Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The lymphomas are a diverse group of hematologic neoplasms arising primarily from lymph nodes. They vary widely in affected age group, clinical course, and prognosis. Hodgkin lymphoma (HL) was first described by Thomas Hodgkin in 1832, and since the early 1990s advances in therapy have been so great that HL is now curable in most patients. The non-Hodgkin lymphomas (NHLs) are far more variable in their clinical course and may be rapidly fatal. As in HL, improvements in survival are largely attributable to advances in therapy, but modern cross-sectional and functional imaging studies have also had significant impact. The accurate delineation of disease extent and identification of risk factors facilitate optimal, individualized, risk-adapted treatment with resultant survival benefits. Accurate imaging is crucial in choosing the most appropriate treatment at the time of diagnosis and staging, in monitoring response to therapy, and in detecting recurrence.
The objectives of initial staging are twofold: to define the local extent of clinically overt disease and to seek for occult disease elsewhere, in the knowledge of the likely pattern of tumor spread. Initial imaging should identify adverse prognostic features and factors that may influence delivery of therapy (e.g., central venous occlusion). Choice of the most appropriate imaging method requires an appreciation of the likelihood of particular sites being affected, the accuracy of specific tests chosen to investigate those sites, and the likely impact of a positive result on management.
Lymphoma is the fifth commonest malignancy in the Western world and in the United States. Within the United States, NHL annually accounts for 5% of new cancers in men and 4% of new cancers in women. It is estimated that there will be 81,560 new cases of NHL and 20,720 deaths in 2021. HL accounts for approximately 15% of all lymphomas. It is estimated that there will be 8,830 new cases in 2021 and 1000 deaths. Men are affected somewhat more than women: for HL, the male-to-female ratio is 1.4:1; for NHL, it is 1.1:1.
The incidence of HL has been stable for years, but that of NHL rose by approximately 60% in the United States after 1960, although it now appears to be stabilizing. This is evident for all age groups, but particularly for older persons. NHL occurs more often in men and in White ethnic groups. The incidence of NHL varies geographically, being more common in the Northeast and Midwest, but the increased incidence has been observed in all geographic areas of the United States. The overall mortality for NHL has also risen over the last few decades, especially in older patients, reflecting the increased incidence, despite the fact that survival rates for each subtype of NHL have improved secondary to advances in treatment.
The reasons for the increasing incidence are only partly understood. Some may be artifactual, in that new lymphoma classifications have led to a diagnosis of NHL in patients who would previously have received other diagnoses, including HL, in up to 15% of cases. Much of the increase from the late 1980s resulted from the increased incidence of lymphomas associated with immune deficiency, notably secondary to human immunodeficiency virus (HIV) infection. However, the occurrence of some HIV-associated NHL has started to fall since the introduction of highly active combined antiretroviral therapy (cART).
HL has a bimodal peak distribution, with one peak in the third decade of life and a second, smaller peak in patients older than 50 years. NHLs are diseases mainly of older persons, with a median age at diagnosis of 65 years.
Lymphomas account for around 11% of pediatric malignancies, and HL accounts for nearly 50% of all pediatric lymphomas.
The incidence of non-Hodgkin lymphoma (NHL) has increased by 60% since the 1960s in the United States and the United Kingdom.
Hodgkin lymphoma (HL) has a peak incidence between the ages of 20 and 40 years, with a second peak in individuals older than 50 years.
NHLs are seen in children and in those older than 50 years.
There is a link between Epstein–Barr virus and lymphoma, and especially between Burkitt lymphoma and HL.
Infective agents are also associated with NHLs.
There is an association between Epstein–Barr virus (EBV) and HL, but the exact etiologic role of the virus is uncertain. Patients with HL have a higher titer of antibody to EBV viral capsular antigen, and the risk of HL among patients who have had infectious mononucleosis is trebled. EBV can also be found in malignant HL cells. Thus, it is classified by the International Agency for Research on Cancer as a cause of HL. Other infective agents such as HIV-1, associated with the mixed cellularity subtype of classic HL, are also implicated in the development of this disease. Infective agents are also associated with the development of certain subtypes of NHL. EBV is found in virtually 100% of cases of endemic African Burkitt lymphoma (BL); in the sporadic form, the incidence is 15% to 30%. The rare primary effusion lymphomas are associated with human herpesvirus–8 (HHV-8), as are some rare diffuse large B-cell lymphomas (DLBCLs). Helicobacter pylori infection is necessary for the development of gastric lymphoma of the mucosa-associated lymphoid tissue (MALT) type. Human T-cell lymphotrophic virus (HTLV-1) has a causal relationship with adult T-cell leukemia/lymphoma, as seen in South Japan and the Caribbean. The disease is believed to represent clonal expansion of HTLV-1–infected T lymphocytes. Additionally, Borrelia burgdorferi infection is associated with cutaneous MALT lymphoma.
