Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Infections of the genitourinary tract can be caused by a number of viral, bacterial, fungal, and parasitic organisms ( Table 16.1 ). Although many of these organisms are also seen in other locations, some of them have a predilection for the genitourinary area. Their clinical manifestations, in conjunction with their gross and microscopic appearances, are useful in characterizing these infections, and ancillary tests are often useful in their diagnosis.
| Disorder | Microbe |
|---|---|
| Chancroid | Haemophilus ducreyi |
| Dermatophytosis | Trichophyton , Epidermophyton , and Microsporum spp. |
| Encrusted cystitis | Corynebacterium urealyticum |
| Granuloma inguinale | Klebsiella granulomatis |
| Lymphogranuloma venereum | Chlamydia trachomatis |
| Malakoplakia | Escherichia coli (frequently) |
| Pityriasis versicolor (tinea versicolor) | Malassezia furfur |
| Syphilis | Treponema pallidum |
Erythrasma is a chronic bacterial infection of the genitourinary region caused by Corynebacterium minutissimum , particularly in humid cutaneous recesses, such as the vulva and inguinal folds, and especially on the inner aspect of the thigh just below the crural fold. Diabetics and institutionalized patients are predisposed. Clinically, it is characterized by a sharply demarcated, brown to red, scaly plaque that fluoresces under a Wood lamp because of the presence of bacterial porphyrins, often giving a coral-red or orange-red color. Corynebacteria are pleomorphic, small, gram-positive rods, also referred to as diphtheroid. The diagnosis is often one of exclusion, after fungal infections are ruled out. Fungal organisms may be visualized with the use of potassium hydroxide (KOH) on various microscopic preparations or with periodic acid–Schiff (PAS) and methenamine silver stains.
Trichomycosis pubis and axillaris is an asymptomatic colonization of hair follicles by various corynebacteria (particularly Corynebacterium tenuis ), which can lead to a red, cream, yellow, or, less commonly, black coating around pubic or axillary hairs and produce an offensive odor. Pubic involvement is less common than axillary involvement. Microscopically, large bacterial colonies are present and may invade the superficial hair cortex. C. tenuis was once thought to be the primary offending organism, but now multiple species are recognized. The so-called corynebacterial triad is the combination of erythrasma, trichomycosis axillaris, and pitted keratolysis. However, pitted keratolysis usually affects the plantar surface of the feet, leading to multiple asymptomatic pits and superficial erosions; it is less common in the genitourinary region.
Cellulitis of the scrotum and penis can result from bacteria (particularly β-hemolytic streptococci but also Escherichia coli , Vibrio vulnificus , Vibrio cholerae , Pasteurella multocida , and others) that gain an entry after a disruption of the epidermal surface, such as after circumcision. Genital skin in this condition is red, hot, and tender, and histologically there is prominent edema with dermal perivascular and interstitial acute inflammation. P. multocida cellulitis may occur after animal bites.
Staphylococcus aureus produces primary lesions in the genital tract, including impetigo, bullous impetigo, furuncles, and folliculitis. Impetigo usually occurs on the face or extremities but may also spread to the genital area; although it was previously thought that group A β-hemolytic streptococci most commonly caused impetigo, S. aureus is currently the most common cause. Bullous impetigo consists of 0.5- to 3-cm in diameter flaccid bullae, with thin roofs and erythematous rims and erosions; the bullae may coalesce. The roof may rupture, forming a thin crust. A furuncle (boil) is an infection of the pilosebaceous unit that occurs at sites of friction, such as the inner aspects of the thighs and the buttocks. Histologically the furuncle consists of an abscess in the deep dermis centered on a hair follicle. Folliculitis, usually caused by S. aureus , has a number of manifestations that clinically consist of small pustules developing around follicular ostia, sometimes pierced by hair. Histologically, folliculitis consists of a subcorneal pustule with abundant neutrophils and some macrophages and lymphocytes overlying the follicular infundibulum. S. aureus can also cause secondary infections in atopic dermatitis, psoriatic plaques, and lichen simplex chronicus, as well as superinfections of candidiasis, genital herpes, or syphilitic chancres.
Group A or B β-hemolytic streptococci can cause superficial erosive intertrigo. In addition, perianal streptococcal dermatitis can be caused by group A β-hemolytic streptococci. Perianal streptococcal dermatitis occurs most commonly in children and is recognized by a perianal erythema with a well-demarcated border that may desquamate as a scale and subsequently heal.
Hidradenitis suppurativa (also known as acne inversa or apocrine acne) is caused by bacterial infection of the apocrine glands, usually in the axilla, but it can also involve apocrine glands in the perineum and vulvar region. Some studies have suggested that coagulase-negative staphylococci are the most common cause, but others have found the microbiologic flora to be variable.
Fournier gangrene is a necrotizing subcutaneous infection of the genital and anorectal regions caused by a mixture of aerobic and anaerobic enteric organisms. Risk factors include genitourinary trauma, periurethritis, diabetes, immunosuppression, poor nutritional status, septic injection into the penile vein, and, rarely, trans -retinoic acid treatment for hematologic malignancies. A rapidly developing cellulitis, fasciitis, myositis, and systemic toxicity may result; however, the external physical findings may not be prominent and may be out of proportion to the severity of the infection. A localized vasculitis results from a Schwartzman-like reaction. The Schwartzman reaction is the inflammatory process brought about by injection of endotoxin, which on first injection results only in mild inflammation but on second injection results in a thrombohemorrhagic lesion, similar to that seen in Fournier gangrene. The mortality rate ranges from 10% to 30%. Treatment is typically with débridement and broad-spectrum antibiotics, as necrotizing fasciitis in other locations.
