Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Non-Hodgkin lymphoma (NHL) is a lymphoid malignancy that represents 4.3% of all new cancer diagnosis in the United States and is estimated to be responsible for over 19,000 deaths in 2020. While newer immunotherapies have emerged that have improved treatment options and outcomes for NHL patients, hematopoietic cell transplantation (HCT) remains an important modality, providing prolonged progression-free survival (PFS) and overall survival (OS), for certain subtypes. This chapter will review the role of autologous and allogeneic HCT (allo-HCT) for the treatment of the more common NHLs based on recent pertinent clinical data and provide recommendations for treatment.
Lymphoma classification continues to evolve over the past several decades with improved knowledge in biology refinement ( Table 24.1 ). In 2008, the fourth edition of the World Health Organization (WHO) classification emerged and was soon revised in 2016. This chapter will focus on HCT treatments for B-cell lymphomas like diffuse large B-cell lymphoma (DLBCL), Burkitt lymphoma, primary mediastinal B-cell lymphoma, follicular lymphoma (FL), and mantle cell lymphoma (MCL), and mature T-cell lymphomas namely peripheral T-cell lymphoma not otherwise specified (PTCL NOS).
| Entity | Clinical Practice Implications |
|---|---|
| Monoclonal B-cell lymphocytosis | Distinguish low-count from high-count MBL |
| Clinical follow-up not required for low-count MBL | |
| In situ follicular neoplasia | Low risk of progression to lymphoma |
| In situ mantle cell neoplasia | Low clinical risk |
| CD8+ T-cell proliferations | Conservative management |
| Pediatric-type follicular lymphoma | Conservative therapeutic approach; must differentiate from high-grade follicular lymphoma grade |
| Duodenal-type follicular lymphoma | Low risk of dissemination |
| EBV+ mucocutaneous ulcer | New entity associated with immunosuppression |
| Diffuse large B-cell lymphoma NOS | Distinction of GCB vs. ABC/non-GC type required |
| Coexpression of MYC and BCL2 recognized as new prognostic marker | |
| High-grade B-cell lymphoma, with MYC and BCL2 and/or BCL6 translocations | New category for double and triple-hit lymphomas |
| Consideration of dose-intensive therapy | |
| ALK– anaplastic large cell lymphoma | Now a recognized entity; prognosis intermediate between ALK+ ALCL and PTCL |
A 54-year-old woman presents with palpable lymph nodes in her neck with associated fatigue, weight loss 10 lbs. Biopsy reveals large B cells, positive for CD20, BCL6, and MUM-1, without presence of MYC gene rearrangement. She is diagnosed with nongerminal center B-cell like DLBCL.
DLBCL is the most common subtype of NHL, though outcomes are poor for relapsed disease. For over two decades, autologous HCT has been offered to fit relapsed or refractory DLBCL, responding to salvage treatments. Autologous HCT became the standard of care when the landmark PARMA trial showed its superiority over standard chemotherapy with decadron, cytarabine, and cisplatin (DHAP) for chemosensitive relapsed DLBCL. The one critique of this trial was that many patients were treated before the approval of the anti-CD20 monoclonal antibody rituximab. Despite this, autologous HCT consolidation continues to show long-term PFS of 45% to 55% for chemosensitive relapse in the rituximab era. Relapses following autologous HCT occur in a subset of patients with adverse clinical characteristics that include MYC gene rearrangement with either BCL-2 and/or BCL-6 gene rearrangement(s) known as double hit lymphoma (DHL), or early relapse within 1 year from diagnosis. As witnessed from the PARMA trial, a significant proportion of patients with R/R DLBCL will not proceed to receive an autologous HCT, often because of refractory disease or comorbidities of age, frailty, or lack of support structure. The Italian Intergroup evaluation of time to relapse in patients enrolled on the PARMA trial indicated that early relapses (within 12 months from diagnosis) had worse PFS and OS requiring improved salvage therapies for these patients. This study further highlighted that patients with chemosensitive disease, lower lactate dehydrogenase, and less bulky disease at the time of HCT had better prognosis. In the Collaborative Trial in Relapsed Aggressive Lymphoma (CORAL), a European intergroup phase III prospective study, relapsed of primary refractory DLBCL patients were randomized to receive salvage chemotherapy with R-ICE or R-DHAP. Responding patients underwent autologous HCT and subsequently randomized to rituximab maintenance versus observation. When comparing the two salvage regimens, there was no difference in response rates or event-free survival (EFS) to either. However, those patients who relapsed within 12 months from diagnoses with prior rituximab exposure had inferior PFS compared with those without rituximab exposure. However, for patients relapsing > 12 months from diagnosis, PFS was not impacted by prior rituximab exposure. This further highlights that disease biology has a greater impact on outcomes overall. Using the traditional Hans algorithm, the CORAL investigators classified 46% patients as germinal center B-cell like (GCB) and 49% activated B-cell like (ABC). The ABC-like DLBCL patients had an unfavorable outcome regardless of the type of salvage regimen unlike GCB-like that had better outcomes with R-DHAP. Despite these findings, both regimens are commonly used salvage treatments based on clinician choice.
