Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The gluteus maximus (GM) flap was initially described as a turnover muscle flap for sacral coverage, in 1974. During the following years, musculocutaneous GM flaps became the gold standard in the treatment of pressure ulcers because of their ability to fill dead space and to provide well-vascularized tissue to the ulcerated, often contaminated defect. The flap was expanded to several variations and was used successfully for reconstruction of sacral, ischial, trochanteric, and perineal defects. Even today, the use of GM myocutaneous flaps is still the first choice of many surgeons for decubitus ulcers of the sacral and ischial regions in paraplegic patients. Microsurgical transfer of a myocutaneous gluteal flap was first described by Fujino et al. in 1975 and, a few years later, Le Quang introduced the inferior gluteus maximus free flap for breast reconstruction. However, the use of GM muscle free flaps has been limited due to the short pedicle and high donor site morbidity.
Since the advent of perforator flaps, harvesting flaps from the gluteal region, without inclusion of GM muscle, as a superior gluteal artery perforator (SGAP) flap or an inferior gluteal artery perforator (IGAP) flap, has supplanted GM muscle flaps in most reconstructions because of the reduced donor site morbidity. Fasciocutaneous flaps seem to be suitable to cover pressure sore defects with a similar outcome to that of muscle flaps. Koshima et al. first described the pedicled SGAP flap for use in pressure sore treatment. Allen has popularized the use of gluteal perforator free flaps in breast reconstruction. Surgeons adept at the use of perforator flaps are rarely in favor of using gluteus muscle flaps and limit their use to patients with lower extremity paralysis that renders these muscles expendable. Although perforator flaps provide a better reconstructive option due to a longer pedicle with a better flap mobility and reduced donor site morbidity, some surgeons continue to use gluteus muscle flaps in patients who are paralyzed and when the techniques of perforator flaps have not been mastered.
Techniques of gluteus maximus muscle flaps, as well as perforator-based flaps, are summarized below.
The gluteus maximus, which is the superficial muscle of the buttock, is a thick muscle (see Fig. 44.1 and Figs 13.1 and 58.1 ). Its function is to extend the hip and to rotate the thigh laterally. The origin of the GM muscle involves the posterior gluteal line of the ilium, the aponeurosis of the erector spinae, the dorsal surface of the lower part of the sacrum, the side of coccyx, and the sacrotuberous ligament. The upper portion inserts into the iliotibial tract and the deep fibers of the inferior portion insert on the gluteal tuberosity of the femur. The GM muscle is completely invested by the deep fascia of the lower limb, which splits into two luminae around the GM muscle; the deep layer is thickened and contains the gluteal vessels and nerve. The piriformis muscle lies under the GM muscle. It is a pyramid-shaped muscle that arises from the 2nd–4th sacral segments and exits the pelvis through the greater sciatic foramen to insert into the top of the greater trochanter. The piriformis muscle acts as a lateral rotator of the hip. The sacral plexus is formed largely on the pelvic surface of the piriformis muscle. The sciatic nerve emerges under the piriformis muscle but may occasionally pass through it.
The GM muscle is type III (Mathes and Nahai classification ), which has two dominant pedicles: the superior and inferior gluteal vessels. Usually the IGA is dominant over the SGA and supplies approximately two-thirds of the muscle.
superior gluteal artery (SGA)
Length: 2.5 cm (range 2–3 cm)
Diameter: 2.5 mm (range 2–3 mm)
The SGA is a terminal branch of the internal iliac artery and exits the pelvis through the greater sciatic foramen. After its exit, the SGA divides into deep and superficial branches. The superficial branch continues above the piriformis muscle into the gluteus maximus muscle. The intramuscular length of these vessels is 4–5 cm, depending on the thickness of the gluteal muscle. The superficial branch gives off perforating vessels (average of four) that go on to supply the upper portion of the gluteus muscle and overlying fat and skin. The perforator vessels pass in superior and lateral directions.
inferior gluteal artery (IGA)
Length: 9 cm (range 8–10 cm)
Diameter: 3 mm (range 2.5–3.5 mm)
The IGA is a terminal branch of the internal iliac artery and exits the pelvis through the greater sciatic foramen. The artery accompanies the greater sciatic nerve, internal pudendal vessels, and the posterior femoral cutaneous nerve. The IGA pierces the sacral fascia and exits the pelvis caudal to the piriformis muscle. It gives several perforators, usually more than the SGA, under the inferior portion of the gluteus maximus muscle to nourish the skin and subcutaneous tissue. Perforators that nourish the medial and inferior parts of the buttock have short intramuscular length (3–5 cm), whereas the perforators that nourish the lateral part of the buttock have a longer intramuscular course (4–6 cm). The IGA descends into the posterior cutaneous surface of the thigh together with the posterior femoral cutaneous nerve, in 90% of the cases.
first perforator off the profunda femoris artery, which courses superiorly to the insertion of the gluteus maximus on the femur.
