Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
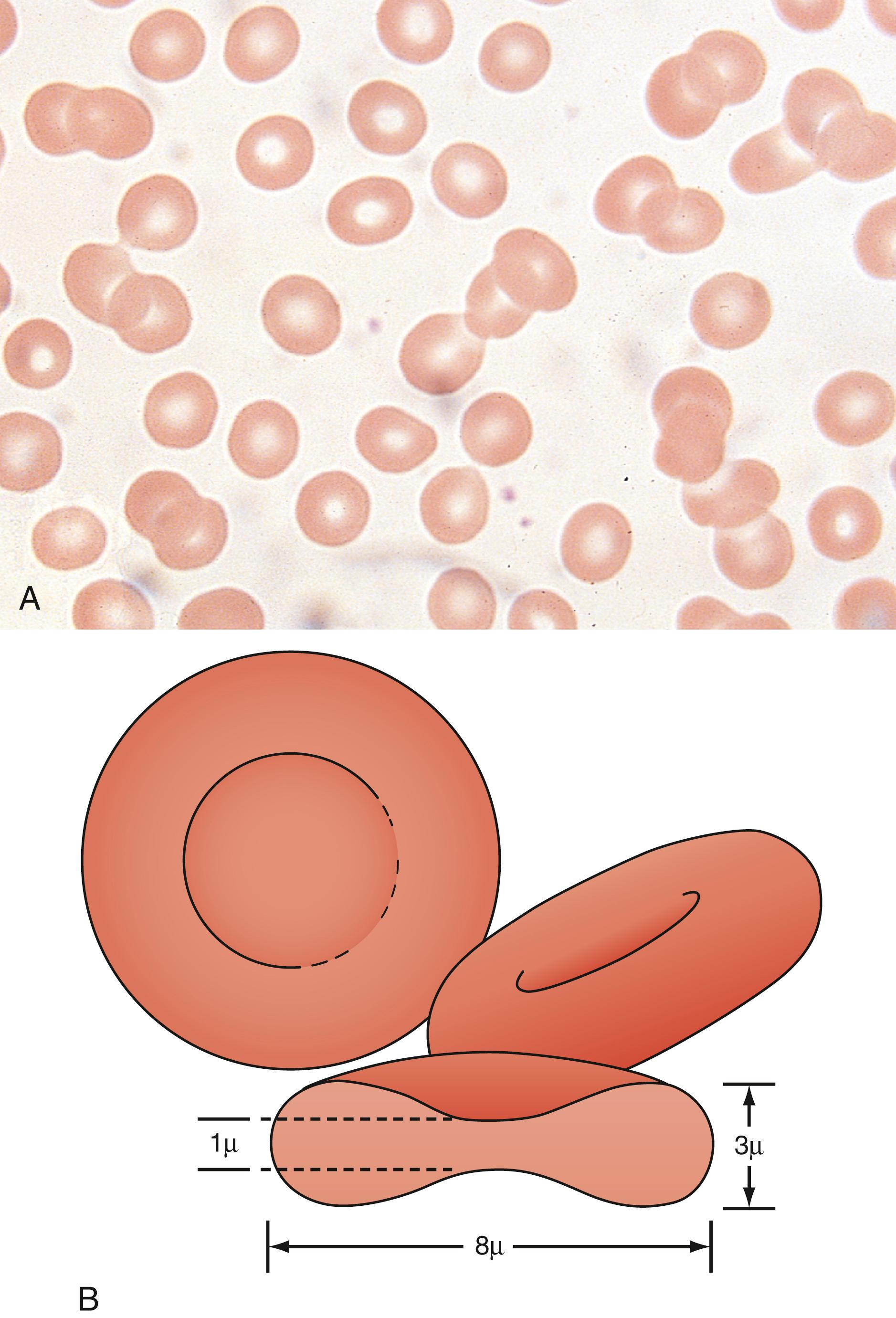
The red blood cell (RBC) is classically described as a biconcave disk. Red cells evolve from immature forms, beginning with pronormoblasts, which are large cells with dark blue cytoplasm and a large centrally located nucleus. As the cell matures, the nucleus becomes pyknotic and is eventually extruded from the cell completely. As the cell begins to make hemoglobin, the cytoplasm undergoes a gradual transformation from dark blue to pink histologically. A peripheral smear is made up of a small droplet of blood on a glass slide. A second slide then is used to smear the blood in a manner that it covers one-half to two-thirds the length of the slide. A well-made smear should not have lines or holes, nor should it reach either end of the slide completely. Commonly, Wright stain is applied after drying to give the cells their characteristic appearance ( Fig. 12.1 ).
A normal RBC has approximately the same size as a lymphocyte nucleus. RBCs should be the same relative shape and size, with an area of central pallor that is approximately one-third the diameter of the cell ( Fig. 12.2 ). A variety of terms are available to describe the physical appearance of mature RBCs. Polychromasia refers to the variability in cell color, with hypochromia specifically indicating an increased area of central pallor. Anisocytosis indicates variability in cell size. Microcytosis describes a small cell size, whereas macrocytosis refers to large cells. Poikilocytosis represents variability in cell shape. The overall number of RBCs in the circulation cannot be described reliably by the peripheral smear. Laboratory values can also aid in assessing RBC morphology. Two of these have wide usage. The mean corpuscular volume (MCV) is a laboratory measure of cell size, and red cell distribution width (RDW) estimates this difference.
The primary function of the RBC is to deliver oxygen to tissues. The effective red cell number commonly is represented by the hemoglobin concentration, in grams per deciliter (g/dL). The oxygen-binding sites of hemoglobin are completely saturated by passage of the RBC through the lungs. As the cells circulate through the systemic capillaries, the hemoglobin releases 25% of its bound oxygen to the tissues. However, the amount of oxygen released to the tissue may be significantly increased, under certain conditions (fever, acidosis, etc.), allowing some compensation for a decrease in hemoglobin. A progressive decrease in hemoglobin eventually leads to tissue hypoxia, which triggers a release of erythropoietin. This induces an increase in RBC production, which brings RBC mass back to normal values.
Different stages of life and disease states impart different oxygen demands, and therefore hemoglobin levels can vary greatly. In general, full-term, newborns are born with relatively high levels of hemoglobin which then fall over the first few months of life. This low point, the physiologic nadir, occurs during the transition in production from fetal to adult hemoglobin. Premature infants may have an earlier, more severe, and more prolonged nadir depending on gestational age. After this time, the average hemoglobin value increases until preadolescence, and reaches adult levels by puberty. Similarly, the MCV is at a low point after the nadir of hemoglobin, and it slowly increases into early adolescence. An estimate of the lower limit of the MCV is 70 femtoliters (fL) plus two times the age in years, from 1 to 10 years old.