Subsequently, several randomized trials have explored the role of upfront consolidation autologous HCT for aggressive DLBCL. A recent metaanalysis of randomized trials evaluating the role of autologous HCT as first-line consolidation therapy for aggressive NHL did not show benefit for EFS or OS. Two randomized trials conducted in the rituximab era tested the role of autologous HCT for newly diagnosed high-risk patients with DLBCL. A Southwest Oncology Group study comparing cyclophosphamide, doxorubicin (Adriamycin), vincristine (Oncovene), and prednisolone (CHOP) or rituximab (R)-CHOP induction followed by either additional cycles of R-CHOP or autologous HCT did not show differences in OS despite higher 2-year PFS, 69% versus 56% ( P = .005) in favor of the autologous HCT arm. However, the benefit of PFS and OS was notable in the high international prognostic index (IPI) group. Similarly, the Italian Lymphoma Foundation conducted a trial for high-risk (aaIPI 2 or 3) DLBCL patients randomizing patients to either RCHOP14 versus RmegaCHOP, followed by a second randomization for responding patients, to either autologous HCT or observation. Once again there was notable benefit if 2-year PFS in the autologous HCT arm 72% versus 59% ( P = .008) but no differences in OS.
Recently, with the advent of novel cellular immunotherapy treatments like anti-CD19 chimeric antigen receptor T-cells (CAR T) for the treatment of R/R DLBCL failing two or more lines of therapy, there is a declining trend in the use of autologous HCT. To appraise the role of autologous HCT in this modern era, Shah et al. explored the registry database of the Center of International Blood and Marrow Transplant Research (CIBMTR) to show the utility and benefit of autologous HCT remains in patients with chemosensitive disease despite positron emission tomography/computed tomography (PET/CT)-positive disease at the time of transplant. Further, patients relapsing within 12 months of diagnosis, identified as early chemotherapy failure, were younger with advanced stage and more primary refractory disease. Although 1-year outcomes for PFS and OS favored patients who experienced late failure, there were no significant differences in 5-year probabilities for PFS and OS between the two groups. Therefore autologous HCT remains an appropriate standard of care treatment option in patients with R/R DLBCL while proving chemotherapy sensitive disease.
Autologous HCT is considered for:
Relapsed/Refractory DLBCL in CR or PR after salvage
DHL DLBCL treated with R-CHOP in CR1
CR , Complete remission; DHL , double hit lymphoma; DLBCL , diffuse large B-cell lymphoma; HCT , hematopoietic cell transplantation; PR , partial remission
The ability of adoptive cell therapy in the form of allo-HCT to recognize and then mount a powerful immune response against unknown tumor antigens (i.e., the graft-versus-malignancy effect) has long supported the role of this modality as a curative therapy in heavily pretreated lymphoma patients. Allo-HCT can provide durable disease control in high-risk lymphomas, with CIBMTR registry data showing continued improved survival of NHL ( Fig. 24.1 ) patients following allografting over the last three decades.
