Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In the United States, about 13% of strokes are caused by intracranial hemorrhage, but the worldwide percentage is about 20% because of countries where hypertension is common but atherosclerosis is less common. Intracerebral hemorrhage, which accounts for about 75% of cases, usually occurs when small penetrating arteries rupture and produce a focal collection of blood within the parenchyma. Subarachnoid hemorrhage, which accounts for about 25% of cases, occurs when a vessel ruptures on the surface of the brain, and blood enters into and spreads throughout the cerebrospinal fluid (CSF). In both of these bleeds, extension of blood into the cerebral ventricles occurs in about 40% of cases.
In subarachnoid hemorrhage, the hemorrhage occurs in the space between the innermost (pia mater) and middle layer (arachnoid mater) of the meninges that cover the surface of the brain.
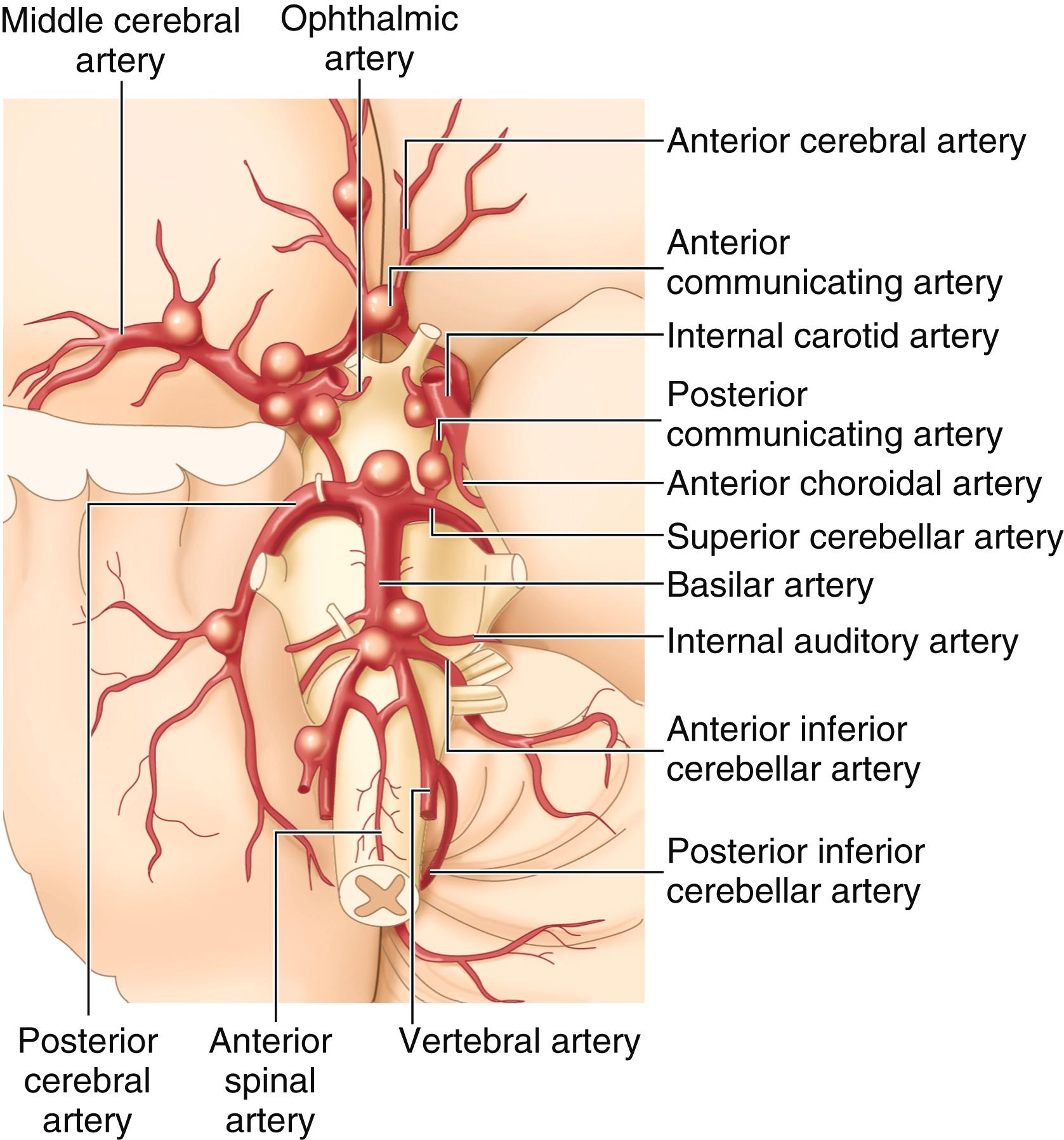
Subarachnoid hemorrhage represents approximately 25% of hemorrhagic strokes and 5% of all strokes. Trauma ( Chapter 368 ) is by far the most common cause of subarachnoid hemorrhage, accounting for perhaps 70% or more of cases. Nontraumatic, spontaneous subarachnoid hemorrhages are commonly caused by rupture of an intracranial aneurysm, almost 80% of which are saccular or berry aneurysms that most often occur at the bifurcations of the major branches of the circle of Willis ( Fig. 377-1 ). Other causes of spontaneous subarachnoid hemorrhage include peri-mesencephalic subarachnoid hemorrhage, cerebral amyloid angiopathy, cerebral venous sinus thrombosis ( Chapter 376 ), cerebral arteriovenous malformation, cerebral vasculitis, mycotic aneurysms, coagulopathies ( Chapter 157 ), dissection of the intracranial carotid or vertebral arteries ( Chapter 376 ), or extension of an intracerebral hemorrhage into the subarachnoid space ( Table 377-1 ).