Anemia is the most common abnormality of RBCs, and it is identified as a decrease in hemoglobin level. At any given age, anemia is defined practically as a value greater than 2 standard deviations, below the mean ( Table 12.1 ). However, physiologically, anemia is best defined as a hemoglobin level that is too low to meet tissue oxygen demands. Recognition that a child’s hemoglobin level is abnormal requires knowledge of age, gender, and race-related normal values. Anemia occurs as a result of one of the following: (1) decreased bone marrow production of RBCs, and reticulocytes ( Fig. 12.3 ), (2) increased destruction of mature RBCs peripherally or as precursors in the bone marrow, or (3) loss of RBCs (hemorrhage). In some situations, a combination of these factors can occur.
| Age | Hemoglobin (g/dL) | Mean Corpuscular Volume (fL) | ||
|---|---|---|---|---|
| Mean | –2 Standard Deviation a | Mean | –2 Standard Deviation a | |
| Birth (cord blood) | 16.5 | 13.5 | 108 | 98 |
| 1–3 days old (capillary) | 18.5 | 14.5 | 108 | 95 |
| 1 weeks old | 17.5 | 13.5 | 107 | 88 |
| 2 weeks old | 16.5 | 12.5 | 105 | 86 |
| 1 month old | 14.0 | 10.0 | 104 | 85 |
| 2 months old | 11.5 | 9.0 | 96 | 77 |
| 3–6 months old | 11.5 | 9.5 | 91 | 74 |
| 0.5–2 years old | 12.0 | 10.5 | 78 | 70 |
| 2–6 years old | 12.5 | 11.5 | 81 | 75 |
| 6–12 years old | 13.5 | 11.5 | 86 | 77 |
| 12–18 years old | ||||
|
14.0 | 12.0 | 90 | 78 |
|
14.5 | 13.0 | 88 | 78 |
| 18–49 years old | ||||
|
14.0 | 12.0 | 90 | 80 |
|
15.5 | 13.5 | 90 | 80 |
a Anemia is defined as a value greater than 2 standard deviations below the mean.
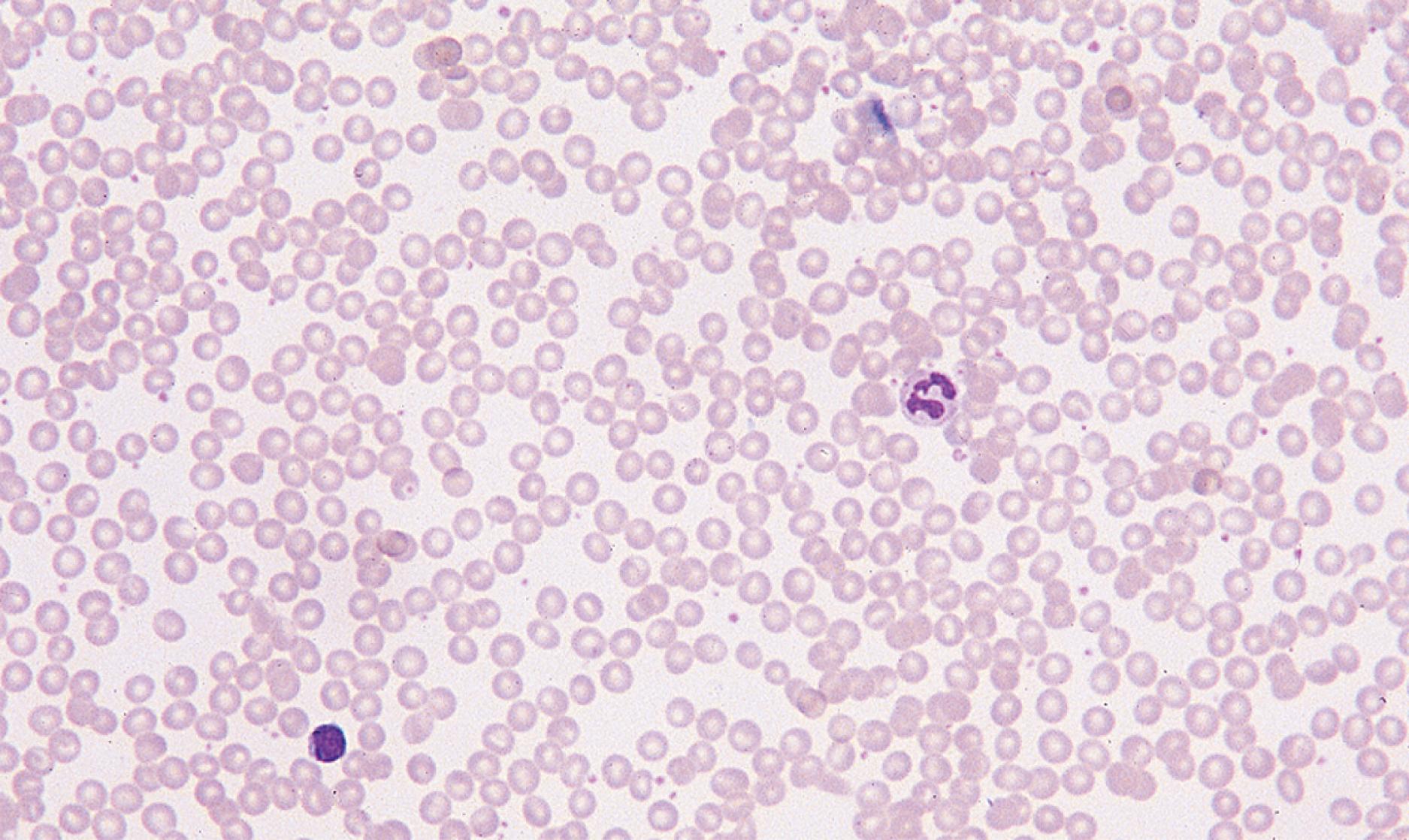
Physical examination of the anemic child may reveal pallor, although in the fair-skinned or dark-skinned child, it may be easily missed, unless palmar creases or conjunctivae are also examined thoroughly ( Fig. 12.4 ). Vital signs may be normal, but tachycardia may be present. Severe anemia from any cause may elicit symptoms of fatigue; decreased appetite; headache; and, in extreme cases, shock, congestive heart failure, or even stroke.
