Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Of all causes of hemophilia, types A and B are most associated with intraosseous and intraarticular bleeding. Hemophilia A (classic hemophilia) results from a deficiency of factor VIII. Hemophilia B (Christmas disease) is due to a functional deficiency of plasma thromboplastin component (factor IX). These disorders are both X linked and, therefore, manifest clinically in men but are carried by women.
Classic hemophilia occurs in 1 in 10,000 males in the United States. Christmas disease is rarer, affecting approximately 1 in 100,000 males. Although both forms are almost exclusive to males, reports exist of the disease occurring in females. Twenty-five percent of hemophilia is due to spontaneous mutation. Acquired hemophilia due to antibodies to factor VIII is rare but severe and can affect women.
Between 75% and 90% of hemophilia patients will suffer from hemarthrosis. Adults are better able to protect their joints from trauma and, consequently, hemarthrosis is more common in young children and adolescents.
Acute intraarticular bleeding will present as pain and dysfunction. Swelling and effusion will be apparent in the more superficial joints. When it is already known that the patient has a coagulation disorder and the clinical findings are apparent, imaging is of limited immediate value. Because these patients are often in severe pain and are prone to worsening of the hemorrhage, it is unwise to perform even the least invasive examination. In the acute phase, the priority is resuscitation and coagulation control with blood factor replacement not radiologic investigation. As soon as the patient is stabilized, radiographs and other imaging may be useful to exclude fracture and evaluate preexisting arthropathy.
Deep and impalpable joints, such as the hip, are difficult to examine, and ultrasonography is the best means of demonstrating or excluding intraarticular bleeding or effusion. This will also be the case in peripheral joints with small effusions or in obese patients. Joints related to the spine may require CT or MRI to detect involvement, although this would be an unusual occurrence.
Hemarthrosis may be first detected by imaging when patients present with less specific complaints, including pain, anemia, and limitation of movement. Therefore, the imaging criterion for the diagnosis of blood in a joint should be known for all of the standard techniques.
Synovial thickening occurs as a result of repeated hemarthrosis. There seems to be a trigger that can turn reactive synovial thickening into an aggressive and progressively destructive synovitis leading to subsequent destruction of the articular surface. Repeated bleeding is the most potent cause, although the exact reason for its onset is not clear. Clinically, the patient will suffer from progressive joint dysfunction, manifest by increased stiffness.
In this stage of disease, imaging may be misleading in distinguishing free fluid from synovial thickening, especially if too much reliance is placed on MRI. Synovial thickening and fluid are indistinguishable on plain films. The synovial tissue will have a rich blood supply, and, although bone scintigraphy may reflect this on blood pool images, it cannot reliably differentiate between the low-grade synovial irritation seen in virtually all joint effusions and significant synovial proliferation. Ultrasonography is arguably the most useful test. Fluid has minimal echoes, whereas synovial thickening is echogenic with blood flow seen on spectral Doppler imaging. CT with intravenous contrast medium enhancement can be used, whereas plain CT may confuse the two intraarticular substances. Regrettably, the MRI appearances of fluid and synovial thickening are the same on T1- and T2-weighted images. The blood supply to synovium will become apparent after intravenous administration of gadolinium diethylenetriamine pentaacetic acid (Gd-DTPA), especially if fat suppression is used on postcontrast, T1-weighted images. It is tempting but inaccurate to rely on conventional MR signal changes to exclude synovitis. Intraarticular contrast would work with either CT or MRI, but, fortunately, there is a better alternative. If ultrasonography is available, this is a less expensive and faster means to diagnose synovial fluid and thickening than either CT or MRI contrast studies.
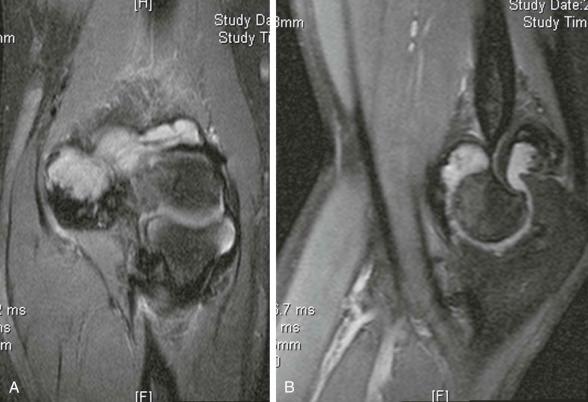
Although radiographs will give an overview of joint degeneration, showing a widened intercondylar notch, MRI is usually the investigation of choice when surgery is being contemplated. MRI will show hemorrhage at varying ages with hemosiderin deposition, which appears as a signal void (i.e., “black”) on all sequences. The differential diagnosis of such a patient's arthropathy would be pigmented villonodular synovitis. Operating on a hemophilic patient is so hazardous and expensive that it would be wise to obtain as much data as possible by noninvasive techniques, even when clinical circumstances may not warrant complex imaging in a similar patient with normal coagulation.
