Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Glycogen storage diseases are a group of disorders resulting from inherited defects in the biochemical pathways of glycogen metabolism. Based on organ involvement, glycogen storage diseases are broadly categorized into two groups: hepatic glycogen storage diseases and muscle glycogen storage diseases.
The individual incidence rate of each glycogen storage disease is very rare. However, the collective incidence of all glycogen storage diseases is approximately 1 case per 10,000 to 25,000 births. Glycogen storage diseases I, II, III, V, VI, and IX account for the majority of cases. High-risk populations include Ashkenazi Jews for type Ia and IV (adult polyglucosan body disease), Faroe Islanders and Native Canadians (Inuit population) for type III, and Mennonites for type IV and VI.
Glycogen is a glucose polymer that is found predominantly in the liver and muscle. In the liver, glycogen maintains glucose homeostasis during periods of fasting. In the muscle, glycogen produces energy during exercise. Tissues other than the liver and skeletal muscle contain lesser amounts of glycogen. Postprandially, blood glucose levels increase, and as a result the expression of glucose transporters (GLUT) is upregulated in response to either the exposure to increased blood glucose levels (GLUT2 in the liver) or insulin release by the pancreas (GLUT4 in the skeletal muscle). GLUT2 and GLUT4 facilitate the transport of glucose into the cells. Intracellular glucose can be used to produce energy (ATP) or to synthesize glycogen , ( Fig. 191-1 ).
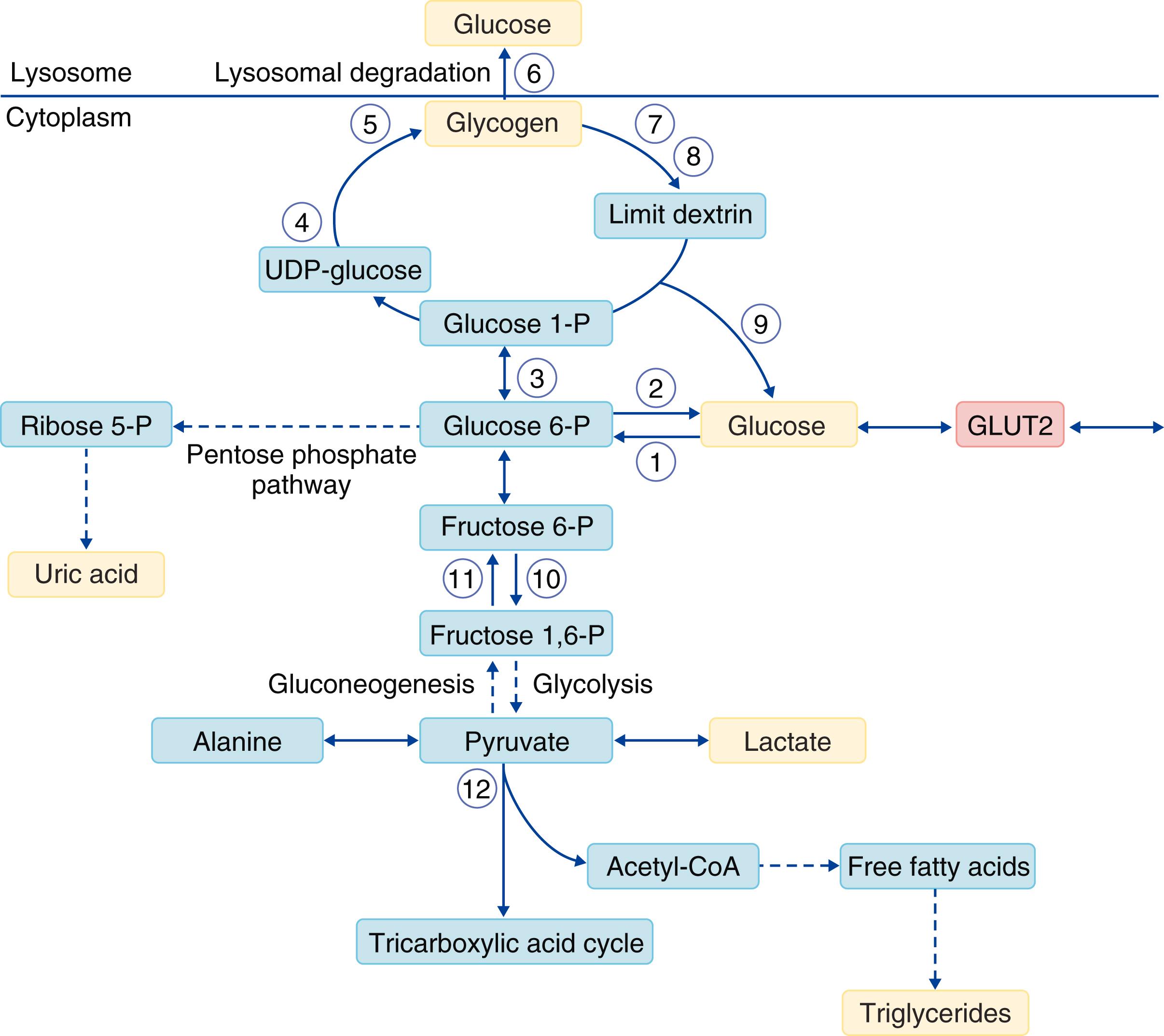
The synthesis of glycogen occurs mainly in the liver and muscle, where intracellular glucose is used to synthesize glycogen that is then stored to be used at times of need. The first step in glycogen synthesis is the phosphorylation of glucose into glucose-6-phosphate by the enzyme glucokinase in the liver and hexokinase in muscle. Next, phosphoglucomutase converts glucose-6-phosphate into glucose-1-phosphate, which then reacts with uridine triphosphate (UTP) to form the nucleotide sugar UDP-glucose. An autoglycosylating protein known as glycogenin then transfers the glucose from UDP-glucose to form an oligosaccharide primer chain, which is then used to form glycogen. Glycogen synthase, which is the primary regulatory enzyme in the synthesis of glycogen, and branching enzyme extend the oligosaccharide primer chain by adding more glucose molecules through α-1,4 and α-1,6 glycosidic bonds, respectively.