Pseudomonal sepsis may be complicated by a cellulitis known as ecthyma gangrenosum, which starts with an erythematous or purpuric cutaneous macule that may rapidly develop into a bulla, which sometimes ulcerates. This is caused by a bacterial vasculitis that may eventually result in cutaneous infarction. Penile ecthyma gangrenosum has been reported in patients with a history of drug abuse and neutropenia.
Genital tuberculosis can result from sexual contact with individuals who have active oral or genital tuberculosis. Mycobacterium tuberculosis is the most common organism, but infections due to Mycobacterium celatum have also been reported. Clinically, subacute or chronic, painful ulcers with or without lymphadenopathy may be present, and the lesions may even mimic tumors grossly, notably in scrotal lesions. Lupus vulgaris, which is the most frequent form of reinfection tuberculosis, can affect the penis. Confluent papules may coalesce into plaques; when viewed by diascopy, these plaques have an “apple-jelly” deposit representing cellular infiltrates. Involvement of the glans may occur. Bacillus Calmette-Guérin (BCG) balanitis after instillation for bladder carcinoma has rarely been reported.
The spirochetal organism, Treponema pallidum , is the causative agent of syphilis. The anogenital area may be affected by primary, secondary, tertiary, or congenital lesions of syphilis. Three weeks after initial exposure, a primary syphilitic chancre occurs at the site of inoculation, usually the inner prepuce, coronal sulcus, penile shaft, vulvovaginal area, or anus.
The painless, buttonlike papule ulcerates centrally, and adjacent “kissing” lesions may also be present. Histologically, there is a prominent lymphoplasmacytic infiltrate in an edematous ulcer bed with prominent capillary proliferation containing swollen endothelial cells. Spirochetes may be identified with the use of Warthin-Starry or Steiner stains or immunohistochemically. Smears can be analyzed by darkfield examination or by immunofluorescence with antitreponemal antibodies. Secondary syphilis occurs if the primary lesion is left untreated and the spirochete spreads systemically. It typically forms 3 to 6 weeks after the chancre but may arise up to 6 months after healing of the primary lesion. Clinically, large, red to pale, moist, flat-topped or verruciform papules may develop in the anogenital region (condyloma lata). In 80% of patients, there is a diffuse, nonpruritic, papulosquamous eruption. In dark-skinned patients, annular polycyclic lesions may occur. Microscopically, many spirochetes are present, explaining the highly contagious nature of secondary syphilis. Pseudoepitheliomatous hyperplasia may be prominent. The composition of the inflammatory cells is similar to that of the primary lesion; in addition, a neutrophilic infiltrate may be present.
Tertiary syphilis targets bones, the central nervous system, the cardiovascular system, and skin with ulcerative and nodular lesions. In tertiary syphilis, the classic lesion is a gumma, a type of granuloma with a focus of necrobiosis surrounded by multinucleated giant cells, histiocytes, plasma cells, lymphocytes, and fibroblasts. Obliterative endarteritis may be present. A nodular presentation consists of tuberculoid granulomas with a mixed inflammatory cell reaction including plasma cells. Treponemes cannot usually be detected with silver stains, but polymerase chain reaction (PCR) may be useful.
Haemophilus ducreyi is the gram-negative, facultative anaerobic bacterium that causes chancroid. It is uncommon in the United States but is a common cause of genital ulcers in Africa. Chancroid is transmitted by sexual contact during the active phase of lesions, which in untreated patients lasts approximately 6 weeks. A papule or pustule appears at the exposure site after approximately 1 week. This usually develops into a painful ulcer or ulcers with a gray-yellow exudate and sharp, undermined, ragged erythematous borders. Many lesions can arise in the location of the original lesion through autoinoculation, and lesions may merge with serpiginous borders. After 1 to 2 weeks, adenopathy with suppuration (buboes) occurs in approximately 25% to 50% of untreated patients, sometimes leading to inguinal abscesses and draining sinuses. Microscopically, microvascular and endothelial proliferation with overlying surface ulceration and necrotic debris is typically seen. Vessels may be thrombosed. There is often a dense lymphoplasmacytic infiltrate. Special culture medium is required for isolation. Chancroid is treated with antibiotics. Large lymph nodes may need to be drained.
Chlamydia trachomatis , an obligate intracellular parasite, particularly serovars L1, L2, and L3, cause lymphogranuloma venereum; the organisms spread through sexual transmission, travel through lymphatic channels, and multiply within draining lymph nodes of the genital and anorectal region. Serotypes D to K are associated with sexually transmitted oculogenital disease.
In the primary stage, eroded papules or groups of small herpetiform ulcers form at the inoculation site (coronal sulcus, frenulum, prepuce, shaft, glans, scrotum, or vulva). Urethritis with a mucopurulent discharge may be present. In the secondary stage, occurring several weeks later, inguinal lymphadenitis and formation of buboes (enlarged lymph nodes, from the Greek boubôn , “groin”) occur, often with concurrent cordlike lymphangitis of the dorsal penis and suppurative inguinal bubo formation. The so-called groove sign may be present, which results from a cleft formed by the inguinal (Poupart) ligament between enlarged involved femoral and inguinal lymph nodes. Proctocolitis may occur.
A third stage comprises various sequelae from inflammation and includes fistula formation, fibrosis, and scarring. This may be complicated by sinuses and tracts between the skin and urethra and deforming scars of the penis. In the anogenital syndrome, proctocolitis and hyperplasia of the perirectal lymph nodes may occur, and fistulas and rectal strictures may form. Obstruction of chronic lymphedema may lead to elephantiasis.