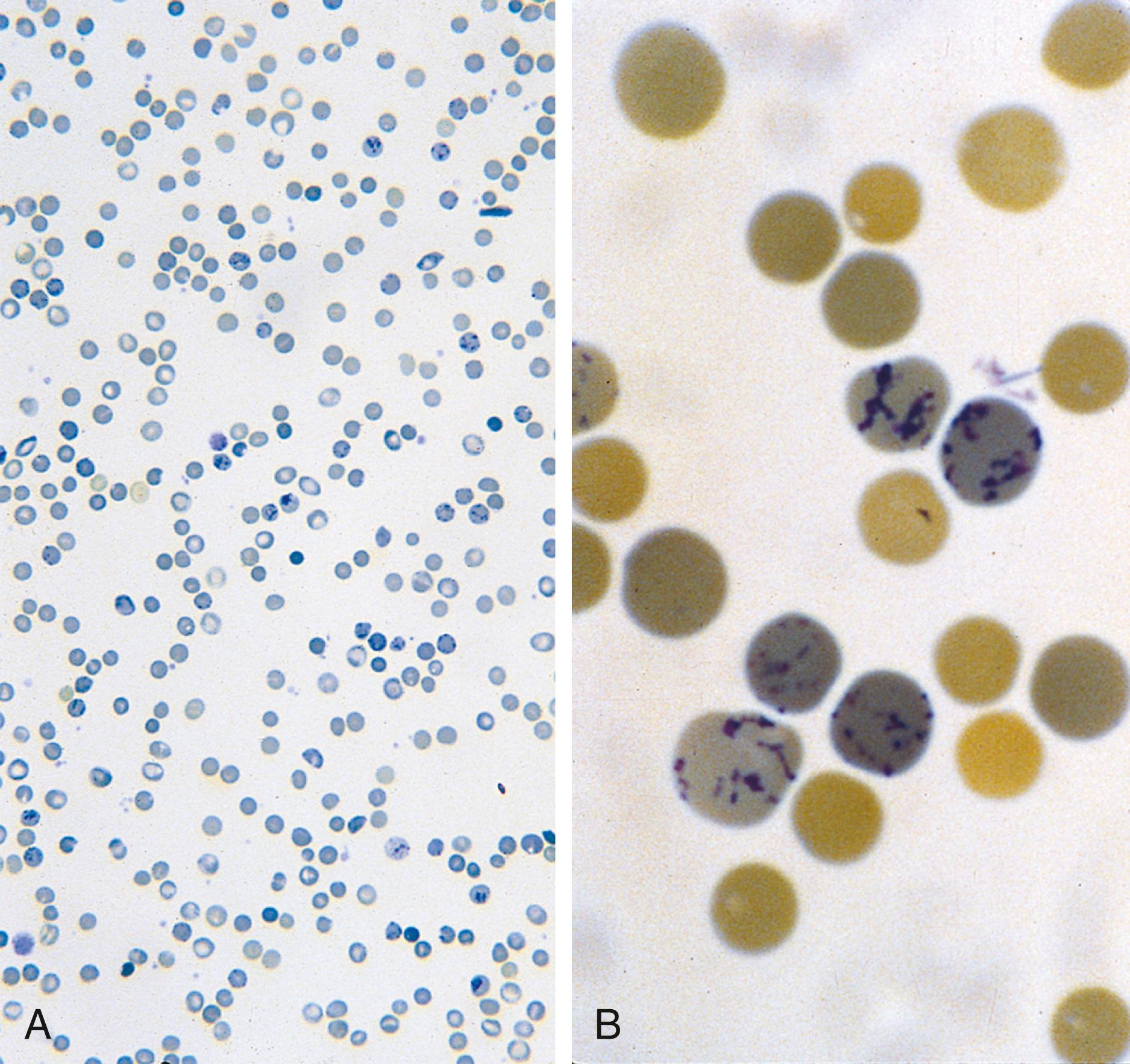
The most common anemia encountered in pediatrics is microcytic anemia, due to iron deficiency. A lack of iron leads to failure of hemoglobin production. Iron deficiency, however, is only a laboratory finding, not a diagnosis. The causes of iron deficiency must be elucidated through a careful history and physical examination. Although poor nutrition is the most common cause, other causes such as hemorrhage or malabsorption must also be considered. Because the normal full-term infant has adequate iron reserve to accommodate the increasing RBC mass through the first 5 months of life, iron deficiency is usually not seen until the second half of the first year of life. It is detected most often in the 10- to 18-month-old child who ingests large volumes of cow’s milk. Whole cow’s milk not only is deficient in dietary iron but often leads to enteropathy with occult gastrointestinal blood loss, exacerbating the child’s iron-deficient status. A second peak of iron deficiency anemia is seen during adolescence in girls, due to nutrition and loss of RBCs.
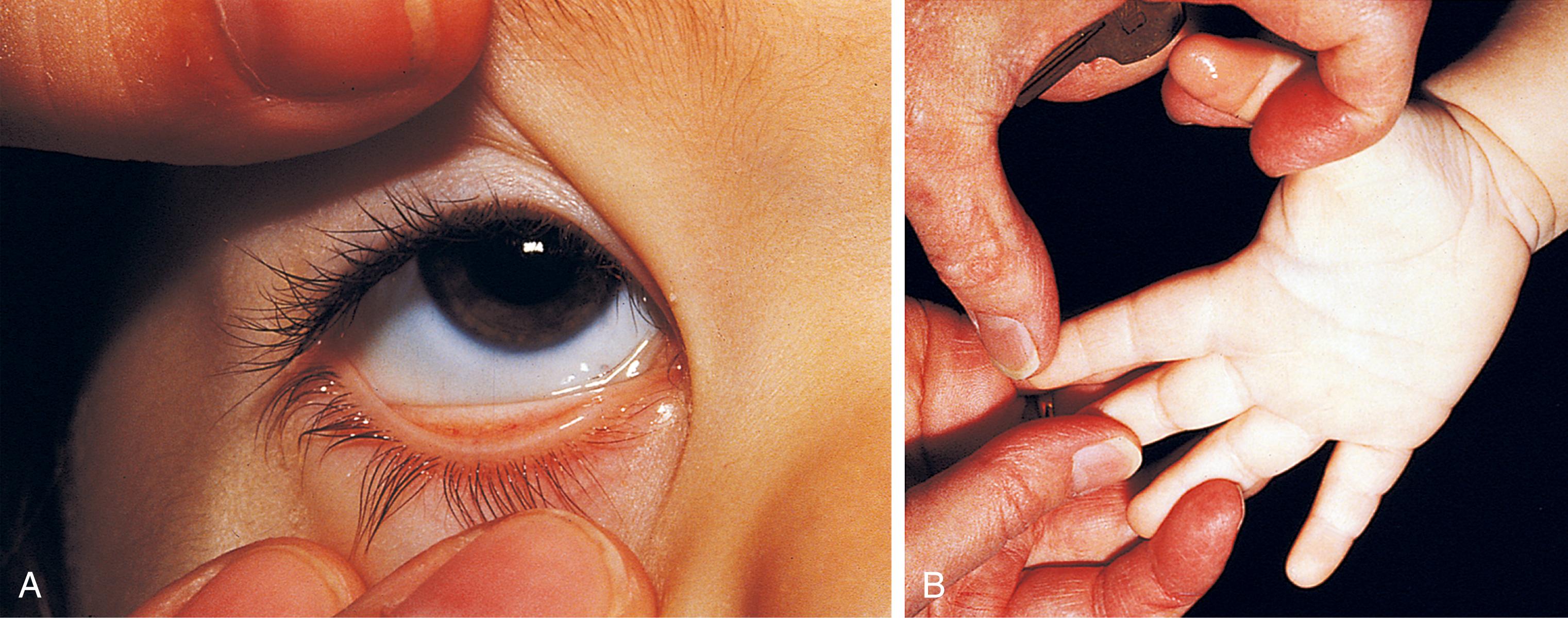
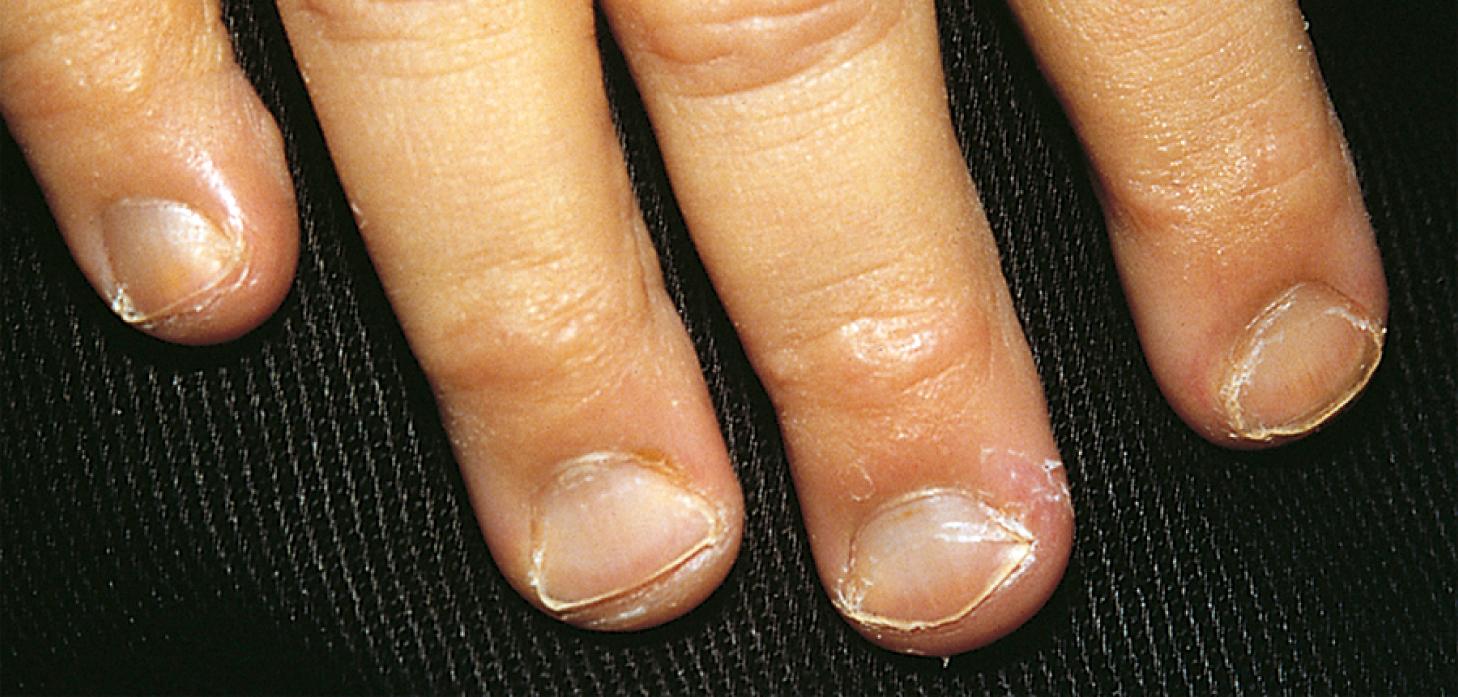