Genetic factors have limited importance in the etiology of HL, with only approximately 5% of cases being familial, whereas familial aggregation of NHL is well recognized.
In NHL, immunosuppression is a very important etiologic factor. The incidence is greatly increased in patients with acquired immunodeficiency syndrome (AIDS) and in patients on long-term immunosuppressant therapy (e.g., following renal transplantation). Up to 25% of patients with congenital immunodeficiency syndromes including ataxia-telangiectasia, Wiskott–Aldrich syndrome, and X-linked immunodeficiencies will develop malignancies, of which lymphoproliferative disorders account for over 50%. Non–organ-specific autoimmune diseases (e.g., rheumatoid arthritis, systemic lupus erythematosus) are associated with HL and DLBCL, whereas organ-specific autoimmune diseases predispose to the development of MALT-type lymphomas within the affected organs (e.g., the thyroid and salivary glands). Finally, occupational exposure to certain chemicals such as pesticides and organic solvents may be a risk factor for the development of NHL.
There have been several pathologic classifications of lymphoma in the past; however, the functional anatomy of the lymph node is key to understanding the pathologic classification of lymphomas ( Fig. 30.1 ).
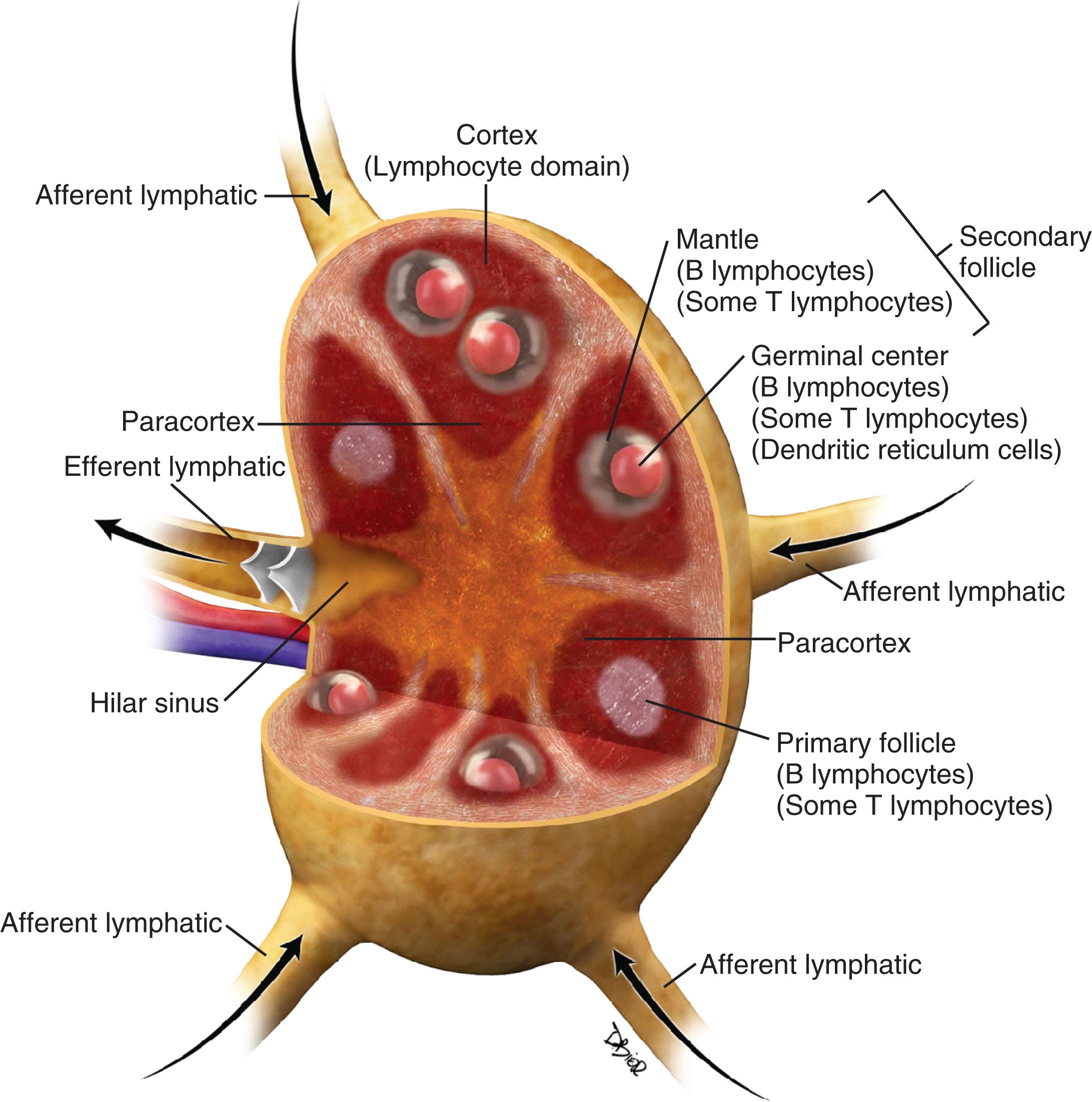
Over 90% of NHLs in the Western world are of B-cell origin. In general, those arising at stages of development within the germinal center of the node have a follicular or nodular architecture, whereas those arising outside the germinal center have a diffuse pattern. New insights into the pathogenesis of NHL, in terms of cellular and immunologic origins, resulted in the introduction of the Revised European American Classification of Lymphoid Neoplasms classification in 1994. This, in turn, led to the adoption of the definitive World Health Organization (WHO) Classification of Tumors of Hematopoietic and Lymphoid Tissues in 1995, which was most recently revised in 2016 ( Table 30.1 ).