Hepatic glycogenolysis (see Fig. 191-1 ) is activated by fasting, which leads to an increase of counter-regulatory hormones such as glucagon, cortisol, and adrenaline. In the muscle, the increase of cAMP during exercise stimulates the breakdown of glycogen. A small amount of glycogen is catabolized in the lysosomes, but the majority of catabolism occurs in the cytosol via a process called glycogenolysis. Glycogenolysis requires the coordinated action of phosphorylase kinase, phosphorylase, and debranching enzyme. Phosphorylase kinase is responsible for activating muscle and liver phosphorylase via phosphorylation. Next, activated phosphorylase releases glucose-1-phosphate from the glycogen branch. This process continues until there are four glucose residues from the α-1,6 branch point. At this point, the debranching enzyme continues the breakdown of glycogen through its two independent catalytic activities: amylo-1,6-glucosidase and 4-α-glucanotransferase. The 4-α-glucanotransferase catalyzes the transfer of three glycosyl residues from one outer branch to another, thereby exposing a single glucose residue that is linked by an α-1,6-glycosidic bond. The α-1,6-glucosidase breaks the α-1,6-glycosidic bond and releases a free glucose molecule.
More than 15 different enzymatic defects in the glycogen metabolic process correspond to the different glycogen storage diseases. Glycogen storage disorders are caused by homozygous, compound heterozygous, or hemizygous mutations in the target genes. Most glycogen storage diseases are inherited as autosomal recessive conditions. However, liver and muscle phosphorylase kinase deficiency, which are caused by mutations in PHKA2 and PHKA1 genes, and Danon disease are inherited as X-linked disorders. Some glycogen storage diseases also can be categorized as metabolic diseases; for example, Pompe disease can be categorized as a lysosomal storage disease, whereas phosphoglucomutase 1 (PGM1) deficiency can be classified as a congenital disorder of glycosylation.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here