Affected children may manifest a peculiar physical finding termed koilonychia, or “spooning” of fingernails and toenails ( Fig. 12.5 ). Glossitis also may be observed in iron deficiency. The RBCs of iron deficiency anemia are microcytic and hypochromic. The number of platelets often is increased (particularly when an enteropathic condition is present), although it may be normal or even decreased. In severe iron deficiency anemia, anisocytosis and poikilocytosis may be prominent. Nonspecific abnormalities of RBC morphology (such as the presence of micro-ovalocytes or basophilic stippling) may also occur. Bizarre RBC shapes are due to a three-dimensional change in structure when viewed in a two-dimensional plane of the light microscope. When viewed in two dimensions, the cells appear ovoid. Fig. 12.6 shows a peripheral blood smear of a child with iron deficiency anemia.
The history and physical examination, blood count, and review of the peripheral blood smear may be sufficient for diagnosis in many instances of hypochromic, microcytic anemia. If the evaluation before obtaining a more specific test strongly suggests iron deficiency, a therapeutic trial of iron may be a reasonable approach. However, additional laboratory studies are often indicated. Importantly, normal values of ferritin, serum iron, total iron binding capacity (TIBC), and transferrin saturation demonstrate age-related variation. Serum iron and TIBC alone may not give an accurate