Histologically the ulcers of lymphogranuloma venereum consist of a chronic inflammatory process with scattered giant cells, plasma cells, lymphocytes, necrosis, and granulation tissue. Nonnecrotizing granulomas composed of epithelioid histiocytes may be present. Fibrosis and, eventually, prominent scarring occur as the process becomes more chronic. Pseudoepitheliomatous hyperplasia may be present. Lymph nodes show accumulation of neutrophils, often with prominent necrosis, particularly in the early stages of the infection. Plasma cell infiltration and lymphocytic hyperplasia follow. Eventually, what are referred to as stellate abscesses form; these are suppurative foci that have coalesced and are surrounded by epithelioid cells and multinucleated giant cells.
Culture of the lesions is the best method to identify the organisms in oculogenital chlamydial infections. The obligate intracellular organisms can typically be identified in vacuoles of vacuolated macrophages. The organisms stain faintly blue with hematoxylin and eosin (H&E), are gram-negative, and can be identified with Warthin-Starry stain. Electron microscopy, immunohistochemistry, and PCR for the 16S ribosomal DNA of the organisms are sometimes needed for a definitive diagnosis. Serology may be useful in confirming the diagnosis.
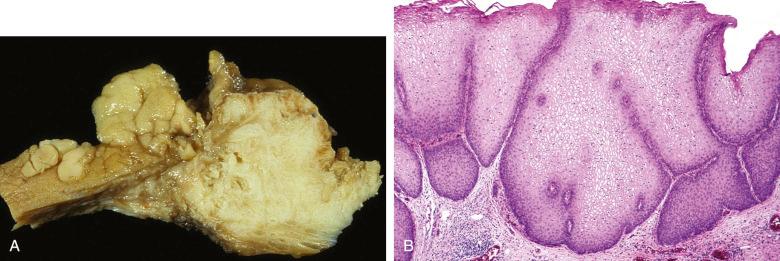
Klebsiella granulomatis (formerly called Calymmatobacterium granulomatis ), a gram-negative bacillus, leads to donovanosis, a sexually transmitted genital ulcer disease. It is rare in the United States and occurs primarily in the southern states. Clinically, well-defined, single or multiple, painless ulcers with beefy and red granulation tissue at the base and elevated, rolled, serpiginous, or hyperplastic borders form the primary lesion. The ulcer may heal with prominent fibrosis. Lymphedema with eventual elephantiasis of the penis and scrotum can result in chronic disease. Subcutaneous nodules (pseudobuboes) can simulate lymph node involvement. Lesions may be nodular, hypertrophic, cicatricial, and ulcerovegetative. Microscopically, there is prominent granulation-type tissue with an inflammatory infiltrate including plasma cells, neutrophils, and histiocytes that contain 0.6- to 2-µm Donovan bodies, which are encapsulated rods with bipolar granules ( Fig. 16.1 ). Pseudoepitheliomatous hyperplasia may be prominent. The gram-negative coccoid to bacillary bacteria may be difficult to identify, requiring Wright/Giemsa, toluidine blue, or Warthin-Starry stains. Cytologic identification of Donovan bodies in granuloma inguinale (see Fig. 16.1 ) can be accomplished with Papanicolaou-stained smears. Ultrastructural studies have identified the organism in greater detail, demonstrating it in the phagosomes of macrophages. Squamous cell carcinomas of the vulva secondary to granuloma inguinale have been reported.
Certain superficial fungal infections, such as dermatophytosis, pityriasis versicolor, and candidiasis, occur in the genitourinary area and are discussed in this section. Deep mycotic infections, including blastomycosis, histoplasmosis, coccidioidomycosis, and paracoccidioidomycosis, also occur in this area. Diagnosis is aided by identification of hyphae in skin scrapings treated with KOH. Fungal infections are usually superficial, but deep infections may occur, and phycomycosis (an older term for zygomycosis) of the vulva has been reported in diabetic women.
Dermatophytosis (tinea) is a superficial fungal infection of keratinized tissues caused by species such as Trichophyton , Epidermophyton , and Microsporum . Tinea cruris is an infection of the inguinal area. An infection of the genital area can result from a transfer by hand from a dermatophytosis of the feet (tinea pedis).
Although pityriasis versicolor is usually located on the upper trunk or upper arms, this superficial fungal infection, caused by the lipophilic yeast of the genus Malassezia , can cause hypopigmented or hyperpigmented, white to red-brown, macular eruptions in genital areas, notably on the penile shaft. Scales show pale yellow fluorescence with a Wood lamp. Pityriasis versicolor is usually considered to be noncontagious. The organism to which this disorder is usually attributed is Malassezia globosa , which was formerly classified as Malassezia furfur .
The normal flora of the intertriginous skin, prepuce, and perineal area includes Candida albicans . Candidal balanoposthitis is the most common fungal infection of the penis. Obesity and uncontrolled diabetes mellitus are predisposing factors, as are other immunocompromised states. The infection can be sexually transmitted. More than 75% of women experience at least one symptomatic episode of vulvovaginal candidiasis. Small pustules may be present. Microscopically, there is epidermal thickening and spongiosis with dermal chronic inflammation. Yeast and pseudohyphae are present on the surface, together with degenerated squamous cells and neutrophils.