| Precursor Lymphoid Neoplasms | |
|
|
| Mature B-Cell Neoplasms | Mature T-Cell and NK-cell Neoplasms |
|
|
| Histiocytic and Dendritic Cell Neoplasms | Hodgkin Lymphoma (HL) |
|
|
| Immunodeficiency-Associated Disorders | |
|
|
The WHO classification is a list of distinct disease entities defined by a combination of morphology, immunophenotype, and genetic and clinical features. It stratifies neoplasms according to myeloid, lymphoid, and histiocytic/dendritic cell lineages and recognizes three major categories of lymphoid neoplasms: B-cell, T-cell/natural killer cell, and HL. Precursor neoplasms, corresponding to the early stages of differentiation and including lymphoblastic leukemias and lymphomas, are separated from the more mature or peripheral neoplasms. For example, chronic lymphocytic leukemia (CLL) is the circulating form of small lymphocytic lymphoma, a mature B-cell neoplasm, and is classified as such.
This comprehensive approach has significantly improved the consistency of classification of lymphoma, such that, if given sufficient material, expert hematopathologists agree on classification of entities in over 95% of cases. Further refinements have drawn on findings from gene expression profiling and facilitate appropriate patient management. For example, gene expression profiling for DLBCL enables recognition of discrete subsets (germinal center B-cell type and activated B-cell type) with independent prognostic significance. Other changes include separation of monoclonal B-cell lymphocytosis into low-count and high-count variants, a new provisional entity of large B-cell lymphoma with IRF4 rearrangement, renaming of in situ follicular lymphoma (FL) to in situ follicular neoplasia, and reclassification of EBV-positive DLBCL in the elderly to “not otherwise specified (EBV+ DLBCL, NOS).” Histologic grade can also influence therapeutic decision-making. For example, FL is graded according to the number of centroblasts per high-power field, and grade III FL may require more intensive chemotherapy.
Investigators have demonstrated that HL is a true lymphoma; hence, the term Hodgkin lymphoma is preferred to “Hodgkin disease.” Indeed, the distinction between HL and NHL is not always straightforward, and composite cases occur. Diagnosis depends upon the demonstration of malignant Reed–Sternberg and Hodgkin cells against a background of nonneoplastic inflammatory cells. The WHO classification recognizes two distinct entities that differ in clinical features, behavior, morphology, and immunophenotype: classical HL (CHL, 95%) and nodular lymphocyte-predominant HL (NLPHL, 5%). CHL is, in turn, divided into four subgroups, based on the proportion of lymphocytes in relation to the number of malignant cells and on the connective tissue background ( Table 30.2 ). All share the same immunophenotype, with expression of CD30 by the malignant cells.
| HISTOLOGY | FREQUENCY (%) |
|---|---|
| Nodular lymphocyte-predominant Hodgkin lymphoma (HL) | 5 |
|
|
In nodular sclerosing HL, nodules of lymphoid tissue are separated by dense bands of collagen. This form of HL accounts for approximately 70% of CHLs and is the only form of HL without a male preponderance. Anterior mediastinal disease occurs in 80% of cases, and bulky disease in approximately 50%. Mixed cellularity HL accounts for 20% to 25% of CHLs and is more common in patients with HIV infection and in developing countries. Advanced-stage disease is common. EBV positivity is frequent. Lymphocyte-rich classic HL accounts for 5% of all HLs; 70% of cases are male, with a higher median age. Stage I or II peripheral nodal disease is typical. Lymphocyte-depleted CHL is the rarest subtype, accounting for less than 5% of cases; the median age at presentation is 35 to 40 years. It is associated with HIV infection and occurs more often in developing countries. Advanced-stage disease at presentation is common (70%), with frequent involvement of abdominal organs, retroperitoneal nodes, and bone marrow. Most cases of NLPHL were probably misclassified as lymphocyte-rich HL in the past. The malignant cells (called popcorn cells because of their striking nuclear convolutions) express CD20, not CD30. Affected patients are mostly male, aged 30 to 50 years, presenting with peripheral adenopathy affecting one or two nodal groups (axillary, cervical, or inguinal).