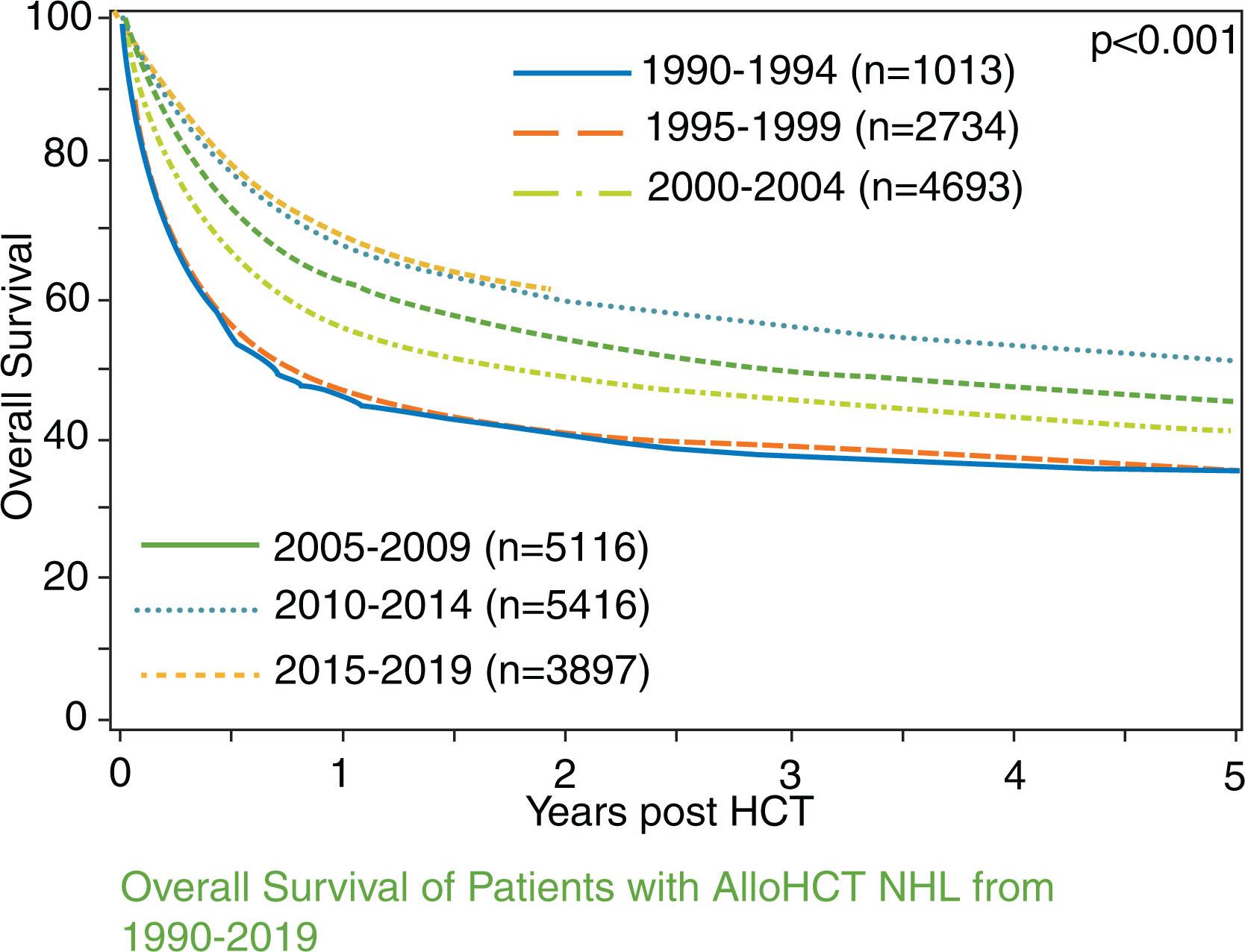
For most DLBCL patients, allo-HCT is used after failure of prior autologous HCT. The European Society for Blood and Marrow Transplantation (EBMT) registry evaluated the role of allo-HCT in DLBCL relapsing after autologous HCT (n = 101; era 1997–2006). Despite aggressive NHL, patients had encouraging 3-year PFS and OS of 42% and 54%, respectively, though accompanied by a relatively high nonrelapse mortality (NRM) rate of 28% at 3 years. To identify patients who would likely benefit from an allo-HCT, the CIBMTR examined clinical factors predictive of outcomes of DLBCL following allo-HCT (n = 503; era 2000–2012). In this report, poor performance status, resistance to chemotherapy, and a short interval between autologous HCT and allo-HCT of less than 1 year were factors predictive of inferior OS. The 3-year OS of patients with none, one, two, or all these risk factors was 43%, 39%, 19%, and 11%, respectively. Therefore patients with two or more of these risk factors were unlikely to benefit from allo-HCT. The use of modified and less intense conditioning regimens before allo-HCT have improved NRM to 15% to 20% for this treatment modality, improving long-term survival in about 45% to 50% patients; however, toxicity concerns related to infections and graft-versus-host disease (GVHD) remain at large. With the advent of CAR T-cell therapies for relapsed DLBCL failing two or more lines of