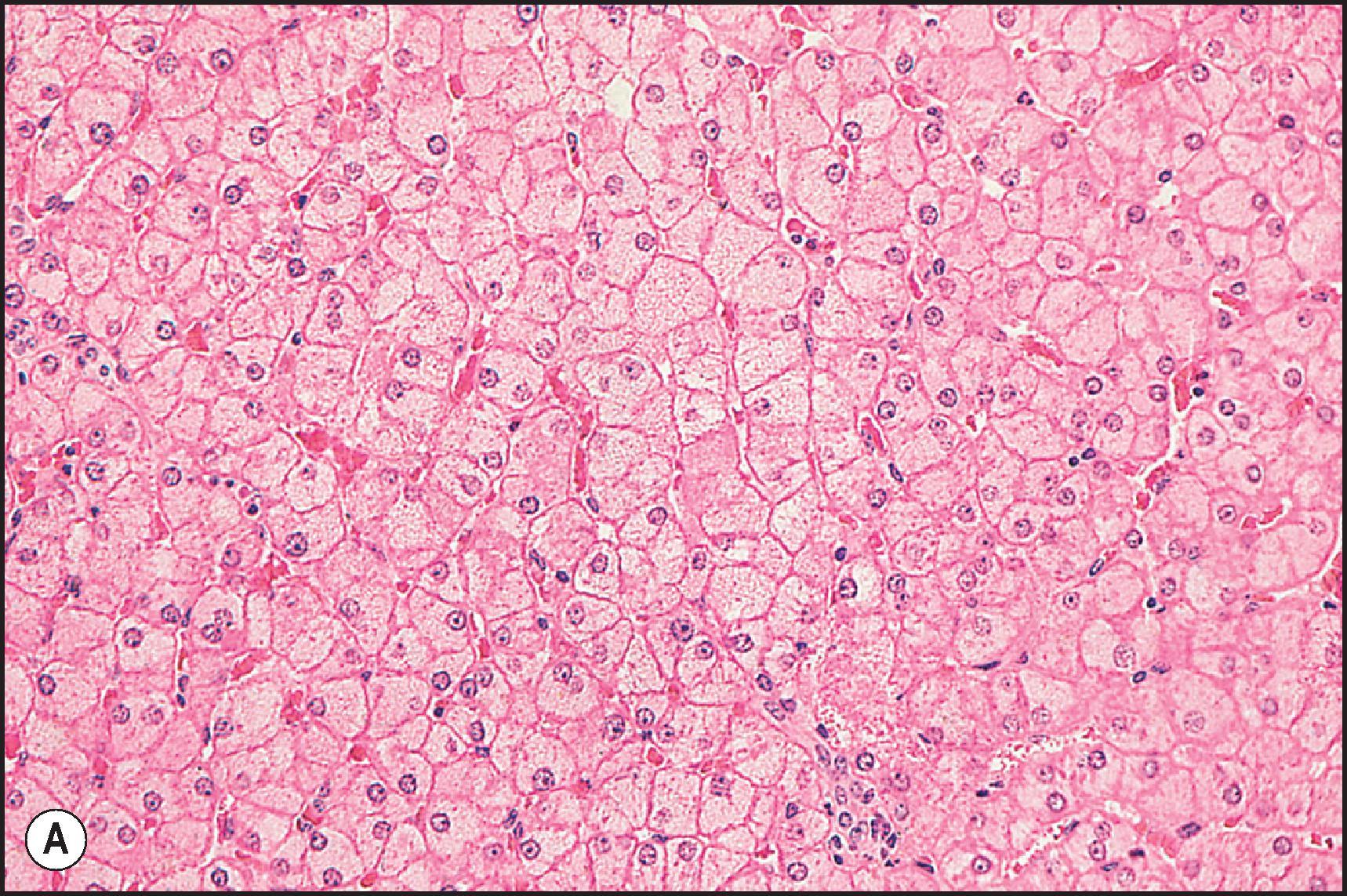
The prognostic significance of drug-induced ground-glass transformation is not known. It is accompanied by elevated levels of γ-glutamyltransferase (GGT) and is believed to account for the hepatomegaly observed in patients on anticonvulsants and other drugs. It is not associated with acute or chronic injury that may be caused by the same drug, and there are no long-term studies of patients with this drug-induced hepatic change. A dramatic form of ground-glass change ( Fig. 12.26 ) has been noted in patients undergoing alcohol aversion therapy. At first attributed to disulfiram, it now appears more clearly associated with cyanamide treatment. The change differs from that described in the foregoing paragraphs, in that the cyanamide-associated altered cells contain ground-glass ‘inclusion bodies’ that are periodic acid-Schiff (PAS)- positive and diastase resistant ( Fig. 12.26 B). Ultrastructurally, the inclusions are composed of glycogen beta granules, secondary lysosomes, lipid vesicles and residues of degenerating organelles; this lesion can apparently lead to cirrhosis. Lefkowitch et al. recently reported a series of patients with PAS-positive, diastase-sensitive inclusions that were termed ‘polyglucosan-like’. These were found in patients on multiple immunosuppressives after transplantation and may represent an adaptive change.
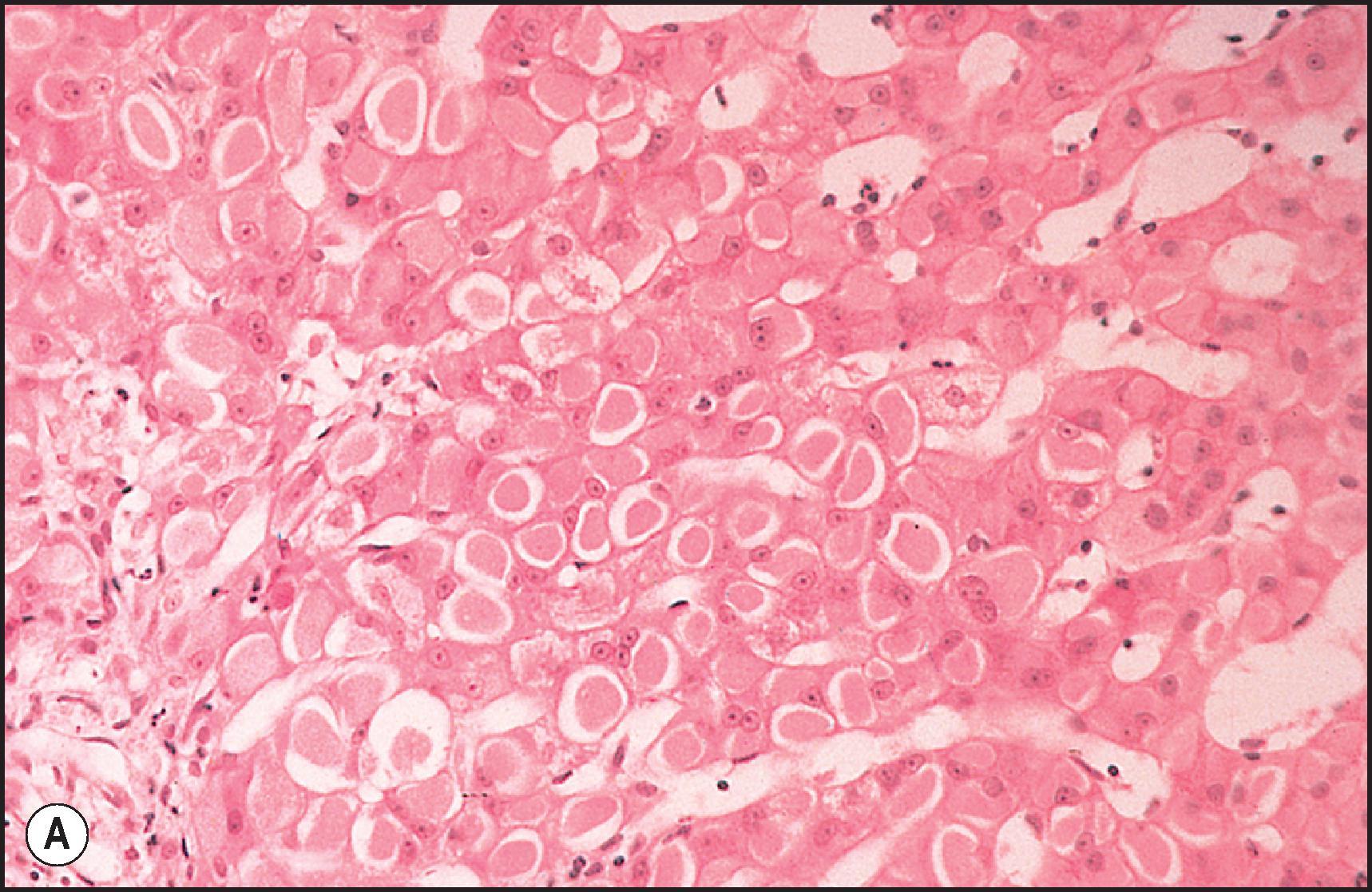
Patients who have been treated with high doses of corticosteroids may develop severe glycogenosis of the hepatocytes. This change is characterized by diffuse cellular enlargement by pale bluish-grey cytoplasm ( Fig. 12.27 ). The cells may contain small lipid droplets, but steatosis may be inconspicuous or absent. An acute rise in transaminases may prompt a biopsy, but the lesion seems to be completely reversible. Glycogenosis has also been reported in association with type 1 diabetes.