The WHO classification stratifies lymphoma according to myeloid, lymphoid, or histiocytic/dendritic lineage and provides distinction between disease entities.
HL comprises two distinct entities: CHL (95%) and NLPHL (5%).
CHL consists of four subtypes with differing presentations: nodular sclerosing (70%), mixed cellularity (20%–25%), lymphocyte-rich (5%), and lymphocyte-depleted (<5%).
Both HL and NHL are predominantly diseases of lymph nodes, which may present as a localized process involving a single nodal group or organ or as widely disseminated disease. However, there are recognizable differences in the clinical presentation of the two diseases ( Table 30.3 ).
| HODGKIN LYMPHOMA | NON-HODGKIN LYMPHOMA | ||
|---|---|---|---|
| Age | Young adults | More common ages 40–70 years | |
| B symptoms | 40% | 20% | |
| Spread | Generally contiguous nodal groups | Multiple remote nodal groups often involved | |
| Stage at presentation | >80% early stage I and II | >85% late stage III and IV | |
| Nodal groups |
|
|
|
| Extranodal disease |
|
|
|
Most patients with HL present with painless asymmetrical lymph node enlargement, which may be accompanied by sweats, fever, weight loss (“B” symptoms), and pruritus in approximately 40% of patients. B symptoms are more common in advanced-stage disease, and thus in the mixed cellularity and lymphocyte-depleted subtypes of CHL. Alcohol-induced pain is rare.
The cervical lymph nodes are affected in 60% to 80% of patients, the axillary nodes in 6% to 20%, and the inguinal/femoral nodes in 6% to 15%. Exclusive infradiaphragmatic lymphadenopathy occurs in less than 10% of patients at diagnosis. Splenomegaly is found on clinical examination in approximately 30% of patients. Some 80% of patients present with early-stage disease, and the incidence of splenic or bone marrow involvement is low, although it varies with the histologic subtype.
Although most patients with NHL present with nodal enlargement, extranodal disease is far more frequent than in HL, as is bone marrow involvement, with over 80% of patients having advanced-stage disease at diagnosis, depending on the subtype. Despite this, B symptoms are less frequent at diagnosis compared with HL, occurring in approximately 20% of patients.
So-called indolent lymphomas account for up to 35% of NHL, and lymphadenopathy may be intermittent. FL is typically indolent, accounting for 25% to 30% of all newly diagnosed NHL. It is usually diagnosed in the sixth decade. Conversely, DLBCL is termed aggressive, but generally responds well to therapy. DLBCL accounts for 35% of all NHL and is the most common B-cell NHL, usually presenting with rapidly enlarging lymph nodes.
The most aggressive subtypes include BL and lymphoblastic lymphoma; these are histologically characterized by very high proliferation fractions, and patients often exhibit rapidly progressive systemic disease that requires urgent treatment.
Most patients with Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL) present with painless enlargement of a group of lymph nodes.
In HL, most patients present with stage I or II disease; in NHL most patients have stage III or IV disease at diagnosis.
Systemic symptoms are twice as common in HL as in NHL.
Because lymphomas are primarily neoplasms of lymphoid tissues (whether nodal or extranodal), the tumor-node-metastasis (TNM) staging system is not appropriate. The Ann Arbor staging system for HL was introduced in 1970 and took into account the extent of nodal disease and the presence of extranodal extension. Increasing recognition of the influence of tumor bulk as an independent prognostic indicator within each stage and the routine application of computed tomography (CT) in the 1980s led to a modification of the classification in 1989, the Cotswolds classification. This system was routinely used until a few years ago. It was similar to the original Ann Arbor classification, but stage III was subdivided, and an additional qualifier “X” denoted bulky disease ( Table 30.4 ).
| STAGE | AREA OF INVOLVEMENT | |
|---|---|---|
| I | One lymph node region or extralymphatic site | |
| II | Two or more lymph node regions on the same side of the diaphragm | |
| III |
|
|
| IV | Extranodal sites beyond those designated “E” | |
| Additional qualifiers | A | No symptoms |
| B | Fever, sweats, weight loss (to 10% body weight) | |
| E | Involvement of a single extranodal site, contiguous in proximity to a known nodal site | |
| X | Bulky disease; mass >10 cm maximum dimension | |
| CS | Clinical stage | |
| PS | Pathologic stage; denoted by a subscript at a given site (M, marrow; H, liver; L, lung; O, bone; P, pleural; D, skin; S, spleen) | |
The prognosis of HL depends upon a number of factors, including:
Age. Older patients have a worse prognosis (for early-stage disease, 5-year survival is 45% in patients older than age 65 years, as opposed to over 90% in younger patients).