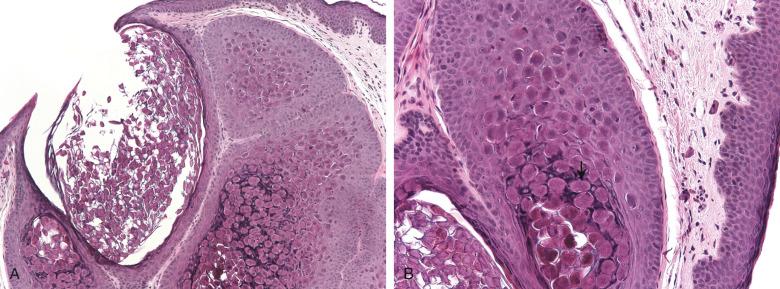
Molluscum contagiosum is caused by a large poxvirus (molluscum contagiosum virus), which is a double-stranded, brick-shaped DNA virus. It manifests as solitary or multiple, 2- to 8-mm, dome-shaped, pearly, centrally umbilicated papules that may cluster, become more keratotic with trauma, and simulate condyloma acuminata. The lesion may express a white caseous material (caseum). Transmission is via fomites or sexual contact. Children often have a primary inoculation in the anogenital region, and autoinoculation may lead to additional lesions. Microscopically ( Fig. 16.2 ), a lobular epidermal acanthosis is present and causes an inverted pattern of epidermal hyperplasia. Intracytoplasmic eosinophilic inclusions, referred to as Henderson-Paterson bodies, can be identified in the stratum spinosum and granulosum. The lesions usually regress spontaneously within 1 year, although scarring may occur. Approximately 60% of individuals with skin lesions have antibodies to the virus, but this fraction is less among patients with acquired immunodeficiency syndrome (AIDS), who, along with other immunocompromised patients, may have numerous lesions, some of which may manifest as tumor-like lesions. Ultrastructurally, large, brick-shaped viral particles can be seen.
Herpes simplex virus type 2 (HSV-2), a double-stranded linear DNA virus, is the most common organism that leads to sexually transmitted genital herpes; however, HSV-1 has been shown to cause almost half of genital herpes cases in developing countries. The incubation period is approximately 5 days, and the virus is harbored for life in sensory ganglia. Vesicles consisting of clear lesions with an erythematous base form the primary lesions, which may rupture and leave behind erosions. The primary lesion may be accompanied by lymphadenopathy, but the lymphadenopathy usually subsides and does not recur. Histologically the lesions start with epidermal cell peripheral nuclear chromatin clumping, ballooning degeneration, and homogeneous ground-glass changes with eosinophilic nuclear inclusions. Cytoplasmic vacuolization may also be present. Eventually, affected cells swell, losing their attachment to and separating from adjacent cells (a process referred to as secondary acantholysis). Cellular cytoplasm becomes homogeneous and intensely eosinophilic, and multinucleated cells (Tzanck cells) may be present. The basal layer of the epidermis may be destroyed, leading to the formation of a subepidermal vesicle. Reticular degeneration, a process whereby the epidermal cells undergo progressive hydropic swelling and cytoplasmic clearing with peripheral cytoplasmic strands remaining, can occur. Eventually, these break, and further vesicle formation occurs. Vesicles may contain neutrophils, and accompanying vasculitis may be present. Late lesions are often ulcerated, with ghosts of acantholytic, multinucleate epithelial cells. Primary infection and viral excretion last approximately 3 weeks. Extragenital lesions occur in approximately 20% of cases, and aseptic meningitis in approximately 10%.
Recurrences are less severe. Burning and tingling may manifest 1 to 2 days before a recurrence. Vesicles are limited in number and heal in approximately 10 days, and virus is shed for only a few days. The glans, prepuce, and shaft are the primary sites of involvement; however, the scrotum may be involved. Perianal lesions may also be present, particularly in men who have sex with men. Chronic HSV disease manifests as painful ulcers with rolled borders. Chronic persistent herpes lesions may suggest and help define the presence of AIDS if they last for longer than 1 month, and they may also occur in other immunosuppressed states (e.g., history of transplantation, chemotherapy, hematologic disorders). The herpes varicella-zoster virus (VZV) may infect the anogenital area in a dermatomal distribution through involvement of the third and fourth sacral sensory nerves.
Immunohistochemical stains may be useful in distinguishing HSV from VZV in biopsy specimens. Monoclonal antibodies to the VZV envelope protein gp1 are sensitive and specific in distinguishing between HSV and VZV. Cultures and molecular probes for viral antigens can also be useful. Viral antigen can be detected in inflamed nerve twigs. Electron microscopy may reveal the 90- to 130-nm viral particles.
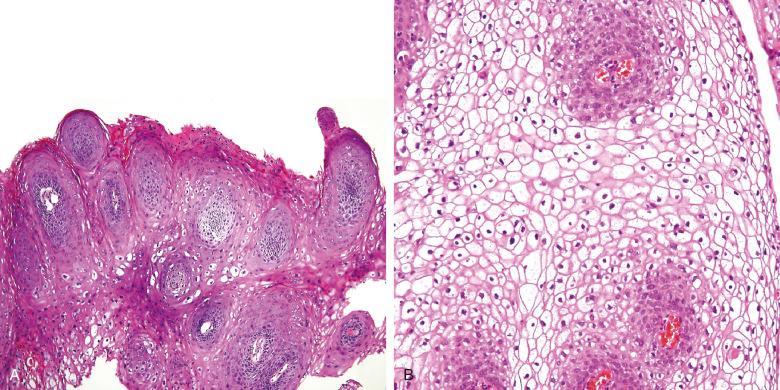
Human papillomavirus (HPV) is a member of the Papovaviridae family, which consists of circular, double-stranded DNA viruses. HPV is involved in a number of pathologic lesions, from the benign end of the spectrum, the common wart (verruca vulgaris) ( Fig. 16.3 ), to frankly malignant squamous cell carcinoma. More than 100 genotypes are known. Relationships between different HPV types and particular lesions have been recognized ( Table 16.2 ).