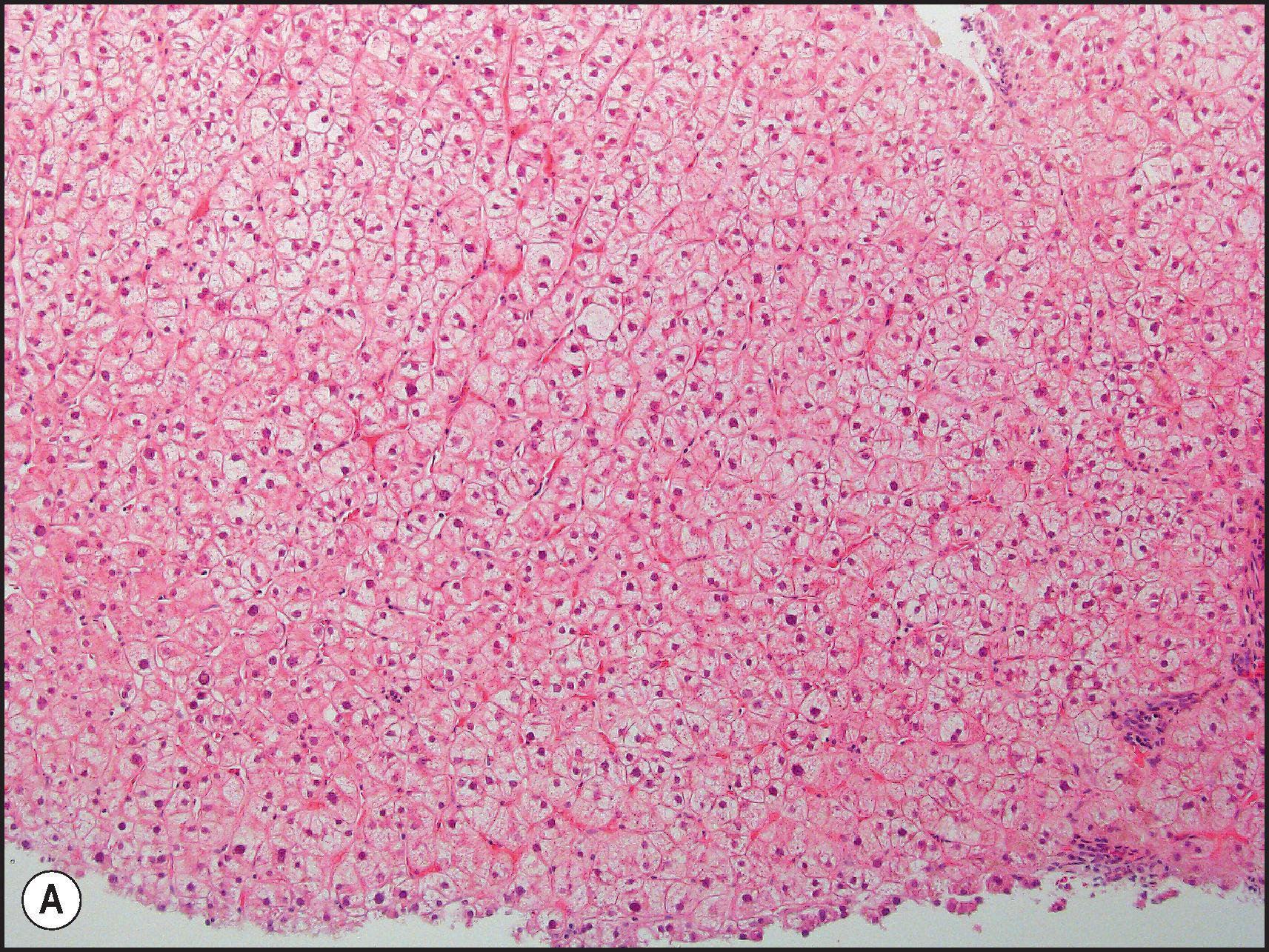
Several types of pigment deposits may follow exposure to exogenous chemicals. These include lipofuscin, haemosiderin, Thorotrast, gold, titanium, mercury, silver, polyvinylpyrrolidone (PVP) and bilirubin (discussed earlier). , ,
Lipofuscin , the least significant of these, occurs normally in the lysosomes of the perivenular hepatocytes; it is more prominent in elderly people. It also is increased in patients on various long-term medications, such as phenacetin, Cascara sagrada and chlorpromazine, anticonvulsant therapy as well as antiretroviral therapy for HIV infection.
Black pigment granules in the liver ( Fig. 12.28 ) may reflect the presence of gold as a residue of use of gold compounds for treatment of rheumatoid arthritis , ; titanium , which may be found in parenteral drug abusers ; or mercuric sulphide in chronic mercury poisoning following ingestion of a laxative preparation containing mercurous chloride. Dark brown pigment granules containing silver were reported in a patient with argyria.
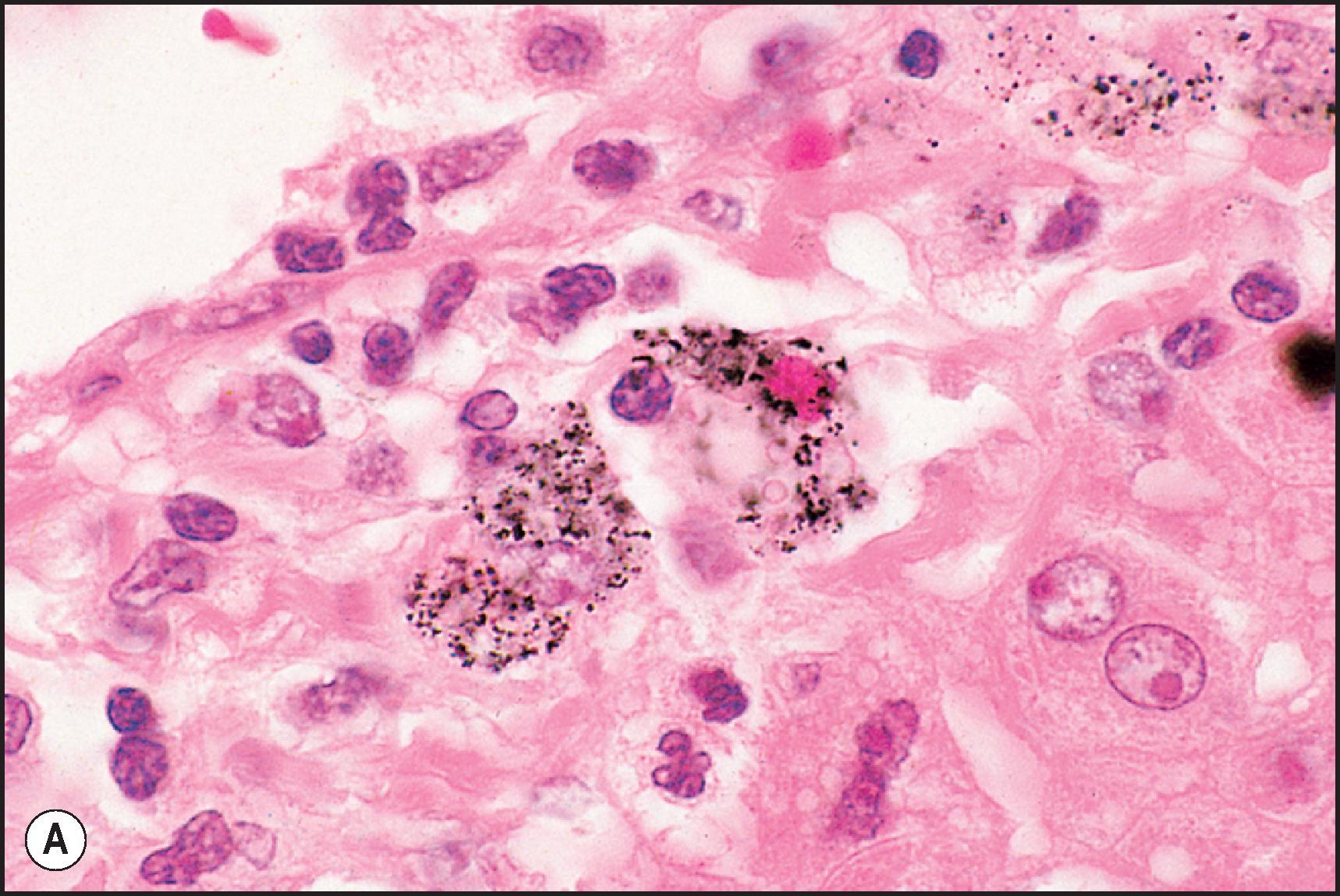
Haemosiderin deposits are seen in several forms of chemical and other causes of hepatic injury. Haemosiderin may be seen in the hepatocytes of patients with porphyria cutanea tarda resulting from drugs or toxins, e.g. hexachlorobenzene. Hepatic haemosiderosis has been reported in patients on long-term haemodialysis who had received parenteral iron. Transfusional haemosiderosis is discussed in Chapter 4 . Rare cases of iron overload have been attributed to excess ingestion of iron preparations.
Thorotrast , a formerly used radioactive contrast medium, remains in the livers of patients many years after its administration. It appears as glistening, greyish-brown granules engorging macrophages or lying free in dense fibrous tissue ( Fig. 12.29 ).