Tumor subtype. Mixed cellularity and lymphocyte-depleted HL have a poorer prognosis.
Raised erythrocyte sedimentation rate (ESR).
Multiple sites of involvement (more than three or four involved regions).
Bulky mediastinal disease.
Systemic B symptoms.
In 2014, the Cotswolds modification of the Ann Arbor system was replaced by the Lugano classification. Here the key distinction is between limited or early-stage disease and advanced-stage disease ( Table 30.5 ).
| STAGE | DESCRIPTION |
|---|---|
| Limited stage | |
| I | Involvement of a single lymphatic site (e.g., nodal region, spleen, Waldeyer’s ring) b |
| IE | Single extralymphatic site, no nodal disease |
| II | Two or more nodal regions on the same side of the diaphragm |
| IIE | Contiguous extralymphatic extension from a nodal site ± involvement of other nodal regions on the same side of the diaphragm |
| II bulky c | Can be limited or advanced stage depending on histology and prognostic factors |
| The maximum diameter of the largest mass should be measured | |
| HL: mass >10 cm or >1/3 thoracic diameter on CT | |
| FL: mass >6 cm | |
| DLBCL: mass >10 cm | |
| A PA CXR is no longer required | |
| Advanced stage | |
| III | Nodal regions on both sides of the diaphragm or nodes above the diaphragm and splenic involvement. IIIE and IIIS are no longer recognized |
| IV | Diffuse/disseminated involvement of ≥1 extralymphatic organ ± nodal involvement or noncontiguous extranodal involvement with stage II nodal disease or any extralymphatic organ involvement in stage III disease |
| NB: includes any involvement of CSF, bone marrow, liver, or lungs except by direct extension in stage IIE disease d | |
a The main differences between the Cotswolds and Lugano classification systems are described.
b Waldeyer’s ring, the thymus, and the spleen are considered nodal or lymphatic sites.
c The “X” subscript is no longer used.
d Any liver involvement by contiguous or noncontiguous spread should be regarded as stage IV disease.
The modified Ann Arbor classification, although applied, was never as useful as for NHL, as the prognosis in NHL is more dependent on histologic subtype, tumor bulk, and specific organ involvement, rather than stage. In NHL, the critical questions are whether or not disease is limited, the effect of the disease on end organs such as bone marrow, and symptomatology. As NHL spreads more randomly than HL, the Cotswolds staging system is less helpful in defining prognostic subgroups. The Lugano classification is also applied to NHL, and the definition of bulky disease is subtype-dependent ( Table 30.5 ).
Considerable effort has been applied to the development of robust prognostic indices in NHL that are simple and discriminant. Such indices not only aid in the management of the individual patient but also facilitate meaningful comparison of results from clinical trials. The International Prognostic Index (IPI) was developed by an international collaborative group. For DLBCL, five factors have prognostic significance: age greater than 60 years, elevated serum lactate dehydrogenase (LDH), Eastern Cooperative Oncology Group performance status greater than 1 (i.e., nonambulatory), advanced-stage (III or IV) disease, and more than one extranodal site of disease.
Four risk groups are recognized depending on the number of adverse prognostic features that are present. Before the monoclonal antibody era, patients in the low-risk group (no or one prognostic factor present) had a 5-year survival rate greater than 70%, whereas patients in the high-risk group (four or five factors present) had only a 25% 5-year survival rate. More recently, the prognostic importance of gene expression profiling within individual subtypes of NHL has become evident, as described previously for DLBCL, where the better prognosis of the germinal center–like subtype compared with the activated B-cell subtype is independent of the IPI. A similar prognostic index has been developed for FL, the FLIPI. In the initial index, prognostic factors were age over 60 years, elevated serum LDH, hemoglobin less than 12 g/dL, stage III or IV disease, and more than four nodal sites of disease. The more recent modification (FLIPI 2) includes elevated serum beta-2 microglobulin and the presence of a lymph node over 6 cm in longest diameter.
For the purposes of the index, nine nodal sites are recognized (right and left cervical; right and left axillary; mediastinal; paraaortic; mesenteric; and right and left inguinal). Histologic factors are also important; grades I, II, and IIIa FL are treated similarly, whereas grade IIIb FL is now treated as DLBCL. Recent work also suggests that gene expression profiling yields significant prognostic information in FL. In other NHL subtypes, other prognostic factors are important. In mantle cell lymphoma (MCL), which has until recently had a dismal prognosis, the Ki-67 proliferation index has greater predictive value than any other histologic or clinical criterion.