| Trauma ( Chapter 368 ) Idiopathic perimesencephalic subarachnoid hemorrhage Arteriovenous malformation Intracranial arterial dissection ( Chapter 376 ) Cocaine and amphetamine use ( Chapter 365 ) Mycotic aneurysm ( Chapter 61 ) Pituitary apoplexy ( Chapter 205 ) Moyamoya disease ( Chapter 376 ) Central nervous system vasculitis ( Chapter 249 ) Sickle cell disease ( Chapter 149 ) Coagulation disorders ( Chapters 158 , 160 , and 161 ) Primary or metastatic neoplasm ( Chapter 175 ) |
Population-based studies indicate that the incidence of spontaneous subarachnoid hemorrhage varies by geographic region. The world age-standardized incidence is approximately 3.3 per 100,000 person-years. The annual incidence of aneurysmal subarachnoid hemorrhage in the United States is about 1 in 13,000, thereby resulting in about 25,000 cases per year, with the highest rates among women ages 40 to 60 years and Black Americans.
The risk factors for aneurysmal subarachnoid hemorrhage include a family history in first-degree relatives, autosomal dominant polycystic kidney disease ( Chapter 112 ), history of prior aneurysmal subarachnoid hemorrhage, and connective tissue or moyamoya disease. Additional modifiable risk factors are smoking ( Chapter 363 ), poorly controlled hypertension ( Chapter 64 ), excessive alcohol use ( Chapter 364 ), and use of sympathomimetic drugs such as cocaine ( Chapter 365 ) or adrenergic stimulants.
The classic presentation of subarachnoid hemorrhage is a “thunderclap headache.” The headache starts suddenly with maximum intensity, and most patients describe it as the worst headache of their lives. The headache is often associated with nausea, vomiting, photophobia, neck stiffness, and back or leg pain. Abrupt elevation of intracranial pressure at the time of aneurysmal rupture may result in brief loss of consciousness, with or without tonic posturing that mimics a seizure.
A sentinel or warning event can occur days to weeks before an overt aneurysmal subarachnoid hemorrhage in about 15 to 40% of cases. This warning event, which often implies minor leaks of blood from the aneurysm or aneurysmal expansion, dissection, or thrombosis, typically is not associated with nuchal rigidity, and the headache tends to be milder.