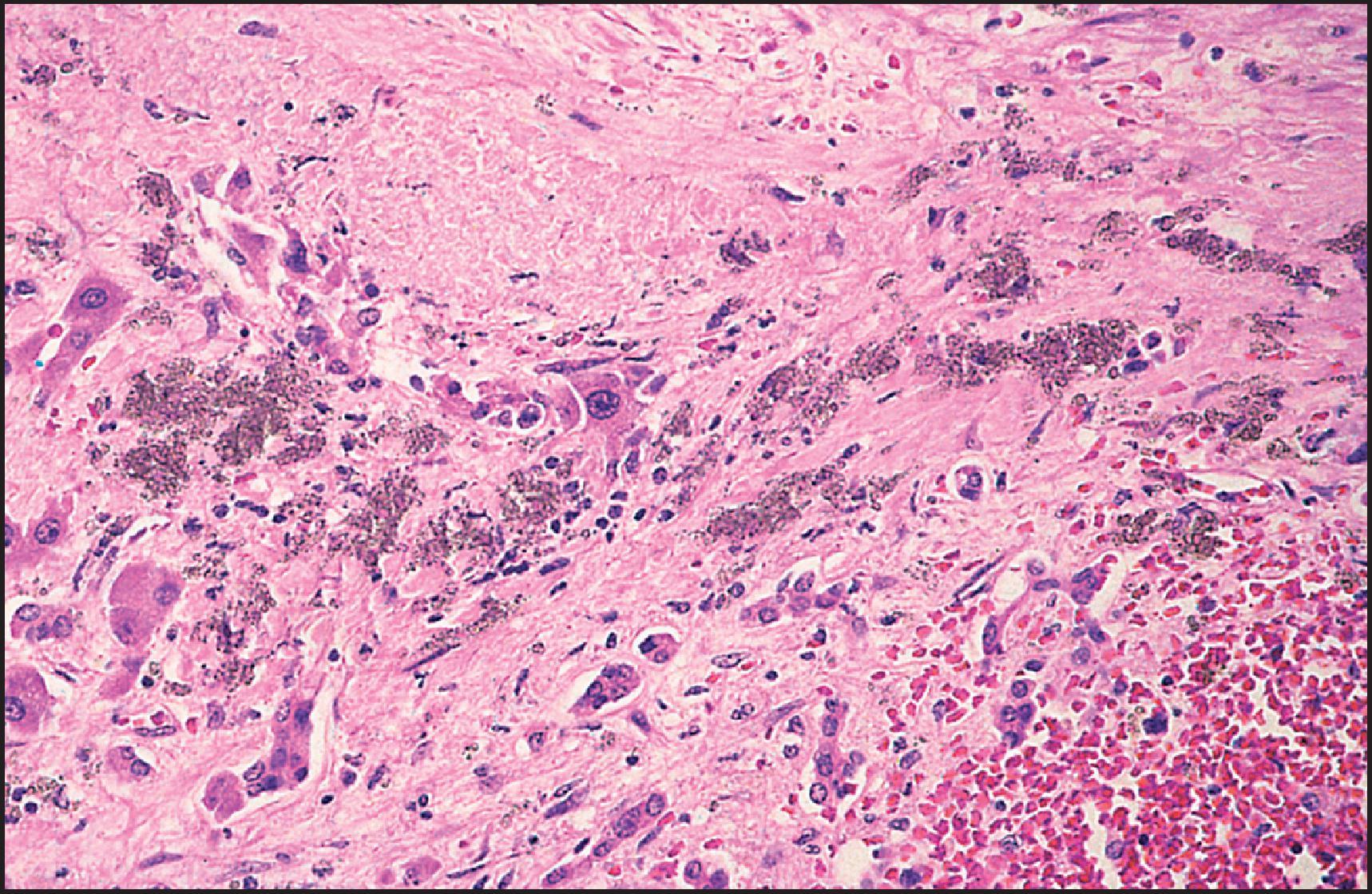
PVP is a water-soluble, hydrophilic, high-molecular-weight product of the polymerization of vinylpyrrolidone. During World War II it was used as a plasma expander and still is used as a binder in oral medications. PVP is stored in reticuloendothelial cells and long-term sequelae have not been reported. The affected cells have a granular, basophilic cytoplasm. The PVP stains positively with Lugol iodine solution (mahogany brown), Congo red, Sirius red and mucicarmine. Unlike amyloid deposits, Congo red-stained PVP does not display apple-green birefringence.
Finally, drugs may cause serum biochemical changes sufficient to generate a liver biopsy, yet not cause much in the way of diagnosable changes. There may be minimal or mild steatosis, rare apoptotic bodies, scattered pigmented macrophages or spotty necrosis. Like the adaptative changes noted previously, it is unclear whether there is potential for more serious injury if the drug is continued. At least in the case of methotrexate, a biopsy showing such mild changes would be reassurance that the drug could be continued, but it is difficult to extend that generalization to therapeutics that are less well understood.
The clinical and biochemical manifestations of hepatotoxicity reflect the histological patterns of injury ( Table 12.15 ) and the various clinical syndromes sometimes take their names from the histological lesions. In the absence of a biopsy demonstrating the actual histological injury, DILI is primarily classified according to biochemical changes. Transaminases (principally ALT) and ALP levels at the onset (or recognition) of injury are used to derive a ratio ‘R’, that is the main biochemical classifier. ALT and ALP levels are normalized by dividing the serum level by the ULN. The normalized value of ALT is divided by the normalized value of ALP to give the value of R. R values greater than 5 are termed ‘hepatocellular injury’ while values less than 2 are termed ‘cholestatic injury’. Intermediate values are termed ‘mixed injury’. The development of jaundice, with the associated high bilirubin level, adds a second dimension to this clinical characterization. For example, a patient with hepatocellular injury and jaundice would be characterized as having hepatocellular jaundice. This categorization is helpful in organizing the clinical investigation to exclude other causes of hepatic injury. Drug-induced acute hepatitis leads to biochemical changes and to clinical features similar to those of acute viral hepatitis. The histological pattern and biochemical signature of drug-induced cholestatic or mixed jaundice can mimic that of biliary tract obstruction. It should be noted that patterns of enzyme elevations cannot predict histologic changes and the degree of enzyme elevation does not always correspond to severity of injury on biopsy.
| Laboratory findings at onset | |||||||
|---|---|---|---|---|---|---|---|
| Clinical syndrome | ALT/AST (×ULN) |
ALP (×ULN) |
Bilirubin (mg/dL) |
Latency (weeks) |
Clinical findings | Histological injury pattern | Main differential diagnosis |
| Acute hepatic necrosis | >20× (often very high) | <2× | <10 initially | <2 | Sudden onset, hepatic dysfunction, nonhepatic organ dysfunction | Zonal necrosis | Ischaemic hepatitis |
| Acute hepatitis | >20× | <3× | >2.5 | 2 to 24 | Insidious onset, fatigue, anorexia, nausea, right upper quadrant tenderness, jaundice | Acute hepatitis with or without cholestasis | Acute viral hepatitis |
| Cholestatic hepatitis | <10× (R < 2) |
>3× | >2.5 | 2 to 24 | Jaundice at onset, nausea, fatigue, pruritus, prolonged course | Cholestatic hepatitis (not overlapping with acute hepatitis) | Biliary obstruction, PBC, sclerosing cholangitis |
| Mixed hepatitis | >6× R < 2, < 5 |
>3× | >2.5 | 2 to 12 | Fatigue, anorexia and nausea followed by pruritus and jaundice | Cholestatic hepatitis (with overlap into acute hepatitis) | Acute viral hepatitis, biliary obstruction, mononucleosis |
| Enzyme elevations without jaundice | >3× but <20× |
>2× but <5× |
<2.5 | 2 to 48 | Generally mild and nonspecific, enzyme elevations may be ALT/AST and/or ALP | Mild acute/chronic hepatitis, nonspecific findings | |
| Bland cholestasis | Normal to <5× | Normal to <2× | >2.5 | 4 to 24 | Prolonged jaundice without evidence of hepatocellular injury | Acute intrahepatic cholestasis | Cholestasis of pregnancy |
| VBDS | >5× | >3× | >2.5 | May present like cholestatic/mixed hepatitis, jaundice, pruritus, fatigue, prolonged ALP elevation | Ductopenia, chronic cholestasis, cholestatic hepatitis | Sclerosing cholangitis, PBC | |
| Acute fatty liver with lactic acidosis | >2× | >2× | Variable | Early symptoms of nausea, anorexia, weight loss, abdominal discomfort. Later lactic acidosis, weakness, shortness of breath, stupor, jaundice | Microvesicular steatosis | Multiorgan failure from nontoxic injury | |
| Nonalcoholic fatty liver | >1× | <2× | Normal | >12 | Nonspecific symptoms, steatosis on imaging | Steatosis, steatohepatitis | Metabolic dysfunction-associated liver disease |
| Chronic hepatitis | >1× | <2× | <2.5 | Minimal nonspecific symptoms | Chronic hepatitis | Chronic viral/autoimmune hepatitis, IBD | |
| Sinusoidal obstruction | Can be high (>10×) | Usually <2× | >2 | Abdominal pain, swelling, weight gain, signs of portal hypertension | VOD/SOS | ||
| NRH | <3× | <3× | Normal | >24 | Insidious onset with signs of portal hypertension | NRH, hepatoportal sclerosis | Other vascular causes of NRH/noncirrhotic portal hypertension |
Hepatocellular necrosis leads to the highest levels of transaminases with only modest changes in ALP. The level of ALT or AST may be increased to values that are 10 to 1000 times the ULN in paracetamol injury. However, the value for ALP increases no more than one- to threefold in response to massive necrosis. Similarly, an acute hepatitis-like pattern of injury from a drug such as INH will also present as hepatocellular injury, with values of R in the range of 10–50, similar to acute viral hepatitis. The tempo of the injury varies between these two presentations. Acute hepatic necrosis has a rapid onset with early evidence of hepatic dysfunction, while acute hepatitis is more insidious, presenting with nonspecific systemic symptoms and jaundice. These differences are represented in the histology as well, as described in the earlier sections.