The clinical spectrum of childhood lymphoma differs from adult lymphoma, with more frequent extranodal involvement, especially of the gastrointestinal (GI) tract, solid viscera including the kidneys and pancreas, and extranodal sites in the head and neck. In these situations, the St. Jude, or Murphy’s, staging classification is applied, because it takes into account the increased frequency of extranodal disease ( Table 30.6 ).
| STAGE | CRITERIA FOR EXTENT OF DISEASE |
|---|---|
| I | A single tumor (extranodal) or single nodal region, except in the mediastinum or abdomen |
| II |
|
| III |
|
| IV | Any of the above with initial central nervous system and/or bone marrow involvement |
For primary extranodal lymphoma (see later), a modified Ann Arbor staging classification may be used. Other staging systems in use include the Rai or Binet systems for SLL/CLL.
HL tends to spread predictably in contiguous fashion from one lymph node group or region to the next adjacent group via lymphatic pathways. Thus, clinical involvement of the cervical lymph nodes should prompt careful scrutiny of the anterior and middle mediastinal nodal groups at staging CT. Similarly, in the presence of bulky mediastinal disease, attention should be paid to the upper retroperitoneal, celiac, portal, and splenic hilar nodal stations. Lymph node involvement is usually the only manifestation of disease. Primary extranodal HL is exceptionally rare and is a diagnosis of exclusion, although it is not uncommon for nodal HL to affect contiguous organs. For example, bulky anterior mediastinal disease can involve the adjacent lung parenchyma or the chest wall, but this does not affect the disease stage or prognosis and would be designated “E” at staging. By contrast, the presence of peripheral subpleural pulmonary nodules discontiguous with the mediastinal mass indicates hematogenous spread and disseminated disease, which would be designated stage IV. With disease progression or relapse, visceral involvement occurs, and tumoral deposits may be seen in the liver, spleen, lungs, and bones.
NHL is generally a disseminated disease at presentation, involving lymph node groups through hematogenous spread, and multiple organs may be involved as well as the bone marrow. Thus, nodal enlargement might be seen at CT in the neck and pelvis, with no nodal abnormality in the chest or abdomen, whereas this distribution would be most unusual for HL. This unpredictability makes a whole-body staging technique imperative. However, individual subtypes of NHL are associated with certain patterns of disease. Marked splenomegaly is a feature of splenic marginal zone lymphoma and MCL, the latter often in association with bowel involvement. The constellation of a large anterior mediastinal mass with central venous obstruction and disease in the liver, kidneys, or adrenal glands but little or no nodal disease makes primary mediastinal large B-cell lymphoma (PMBL) the most likely diagnosis, whereas widespread peritoneal disease at presentation, with involvement of the viscera and female genital tract, suggests BL. Certain types of NHL are strongly associated with central nervous system (CNS) or meningeal disease, especially testicular and head and neck lymphoma, and this association may warrant screening of the craniospinal axis or prophylactic intrathecal therapy. With disease progression, nodal lymphoma may spread to involve adjacent structures. In the retroperitoneum, this can affect the paravertebral and paraspinal structures, with resultant neural compression. In the mesentery, spread into adjacent bowel loops is common, causing displacement, encasement, or compression. As disease advances, peritoneal involvement can occur that is radiologically indistinguishable from peritoneal carcinomatosis.
Often, the pattern of disease at CT may suggest the diagnosis. Thus, involvement of cervical lymph nodes and the tissues of Waldeyer’s ring suggests NHL rather than HL. Nodal enlargement in the anterior and middle mediastinum suggests HL, whereas disease in the mesentery, with or without concomitant bowel involvement, strongly favors NHL (see Table 30.3 ).