Mental status may vary from normal to coma depending on the severity of the subarachnoid hemorrhage. The physical examination may reveal meningismus and/or preretinal hemorrhages as a result of abrupt increase in intracranial pressure. Most patients do not have focal neurologic deficits, but focal neurologic deficits can occur based on the location of the aneurysm and hemorrhage. For example, cranial nerve III (oculomotor nerve) palsy suggests a posterior communicating artery aneurysm.
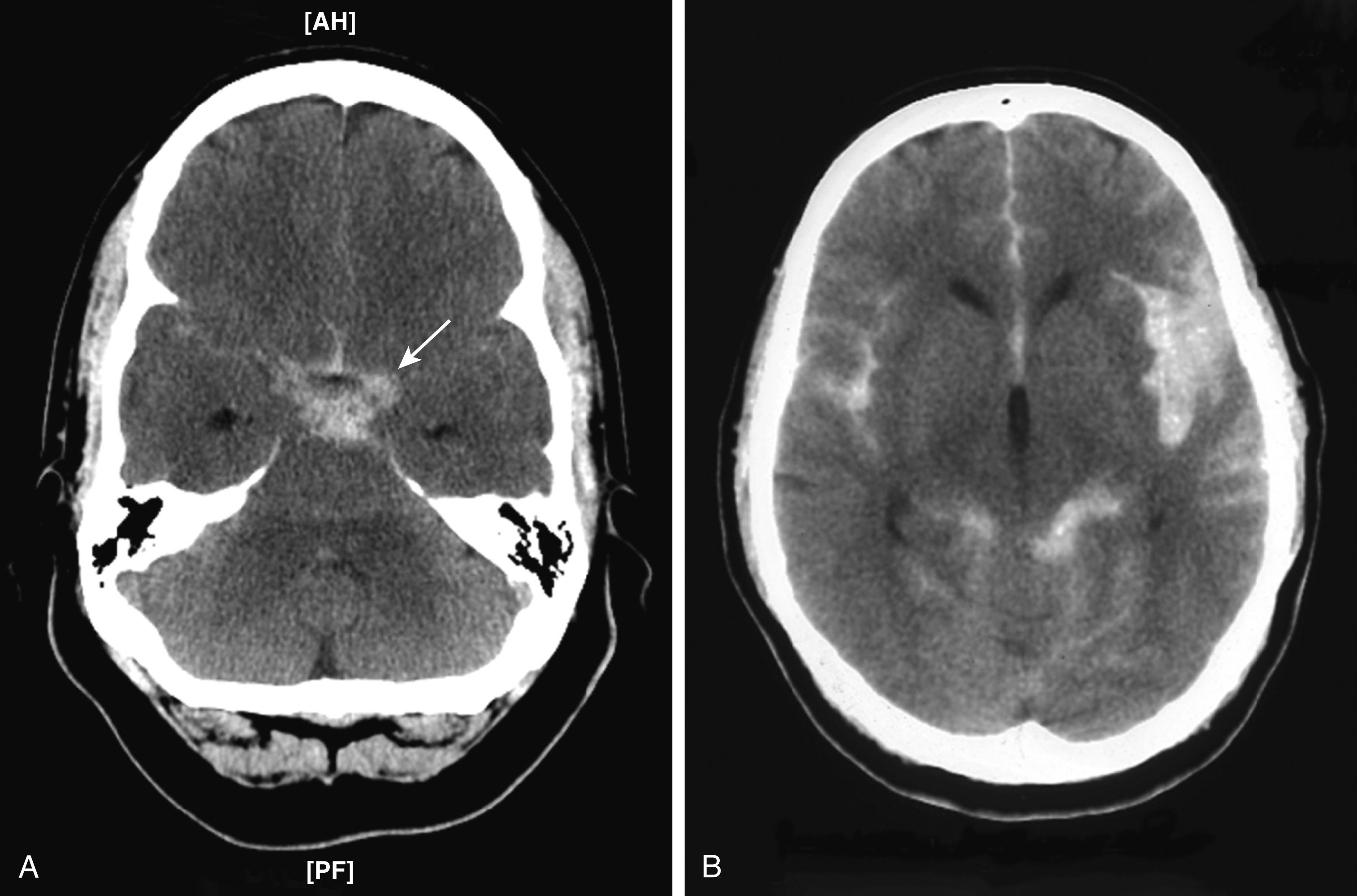
All patients who present with a new atraumatic headache with a maximum intensity within 1 hour should be questioned about characteristics that could be consistent with a subarachnoid hemorrhage ( Fig. 377-2 ) and further evaluated if such characteristics are present. The utility of diagnostic tests for subarachnoid hemorrhage is time-dependent. A noncontrast computed tomographic (CT) scan of the brain is the preferred initial diagnostic test, with an overall sensitivity of 97.5% for detecting subarachnoid hemorrhage ( Fig. 377-3 ). The sensitivity is even higher during the first 6 hours after the onset of symptoms (97% to 99%) but declines substantially (to about 75% by day 3) if the scan is delayed for 24 to 72 hours after the onset of symptoms.