treatment, the role of allo-HCT is further debatable because of the known toxicities of the treatment. However, it remains, to date, the only potentially curative approach when used in the right clinical setting, even though from 2017 onward, the CIBMTR trends data show a decline in the use of allo-HCT for DLBCL, which coincides with the rise in utilization of CAR T-cell therapy for this disease ( Fig. 24.2 ). While details of CAR T-cell therapy will be covered in Chapter 26 , it is important to note here that despite the remarkable response to CAR T-cell therapies, more than 50% patients will ultimately relapse with extremely poor prognosis. For these patients, allo-HCT remains a consideration. Early identification of patients at risk of relapse after CAR T-cell therapy, by testing measurable residual disease (MRD) with circulating tumor deoxyribonucleic acid assays, can ease the burden of time-sensitive and complex logistics of planning for the allo-HCT process.
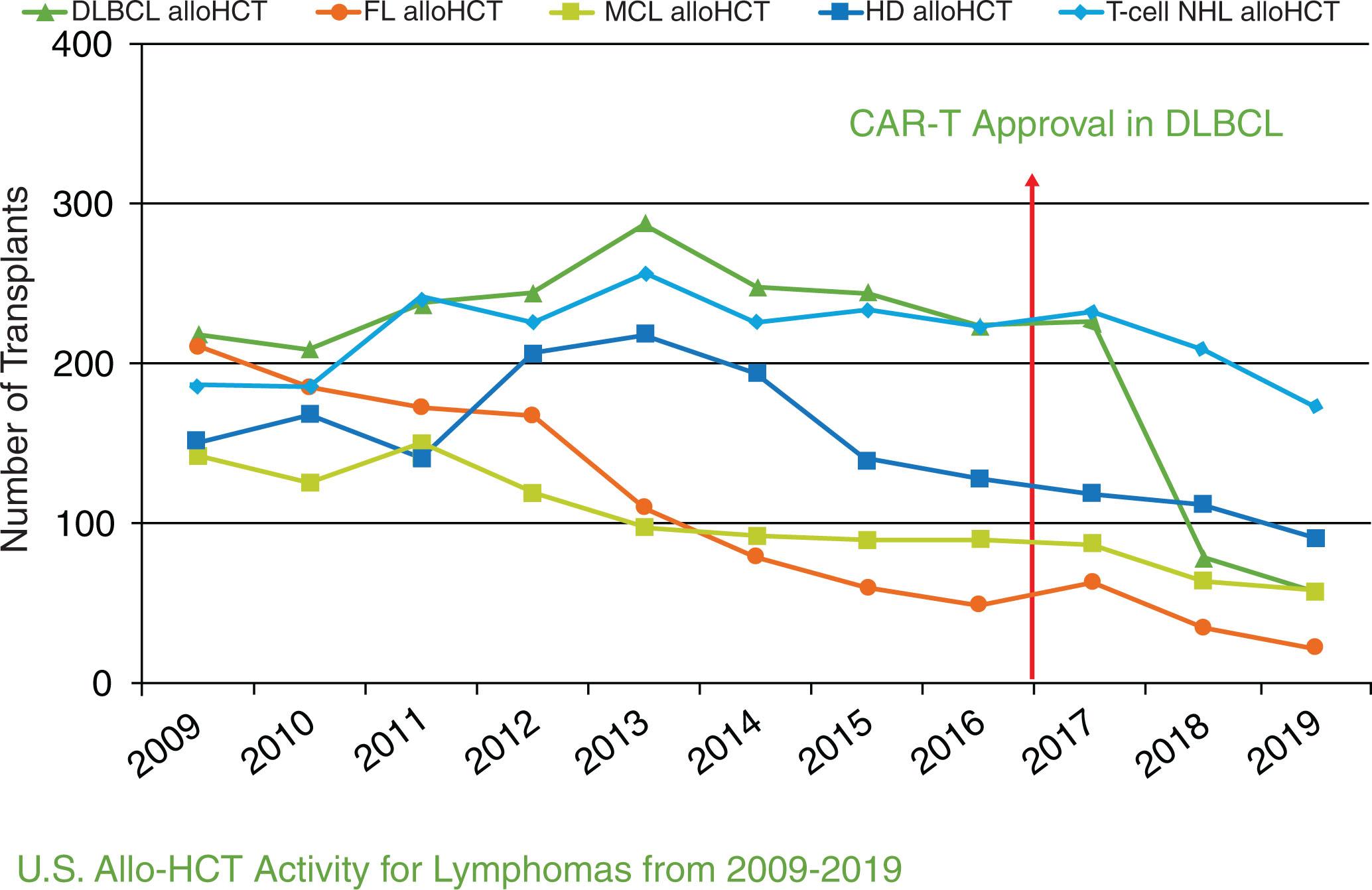
Table 24.2 shows select studies and outcomes following allo-HCT for DLBCL. Importantly, when considering allo-HCT for DLBCL, multiple studies now show the value of using reduced-intensity regimens (RIC), which are sufficient for graft-versus-lymphoma (GVL) effect and in turn reduced NRM.
Allogeneic HCT is considered for:
DLBCL relapsing after autologous HCT or CAR T-cell therapy (if this treatment is available to the patient).