| Human Papillomavirus Type | Lesion |
|---|---|
| 1 | Plantar, common (verruca vulgaris) warts (see Fig. 16.3 ) and anogenital warts |
| 2 | Common warts; also plantar, oral, and anogenital lesions |
| 3 | Plane warts (verruca planae) and epidermodysplasia verruciformis |
| 4 | Plantar and common warts |
| 5 | Epidermodysplasia with subsequent development of carcinoma |
| Psoriatic skin lesions (detected in research setting) | |
| 6 | Anogenital warts and epidermodysplasia |
| Condyloma acuminatum (other types detected include HPV-2, 6, 11, 16, 18, 31, 33, 35, 39, 41-45, 51, 56, 59, and others) (see Fig. 16.4 ) | |
| Oral and laryngeal squamous papilloma | |
| Verrucous carcinoma | |
| 7 | Warts in meat and fish handlers |
| 8, 9, 10, 12, 14, 17, 19, 22, 24, and others | Epidermodysplasia |
| 10 | Plane warts |
| 11 | Anogenital lesions (e.g., condyloma acuminatum) |
| Oral and laryngeal squamous papilloma | |
| Verrucous carcinoma | |
| 16 and 18 | Bowenoid papulosis (which sometimes progresses to invasive carcinoma) |
| Anogenital warts | |
| Cervical squamous dysplasia and invasive squamous cell carcinoma (types 16, 18, 31, 33, and 35) | |
| Cervical AIS and invasive cervical adenocarcinoma (type 18 > type 16) | |
| Squamous cell carcinoma in other nongenital sites | |
| Verrucous carcinoma | |
| 13 and 32 | (Oral) Focal epithelial hyperplasia (Heck disease) |
| 57 | Plantar epidermoid cysts and nail dystrophy |
| 60 | Plantar warts and epidermal cysts |
| 63, 65, 66 | Plantar warts |
| 75, 76, 77 | Common warts in immunosuppressed patients |
HPV was first recognized to have oncogenic potential in patients with epidermodysplasia verruciformis. This is an autosomal recessive condition, associated with a gene locus on chromosome 17, that leads to decreased defenses against several HPV subtypes (HPV-2, 3, 5, 10, and 8), resulting in exophytic lesions on the trunk and upper extremities which appear in the first decade of life. These lesions may undergo malignant transformation into squamous cell carcinoma during young adulthood. Unlike common warts caused by HPV, epidermodysplasia verruciformis lesions are not transmissible to healthy individuals. Lesions resembling epidermodysplasia verruciformis are sometimes seen in organ transplant recipients.
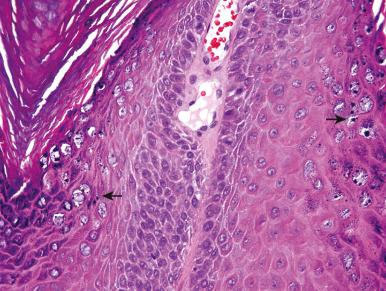
Condyloma acuminatum is one of the most common sexually transmitted diseases. In males, the frenulum, corona, glans, shaft, and scrotum are affected. Multiple lesions may become confluent, forming a cobblestone appearance. Microscopically, condyloma acuminatum typically has some degree of epidermal hyperplasia or hyperkeratosis, acanthosis, parakeratosis, and numerous koilocytes which cytologically are characterized by perinuclear clearing and wrinkled or crenated (“raisinoid”) nuclei ( Fig. 16.4 ). Condyloma acuminatum is usually caused by HPV types 6 and 11, and there is little risk of squamous cell carcinoma from infection with these HPV types. HPV types 16 and 18 are the high-risk types, leading to anogenital squamous intraepithelial lesions and squamous cell carcinomas (e.g., penile squamous cell carcinoma, cervical carcinoma; Fig. 16.5 ). HPV 16 and 18 account for more than 70% of all malignancies in the anogenital region and are present in more than 98% of cases of high-grade dysplasia and squamous cell carcinoma of the cervix. After treatment, latent HPV in perilesional skin may lead to recurrent lesions.
Generic intraepithelial neoplasia terminology has been defined by consensus recommendations in the Lower Anogenital Squamous Terminology (LAST) Project, which was cosponsored by the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. In this terminology, “-IN” can be used to refer to intraepithelial neoplasia of particular sites. Thus for particular sites the following designations are used: anus = AIN; perianus = PAIN; penis = PeIN; cervix = CIN; and vagina = VaIN; and, for example, the corresponding designation of -IN3 of each of these sites would be AIN 3, PAIN 3, PeIN 3, CIN 3, and VaIN 3, respectively. In this system, low-grade dysplasia is referred to as grade 1, which is also termed low-grade squamous intraepithelial lesion (LSIL); and grade 2 or 3 is considered to precancerous or high-grade dysplasia, which is also termed a high-grade squamous intraepithelial lesion (HSIL). Thus a three-tiered system formerly used (“-IN1” through “-IN3”) was converted to a two-tiered system (LSIL and HSIL) by the LAST Project.
Immunohistochemistry for p16 can be used as a surrogate marker of HPV infection and can be used in the assessment of dysplasia. So-called “block” p16 positivity, consisting of strong diffuse p16 staining, is considered indicative of an HSIL. If the H&E morphologic differential diagnosis is between negative for dysplasia and low-grade dysplasia (an intraepithelial lesion grade 1 [-IN1]), then p16 should not be used because -IN1 lesions can be p16-negative. Although p16 can be an aid for the diagnosis of HSIL, p16 positivity in and of itself does not define -IN. Ki67 staining has been used in conjunction with p16 staining, and some studies have shown an increased specificity and sensitivity for dysplasia when these stains are used together. Studies have indicated that Ki67 positivity increases with increasing dysplasia.