If the CT scan is negative for subarachnoid hemorrhage but the index of suspicion is high, a lumbar puncture should be performed as soon as possible to examine CSF for cell counts and xanthochromia (a yellow-tinged appearance owing to hemolysis and usually indicative of a bleed 2 hours to a week or more earlier of the supernatant fluid after centrifugation. The classic findings in subarachnoid hemorrhage are an elevated opening pressure, an elevated red blood cell count typically greater than 100,000 cells/µL that does not decrease from tube 1 to 4, and xanthochromia. However, xanthochromia may take up to 12 hours to appear. Other conditions that can result in xanthochromia include systemic hyperbilirubinemia, a traumatic lumbar puncture with more than 100,000 red blood cells/µL, and increased concentrations of protein in the CSF.
About 15% of subarachnoid hemorrhages are initially overlooked, particularly in less symptomatic patients, and about 40% of these misdiagnosed patients experience subsequent complications such as vasospasm, rebleeding, hydrocephalus, or even death before returning for further evaluation. Therefore, a high degree of suspicion and vigilance is required to obtain a head CT scan and lumbar puncture in all patients who present with a thunderclap headache.
Laboratory testing including a complete blood count, platelet count, partial thromboplastin time, and international normalized ratio (INR) should be performed to evaluate for an underlying coagulopathy ( Chapter 157 ). An electrocardiogram showing peaked T-waves with QTc segment prolongation or an elevated troponin level can diagnose sympathetically mediated cardiac injury, which warrants an echocardiogram, and a chest radiograph can assess for possible aspiration pneumonitis or neurogenic pulmonary edema.
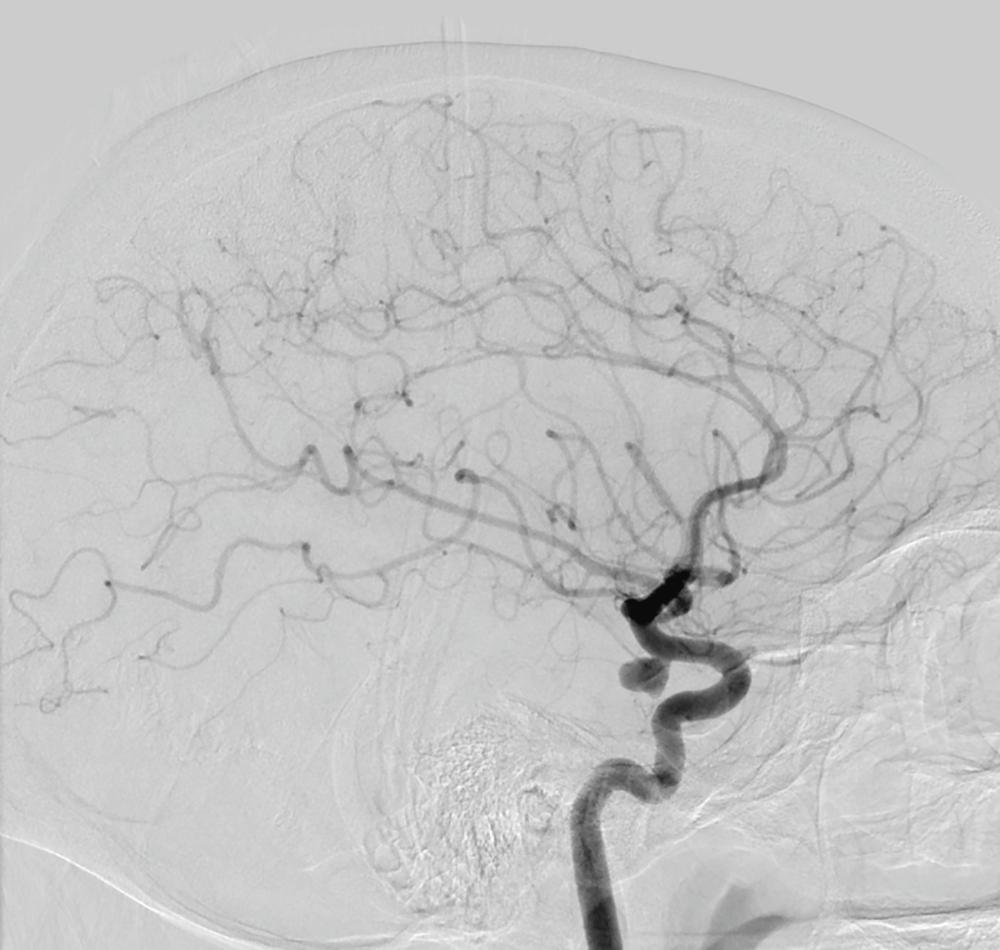