The clinical syndromes of cholestatic hepatitis and mixed hepatitis present in similar ways with fatigue and nausea, followed by elevated bilirubin levels and jaundice. The main clinical difference is in the relative amount of hepatocellular injury, with mixed hepatitis having higher serum transaminase levels. The clinical differential diagnosis also depends on the degree of hepatocellular injury. At high levels of transaminases, the differential includes acute hepatitis, while at lower levels, the differential diagnosis includes chronic cholestatic conditions. Obstructive causes of jaundice are a consideration in both syndromes. Biopsies typically show a cholestatic hepatitis with varying degrees of zone 3 cholestasis and portal and lobular inflammation. There is overlap in the agents that cause these conditions, but at the more cholestatic end of the spectrum are the antibiotics, including amoxicillin-clavulanate, cephalosporins and the sulpha drugs, while at the more hepatocellular end are the anticonvulsants and nonsteroidal anti-inflammatory agents.
Bland cholestasis is characterized mainly by jaundice with little or no elevation in transaminases or ALP. Patients usually feel well, although there may be pruritus. The jaundice may be prolonged despite stopping the medication. Biopsies typically show zone 3 cholestasis without much inflammation or duct injury. This clinical syndrome is classically associated with the anabolic steroids, although high-dose oestrogen therapy and drugs more often associated with cholestatic hepatitis can also present in this fashion.
A particularly dramatic form of DILI is the acute development of fatty liver associated with lactic acidosis. This type of injury is associated with mitochondrial dysfunction, sometimes in multiple organs, including the skeletal muscles, pancreas and kidneys, along with the liver. The mechanism of injury can include uncoupling of oxidative phosphorylation, damage to mitochondrial membranes, damage to mitochondrial DNA, inhibition of mitochondrial DNA replication and impairment of mitochondrial transcription and translation. Early symptoms may be nonspecific or related to one of the other damaged organ symptoms. LAEs are elevated, although not as high as seen in acute hepatic necrosis. Hepatomegaly may develop. With severe hepatic injury there may be evidence of hepatic failure, with elevated bilirubin, prolonged prothrombin times and hepatic encephalopathy. Biopsies show microvesicular steatosis, sometimes admixed with macrovesicular steatosis. Inflammation and necrosis may be minimal. Classical causes of this syndrome include tetracycline, aspirin and some of the antiretroviral nucleoside analogues. A recent addition is the antimicrobial linezolid.
SOS and NRH both result from vascular/endothelial injury but present in very different ways. Patients with acute SOS present with signs of portal hypertension (ascites, splenomegaly) along with hepatomegaly and abdominal pain. Transaminases may be significantly elevated. There is usually no difficulty with the diagnosis when patients present with these findings following haematopoietic stem cell transplantation. However, there are other causes, including a well-described association with natural products containing PAs. NRH also results in symptoms of portal hypertension but the onset is insidious. Transaminases are normal or minimally elevated while ALP is typically elevated, mimicking a cholestatic injury.
Sometimes a drug will cause hepatic injury characterized by abnormalities of transaminases or ALP without bilirubin elevations or systemic symptoms. Patients who develop fatty liver disease or chronic hepatitis from drugs have subtle presentations that may not be recognized unless blood tests are done. Bilirubin elevations are typically not observed and systemic symptoms may be nonspecific or absent. Because both chronic hepatitis and fatty liver disease have common nondrug aetiologies, the burden of proof rests in excluding these alternatives. A biopsy may also show minimal findings: clusters of pigmented, PAS-positive macrophages, minimal lobular or portal inflammation or hepatocellular alterations such as glycogenosis or ground-glass cell change.
Many drugs capable of causing acute hepatocellular injury lead to modest elevations of transaminase values that either do not become severe or even normalize despite continuation of the agent. This phenomenon has been called hepatocyte ‘tolerance’, a type of adaptation that takes place to prevent more severe injury. Failure of tolerance may be one explanation for idiosyncratic DILI. Although the actual mechanism of such tolerance is unknown, it is thought to represent an upregulation of cytoprotective mechanisms or suppression of necrogenic factors. , Usually no histologic correlation of these mild enzyme elevations is available; liver biopsy is rarely done. Table 12.16 lists the drugs associated with ALT elevations that have demonstrated evidence of tolerance.
| Prevalence | Examples |
|---|---|
| 25–50% | Tacrine |
| 20–25% | Chlorpromazine, triacetyloleandomycin, phenytoin, amiodarone, perhexiline, papaverine, cisplatin, nicotinic acid, valproic acid, 6-MP |
| 10–20% | Isoniazid, ketoconazole, androgens, erythromycin estolate, etretinate, ximelagatran |
| 5–10% | Penicillamine, chenodeoxycholate, flucytosine, disulfiram, most NSAIDs |
| <5% | HMG-CoA reductase inhibitors (statins), salicylates, gold salts, sulphonamides, antrolene, sulfonylureas, quinidine, thiabendazole, ticarcillin, ethionamide, tricyclic antidepressants |
DILI has several possible outcomes. Patients may recover over days to weeks, develop evidence of chronic injury or may develop acute liver failure that results in transplantation or death. Acute liver failure presents in the same way as lesser forms of acute liver injury. There may be nonspecific symptoms of fatigue and nausea with jaundice and dark urine. Depressed levels of plasma coagulation factors (international normalized ratio [INR], factor VIII levels, etc.) are characteristic of severe hepatic necrosis, with the most useful clinical clues to the severity of necrosis being the prothrombin time (INR) and bilirubin levels. Liver failure is diagnosed when the patient develops symptoms of hepatic encephalopathy. Ascites and peripheral oedema may develop. Albumin levels usually remain normal in the early phase of acute necrosis. Acute liver failure can develop out of the syndromes of acute hepatic necrosis and acute hepatitis and less often from mixed or cholestatic hepatitis.
In acute liver failure due to the intrinsic toxins CC1 4 , phosphorus, iron, hepatotoxic mushrooms and acetaminophen (paracetamol), there are three distinct phases: (i) immediate with severe neurological or gastrointestinal (GI) manifestations; (ii) a period of relative well-being; and (iii) a phase of overt hepatic injury often accompanied by renal failure ( Table 12.17 ). These acute hepatotoxins produce initial symptoms within a few hours of exposure, although those associated by ingestion of toxic mushrooms may be delayed for several hours.
| Phase of injury | APAP | Phosphorus | Amanita phalloides | CCl 4 |
|---|---|---|---|---|
| I. 1–24 hours | ||||
| Onset | Immediate | Immediate | Delayed 6–20 hours | Immediate |
| A, N, V, D | + | 4+ | 4+ | + |
| Shock | − | + | ± | − |
| Neuro symptoms | − | + | ± | − |
| II. 24–72 hours | ||||
| Latent period | + | ± | + | + |
| III. >72 hours | ||||
| Jaundice | + | + | + | + |
| Hepatic failure | + | + | + | + |
| Renal failure | + | + | + | + |
| Maximum AST/ALT | 1000× | 100× * | 500× | 500× |
| Zonal necrosis | 3 | 1 | 3 | 3 |
| Steatosis | No | 4+ | + | + |
| Case-fatality rate | 5–15% | 25–50% | 20–25% | 20–25% |
* Often <10×. A , Anorexia, ALT , alanine aminotransferase; APAP , paracetamol, acetaminophen; AST , aspartate aminotransferase; CCl 4 , carbon tetrachloride; D , diarrhoea; N , nausea; V , vomiting.
If a patient does not develop acute failure then recovery is the usual outcome, although a minority of patients will have persistent laboratory and clinical abnormalities. In the Spanish DILI registry, the mean recovery time to normalize LAEs was 81 to 104 days depending on the biochemical presentation. Acute cholestatic injury leads to the most prolonged recovery—at times taking several months and even years. , Prolonged abnormalities (beyond at least 6 months) of LAEs are taken as evidence of chronic DILI. This is a relatively rare outcome. Only 6% of a Swedish cohort with DILI had abnormal LAEs persisting at a median follow-up of 48 months, and only 3.4% of 685 patients who survived acute DILI (all with jaundice initially) were hospitalized for liver disease over a 10-year study period. In the U.S. DILIN series, 17% of patients had persistence of abnormal LAEs beyond 6 months, most often seen in cholestatic cases. DILIN also studied the cohort of patients that still had persistent abnormalities 2 years after the onset of drug injury. When compared with patients whose biochemical injury resolved within 12 months, those with persistent injury were more likely to be older and have heart disease but were less likely to have drunk alcohol (any degree) and were no different with respect to diabetes or body mass index (BMI). Patients with persistent injury also tended to have biochemically cholestatic injury at presentation. Seventeen of the patients had liver biopsies a median of 387 days after onset. Nine of these showed chronic cholestatic liver injury, some with VBDS. Three showed chronic hepatitis (without apparent viral infection) and three showed steatohepatitis. The finding of steatohepatitis suggests that in at least some cases, there could be other explanations for persistently abnormal LAEs. Drugs that cause fibrosis as part of their injury pattern (e.g. methotrexate, tamoxifen, nitrofurantoin and amiodarone) may show persistent biochemical abnormalities for months. Twelve patients in the DILIN study had more than one biopsy, eight of which demonstrated fibrosis progression between biopsies, suggesting a true chronic (and potentially progressive) injury. As with other chronic liver diseases, fibrosis may regress if the causative agent is removed. Tamoxifen-induced steatohepatitis with fibrosis can slowly resolve once the tamoxifen is stopped.
There have been several studies looking for histological indicators of good or bad prognosis. The presence of eosinophils in the inflammatory infiltrate has been associated with a good short-term prognosis. Peripheral and hepatic eosinophilia is associated with lower total bilirubin levels and improved outcomes among patients with fulminant hepatic failure due to DILI. Pachkoria et al. have postulated that the absence of eosinophilia is associated with an accelerated T-helper type 1 cytotoxic response leading to more severe liver injury. The degree of necrosis has been associated with poor outcome in several studies. , , A study of liver biopsies in DILIN confirmed these observations and extended the list of histological associations with outcome. In addition to eosinophils, the presence of granulomas (even just microgranulomas) favoured a better outcome. Patients who developed acute liver failure, were transplanted or died more often had high-stage fibrosis, microvesicular steatosis (even when present with macrovesicular steatosis) and ductular reaction. Ductular reaction has been noted to be a poor prognostic marker in other cases of acute liver failure and it is postulated that this is a surrogate marker for replicative failure in the hepatocyte compartment.