Bowenoid papulosis due to HPV (mostly type 16) manifests on the anogenital skin as multiple small (2 to 3 mm), pearly papules. Histologically, it resembles squamous cell carcinoma in situ (also known as Bowen disease). Bowenoid papulosis rarely results in invasive carcinoma and often spontaneously regresses. It can usually be distinguished clinically from Bowen disease (also known as erythroplasia of Queyrat).
Verrucous carcinoma (giant condyloma of Buschke and Lowenstein) is sometimes associated with HPV. It is a slow-growing, well-differentiated variant of squamous cell carcinoma with an exophytic papillary appearance, typically 1 to 3 cm in diameter, that arises in the skin (usually the genital skin), oral cavity, larynx, and esophagus. It is defined by a broad base between the tumor and the underlying stroma with minimal atypia and mitoses at the base, although focal stromal invasion may be observed; dense chronic inflammation may also be present at this interface. Microscopically, verrucous carcinoma is hyperkeratotic and acanthotic, with orthokeratosis (the presence of keratohyaline granules), parakeratosis, and papillomatosis. Papillae may have fibrovascular cores, and papillae may also have a central keratin plug with peripherally located tumor cells. Occasional vacuolated (nonkoilocytotic) clear cells may be present. Verrucous carcinoma has been associated with HPV-6, 11, 16, and 18, although koilocytosis is not present. Verrucous carcinoma is sometimes distinguished from giant condyloma by the absence of koilocytic change, and it has been suggested that the term “warty (condylomatous) squamous cell carcinoma” should be used for exophytic warty tumors of the penis and similar lesions that occur on the vulva and are related to HPV-16. These warty carcinomas are described as having more prominent, long, condylomatous papillae and fibrovascular cores with rounded, irregular, jagged bases, and more obvious koilocytotic atypia. Penile warty carcinoma appears to have a better prognosis than typical squamous cell carcinoma.
Both in situ hybridization and immunohistochemistry are available for the detection of HPV in formalin-fixed tissue. In situ hybridization, which is typically considered to be more sensitive, has largely replaced immunohistochemistry for this purpose (except for specific diagnostic purposes in the cervix, discussed later). Immunohistochemistry detects only productive and not latent infections, and it cannot be used to determine the type of virus present, whereas other molecular techniques are useful for some of these purposes.
The perineal skin can be affected by Entamoeba histolytica when infection extends from the anus in a patient with E. histolytica colitis. This can result from spread by anal intercourse and may also involve the penis. Clinically, ulcerated, painful nodules with serpiginous borders result. An amebic ulcer may present as balanoposthitis resistant to antibiotics.
Wuchereria bancrofti , Brugia malayi , and Brugia timori are filarial worms that can involve lymphatic vessels, resulting in obstruction, chronic lymphedema leading to elephantiasis, and chronic inflammation. Up to 25% of the male population are affected in tropical areas. The scrotum is often swollen, erythematous, and tender. Histologically, there may be thickening of the scrotal skin and verruciform epidermal hyperplasia. Vulvar elephantiasis may also occur from lymphatic obstruction by filariform worms.
Schistosoma mansoni is a fluke that can colonize pelvic veins. Adult organisms within abdominopelvic veins can involve surrounding visceral or cutaneous tissue with a granulomatous reaction. Eggs are typically conducted to the bladder lumen and are excreted during micturition. Sinuses, fistulas, and masses may be present. Ulcers and papillary nodules may simulate condyloma acuminata.
Scabies (caused by the mite Sarcoptes scabiei ) can form tunnels under the stratum corneum of the penis, scrotum, buttock, and waist. Immunocompromised patients may have crusted lesions (known as Norwegian scabies). Lice may also infect the genital region.
Acute interstitial nephritis was first thoroughly defined by Councilman in 1898. Before the advent of widespread antibiotic use, acute interstitial nephritis was most commonly caused by bacterial infections, such as scarlet fever (scarletina, caused by Streptococcus spp.) or diphtheria. Currently, acute interstitial nephritis is most commonly observed in the setting of an allergic reaction (such as to a drug ) or acute transplant rejection; however, bacteria and viruses are still important causes of acute interstitial nephritis. Councilman was astute in observing that many cases of acute interstitial nephritis do not arise as a direct infection by organisms filling the kidney but rather from an indirect influence in which inflammatory cells are attracted to the kidney by, as he surmised, “soluble substances that may exert a positive chemotaxis for them.”
Grossly the kidneys are enlarged, soft, and pale due to edema. Microscopically, edema expands the interstitium, and there is usually a mixed inflammatory cell infiltrate composed primarily of lymphocytes and monocytes with a smaller number of plasma cells and eosinophils. Granulomas may be present in rejection, allergic reactions, or sarcoidosis, in which they are usually noncaseating. Tubulitis, a lymphocytic infiltration of tubular epithelial cells, and vasculitis may be present.
Bacterial organisms, particularly Streptococcus , were formerly the common causes of acute interstitial nephritis. Glomerulonephritis was formerly rare compared with interstitial nephritis in streptococcal infections. However, interstitial nephritis due to Streptococcus declined around the beginning of the 20th century. Specific bacteria involving the kidney are discussed in the following paragraphs. In general, acute interstitial nephritis due to bacterial organisms contains a mixed infiltrate of neutrophils, plasma cells, and eosinophils amid interstitial edema and tubular injury.