perspective of total body iron stores, as they are relatively short-term measures, and are affected by recent intake. Ferritin is a superior measure of long-term iron stores, but it also is an acute-phase reactant and may be elevated into the low normal range in a child with a concurrent inflammatory process. However, if ferritin is low, it is diagnostic of iron deficiency.
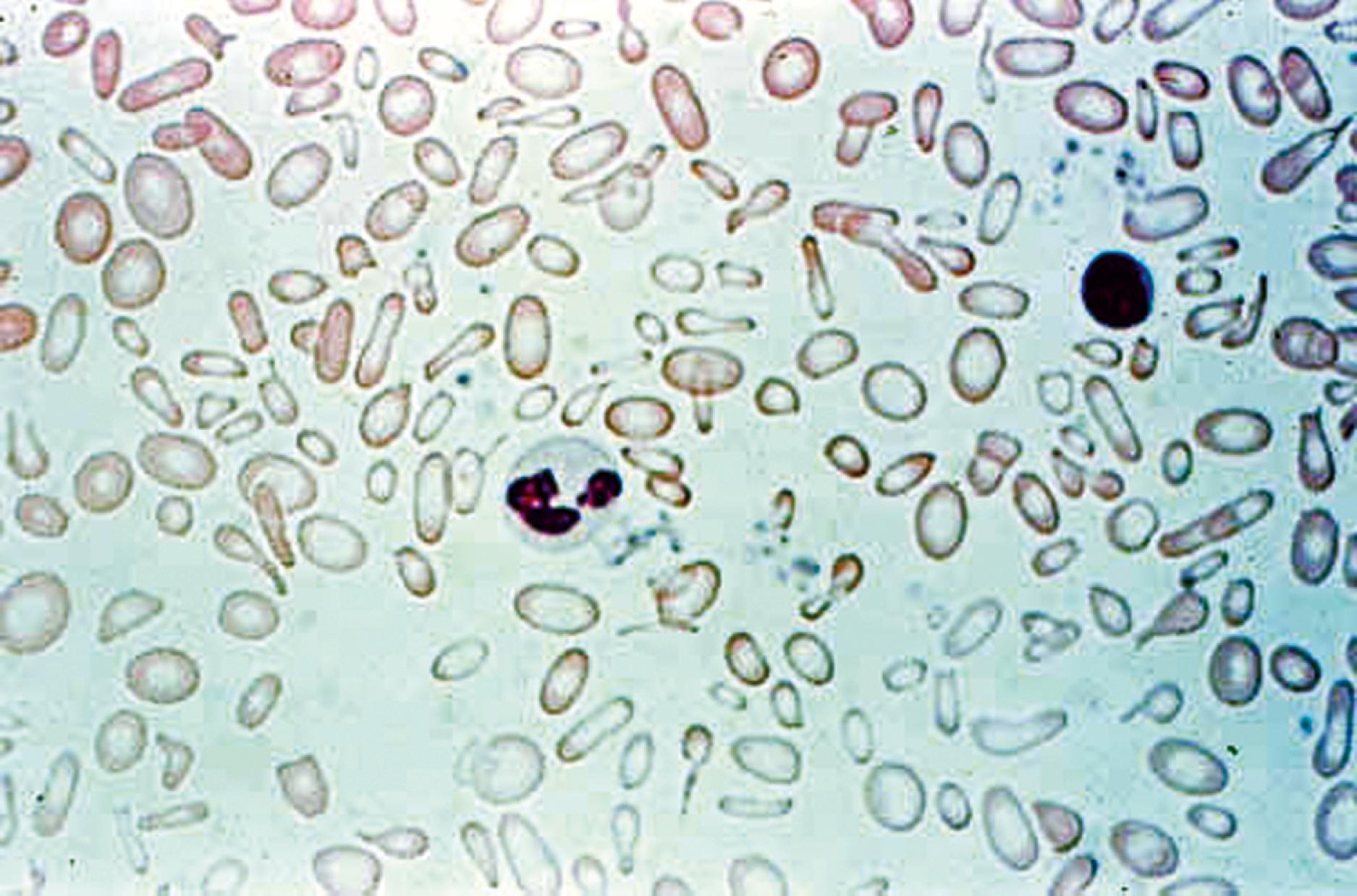
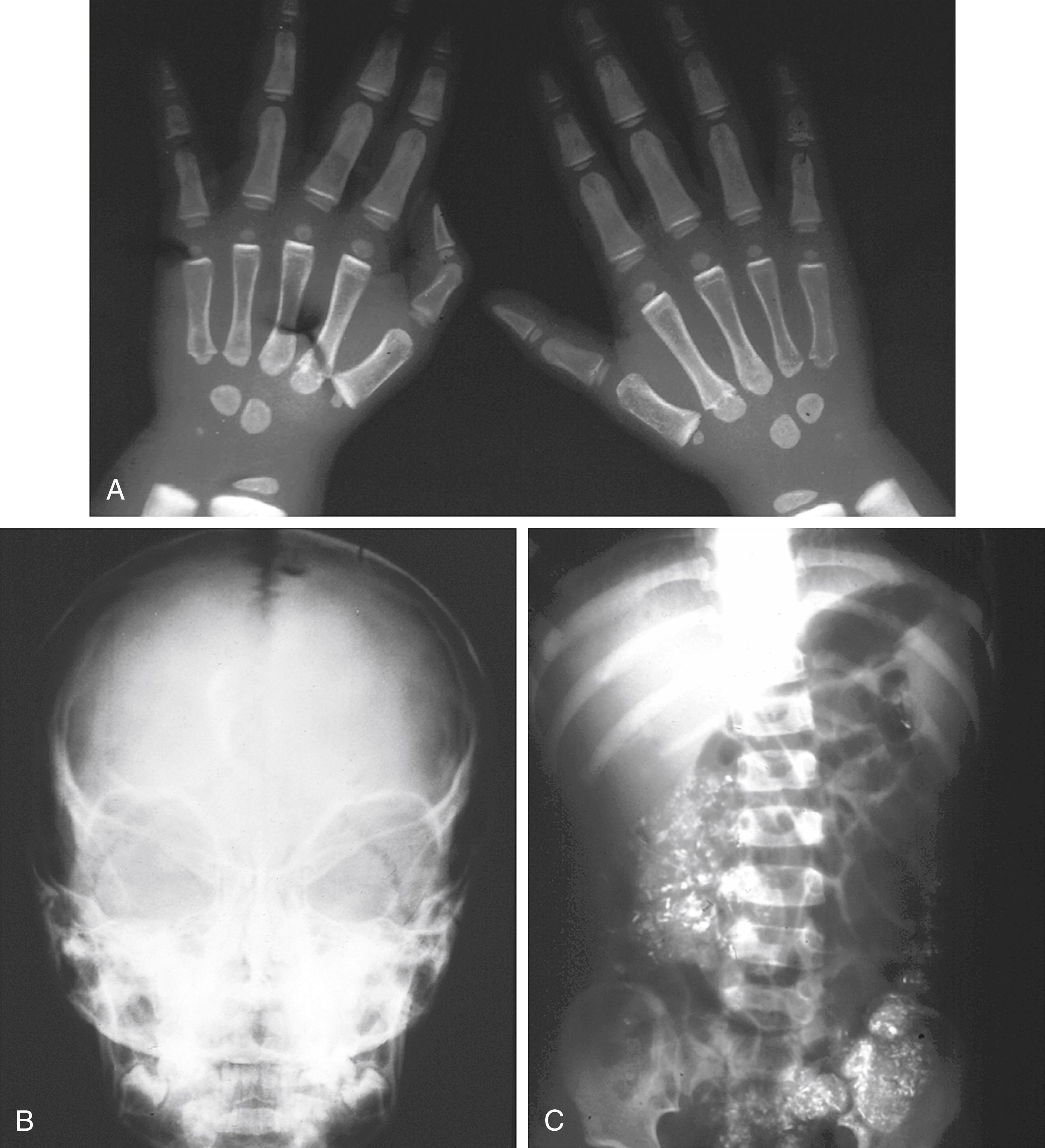
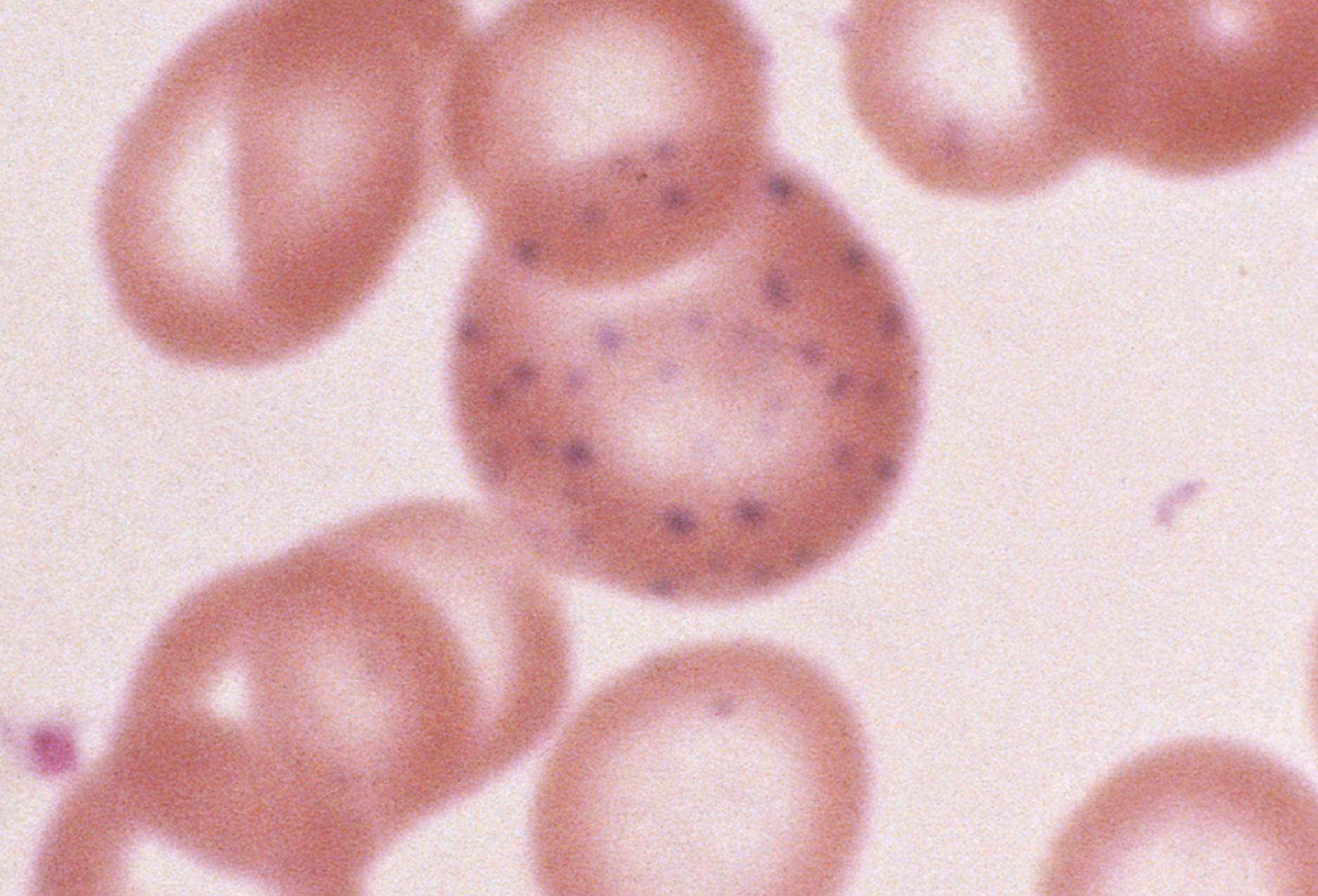
The coexistence of iron deficiency and lead intoxication is well documented; though in practice, an increased lead level more commonly is secondary to pica in children with iron deficiency, and it is not the cause of anemia. Pica is the ingestion of non-food items, such as chalk, carpet fibers, or other debris. In primary lead intoxication, nonhematologic manifestations, particularly neurologic complications, appear long before an anemia develops. Significant radiographic changes are also seen in primary lead intoxication ( Fig. 12.7 ). When they do occur, hematologic abnormalities of lead intoxication such as basophilic stippling ( Fig. 12.8 ) are a direct result of the effect of lead on several cellular enzymes involved in heme production. Lead inhibits these enzyme systems, impairing iron use and globin synthesis.