Once the diagnosis of subarachnoid hemorrhage is established, its cause must be determined (see Table 377-1 ). Aneurysmal subarachnoid hemorrhage is always the primary consideration in patients who present with a sudden nontraumatic headache. A CT angiogram or brain magnetic resonance image (MRI) with and without gadolinium and susceptibility-weighted imaging often can detect the bleed or a thrombosed aneurysm. However, the gold-standard for diagnosis of an aneurysm ( Fig. 377-4 ) is a four-vessel cerebral angiogram, which is indicated if noninvasive imaging is not revealing. Even with cerebral catheter-based angiography, a cause for the subarachnoid hemorrhage may not be evident on the initial study in 14 to 22% of cases. If the initial angiogram is negative, a repeat angiogram within 14 days should be considered. Angiography can also reveal an arteriovenous malformation, which is more likely to cause intracerebral than subarachnoid hemorrhage (see later) or features suggestive of CNS vasculitis.
The goals of treatment are to minimize early brain injury, prevent rebleeding, perform definitive surgical or endovascular interventions quickly, and prevent and treat secondary complications. Management of subarachnoid hemorrhage begins with early diagnosis and, if feasible, transfer to a high-volume, specialized center for emergency management and subsequent neurocritical care ( Table 377-2 ).
| Blood pressure |
|
| Rebleeding prophylaxis |
|
| Intravenous hydration |
|
| Laboratory testing |
|
| Seizure prophylaxis |
|
| Vasospasm prophylaxis |
|
| Physiologic homeostasis |
|
| Ventricular drainage |
|
| Vasospasm diagnosis |
|
| Therapy for symptomatic vasospasm |
|
Supplemental oxygen should be given to maintain normocapnea, and an oxygen saturation greater than 90%. Intubation should be considered if the patient’s Glasgow Coma Scale score (see Table 368-1 ) is less than 8, or if there is concern about patient’s ability to protect the airway (e.g., if excessive secretions or vomiting are present, or if gag or cough reflexes are absent). Maintenance of the circulation begins with establishing peripheral venous access and administration of isotonic fluids to maintain a mean blood pressure of 90 mm Hg.