DLBCL in partial remission after CAR T-cell therapy.
Physically fit patient with chemotherapy sensitive disease.
CAR , Chimeric antigen receptor; DLBCL , diffuse large B-cell lymphoma; HCT , hematopoietic cell transplantation.
| Author/Year Design | N | Median Age (Range) | Conditioning | aGVHD (Grade 2–4) | NRM | EFS/PFS | OS | Comments |
|---|---|---|---|---|---|---|---|---|
| Dreger (2019) Registry | 1438 | 58 (20–75) ⁎ | RIC 100% | 34% ⁎ | 3-year 22% ⁎ | 3-year 38% ⁎ | 3-year 46% ⁎ | Haploidentical vs. MRD/MUD w/w/o TCD |
|
48 |
|
|
17% | 4-year 32% | 4-year 48% | 4-year 47% | Improved OS in chemosensitive patients |
|
68 |
|
|
39% | 1-year 23% | 2-year 44% | 2-year 49% | Improved outcomes in CR patients at allo-HCT |
|
79 |
|
|
100-day 42% | 5-year 45% | 5-year 22% | 5-year 22% | All matched sibling donors. Upfront allo-HCT |
|
101 |
|
|
33% | 3-year 28% | 3-year 42% | 3-year 52% |
|
|
396 |
|
|
|
|
|
|
Lower NRM with RIC or NMAC, with higher risk of lymphoma progression |
|
503 |
|
|
|
|
|
|
All patients had prior auto-HCT. MAC associated with worse OS |
While keeping in perspective the recent advances in the treatment of relapsed DLBCL, consolidation with autologous HCT is still considered standard of care for relapsed DLBCL patients responding to salvage chemotherapies. Upfront consolidation with autologous HCT is not recommended routinely, with the possible exception of DHL patients treated with R-CHOP in first-line setting.