Inflammatory diseases, such as chronic infection, inflammatory bowel disease, or rheumatologic conditions, may lead to microcytic anemia. Although the symptoms of the child’s primary illness usually clarify the diagnosis, there are occasionally cases in which a chronic subclinical infection (particularly of the urinary tract) may go undiagnosed. Clinical factors often can make this diagnosis, although one laboratory study that may be of value is the serum ferritin determination. Ferritin is decreased in iron deficiency, whereas in chronic inflammatory states, it is usually elevated. Also, though serum iron can be low in both iron deficiency and anemia of chronic disease, TIBC is increased in iron deficiency and decreased in anemia of chronic disease. These changes are felt to be due to the upregulation of the enzyme hepcidin, which redirects total body iron to storage pools rather than RBC production.
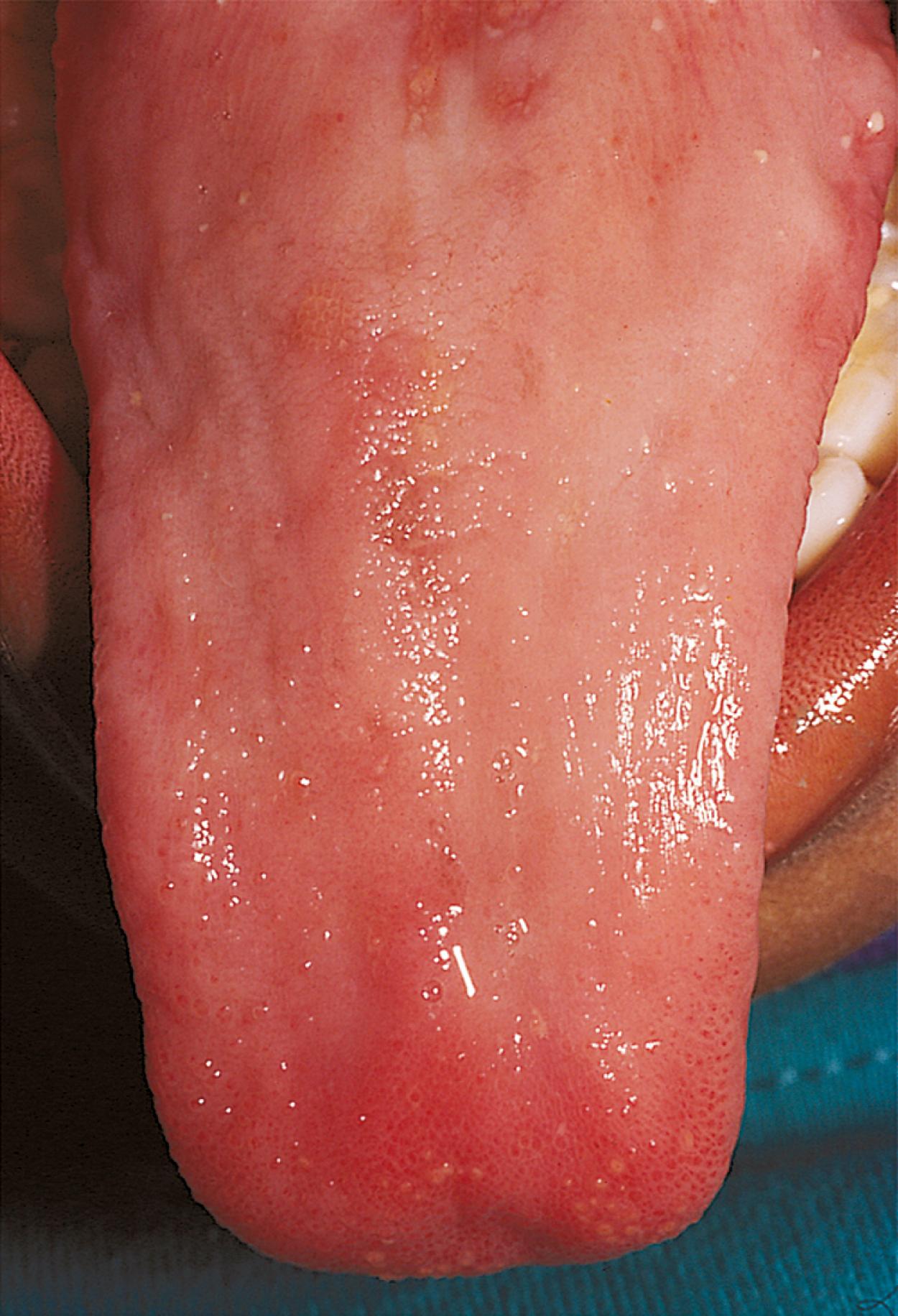
Although the etiology of macrocytic anemias varies, common morphologic abnormalities of erythropoietic cells exist. In the bone marrow, the hallmark is the megaloblast, a nucleated RBC with a lacy chromatin pattern and asynchrony of maturation between cytoplasm and nucleus. The morphologic alterations are a direct result of decreased nuclear protein deoxyribonucleic acid (DNA) synthesis compared with cytoplasmic protein synthesis, which stems from a relative decrease in the factors needed in DNA replication, namely folate and cobalamin (vitamin B 12 ). Laboratory diagnosis requires the measurement of folate and vitamin B 12 levels in the serum and RBCs. Glossitis ( Fig. 12.9 ) or angular stomatitis, commonly seen in vitamin B 12 deficiency, can be helpful during physical findings in the diagnostic process.