Occupational and environmental hepatotoxicity has been recognized for more than a century, with workers exposed to a wide variety of solvents and chemicals used in the production of munitions and other materials during the First and Second World Wars, emphasizing the risks involved. , , , While industrial exposure to hepatotoxic chemicals is less of an occupational hazard today, many of these agents remain important experimental tools in the study of mechanisms and the prevention of hepatotoxicity, and reports of toxicity from chemical agents have not disappeared. Workers exposed to petrochemicals during the gulf oil spill showed higher levels of transaminases and ALP than controls did. Cases of acute hepatitis related to exposure to chlorinated organic solvents continue to be reported. Recent studies have focused on tools to screen potentially exposed workers for hepatoxicity. , Toxin-associated steatohepatitis has been related to exposure from a variety of chemicals, including vinyl chloride, CC1 4 , chloroform, dimethylformamide, methanol, toluene, trinitrotoluene (TNT) and tetrachlorethane.
Among thousands of chemical compounds in commercial and industrial use, several hundred are listed as causing liver injury by the National Institute for Occupational Safety and Health. The National Library of Medicine also maintains a database on chemical toxins in its Toxicology and Environmental Health Information Program (TEHIP) that serves as another important resource of information. Toxic exposure to chemical agents occurs most often from inhalation or absorption from the skin, and less often after oral ingestion via the GI tract or via parenteral means. As a majority of chemical toxins are lipid soluble, once absorbed they can easily cross biological membranes to reach their target organ(s), including the liver. , The clinical syndrome associated with hepatotoxic chemical exposure is usually acute cytotoxic injury (as seen with CC1 4 and phosphorus) that typically consists of three distinct phases similar to that observed after an acetaminophen overdose or ingestion of toxic mushrooms ( Table 12.17 ) or less commonly as acute cholestatic injury (as seen with methylenedianaline [MDA], the agent responsible for Epping jaundice). , Many chemicals (e.g. vinyl chloride) are also carcinogenic, and the development of hepatic malignancies has been part of the clinicopathologic spectrum of chemical injury. , The clinicopathological spectrum of several chemical hepatotoxins is given in Table 12.18 .
| I. Acute injury | ||
|---|---|---|
| A. Necrosis | CCl 4 and other haloalkanesHaloaromatics, nitroaromatics, nitroaliphaticsHydrochlorofluorocarbonsPhosphorus, iron, copper salts, inorganic arsenicCocaine, ecstasy, phencyclidine | |
| B. Microvesicular steatosis | DimethylformamideHypoglycinHydrazineToluene, xyleneChlordeconeCocaine, boric acid, thallium | |
| C. Cholestasis | Methylene dianilineParaquatAniline-rapeseed oilDinitrophenolAlpha-naphthylisocyanate (ANIT) | |
| II. Subacute injury | ||
| A. NecrosisB. VOD | Trinitrotoluene (TNT)Pyrrolizidine alkaloids, arsenic, thorium dioxide | |
| C. Toxic cirrhosis | TetrachloroethaneHexachlorobenzene, PCBs | |
| D. Peliosis | Dioxin | |
| III. Chronic injury | ||
| A. | Cirrhosis | Chloroaliphatics, TNT, arsenic, pyrrolizidine alkaloids |
| B. | Hepatoportal sclerosis | Arsenic, vinyl chloride |
| IV. Neoplasia | ||
| A. Hepatocellular carcinomaB. Angiosarcoma | Arsenic, aflatoxins, thorium dioxideVinyl chloride, thorium dioxide, arsenic | |
| C. Haemangioendothelioma | Arsenic | |
CCL 4 is a classic example of a zone 3 hepatotoxin ( Fig. 12.30 ) causing necrosis leading to hepatic failure. This chemical is metabolized to a toxic trichloromethyl radical catalysed by CYP 2E1. Alcohol potentiates the injury via induction of this cytochrome. Most cases have been the result of industrial or domestic accidents, such as inhalation of CCl 4 -containing dry cleaning fluids and use as a household reagent, or after ingestion by alcoholics who have been known to mistake it for a potable beverage. At the cellular level, direct damage to cellular membranes results in leakage of intracellular enzymes and electrolytes leading to calcium shifts and lipid peroxidation. Hepatic steatosis develops as a result of triglyceride accumulation from haloalkylation-dependent inhibition of lipoprotein micelle transport out of the hepatocyte and increases with the continuous supply of free fatty acids from the blood. Activation of endonucleases causing chromosomal damage and mutations may result in carcinogenesis. The relative toxicity of CCl 4 compared to other haloalkanes and haloalkenes correlates inversely with the level of bond dissociation energy, the number of halogen atoms and the chain length. Although complete recovery from CCl 4 -induced liver damage is typical in moderate exposures, renal failure from acute tubular necrosis and GI haemorrhage leads to a case-fatality rate of 10–25%.
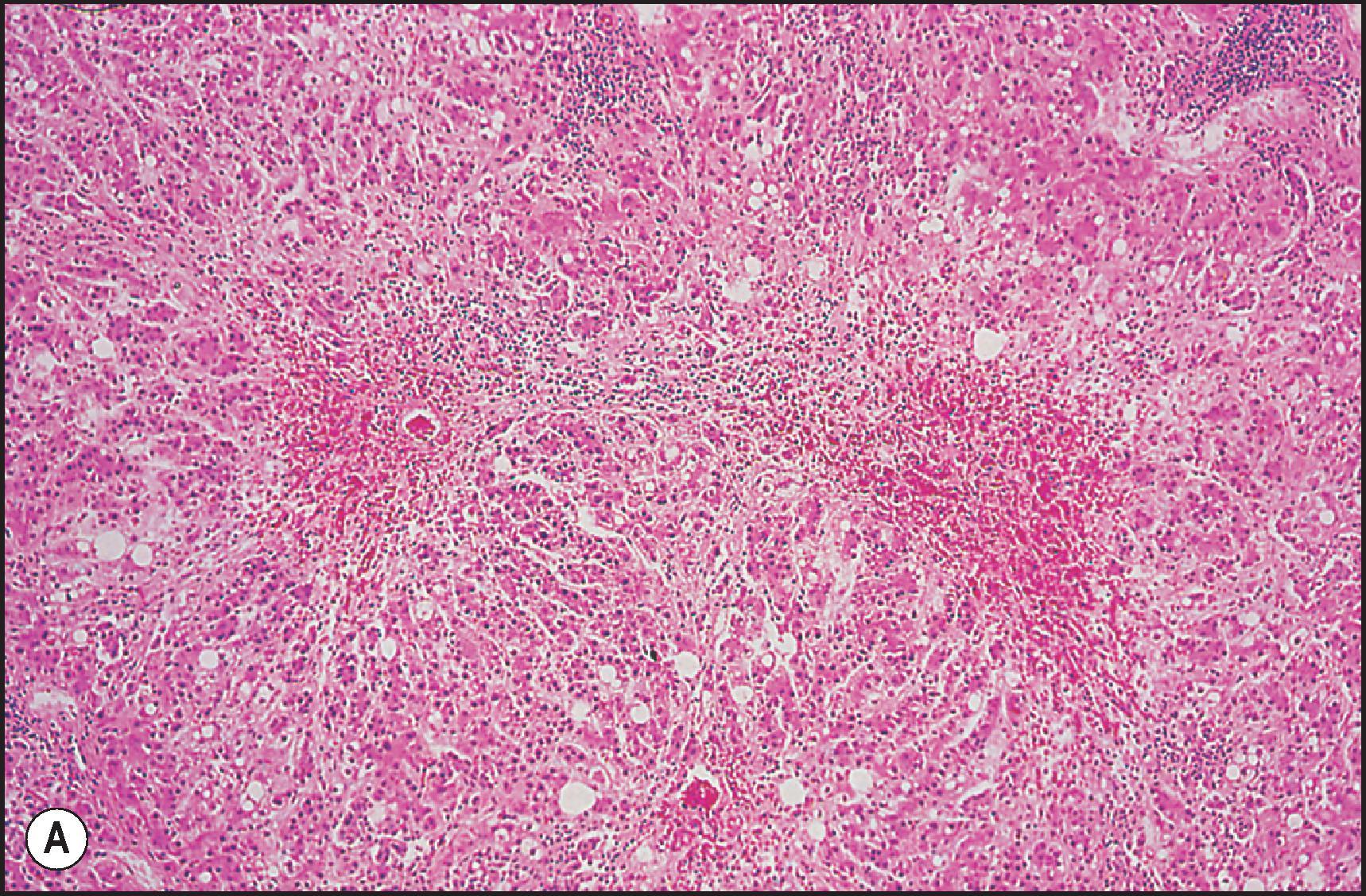
Chloroform is an important experimental hepatotoxin; its use as an anaesthetic having been abandoned in the 1950s. 1,1,2-Trichloroethane (methylchloroform) and tetrachloroethane are less potent haloalkane hepatotoxins that found use as solvents for components of varnish used to cover fabrics of airplane covers during the First and Second World Wars. Inhalational or percutaneous exposure led to subacute hepatic necrosis, with susceptibility increased in females and alcoholics. There are a number of reports of injury with 1,1,1-trichloroethane , including chronic hepatitis.