Notwithstanding the risk of toxicities and availability of a donor, allo-HCT is considered for those DLBCL patients relapsing after autologous HCT and where CAR T-cell therapies are not available. Advanced age alone, in otherwise healthy individuals, is no longer a contraindication for HCT, particularly when using RIC or nonmyeloablative regimens. A currently enrolling Blood and Marrow Transplant Clinical Trials Network (BMT CTN) clinical trial (NCT03992352) uses a composite health assessment model to predict NRM in older patients undergoing allo-HCT. If successful, this tool will help identify appropriate older patients for allo-HCT and may further improve clinical outcomes.
Upfront allogeneic HCT is only rarely considered in the young, high-risk patient with aggressive DLBCL (double-hit, double-expressors) in the appropriate clinical setting, though this is not a standard approach.
FL is the second most common type of NHL accounting for 22% of the cases. The median survival of patients with FL ranges from 8 to 12 years, but there is considerable variability given the heterogeneous presentation and not infrequent development of resistance or transformation to a more aggressive histology. Therefore management strategies vary from surveillance to immunotherapy, combination chemoimmunotherapy, radioimmunotherapy, as well as autologous or allogeneic HCT. Front-line therapies utilizing rituximab or radioimmunoconjugates have dramatically improved the results for this indolent lymphoma, but relapses are still common.
A 63-year-old man without any constitutional symptoms, presents with palpable lymphadenopathy in his left axilla. PET/CT reveals enlarged lymph nodes in bilaterally axilla, the largest measuring 2.5 cm x 3 cm, as well as enlarged inguinal lymph nodes bilaterally, largest measuring 3.5 cm × 3 cm. Biopsy of left inguinal lymph node reveals small follicular pattern of cells staining positive for CD20, BCL2, CD10. Patient is diagnosed with grade 1 FL.
The role of autologous HCT as consolidation therapy for frontline treatment of FL has been evaluated in several key clinical trials ( Table 24.3 ). Unfortunately, lack of prospective randomized trials comparing novel immunotherapies with autologous HCT makes clinical practice variable for this indolent lymphoma, and most patients will often face multiple different lines of chemo- and/or immunotherapies before receiving an HCT. Moreover, while autologous HCT in indolent lymphomas has low treatment-related mortality (TRM), there is a risk of disease relapse and concerns for long-term toxicities like second malignancies. In the German (German Low Grade Lymphoma Study Group [GLSG]) and two French ( Groupe d’Etude des Lymphomes de l’Adulte [GELA], & Groupe Ouest-Est des Leucémies et Autres Maladies du Sang [GOELAMS]) cooperative group studies; advanced-stage FL patients in first remission were randomized to receive either consolidation with auto-HCT or interferon maintenance. Autologous HCT improved PFS in all expect the GELA study as seen in Table 24.3 . No study reported an OS benefit, and more disturbing was the increased risk of therapy-related neoplasms (secondary myelodysplastic syndrome [sMDS]/acute myeloid leukemia [AML]) in the autologous HCT group. To note, these three trials were conducted in the prerituximab era, therefore the relevance to contemporary times is unclear.
| Study (Year) | No. | TRM Auto vs. Chemotherapy | EFS/PFSAuto vs. Chemotherapy (years) | OSAuto vs. Chemotherapy (years) | Comments |
|---|---|---|---|---|---|
|
307 | < 2.5% in both arms |
|
|
Significantly more sMDS/AML with Auto (3.5% * vs. 0%; P = .02) |
|
402 | Not reported |
|
|
Second malignancies similar in both groups; 14 with chemotherapy and 11 Auto |
|
166 | Not reported |
|
|
Auto associated with significantly more second malignancies (n = 12 * vs. 1; P = .01) |
|
136 |
|
|
|
4–year sMDS/AML was higher with HDT (6.6% vs. 1.7%) |
The Gruppo Italiano Trapianto di Midollo Osseo/Intergruppo Italiano Linfomi (GITMO/IIL) trial addresses the role of upfront autologous HCT in the rituximab era comparing chemoimmunotherapy with R-CHOP to rituximab supplemented dose intense therapy and autologous HCT. Like the trials of the prerituximab era, this study showed improved PFS toward autologous HCT without OS benefit (see Table 24.3 ). Thus, based on results of the studies mentioned, autologous HCT is not recommended as consolidation in FL patients achieving first remission.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here