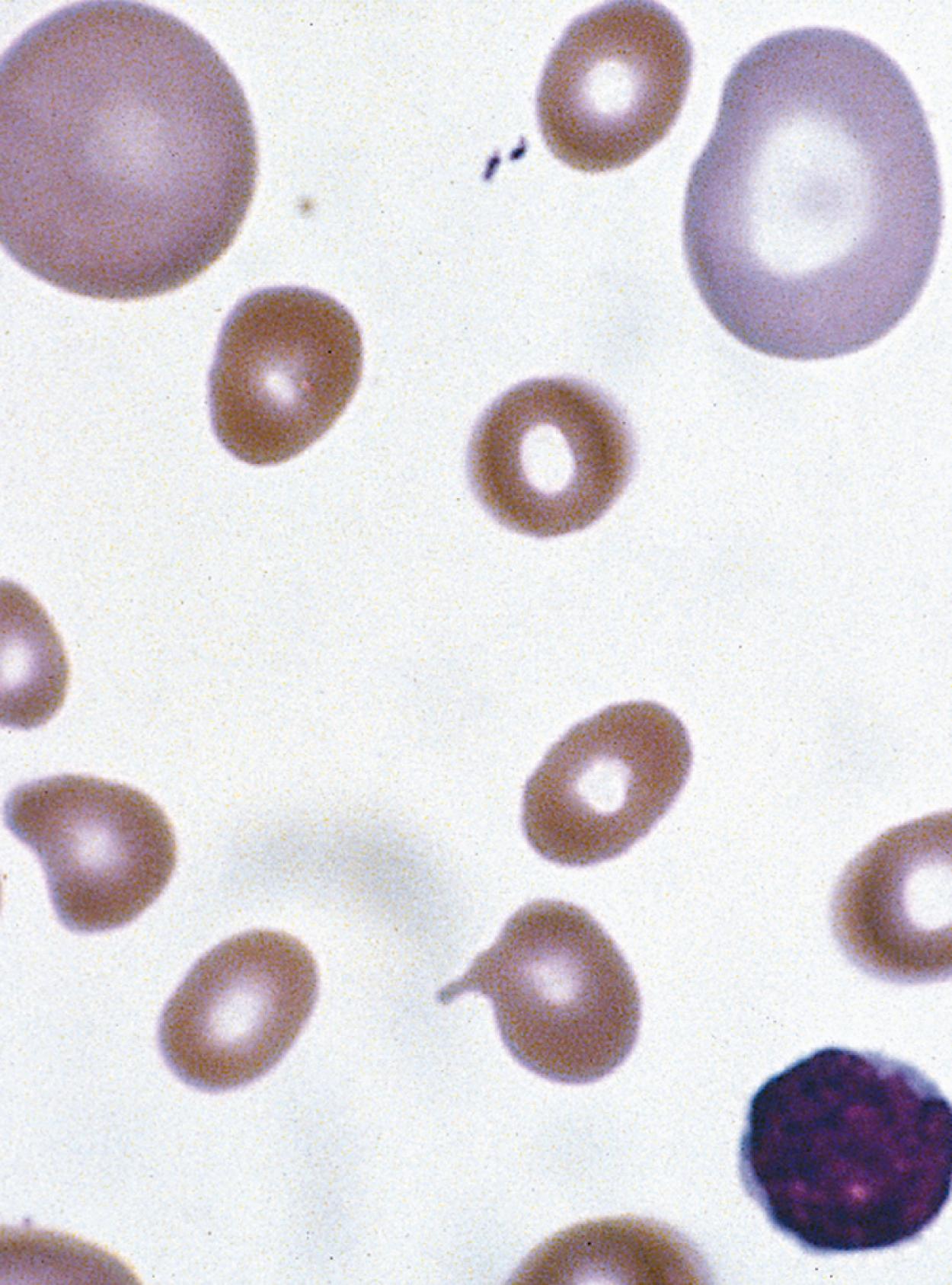
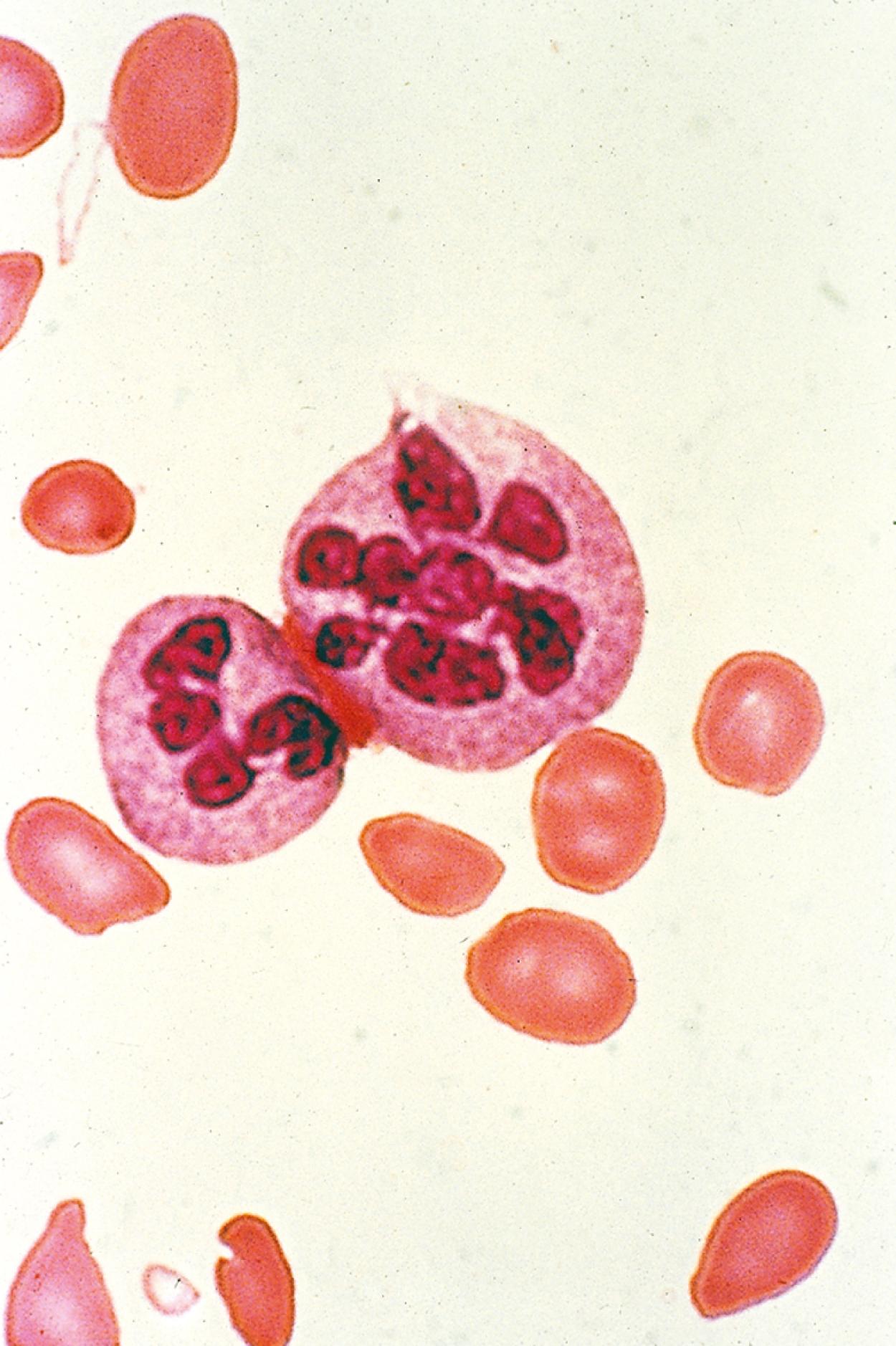
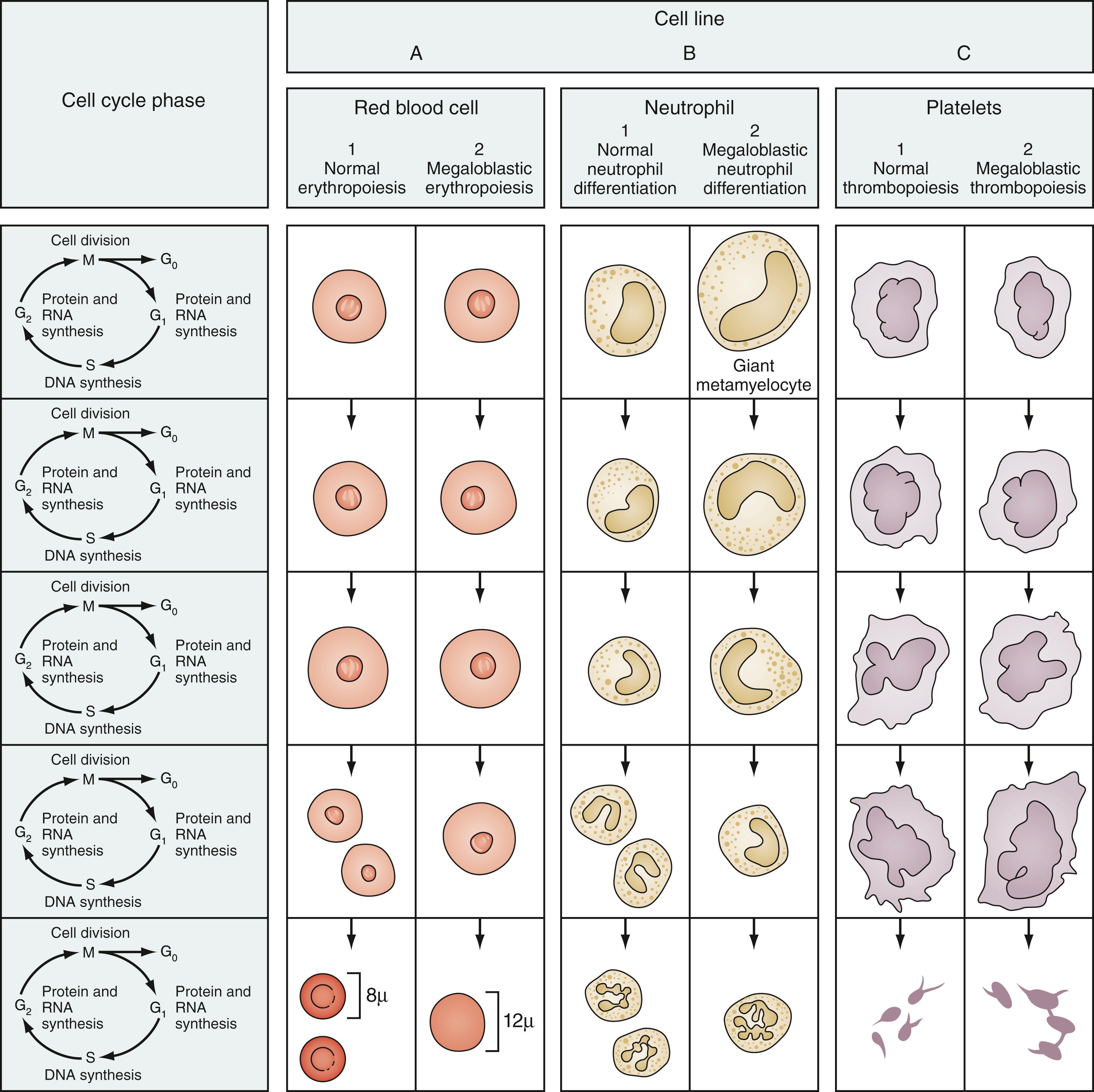
On the peripheral blood smear, the RBCs are large, and display a great deal of variation in their shapes ( Fig. 12.10 ). These macrocytic cells are generally normochromic. In addition, actively dividing marrow cells may become involved in the pathologic process. Neutrophils are the second most likely cells to display morphologic abnormalities. These cells become large and show pathognomonic hypersegmentation of the nucleus ( Fig. 12.11 ). Neutropenia is common. The more severe and prolonged megaloblastic anemias ultimately may lead to moderate thrombocytopenia, with large, bizarre platelets on the peripheral blood smear. Megaloblastic changes in each cell line are shown in Fig. 12.12 .
Because a healthy diet is unlikely to lead to folate or vitamin B 12 deficiency, a low level of either should raise questions of altered bioavailability or unusual diet. Goat milk is folate deficient, and an infant on a goat milk diet may become folate depleted over time. Various drugs that decrease folate absorption (phenytoin) or interfere with folate metabolism (methotrexate) may also lead to megaloblastic changes. A pathologic condition of the gastrointestinal tract may cause malabsorption of folate or vitamin B 12 . This condition may be iatrogenic if a portion of the intestines is removed for medical reasons. Pernicious anemia is a specific cause, due to deficiency in intrinsic factor, which is required for vitamin B 12 absorption. Elevated levels of vitamin B 12 are generally not indicative of a problem in children, though there are exceptions.
Acquired forms of pediatric pure red cell aplasias (PRCAs) are rare, although well recognized. Transient erythroblastopenia of childhood (TEC) occurs in 1- to 4-year-old children and appears 2 weeks to 2 months after a respiratory or gastrointestinal illness. Fortunately, TEC is transient and self-limited. It has been noted that the RBCs in congenital PRCA have fetal characteristics, but the RBCs of TEC have