Cytomegalovirus (CMV), adenovirus, and polyomavirus produce characteristic intranuclear viral inclusions in tubular epithelial cells and occasionally in the endothelium and glomerular epithelium. These infections are particularly important offenders in transplant patients. Adenovirus may produce smudgy intranuclear inclusions that may have a glassy appearance. Intranuclear inclusions may have a distinct halo surrounded by a ring of marginated chromatin (Cowdry A inclusion). Necrotizing tubulointerstitial nephritis due to adenovirus has been reported, notably in patients who are immunosuppressed. Mass-forming lesions and granulomatous inflammation have also been reported. Immunofluorescence or immunohistochemistry for viral antigens may aid the diagnosis.
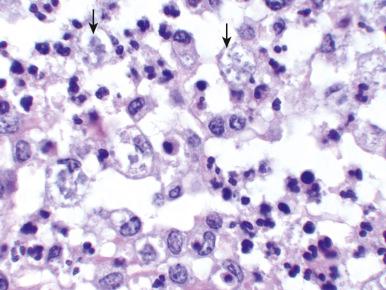
CMV produces large eosinophilic intranuclear inclusions with halos, most often in proximal tubules but also in the loops of Henle and collecting ducts. Microscopically, interstitial inflammation is typically observed to be composed of lymphocytes, plasma cells, and macrophages. CMV-infected renal tubular cells or stromal cells may show enlargement and a single eosinophilic nuclear inclusion surrounded by a clear halo ( Fig. 16.6 ). CMV acute interstitial nephritis can occur as a congenital infection during the neonatal period. Another setting for CMV acute interstitial nephritis is in renal allografts. CMV may result in impairment of renal graft function and glomerular and interstitial injury. Immunohistochemistry for viral antigens may help to solidify the diagnosis.
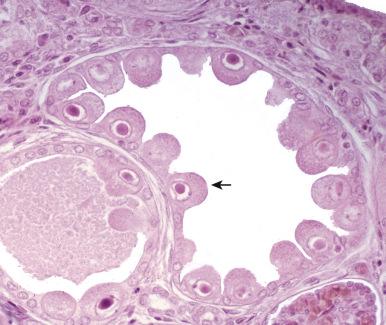
Epstein-Barr virus (EBV), the cause of infectious mononucleosis, may also produce an acute interstitial nephritis. In EBV acute interstitial nephritis, there is usually a prominent lymphocytic infiltrate with plasma cells and macrophages producing tubular injury and necrosis. Lymphocytes may be atypical and are usually CD8 + . Granulomas may be present. It has been argued that EBV acute interstitial nephritis is simply an immunologic phenomenon. In situ hybridization for EBV RNA and immunohistochemistry findings may be negative; however, EBV DNA has been identified on PCR studies in biopsy material involved by EBV-associated acute interstitial nephritis.
Other infectious agents that may cause acute interstitial nephritis include viruses, such as HSV, rubeola virus (measles), and human immunodeficiency virus (HIV); bacteria, including Brucella spp., Legionella , Mycoplasma , and Yersinia ; and parasites, including Toxoplasma gondii and Leishmania (acute visceral leishmaniasis or kala-azar). Only rare cases have been reported of HSV and measles leading to acute interstitial nephritis. Brucella may produce a caseating granulomatous infiltrate with neutrophils, lymphocytes, and interstitial edema. Legionella often has an infiltrate rich in lymphocytes, plasma cells, macrophages, and sometimes even neutrophils. T. gondii can lead to an acute interstitial nephritis rich in lymphocytes and neutrophils. Acute visceral leishmaniasis results in an inflammatory infiltrate of plasma cells and lymphocytes without eosinophils. The renal parenchyma is spared. Patients with HIV infection (AIDS) can have a severe interstitial nephritis composed of an infiltrate rich in lymphocytes and plasma cells. AIDS also predisposes individuals to infections with a number of organisms such as Pneumocystis .
Certain disorders are in the differential diagnosis of granulomatous interstitial nephritis and other granulomatous diseases of the kidney; they include xanthogranulomatous pyelonephritis, malakoplakia, brucellosis, mycobacterial infection, fungal infection, parasitic infection, urate nephropathy, oxalosis, sarcoidosis, vasculitis, drug hypersensitivity, tubulointerstitial nephritis with uveitis syndrome (TINU), granulomatous vasculitis (i.e., Wegener granulomatosis, or the recently preferred term granulomatosis with polyangiitis [GPA]), and cholesterol granulomas. Many infectious agents that may produce a granulomatous interstitial nephritis are listed in Table 16.3 . Ultimately, many cases are idiopathic.
| Causes | Selected Drugs |
|---|---|
| Viral infection | Antibiotics |
|
|
| Bacterial infection | Penicillin G |
|
|
| Others | |
|
|
| Fungal infection | Rifampin |
| Histoplasma | Sulfonamides |
| Parasitic infection | Tetracyclines |
|
Diuretics |
| NSAIDs Allopurinol |
|
| Systemic disorders | Rifampicin |
| Connective tissue disorders | Cimetidine |
| Systemic lupus erythematosus | Sulfa drugs |
| Sjögren syndrome | Phenytoin |
| Sarcoidosis | |
| Transplant rejection | |
| Irradiation | |
| Ischemia | |
| Protein accumulation (e.g., amyloid, light chain, myeloma) | |
| Hereditary (e.g., Alport syndrome, medullary cystic disease, nephrophthisis, familial interstitial nephritis) | |
| Metals (e.g., cadmium, lithium, gold) | |
| Metabolic disorders (e.g., oxalosis, hypercalcemia, gout, cystinosis) | |
| Toxins (e.g., snake bite, CCl 4 , mushroom) | |
| Anti-tubular basement membrane disease | |
| Tubulointerstitial nephritis-uveitis syndrome (Dobrin syndrome) | |
| Idiopathic |
* Organisms, agents, and disorders listed include entities that affect the kidney directly (e.g., through direct infection) or indirectly (e.g., through an immunologic reaction or other reactive state).