Veins usually accompany the arteries. The superior gluteal vein (SGV) receives multiple tributaries from other pelvic veins at the level of entering the pelvis. Veins accompanying the IGA are larger but are fragile and prone to bleed easily during dissection.
venae comitantes of the SGA
Length: 2.5 cm (range 2–4 cm)
Diameter: 3 mm (range 2–4 mm)
venae comitantes of the IGA
Length: 8 cm (range 7–10 cm)
Diameter: 3 mm (range 2.5–4 mm)
none.
cluneal nerves (nervi clunium superiores) – direct branches off the spinal cord (L1–S3) – can be identified at the superior edge of the flap above the muscle fascia.
the inferior gluteal nerve (IGN) nerve enters the deep surface of the gluteus maximus, but it is not of reconstructive use and is usually left intact.
For small sacral pressure sores, a pure skin rotation flap design may be sufficient. For larger pressure sores, a segment of GM muscle is incorporated, based on the SGA as a musculocutaneous flap. A bone component from the sacral region can be incorporated; however, the donor site morbidity does not justify its use.
The gluteal flap can also be raised as a skin-fat perforator flap or as a compound flap (skin and muscle), where a segment of GM muscle is harvested based on a muscular branch from the main pedicle or as a side branch from the perforator.
Due to its abundant blood supply, the GM muscle flap provides a generous amount of well-vascularized soft tissue.
Thick muscle and subcutaneous tissue allow for adequate coverage of bony prominences and filling of defects.
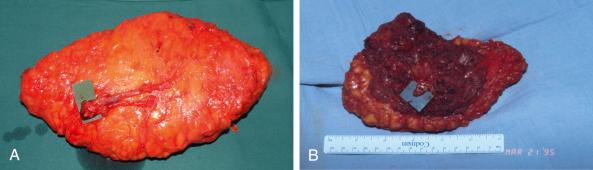
When harvested as a free flap, a sufficient amount of soft tissue with a constant vascular pedicle can be used for breast reconstruction, leaving a well-concealed scar on the donor site.
Raising a GM flap requires a muscle transection, which may lead to important blood loss. Furthermore, dissection of the pedicle under the muscle is tedious because of multiple side branches and fragile veins. Bleeding in that area can be difficult to control.
In the case of a free transfer of a GM musculocutaneous flap, the superior gluteal vessels are short and can result in the need to use an intertransposition vein graft to reach the recipient vessels.
An area of numbness over the posterior surface of the thigh is possible due to sacrifice of the posterior cutaneous nerve of the thigh during dissection of an inferior gluteal musculocutaneous flap.
The skin paddle cannot be >10 cm if the donor site is to be closed primarily.
The patient's position during flap harvesting can complicate simultaneous work by a second team of surgeons.
The firmer consistency of gluteal fat makes it more difficult to mold the flap, especially for breast reconstruction. Additional minor secondary corrections might be necessary to obtain the desired shape.
At the donor site, a contour deformity or asymmetry can occur in unilateral harvesting and may necessitate a secondary correction of the contralateral side.
Although there is no functional advantage of sparing GM muscle in paralytic patients, harvesting part of the GM can result in functional weakening of hip extension in ambulating patients.
Some of the above-described disadvantages are reduced by harvesting the flap based only on the perforators. The gluteal perforator flap has the following advantages:
Donor morbidity after raising gluteal perforator flaps is the lowest that can be offered to the patient today. This itself will lead to a shorter hospital stay and therefore reduced costs.
A better intraoperative exposure, less blood loss, and a longer vascular pedicle compared with its myocutaneous predecessor ( Fig. 44.1 ). The length of the pedicle gives the flap an impressive mobility and allows for easy tissue transposition without tension.
The possibility of including a sensate branch of the nervi clunium superiores within a gluteal perforator free flap provides better quality and quantity of sensation to the flap.
In the case of a pedicled flap, the tissues used in the gluteal perforator flap come from a further distance from the ulcer or wound and, as such, they are less inflamed, less edematous, and less prone to dehiscence. The inset of the flap is always tension-free and the donor defect can always be closed primarily.
Preoperative bowel preparation is performed on patients who are undergoing this procedure for breast reconstruction. However, a temporary intestinal stoma may be required to avoid wound contamination in complicated cases of large sacral defects. Wound culture and bone biopsy are necessary for chronic cases with signs of osteomyelitis. Spasticity contractures and vascular disease that can compromise the flap or wound healing and nutritional factors must first be managed. Intrinsic factors in bed sore physiopathology include sensory loss, diminished muscle strength or mobility, incontinence, infection, anemia, protein malnutrition, and advancing age. Obviously, if possible, these factors must be considered and corrected before surgery. Antibiotic therapy is given at the beginning of surgery and continued at least for 24 h postoperatively. Depending on perioperative wound culture, antibiotics should be adjusted if necessary.
The initial points marked are the posterior superior iliac spine (PSIS), the prominence of the greater trochanter (T) of the femur, the superior edge of the greater trochanter, and the coccyx (C). The location where the SGA exits the suprapiriform foramen is marked on the skin at the proximal one-third of a line connecting the posterior superior iliac spine and the apex of the greater trochanter. Figure 44.2 shows the territories of the superior gluteal and the inferior gluteal flaps and their relationships with the above-mentioned landmarks.