To prevent rebleeding, the targeted systolic blood pressure should be less than 160 mm Hg, with a preferred goal of 140 to 160 mm Hg. The timing and need for vasopressors, central access, and invasive blood pressure monitoring should be individualized. To avoid rapid and large fluctuations in blood pressure, continuous intravenous infusions of labetalol or nicardipine (see Table 377-2 ) can be titrated. Vasodilators such as nitroprusside or nitroglycerin should be avoided because of their propensity to increase cerebral blood volume and intracranial pressure. Desmopressin (0.3 µg/kg intravenous push) may be beneficial in patients who have been taking antiplatelet agents. Epsilon aminocaproic acid (4 g followed by 1 g/hour for up to 72 hours) may be of modest benefit.
Environmental stimuli should be minimized to decrease the risk of rebleeding. Bed rest, low lighting, minimizing stress/pain-inducing interventions, use of antiemetics, and adequate pain control are important supportive measures to achieve this goal.
Emergent endovascular coiling or surgical clipping of a ruptured aneurysm is usually required to prevent rebleeding, except in patients with extremely poor prognoses. For small- to medium-sized anterior circulation aneurysms in patients with good Hunt-Hess grades of 3 or less ( Table 377-3 ), endovascular coiling provides better results than surgical clipping.
| GRADE | CLINICAL FINDINGS | HOSPITAL MORTALITY (%) |
|---|---|---|
| I | Asymptomatic or mild headache | 3 |
| II | Moderate to severe headache or oculomotor palsy | 3 |
| III | Confused, drowsy, or mild focal signs | 9 |
| IV | Stupor (localized to pain) | 24 |
| V | Coma (posturing or no motor response to pain) | 70 |
| TOTAL | 18 |
Vasospasm, manifested by focal ischemic findings and diminished consciousness, is a common complication that may occur 5 to 14 days after an aneurysmal subarachnoid hemorrhage when blood degradation products irritate the walls of an artery, thereby leading to spasm with subsequent reduction in blood flow and cerebral ischemia ( Table 377-4 ). Transcranial Doppler ultrasonography is the primary noninvasive method to detect vasospasm by measuring elevations of mean cerebral blood flow velocities within the cerebral arteries. CT angiography also can be helpful to establish the diagnosis, but catheter-based cerebral digital subtraction angiography remains the gold standard to diagnose radiographic vasospasm.
| PERCENTAGE OF AFFECTED PATIENTS | ||||
|---|---|---|---|---|
| GRADE | CRITERIA | FREQUENCY | DELAYED CEREBRAL ISCHEMIA ∗ | INFARCTION |
| 0 | No subarachnoid hemorrhage or intraventricular hemorrhage | 5% | 0% | 0% |
| 1 | Minimal/thin subarachnoid hemorrhage, no biventricular intraventricular hemorrhage | 30% | 12% | 6% |
| 2 | Minimal/thin subarachnoid hemorrhage, with biventricular intraventricular hemorrhage | 5% | 21% | 14% |
| 3 | Thick subarachnoid hemorrhage, † no biventricular intraventricular hemorrhage | 43% | 19% | 12% |
| 4 | Thick subarachnoid hemorrhage, † with biventricular intraventricular hemorrhage | 17% | 40% | 28% |
| All patients | 100% | 20% | 12% | |
∗ Defined as symptomatic deterioration, cerebral infarction, or both resulting from vasospasm.
† Thick subarachnoid hemorrhage is defined as completely filling at least one cistern or fissure.