age-appropriate characteristics ( Table 12.2 ). The cause of TEC remains obscure. TEC may occur after many different viral infections and most likely represents an altered immunity from the infection that affects erythropoiesis. Treatment is supportive and determined by the degree of symptoms. Non–TEC-acquired PRCA is rare in pediatrics, although these can result from infectious suppression of marrow precursors.
| Red Blood Cell Characteristic | Disease | |
|---|---|---|
| Pure Red Cell Anemia | Transient Erythroblastopenia of Childhood | |
| Hemoglobin | Increased fetal | Normal fetal |
| Mean corpuscular volume | Increased | Normal |
| Red blood cell enzyme activity | Normal or high | Low |
Hemolytic anemias are defined by the premature destruction of RBCs. This can occur by mechanical, infectious, enzymatic, or immune-mediated mechanisms. Symptoms and severity vary not only among individuals, but also between diagnoses. Jaundice, especially scleral icterus, splenomegaly, and dark urine are common presentations during hemolytic episodes. Although all hemolytic anemias may have acute exacerbations, some also feature in more chronic phases of red cell breakdown. The history of individual patients, laboratory studies, and blood cell morphology are important factors in making the accurate diagnosis. Some of the most common morphologic features are described in Box 12.1 . Treatments can vary, and splenectomy may be a consideration. However, splenectomy in children is fraught with infectious, vascular, and thrombotic complications.
a Nonhemolytic disorders of similar morphology are enclosed in parentheses for reference.
RBC fragmentation syndromes (microangiopathic and macroangiopathic hemolytic anemias)
Hereditary elliptocytosis in neonates
Hereditary elliptocytosis
Thalassemia
(Other hypochromic microcytic anemia)
(Megaloblastic anemia)
Acute hepatic necrosis (spur-cell anemia)
Uremia
Abetalipoproteinemia
Thalassemia
Unstable hemoglobin levels
Lead poisoning c
c Disease sometimes associated with this morphology.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here