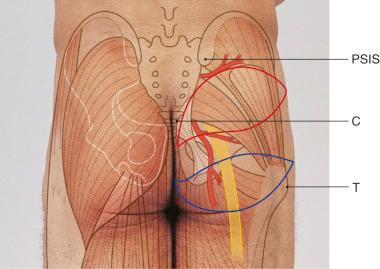
Since the gluteus maximus is a type III muscle, it can be harvested based on either the superior or the inferior gluteal artery. For free tissue transfer, usually a small part of the muscle is taken along with an overlying island of skin and subcutaneous tissue. The skin paddle is oriented along the inferior gluteal crease in the case of an inferior gluteal flap, whereas it is oriented obliquely from the sacrococcygeal junction to the anterior superior iliac spine for a superior gluteal flap ( Fig. 44.3 ). This orientation of the skin paddle minimizes distortion of the anatomy upon donor site closure.
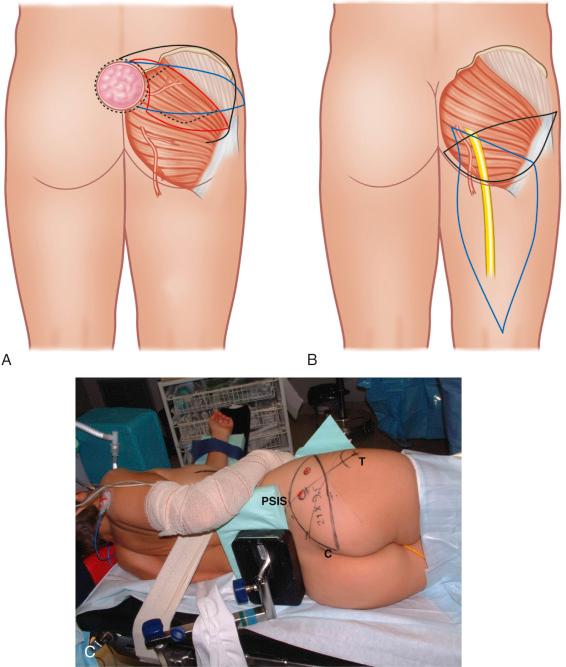
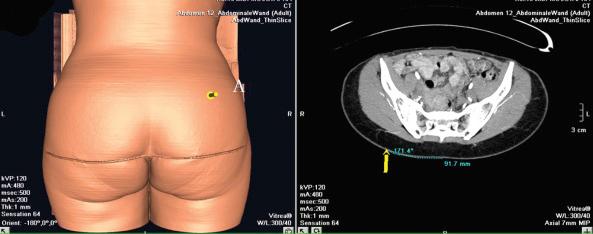
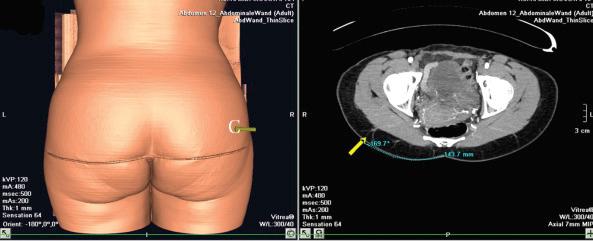
In the case of a perforator flap, a fusiform skin island is drawn over the perforators, ideally centered on selected perforators. The main perforators are localized by a handheld 5–8 MHz Doppler probe in an area above or under the piriform muscle, for SGAP or IGAP flaps, respectively ( Fig. 44.3C ). Currently, we are using a new method for perforator mapping with a 64-slice spiral computed tomography (CT) scan ( Figs 44.4 , 44.5 ). This provides more accurate information concerning the vascular network and perforators. Choice of the perforator depends not only on the strength of the Doppler signal or the findings of the CT scan but also on the location of the perforator. Choosing the most remotely located perforator from the SGA exit will provide a longer pedicle, which is more suitable for microanastomosis ( Figs 44.4 , 44.5 ).
In the case of a superior gluteal free flap, a skin paddle of 24 × 9 cm is usually placed obliquely with a horizontal orientation at the lateral aspect to produce a scar that is easy to conceal in a bathing suit. Also, we add a small fishtail at the medial tip of the flap to avoid dog-ear formation in the median gluteal crease ( Fig. 44.3 ). The flap is also designed longer to be able to shape the breast in the form of a hammock, taking care to reconstruct both the anterior axillary fold and the full width of the inframammary crease.
The oblique/horizontal axis of the flap provides a tension-free closure with buttock lift and minimal contour deformity. The flap island can be designed much higher on the buttock than traditionally described, to allow for the scar line to ultimately rest at the junction of the aesthetic units of the lower back and the upper buttock. This essentially results in only the medial half of the flap being centered over the GM muscle. A careful Doppler examination is then mandatory to ensure inclusion of SGA perforators in the flap. In the case of the inferior gluteal free flap, a transverse skin ellipse is marked on the ipsilateral gluteal crease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here