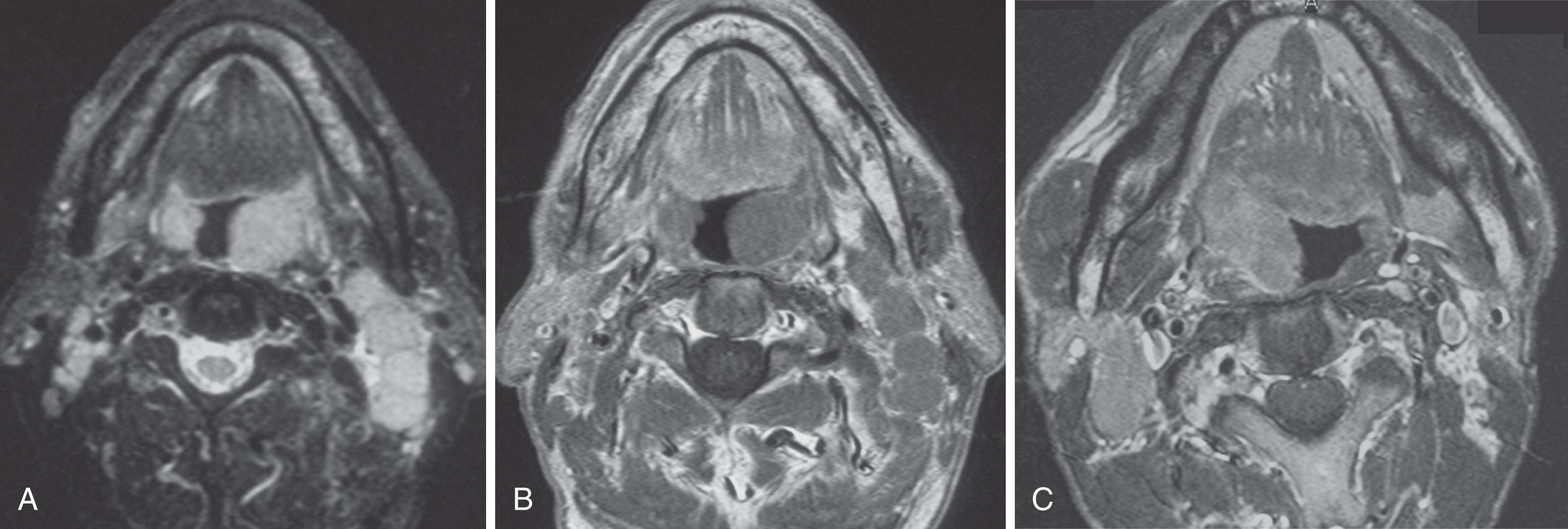
For some time, contrast-enhanced CT (CE-CT) has been the modality of choice for the staging and follow-up of lymphoma. It enables localization of the most appropriate lesion for percutaneous image-guided biopsy. Ultrasound has limited value in staging. Involved lymph nodes have nonspecific appearances, although the pattern of nodal vascular perfusion on power Doppler sonography may suggest the diagnosis. The main value of ultrasound is in providing image guidance for biopsy. Although magnetic resonance imaging (MRI) is as accurate as CT in detecting lymph node enlargement, its role has been essentially adjunctive. As with CT and ultrasound, involved lymph nodes cannot be diagnosed other than by size criteria ( Fig. 30.2 ). Advances in scanner technology including high–field strength magnets enable whole-body MRI studies, and in the last decade there has been much research into the use these techniques as a staging tool, particularly in pediatric patients, where radiation dose is an important consideration. Whole-body diffusion-weighted imaging (DWI) of lymphoma may have a role in the differentiation of lymphoma from other causes of malignant nodal enlargement.
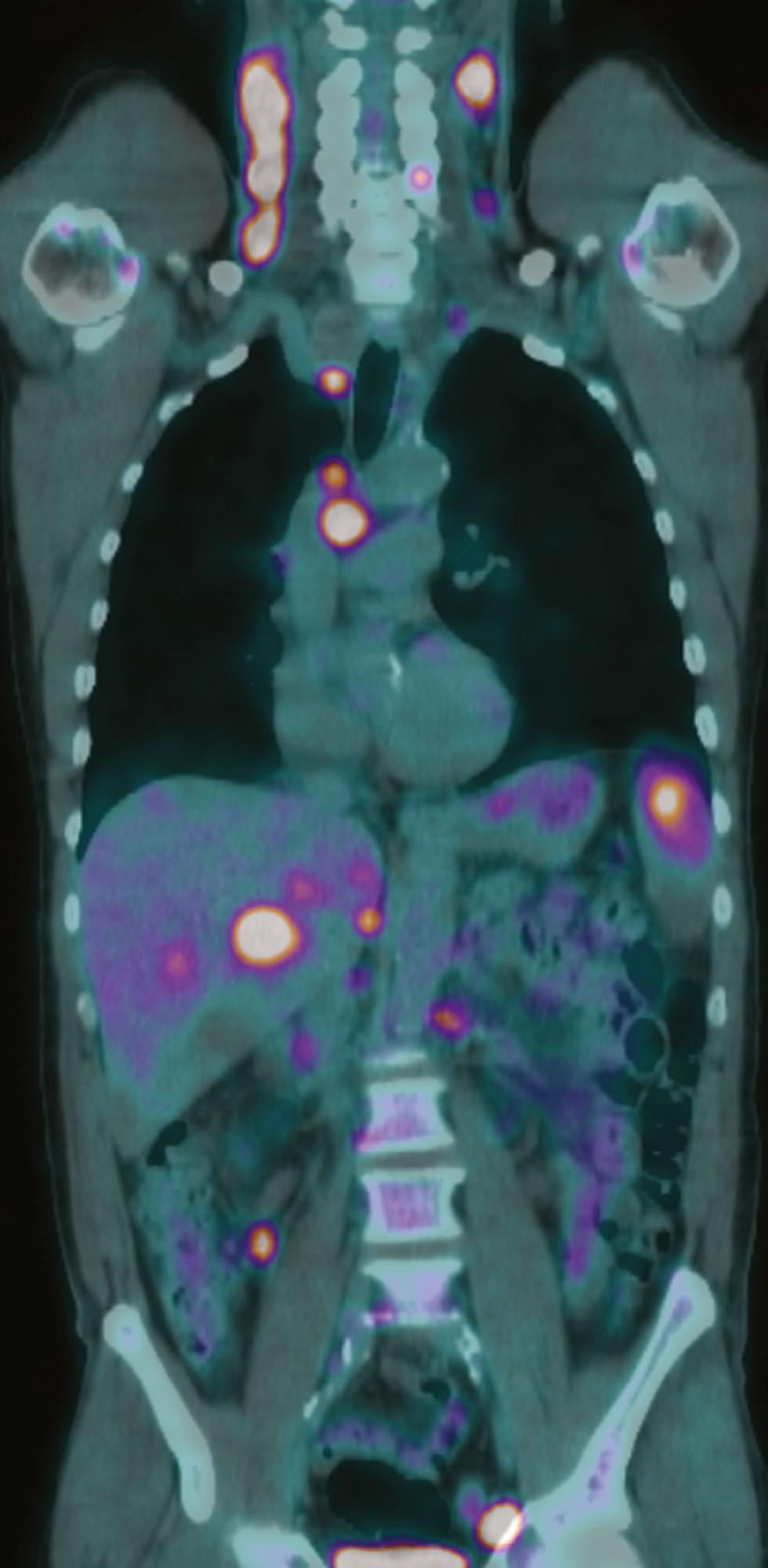
Detection of disease in normal-sized nodes is not possible with cross-sectional imaging, nor is it possible to differentiate between nodal enlargement secondary to lymphoma or reactive hyperplasia. Functional radioisotope studies permit this distinction. Gallium-67 citrate scintigraphy has been superseded by 2-[ 18 F] fluoro-2-deoxy-D-glucose–positron emission tomography (FDG-PET). In most lymphomas, increased glucose metabolism results in increased cellular uptake of FDG with an accuracy comparable with or better than CT for the detection of nodal and extranodal disease. Upstaging with PET/CT occurs in up to 40% of patients, although changes of therapy as a consequence are more common in HL than in NHL. In NHL, staging FDG-PET gives important information by indicating tumor burden as well as the presence of extranodal disease ( Fig. 30.3 ). Most NHLs show increased