Careful control of blood pressure and fluid status are important to prevent and treat vasospasm. After the aneurysm has been secured, blood pressure can be liberalized to a mean arterial pressure of 100 to 110 mm Hg to maintain adequate cerebral perfusion pressure of 60 to 80 mm Hg. Intravascular fluid management should focus on avoiding hypovolemia and maintaining euvolemia with isotonic fluid resuscitation (0.9% normal saline at 1 to 1.5 mL/kg/hour). Nimodipine (60 mg every 4 hours for 21 days orally or via nasogastric tube) is also indicated beginning within 48 hours of the hemorrhage and continuing for 21 days to reduce the risk of delayed cerebral ischemia. Some data suggest that local administration of an antifibrinolytic drug at the site of the clot might reduce the risk of symptomatic vasospasm and its adverse effects, but the treatment is not widely advocated. If hypotension or symptomatic vasospasm occurs, bolus doses of isotonic fluid can be given in combination with pressors, (e.g., norepinephrine, phenylephrine, or milrinone) to elevate systolic blood pressure above 180 mm Hg. In refractory symptomatic cases, intra-arterial infusion of verapamil or nicardipine may be beneficial, and sometimes intraluminal balloon angioplasty may be warranted.
Obstructive or nonobstructive hydrocephalus can develop acutely after subarachnoid hemorrhage and result in increased intracranial pressure or herniation. Initial life-saving treatment requires placement of an external ventricular drain to remove CSF and to monitor intracranial pressure. Prophylactic lumbar drainage (5 mL/hour starting within 72 hours) can reduce the risk of secondary infarction and unfavorable outcomes. Many patients with acute hydrocephalus will develop chronic hydrocephalus and require placement of a permanent ventriculoperitoneal shunt.
Patients who suffer an aneurysmal subarachnoid hemorrhage are at high risk of seizures, which can raise intracranial pressure and increase the risk of rebleeding. To reduce the risk of seizures, prophylactic therapy with levetiracetam (2000 mg IV loading dose followed by a maintenance dose of 1000 mg IV twice daily) is commonly started at the time of diagnosis. In the absence of acute seizures, antiepileptic therapy can be safely discontinued in good-grade (Hunt-Hess grade 3 or less) patients after the ruptured aneurysm has been repaired. In poor-grade, comatose or stuporous patients, continued antiepileptic therapy and continuous EEG monitoring may be considered to evaluate for subclinical seizures. A benzodiazepine or propofol may be used to treat seizures ( Table 372-6 ).
Patients who suffer a subarachnoid hemorrhage are at risk for medical complications. Hyponatremia ( Chapter 102 ) is common and may be caused by the syndrome of inappropriate antidiuretic hormone (SIADH) or by cerebral salt wasting. Sodium levels should be monitored closely, and hyponatremia should be corrected slowly at a rate not to exceed 1.3 mEq/L or more than 10 Eq/L in 24 hours. Unlike hyponatremia in other settings, fluid restriction should be avoided in patients who have a subarachnoid hemorrhage because it may result in increased blood viscosity and predispose to cerebral ischemia from vasospasm. Early treatment with fludrocortisone (0.05 mg per day) is recommended to prevent natriuresis and hyponatremia, and small doses (e.g., 250 to 500 mL) of hypertonic saline may be considered.
Fever, which is common after subarachnoid hemorrhage, may be related to loss of autoregulation or to a systemic inflammatory response. Acetaminophen (650 mg every 6 hours as needed) is preferred over agents that may aggravate bleeding. Surface or intravascular cooling may be considered as a second-line option if needed.
Massive sympathetic outflow after subarachnoid hemorrhage, particularly in poor-grade patients, may lead to neurogenic pulmonary edema ( Chapter 45 ), cardiac dysfunction, electrocardiographic changes, an elevated cardiac troponin level, and a reduced left ventricular ejection fraction with wall motion abnormalities. These consequences are usually reversible with supportive treatment.
Preventive screening for intracranial aneurysms is reasonable in first-degree relatives of patients who have suffered an aneurysmal bleed and in patients with polycystic kidney disease ( Chapter 112 ). Intracranial aneurysms that show growth on serial studies have about a 2.5% risk of rupture within the next year and may be appropriate for definitive treatment (coiling or clipping). Otherwise, however, intervention appears to be no better than careful monitoring and control of risk factors, such as hypertension ( Chapter 64 ), and cessation of smoking ( Chapter 363 ) and heavy alcohol use ( Chapter 364 ), probably even in patients with polycystic kidney disease ( Chapter 112 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here