Patients present with acute pain, swelling, and dysfunction. There may be secondary complications resulting from the enlargement of a hematoma. For example, there may be compression of adjacent nerves or vascular structures. In this phase of disease, acute hematomas or soft tissue collections can cause severe compression of a nerve or vessel so that urgent surgery will be considered.
It may be argued that MRI should be the first and only investigation for the investigation of soft tissue hemorrhage and its complications. CT with ultrasonography should only be used when MRI is not available ( eFig. 73-1 ).
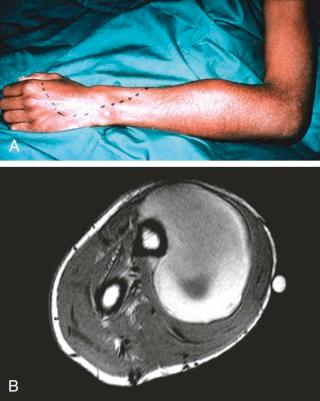
For the reasons listed earlier, ultrasonography or MRI is the best means of showing a recurrent soft tissue hemorrhage.
Pseudotumors are a rare complication of hemophilia, occurring in 1% to 2% of patients with severe hemophilia (clotting factor level < 1% of normal). The pseudotumor grows as a chronic, slowly expanding, encapsulated cystic mass as a result of recurrent hemorrhage into extra-articular soft tissue sites and, therefore, presents as a slowly enlarging mass. Pseudotumors are categorized as osseous and soft tissue lesions, on the basis of anatomic location. The sites most frequently affected are, in order of descending frequency, the femur, pelvis, tibia, and small bones of the hand. Radiographic findings vary greatly with the extent, location, and different stages of hemorrhage and reflect the presence of medullary bone destruction, cortical changes, internal opacities, various types of periosteal reaction, and surrounding soft tissue abnormalities. With these variable radiographic appearances, osseous hemophilic pseudotumors can be confused with other tumorous or infectious conditions.
MRI is unrivaled in the detection, staging, and follow-up of soft tissue bleeding with mass effects.
Because transfusion factor replacement and immobilization are the emergency treatments for massive blood loss, imaging has little immediate role and probably should be avoided. Exceptions to this rule would be in a penetrating injury or fracture when the same protocols that would be considered for those with normal coagulation should be applied.
When the nature of the injury might have caused a fracture, then radiographs are always indicated. CT may be used to support the diagnosis and further characterize complex fractures if surgery is necessary.
It is well recognized that repeated bleeding into the joint is responsible for synovial and cartilage changes. Blood appears to have three effects that result in joint destruction: a direct effect on the cartilage and indirect effects of synovial proliferation and induction of inflammatory cells.
Blood appears to have a direct toxic effect on cartilage before and independent of its other effects. The direct effects of blood on cartilage precede synovial changes, and synovitis may be secondary to articular cartilage damage. Studies show that, after exposure to blood, there is inhibition of matrix formation and increased matrix breakdown, resulting in a loss of matrix.
Iron deposition seems to be the mechanism by which bleeding into a joint exerts its indirect effects. Acute episodes of bleeding result in the formation of a dark clot containing fluid within the joint. After repeated episodes, resorption is incomplete, resulting in the synovial membrane taking on a brown tint caused by blood pigment absorption. This progressive accumulation of iron deposits in the synovium is an important characteristic and thought to be the trigger for synovial inflammation with proliferation in number and hypertrophy of the synovial villi. The hemosiderin deposits may attract lymphocytes. The inflammatory changes are mild in comparison with true inflammatory arthropathies, such as rheumatoid arthritis. The presence of synovial iron deposits is also seen in other joint disorders, such as pigmented villonodular synovitis, hemangiomas, and hemosiderotic synovitis. All of these disorders share a pattern of joint destruction that resembles hemophilic arthropathy, suggesting iron deposition is important. Therefore, it is thought that the accumulation of iron within the joint is a stimulus for synovial proliferation and an attractant for inflammatory cells that produce enzymes and cytokines that cause destruction of articular cartilage.
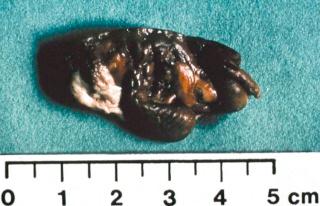
Pseudotumors represent an uncommon complication of hemophilia and occur in 1% to 2% of patients with severe forms of the disease. Pseudotumors represent chronic, slowly expanding hematomas. The lesions are encapsulated and often occur in the soft tissues but may arise in bone or subperiosteum. Pseudotumors that occur in muscles with broad tendon insertions progress to cause severe pressure erosion of adjacent bone. The bones most commonly affected are the femur, pelvis, tibia, and bones of the hand.