Hydrochlorofluorocarbons (HCFCs) caused liver injury in industrial workers exposed to dichlorotrifluoroethane (HCFC 123) and 1-chloro-tetrafluoroethane (HCFC 124), both of which are metabolized to reactive trifluoroacetyl halide intermediates similar to those implicated in halothane hepatoxicity. Zone 3 necrosis was present on biopsy, and autoantibodies against cytochrome P450 2E1 or P58 were detected in the serum. As with halothane, potentiation of HCFC liver toxicity is seen with ethanol-induced CYP 2E1 activity in animal models.
Vinyl chloride monomer ( VCM , monochloroethylene) exposure previously occurred in polymerization plants where vinyl chloride was heated to form polyvinyl chloride (PVC) in the manufacturing of plastics, leading to inhalation of the toxic gas in the process. VCM is ubiquitous in the environment and has been estimated by the Environmental Protection Agency (EPA) to exist in at least 10% of toxic waste sites. Although PVCs appear to be nontoxic per se , liver damage from VCM has been described in the form of several types of chronic liver injury from long-term exposure, including nodular subcapsular fibrosis, sinusoidal dilatation, peliosis hepatis and periportal fibrosis associated with portal hypertension. , Milder forms of injury have also been described, which were reversible within 6–24 months of avoiding exposure with instances of relapse after re-exposure. In workers with hepatitis B or C infection, a higher prevalence of elevated ALT values was seen in those with a high exposure to VCM compared to those with a lower exposure, a finding not observed among those without a viral hepatitis infection. NAFLD, as well as hepatomegaly and periportal fibrosis, has been recently described in a third of nonobese chemical workers with high exposure levels, some of whom continued to have NASH up to 6 years later.
VCM is carcinogenic in animals and humans, with the first reports of angiosarcoma among workers in rubber plants appearing in 1974. A history of vinyl chloride exposure was found to be present in 15% to 25% of all cases of hepatic angiosarcoma reported in the late 1970s. HCC develops after a latency of decades, with the risk related to the time and extent of contact. , Efforts to reduce occupational exposure to VCM have resulted in a marked decrease in the incidence of angiosarcoma.
Trichloroethylene (TCE) leads to a similar but often less severe clinical syndrome as seen with CCl 4 , with exposure the result of accidental industrial exposure or from sniffing the chemical. Once used as an anaesthetic, it now has a role as a solvent, spot remover and degreaser. Vinylidene chloride (VDC) is another intermediate used in the production of plastics that targets the liver.
In general, these agents, including aliphatic hydrocarbons, cycloparaffins, esters, ethers and ketones, produce little or no hepatic injury in animals and humans. Most aromatic hydrocarbons also appear devoid of significant hepatotoxic potential. Children exposed to benzene during a flaring incident at a refinery developed elevated LAEs. Toluene led to steatosis and necrosis in a ‘glue sniffer’. It caused elevations in γ glutamyl-transpeptidase after industrial exposure. Xylene can cause mild steatosis and styrene (vinyl benzene) has led to elevated transaminases after prolonged exposure.
TNT (nitroglycerin) has been known to be hepatotoxic since World War I when munitions workers developed severe acute and subacute necrosis, with a case-fatality rate of more than 25%. The incidence of hepatotoxicity during World War II was lower, with approximately 1 in 500 workers affected, with the estimated incidence of associated toxicities, methemoglobinemia and aplastic anaemia being as much as 50 times higher than liver injury. The syndrome of subacute hepatic necrosis was preceded by a latent period of 2–4 months of regular exposure to TNT. Percutaneous absorption through the skin was the major source of exposure, with lesser contributions from inhaling toxic fumes and ingesting contaminated food. In some patients, rapidly progressive liver failure and death within days to months were observed, with massive hepatic necrosis at autopsy.
Nitrobenzene and dinitrobenzene are also potent hepatotoxins described during World War I. Ingestion of dinitrophenol has been associated with necrosis and intrahepatic cholestasis, possibly owing to its uncoupling of oxidative phosphorylation. , Nitrobenzene and nitrotoluene exposures have also been associated with steatosis and steatohepatitis.
Nitromethane, nitroethane and nitropropane cause variable degrees of hepatic injury. 2-Nitropropane (2-NP) caused fatal massive hepatic necrosis after occupational exposure as a solvent, fuel additive, varnish remover and rocket propellent. Toxic hepatitis associated with the chronic inhalation of propane and butane has been reported. Both 1-NP and 2-NP are mutagenic potential human carcinogens.
Polychlorinated biphenyls (PCBs) are mixtures of trichloro-, tetrachloro-, pentachloro- and hexachloro- derivatives of biphenyls, naphthalenes and triphenyls used in the manufacture of electrical transformers, condensers and industrial fluids. Acute and chronic hepatotoxicity was seen during World War II and resembled that due to TNT. , Inhalation of toxic fumes released by the melting of PCBs and chloronaphthalene mixtures during soldering of electrical materials was the most common means of exposure. Liver injury appeared to correlate with the number of chlorine molecules. Liver damage appeared as early as 7 weeks after initial exposure. Skin lesions, known as chloracne, usually preceded the hepatic injury. Death occurred within 2 weeks in fulminant cases with massive necrosis, or after 1–3 months in subacute injury; cirrhosis developed in some who survived the acute injury. A study of the liver enzyme data from the 2003–2004 National Health and Nutrition Examination Survey (NHANES) demonstrated an association between several different PCBs and elevated LAEs. Polybrominated biphenyls (PBBs) appear to more toxic and more potent enzyme inducers compared to PCBs. Consumption of milk and meat from livestock given feed mistakenly contaminated by a PBB led to hepatomegaly and minor abnormalities of liver function tests in exposed persons.
Dimethylformamide is a solvent widely used in the synthetic resin and leather industries that causes dose-related massive necrosis in animals and is capable of producing focal hepatic necrosis and microvesicular steatosis in humans, the severity of which correlates with the duration of occupational exposure. Most individuals with prolonged exposure (greater than 1 year) had symptomatic disease that slowly resolved when they were removed from the workplace. Re-exposure in one individual produced recurrent injury. In a series of workers from Italy, 23% had liver function test abnormalities and 50% reported disulfiram-like symptoms. Workers in a Chinese organic chemical factory who were exposed to DMF had levels of DMF metabolites in their urine that correlated with elevations in LAEs.
Hydrazine and its derivatives are experimental hepatotoxins and carcinogens. In humans, steatosis and focal necrosis have been reported. Bromoalkanes and iodoalkanes have been used in insecticides and aircraft fuels, with rare reports of hepatic injury. Ethylene dibromide (dibromoethane) produces zone 3 necrosis in experimental animals with at least one instance of necrosis after an attempted suicidal ingestion. Occupational exposure has been linked to fatal toxicity, which may be potentiated by the concomitant use of disulfiram.
Accidental iron poisoning has been described for more than half a century, with upwards of 5000 cases reported to occur in the United States annually. Most cases occur in young children who mistake iron supplements for sweets and the severity of injury depends on the dose ingested. Indeed, severe injury was seen only with serum iron concentrations above 700 µg/dL measured within the first 12 hours. Ingestion of <20 mg/kg of elemental iron is unlikely to produce serious toxicity, whereas doses >200 mg/kg can be fatal. Iron, per se , is not hepatotoxic, but ferric and ferrous ions acting through free radicals and lipid peroxidation can cause membrane disruption and necrosis. Clinically evident liver injury is uncommon in most instances. Periportal necrosis may be seen in the most severe cases ( Fig. 12.3C, D ). The oral iron chelator deferiprone was implicated in causing worsening hepatic fibrosis in a long-term study of patients with thalassemia ; however, these findings were not substantiated by subsequent histopathological analyses, and injury from this agent appears unlikely.
Poisoning by white phosphorus is rare since its use in fireworks and matches was outlawed more than 60 years ago, , although case reports continue to appear. , Symptoms of severe GI and neurotoxicity develop shortly after ingestion, with death occurring within 24 hours. Phosphorescence of the vomitus and stools and a typical garlic-like odour on the breath are characteristic when present. Histologically, the liver may show only steatosis, initially in the periportal and then diffuse ( Fig. 12.3B ). In severe cases there may be massive necrosis, which has sometime been reported to be periportal in distribution.
Acute poisoning by copper is usually associated with abdominal pain and nausea but can cause hepatic toxicity in larger doses. Toxicity is related to free radical production that overwhelms intracellular protective mechanisms. Ingestion of toxic amounts (1–10 mg) is usually seen with suicidal intent, especially on the Indian subcontinent. Acute copper toxicity may result from ingestion (either suicidal or accidental) of copper sulphate, from its use (in the past) as an emetic or for the treatment of burns and from the release of copper ions (from equipment such as tubing made of copper) in an acid pH, for example, malfunctioning haemodialysis equipment, or from high levels of copper in drinking water. Histologically there is perivenular necrosis and cholestasis in deeply jaundiced patients, but only focal necrosis or no changes in mildly jaundiced patients; noncirrhotic portal hypertension has been described. Serum copper levels are markedly elevated in acute copper intoxication, as is serum caerulo-plasmin; the concentrations of copper in the liver can be very high. Mortality in a recent series of copper sulphate poisoning was 23%.
Chronic copper toxicity can result from occupational or domestic exposure. An example of the former is the chronic exposure of vineyard workers to fungicide sprays containing copper sulphate. The resultant hepatic injury includes noncaseating granulomas, parenchymal and periportal fibrosis, cirrhosis and, rarely, angiosarcoma. Copper poisoning remains a hazard in rural parts of the world. Of interest are reports of several children who developed an illness, clinically and histopathologically resembling Indian childhood cirrhosis, from the chronic ingestion of well water contaminated with high levels of copper.