uptake, the exceptions being some MALT, cutaneous, and small lymphocytic lymphomas. In FL, the presence of relatively high FDG uptake in a given nodal group may suggest histologic transformation, which can then be confirmed by targeted percutaneous biopsy. PET/CT allows accurate co-localization of morphologic abnormalities and their associated functional changes. Debate continues as to whether it is always necessary to carry out a full diagnostic CE-CT as part of the PET/CT study or whether a low-dose CT for the purposes of attenuation correction and anatomic correlation is sufficient. Current guidelines recommend performing a diagnostic CE-CT at presentation, after which it can be omitted if noncontributory. For nonavid and variably FDG-avid histologies, CE-CT remains the staging technique of choice. CE-CT is also recommended for enumerating nodes for clinical trials and for radiotherapy planning. In these guidelines, a lymph node with longest diameter (LDi) greater than 1.5 cm is considered enlarged, and it is recommended that up to six nodes measurable in two orthogonal diameters are used as target lesions.
Recognition of nodal disease with computed tomography (CT) and magnetic resonance imaging (MRI) depends on size criteria alone.
CT and MRI have equal sensitivity below the diaphragm.
Ultrasound has a role in problem-solving and biopsy guidance.
FDG PET/CT may enable identification of disease in normal-sized lymph nodes.
Nodal disease in the lymphomas may involve any anatomic lymph node site.
Cervical lymph node enlargement is seen in 60% to 80% of patients with HL at presentation. It typically involves the internal jugular chain of nodes initially, with further spread to the spinal accessory and transverse cervical chains. In NHL, the pattern of involvement is more haphazard than in HL ( Fig. 30.2 ). Enhancement after administration of contrast medium is usually mild to moderate, and central necrosis within a lymph node is rare. MRI may be particularly useful for defining the extent of lymphomatous masses in the lower neck and supraclavicular fossa.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here