Pathologically, pseudotumors consist of blood products in various stages of evolution with a fibrous capsule ( eFig. 73-2 ). Hemosiderin-laden macrophages are seen within the fibrous capsule. As the pseudotumor enlarges, there may be increasing pressure on adjacent structures that may undergo necrosis. The result can be destruction of adjacent bone or necrosis of muscle and skin. Compartment syndromes and joint contractures can also occur. The tumors are usually painless, but there may be compression of adjacent nerves resulting in pain or neurologic dysfunction. Pain can also result from pathologic fractures (see eFig. 73-23B ). As pseudotumors progress, function of the extremity will be compromised. Rarely, pseudotumors have ruptured, resulting in exsanguination. These complications may develop suddenly in patients who previously exhibited no symptoms or appeared clinically stable.
It is usually difficult on clinical and imaging grounds to determine whether an effusion is composed of fresh blood as opposed to transudate or pus. It would normally be necessary to aspirate the joint to confirm hemarthrosis. This may be a hazardous procedure without factor replacement capability, and this will always occur when the coagulation disorder is not yet discovered. Those who aspirate joints must be aware of this risk. Imaging control or guidance is useful to ensure that the joint was punctured and not an adjacent vessel. Joint aspiration techniques are described elsewhere, but there are specific precautions to be taken when a coagulopathy is known or suspected. These include adequate cover with blood factor replacement, strict aseptic technique to minimize the risk of infection, and immobilization after the procedure in those with antibodies to injected coagulation factor. The risk of introducing infection or precipitating fresh hemorrhage should be balanced against the risk of failing to detect septic arthritis. A principal role of imaging is to avoid unnecessary joint puncture when there is no fluid present. CT, MRI, or ultrasonography can all be used to guide the needle placement. After the procedure, CT, ultrasonography, or MRI may be used to assess how much fluid remains. For practical and financial reasons, ultrasonography is the method of choice.
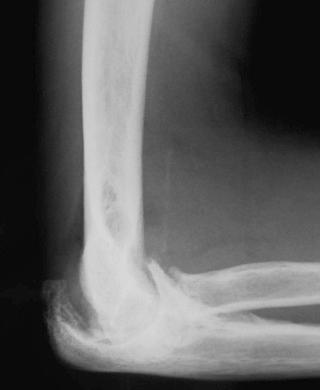
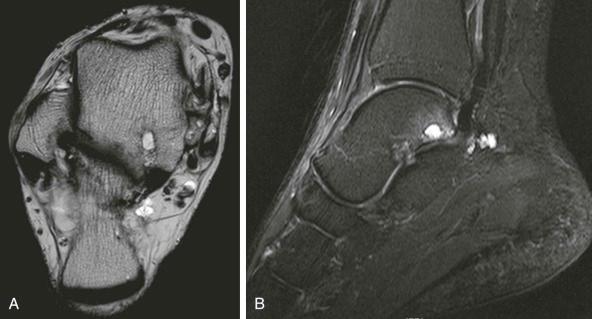
If careful attention is paid to the soft tissue planes, radiographs may show joint effusion, but, in practice, these signs are of minimal value. When radiographic signs are apparent, joint distention is clinically obvious. In the early stages, when the effusion is small and difficult to detect clinically, the assessment of fat stripes next to joints may be very misleading and, with the exception of the elbow ( eFig. 73-3 ), prone to overinterpretation. The availability of other imaging methods means that the plain radiographic signs of acute disease are now largely of historical interest.
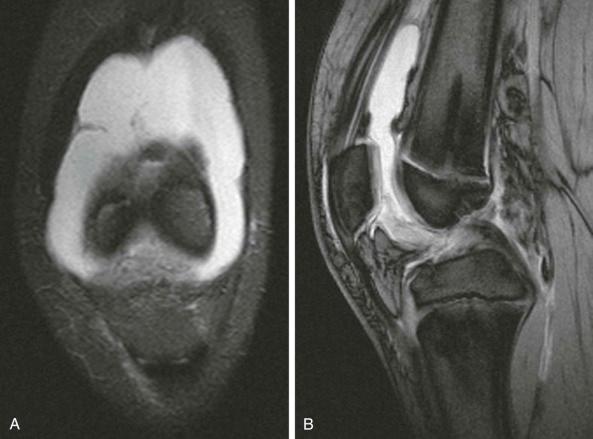
Magnetic resonance imaging is very sensitive to joint fluid, especially on T2-weighted images where fluid returns a high signal ( eFigs. 73-4 through 73-6 ). If a fat-suppression technique is used, the signal from fat cannot obscure the details of the joint effusion. Unenhanced MRI cannot determine the degree of synovitis. The swollen synovium has the same signal as fluid within the joint on most conventional images. Some would argue that magnetization (or saturation) transfer contrast in MRI is an improvement, but there are still problems, and enhancement with intravenous Gd-DTPA is necessary for precision.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here