Immune complex deposits may be seen on immunofluorescence in tubular basement membranes or peritubular capillaries in cases of interstitial nephritis, but this is a nonspecific finding that merely indicates that an immunologic reaction has taken place. Immunoperoxidase stains may be useful to confirm the presence of adenovirus, CMV, or BK polyomavirus. However, in many cases of acute interstitial nephritis, a specific etiology cannot be determined.
Acute pyelonephritis occurs as a result of infection of the kidney, usually from bacteria. Most pediatric cases occur in boys, and most adult cases occur in women. Infections can be ascending, from lower urinary tract infection, or hematogenous, from sepsis or endocarditis.
Lower urinary tract infections that can involve the kidney include E. coli , followed by Proteus , Klebsiella , and Enterobacter . Vesicoureteral reflux in cases of bacterial cystitis can lead to pyelonephritis. Congenital predisposition toward vesicoureteral reflux leads to more frequent cases of pyelonephritis. Incompetence of the vesicoureteric junction causes congenital (primary) reflux. Some cases of congenital obstruction are familial. Reflux may also result from neurogenic bladder, cystitis, or other causes of distal obstruction. Intrarenal reflux leads to infection of the kidney.
Hematogenous infections involve organisms such as S. aureus and are usually composed of numerous small abscesses scattered throughout the parenchyma, particularly the cortex; this is sometimes termed multiple cortical abscesses, diffuse suppurative nephritis, or diffuse bacterial nephritis. This process is sometimes considered distinct from acute pyelonephritis. Prolonged infectious endocarditis or sepsis is a common cause of hematogenous pyelonephritis, and gram-positive bacteria and fungi are the most common culprits in these instances. Pyonephrosis is the term used for near-total destruction of an obstructed kidney by acute pyelonephritis. Pyonephrosis or renal abscesses may lead to perinephric abscesses.
Clinically the patients are often febrile with flank pain and have leukocytosis. Urinalysis usually shows pyuria and white blood cell casts. Treatment is usually medical with antibiotic therapy, although surgical intervention with either débridement or nephrectomy may be required in severe cases.
Grossly the kidneys are often yellow-white with suppurative foci and sometimes frank abscesses, particularly in ascending acute pyelonephritis. The abscesses may be smaller and scattered, with a concentration in the cortex, in the hematogenous form. Microscopically, there are numerous neutrophils in the tubules and interstitium. Neutrophils and bacteria can be seen in the tubules and collecting ducts of the cortex and medulla ( Fig. 16.7 ). Glomeruli are affected only in severe cases in which there is extensive parenchymal destruction with abscess formation. Vascular thromboses, ischemic necrosis, and cyst formation may be seen.
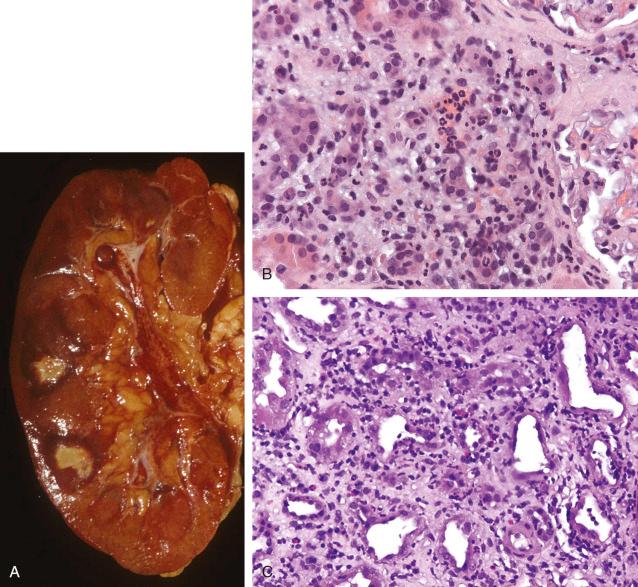
Acute focal bacterial nephritis (lobar nephronia) is a rare form of renal infection. It typically involves the renal cortex and may manifest as a renal mass and be mistaken for a neoplasm, even after cytologic examination. It is thought to be the early stage of an intrarenal abscess. Clinically, flank pain, fever, leukocytosis, pyuria, and bacteriuria may be present; all of these features may also be present in renal abscesses or acute pyelonephritis. Radiologic studies may detect lobar nephronia as focal imaging abnormalities. Microscopically, microabscesses with foci of hemorrhage and necrosis can be appreciated. E. coli is thought to be the most common causative organism, but other organisms have also been implicated, including S. aureus and Klebsiella pneumoniae . Lobar nephronia has been reported in renal allografts.
Emphysematous pyelonephritis is an uncommon, life-threatening condition in which bubble-like collections of gas develop in the renal parenchyma, particularly in areas of coagulative necrosis, and extend into the perinephric space or retroperitoneum or both. The emphysematous foci may be cystic and multiple. Approximately 90% of the patients have diabetes mellitus, and signs of diabetic glomerulosclerosis may be present. Urinary tract obstruction is present in approximately 40% of patients. Cortical and papillary necrosis and vascular thromboses may result. E. coli is the most common organism (approximately 68% of the cases), and Klebsiella is present in approximately 9% of patients. Mixed infections are detected in 19% of patients. The infectious route is usually thought to be hematogenous, and gas-forming bacteria, high tissue glucose levels, decreased tissue perfusion, and an inadequate immune response are the four factors that are considered important in the development of emphysematous pyelonephritis. Emphysematous pyelonephritis has also been described in the parenchyma of tumors. Surgical intervention is ultimately required because antibiotics are usually not effective.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here