The colloidal preparation of thorium dioxide was used as an intravenous contrast medium for radiographic procedures in the first half of the 20th century, with estimates of >50,000 patients being exposed before it was abandoned in 1955. Rare cases continue to appear, long after exposure. A large number of reports have appeared describing tumours of the liver as well as lympho- and myeloproliferative syndromes and non-neoplastic lesions of the liver. , The lesion that has drawn the most attention is angiosarcoma, , but cholangiocarcinoma and HCCs have been attributed to Thorotrast with almost equal frequency. , Hepatic leiomyosarcoma has also been reported. Nonmalignant hepatic lesions attributed to Thorotrast include periportal fibrosis, veno-occlusive lesions and cirrhosis. The fibrosis is particularly marked in the subcapsular region. An additional lesion of note is peliosis hepatis, which may be fatal. Histologically, thorium dioxide is found in Kupffer cells and macrophages as dark brown refractile granules approximately 10 µm in diameter ( Fig. 12.29 ), often in clusters, which can be confirmed by spectrographic analysis.
Cadmium is an experimental hepatotoxin that produces hepatic necrosis and cirrhosis and that can enhance the hepatotoxicity of endotoxin in laboratory animals. Recent epidemiological studies in the United States and Korea have linked cadmium exposure to elevated LAEs and an increased incidence of steatosis and steatohepatitis in men. , A study of liver biopsies in patients with chronic cadmium toxicity demonstrated increased portal and bridging fibrosis as compared to controls.
Beryllium has led to midzonal (zone 2) necrosis, the result of phagocytosis of insoluble beryllium phosphate (formed by the reaction of beryllium with phosphate in the blood) by Kupffer cells. The Kupffer cells undergo necrosis, releasing beryllium and possibly cytokines. Chronic industrial exposure, usually by inhalation of high concentrations of oxide or phosphorus mixtures, is associated with the formation of hepatic (and pulmonary) granulomas.
Lead hepatotoxicity may be seen as part of the larger symptom complex of abdominal pain, constipation and encephalopathy that occurs with chronic ingestion or environmental exposure. Histologically, lead poisoning has been reported to cause canalicular cholestasis and mild portal inflammation.
Hepatotoxins still encountered in the home include some of the well-known industrial toxins, e.g. CC1 4 , and other chemicals such as phosphorus, copper salts and camphor. Camphor is a home remedy that is highly toxic to the GI and nervous systems and its use has been reported to lead to abnormal liver function tests and hepatic steatosis.
Mycotoxins and other botanical agents such as poisonous mushrooms are still an important cause of acute hepatic injury in some parts of the world. , , Food contaminated with aflatoxins has been implicated in acute hepatic disease and, on epidemiological grounds, in the aetiology of HCC. , Consumption of plants containing PAs has long been known to cause acute and chronic hepatic disease and continues to be responsible for epidemics of veno-occlusive injury. , Herbal remedies and agents used in illicit substance abuse are discussed here, as are foods contaminated with toxic chemicals and various pesticides and herbicides.
Microcystins , produced by cyanobacteria (blue-green algae) growing in water used for haemodialysis in a haemodialysis centre in Brazil, led to liver failure and death of 26 patients. Liver tissue from 16 of the patients revealed disruption of liver plates, extensive necrosis, apoptosis, severe cholestasis, cytoplasmic vacuolization and a mixed leucocyte infiltrate. An acute case of microcystin toxicosis from swallowing contaminated lake water has been reported.
While exposure to insecticides, herbicides and other pesticides in the environment is common, acute liver injury from these compounds, many of which are chlorinated hydrocarbons, is rare. A study of data from the 2003–2004 NHANES found associations between elevations in transaminases and exposure to a limited group of chemicals, including dioxins, non-dioxin-like PCBs and mercury. Ingestion of large amounts of dichlorodiphenyltrichloroethane (DDT) or chlordane has occasionally been reported to cause a clinical syndrome of hepatic necrosis similar to that seen with CCl 4 . The P-450 cytochrome enzyme inducing effects of some of these agents can enhance the toxicity of other agents.
DDT and other organochlorines (aldrin, amitrole, chlordane, dieldrin, lindane, mirex) accumulate in fat deposits and, to a lesser extent, in the liver after prolonged exposure. Despite the concern that DDT might lead to occupational hepatotoxicity, there is almost no evidence of liver damage from chronic contact. An outbreak of hepatotoxicity in the Hirmi Valley in Ethiopia was linked to a combination of DDT and PAs, although the histologic injury described is more typical of the latter agent. Similarly, there is scant direct evidence that DDT is carcinogenic in humans. Much attention has been focused on Agent Orange (2,4-dichlorophenoxyacetic acid), the defoliant widely used in Vietnam. Acute hepatitis has been reported after chronic exposure from licking a contaminated golf ball. In animals, dioxin (tetrachlorodibenzo- p -dioxin) has produced progressive sinusoidal dilatation and peliosis hepatis.
Dichloridedimethyldipyridlium (paraquat) has been implicated in several instances of poisoning, many as the result of attempted suicides and occasionally due to homicides. , Ingestion of 2–25 g of the liquid concentrate has produced severe toxicity, and dermal exposure has also been sufficient to cause liver injury. , Many patients have severe vomiting, and profuse diarrhoea leading to hypokalaemia, and often show evidence of oral, pharyngeal and oesophageal caustic lesions (stomatitis) after ingestion. Death results from a combination of renal, respiratory, cardiac and hepatic failure with mortality rates as high as 70%, often within the first 48 hours. Histopathological changes include zone 3 necrosis and rather unique injury to small and medium-sized interlobular bile ducts that occurs in a biphasic pattern ( Fig. 12.13 ). It is thought that hepatocellular injury becomes cholangiocellular in nature with jaundice after the initial 48 hours, with bile duct injury being a direct corrosive effect of paraquat.
Chlordecone (kepone) has been shown to impair biliary excretion and lipid transport and storage, but neurological toxicity appears to dominate the clinical picture. Occupational exposure has led to hepatic steatosis and abnormal liver function tests, and chronic exposure is hepatocarcinogenic in rodents. Steatosis is seen in rodents exposed to chlordane . Hexachlorobenzene was associated with an epidemic of porphyria cutanea tarda and liver injury from contaminated grain.
Inorganic arsenic has long been used as a homicidal or suicidal agent, and toxic exposure also followed ingestion of Fowler solution (arsenic trioxide) used in the past as a treatment for psoriasis and asthma. Other sources of exposure come from contaminated ground and well water and homemade alcohol. Necrosis of capillary walls causes congestion and haemorrhagic necrosis of the GI tract leading to severe nausea, vomiting and diarrhoea and is also thought to produce a lesion in the liver resembling VOD. Severe steatosis and varying degrees of necrosis have been observed. More than 90% of a group of 248 patients who consumed contaminated drinking water for up to 15 years were shown to develop noncirrhotic portal hypertension. A study of children in India with noncirrhotic portal hypertension also found an association with exposure to arsenic.
Occupational exposure to arsenic is still observed among vineyard workers, farmers and gold miners, although its use as an insecticide has been curtailed since the 1940s. Lumber treated with chromated copper arsenate as a preservative may be an additional source of exposure. Chronic hepatic injury, including cirrhosis and noncirrhotic portal hypertension, may be precursor lesions to hepatic malignancies, including angiosarcoma, hemangioendothelioma and HCC. ,
There are about 100 poisonous varieties of mushrooms among the more than 5000 species, with reports of serious and fatal mushroom poisonings found worldwide. In the United States, between 1999 and 2016 about 7400 mushroom poisonings were reported per year, with two-thirds of fatal poisonings due to A. phalloides (death cap) or Amanita verna (destroying angel). A fatal dose can involve the ingestion of a single 50 g mushroom, representing a dose of about 21 mg of amatoxin. Alpha amatoxin is thermostable, can resist drying for years and is not inactivated by cooking. Rapidly absorbed via the GI tract, it inhibits production of mRNA and protein synthesis in hepatocytes, leading to necrosis. A second toxin, phalloidin, is responsible for the GI distress that precedes hepatic and central nervous system (CNS) injury. , Phalloidin disrupts cell membranes by interfering with actin polymerization and leads to severe gastroenteritis. In contrast to the injury seen with CCl 4 and phosphorus, the latent period is often longer (6–20 hours). Intense abdominal pain, vomiting and diarrhoea develop, with hepatocellular jaundice and renal failure occurring over the next 24–48 hours, followed by convulsions and coma by 72 hours. , In a case series of eight patients reported by Rengstorff et al., mean AST was 5488 IU/L, mean ALT was 7618 and bilirubin was 10.5 mg/dL, peaking at 4 to 5 days after ingestion. Mortality is generally high, especially when the ALT exceeds 1000 IU/L, and emergency liver transplantation is often required. , The characteristic hepatic lesion is prominent steatosis and zone 3 necrosis ( Fig. 12.31 ).
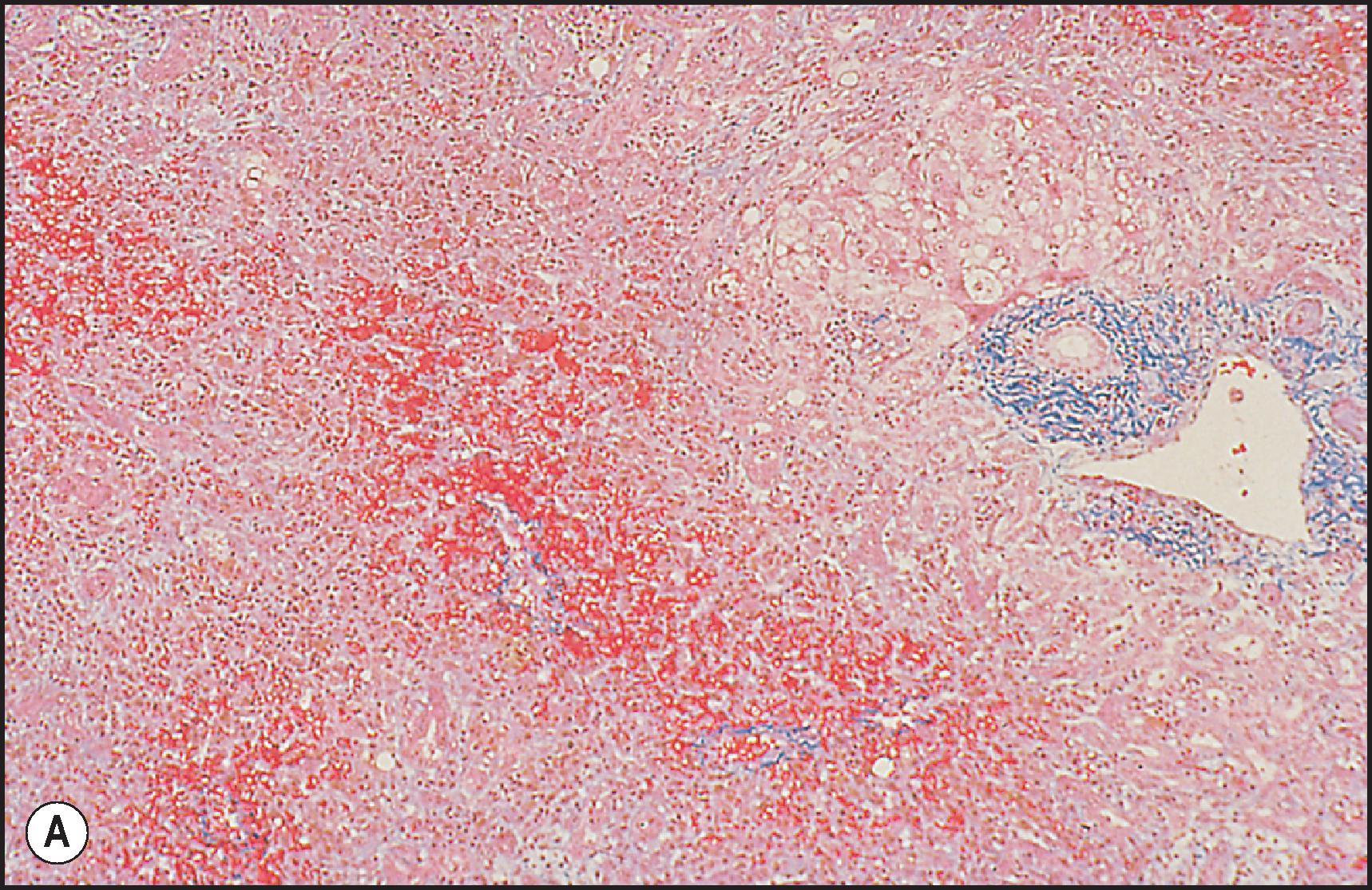
The unripe fruit of the ackee tree ( Blighia surpida ), native to Jamaica, contains a cholestatic hepatotoxin, hypoglycin A, which produces a clinical syndrome of GI distress and microvesicular steatosis known as Jamaican vomiting sickness that resembles Reye syndrome. Cholestatic jaundice has been described after chronic ingestion. Cycasin is a potent hepatotoxin and carcinogen found in the fruit of the cycad tree ( Cycas circinalis , Cycas revoluta ). A small epidemic of acute hepatic injury attributable to the ingestion of cycad nuts was reported from Japan.
Aflatoxins are a family of mycotoxins found in Aspergillus flavus and related fungi that are ubiquitous in the tropics and subtropical regions. They contaminate peanuts, cashews, soybeans and grains stored under warm, moist conditions and are well-known hepatotoxins and hepatocarcinogens. Occupational exposure to aflatoxins has been documented in Egyptian wheat handlers. Aflatoxin B1, a potent inhibitor of RNA synthesis, is the most hepatotoxic. Reactive metabolites are formed by P450 metabolism; malnutrition is a possible potentiating factor (perhaps due to glutathione depletion). When consumed in large quantities, it is responsible for a clinical syndrome characterized by a prodrome of fever, malaise, anorexia and vomiting followed by jaundice. Portal hypertension with splenomegaly and ascites may develop over the next few weeks. In large epidemics, mortality rates have approached 25% and are dose related. Zone 3 necrosis without significant inflammation is the characteristic lesion. Other histological findings include cholestasis, microvesicular steatosis and bile duct proliferation.
The risk of HCC has been well confirmed and is directly related to the amount of aflatoxin consumed, especially in sub-Saharan Africa and eastern China, where wheat often exceeds rice as a staple in the diet. Alcohol and possibly exposure to DDT appear to play an enhancing role in the hepatocarcinogenesis of rats exposed to aflatoxin B978-0-7020-8228-3. Perhaps an even more important cofactor is the interaction between aflatoxin and the hepatitis B virus (HBV). Aflatoxin is associated with a specific mutation in the p53 tumour suppressor gene leading to an amino acid change (R249S) that has been directly correlated to the development of HCC in these regions as compared to the rarity of this mutation in HCC from Western countries.
Kodo millet ( Paspalum scrobiculatum ) is a staple food found in northern India that is frequently contaminated by Aspergillus tamarii , which produces acute hepatic injury and preneoplastic changes due to another mycotoxin, cyclopiazonic acid.
A number of contaminated foodstuffs and cooking oils have been associated with epidemics of hepatotoxicity. While they are now largely of historical interest they are included as examples of the types of contaminations that can occur.
Spanish toxic oil syndrome occurred in 1981 after exposure to rapeseed cooking oil that was contaminated by anilines and acetanilides. Nearly 20,000 individuals became ill, many of whom had hepatic injury. The histopathological changes were those of a cholestatic hepatitis that resembled chlorpromazine-induced liver injury. Of 24 patients studied by Solis-Herruzo et al., 11 had necroinflammatory bile-duct lesions; NRH was also noted.
Epping jaundice refers to an epidemic of toxic liver injury that occurred in Epping, England in 1965. It involved 84 persons who had eaten bread contaminated with MDA. The clinical syndrome consists of cholestatic jaundice preceded by abdominal pain and fever, resembling biliary colic, beginning a few hours after ingestion. Liver biopsies performed in some patients revealed cholestatic hepatitis with cholangitis but little or no necrosis. Most individuals recovered within 4–6 weeks, with jaundice lasting up to 4 months in a few.
Yusho oil disease in western Japan, and a related epidemic referred to as Yu-Cheng in Taiwan, involved nearly 2000 individuals who had eaten rice prepared in oil contaminated by PCBs, dioxins and polychlorinated dibenzofurans (PCDFs) in 1968. It was characterized by acne-like eruptions, skin hyperpigmentation and neuropathy, with jaundice reported in approximately 10%. Exposed individuals still harboured high levels of these agents more than 26 years after the outbreak and have an increased risk of liver cancer among other malignancies after 40 years of follow-up.
Cocaine is a dose- and time-dependent hepatotoxin in mice manifested by elevated transaminase levels and by necrosis and steatosis. Numerous reports attest to its hepatotoxic potential in humans. , Acute cocaine intoxication involved the liver in 60% of patients reported by Silva et al., many of whom had elevated ALT values >1000 IU/L. Associated features include rhabdomyolysis, hypotension, hyperpyrexia, disseminated intravascular coagulation and renal failure. , , Hepatic and other organ injury is likely the result of toxic metabolites (e.g. norcocaine nitroxide) formed via CYP2E1 and CYP2A, which helps explain the enhanced hepatotoxicity seen in those who also abuse alcohol. Severe hepatic injury is characterized by coagulative necrosis involving the perivenular and midzones or all three zones, as well as microvesicular steatosis. , ,
Amphetamines have long been suspected to cause hepatic injury when taken as a form of drug abuse, but evidence for this is scant. However, ecstasy (3,4 methylenedioxymethamphetamine, MDMA) is an amphetamine derivative that can lead to hepatic necrosis as part of a heat-stroke-like syndrome that occurs as a result of exhaustive dancing in hot nightclubs. , The injury can be fatal and has necessitated liver transplantation in some instances. The role of P450 enzymes in the toxicity of this and other so-called designer drugs may be related to specific genetic polymorphisms of CYP2D6 or other cytochromes. Both cholestatic and hepatocellular injuries have been described. Chronic hepatitis with and without autoimmune features has also been attributed to this agent. ,
Phencyclidine (‘angel dust’) is another stimulant that has led to hepatic injury as part of a malignant hyperthermia syndrome producing zone 3 necrosis with congestion reminiscent of shock liver. Phencyclidine potentiates cocaine hepatotoxicity in animals, in part by depleting glutathione.
Although in general opioids have not been associated with hepatotoxicity, perivenular fibrosis and zone 3 sinusoidal dilation have been described in intravenous abusers of heroin . The changes were associated with inflammation of the wall of terminal hepatic venules. Patients who stopped abusing heroin had fibrous thickening of the veins and perisinusoidal fibrosis.
Although there are few reports of serious toxic effects from smoking or ingesting marijuana , intravenous usage has been associated with liver injury. The findings included fulminant gastroenteritis, hypoalbuminaemia, acute renal failure, pancytopenia and cholestatic hepatitis with portal inflammation, interface hepatitis and zone 3 cholestasis confirmed by biopsy. Cannabidiol has been associated with transaminase elevations in clinical trials but not with overt liver injury.
Ketamine is a short-acting anaesthetic with limited clinical use due to its psychological side effects. However, these side effects have made it attractive for abuse. Ketamine has been associated with biliary injury with dilations and narrowing of the biliary tree on images. Biopsies show varied bile duct injury, including periductal sclerosis and reactive changes with vacuolation.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here