Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter addresses general concepts on MRI of the bone marrow, with emphasis on common normal and abnormal marrow patterns. MRI plays a key role in marrow imaging because of its high sensitivity for detecting focal or diffuse alterations in marrow content. MRI of the bone marrow is also performed for other purposes, including medullary lesion characterization, disease staging, prognosis, and monitoring of treatment response.
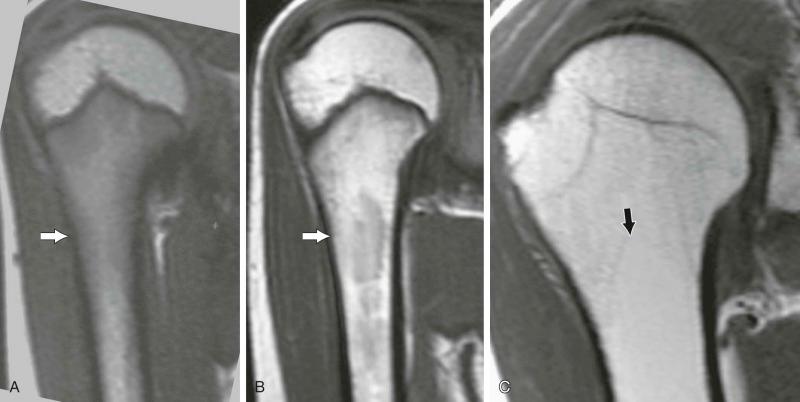
MRI of the skeleton yields information on the medullary content of the bones ( Fig. 71-1 ). The calcified components of the skeleton—either cortical or cancellous—are best depicted by using ionizing imaging modalities (radiographs, CT, and bone scintigraphy). Therefore, it is not surprising that MR and CT images of the same lesion occasionally yield different information ( eFig. 71-1 ).
The cellular components of bone marrow, which are critical for maintaining oxygenation, immunity, and coagulation, include stem cells, erythrocytes, myeloid cells, and megakaryocytes. They are found around a network of cancellous bony trabeculae, lined by a fibrous tissue reticulum, whereas the remaining marrow space is occupied by fat. The medullary cavity of the adult human skeleton contains red and yellow marrow.
In adults, red marrow occupies the cranial vault, the spine, the ribs, the sternum, the pelvic region, and the proximal aspects of the femur and humerus. Red marrow contains about 40% to 50% fat cells and 50% to 60% hematopoietic cells embedded in a network of highly permeable sinusoids ( eTable 71-1 ), with a chemical composition of 40% to 60% lipids, 30% to 40% water, and 10% to 20% proteins. The relative proportion of fat and nonfat cells varies among individuals according to poorly understood parameters but also according to age and sex; it also depends on the bone considered and on the anatomic region of each individual bone ( eFig. 71-2 ).
| Yellow Marrow | Red Marrow | |
|---|---|---|
| Chemical composition | 80% lipids, 15% water | 40% lipids, 40% water |
| Main cellular composition | Fat cells | Hematopoietic and fat cells |
| Vasculature | Few capillaries | Permeable sinusoids |
| Main distribution | Appendicular skeleton | Axial skeleton |
Yellow marrow almost exclusively contains fat cells (95%) and few capillaries, with a chemical composition of 80% of lipids, 15% water, and 5% proteins. It is mainly found in the appendicular skeleton of human adults. All long-bone epiphyses, except the proximal humeral and femoral epiphyses, contain yellow marrow.
The possibility of red marrow converting to yellow marrow and, conversely, of yellow marrow transforming into into red marrow is a unique feature of the marrow. It occurs as a physiologic process during growth and until adulthood: At birth, red marrow occupies almost the entire skeleton. It progressively converts to yellow marrow, in a predictable manner, starting distally in the limbs and progressing in a symmetric, centripetal pattern in the long bones. In the late third decade of life, the distribution of bone marrow attains its mature state, where red marrow can be found in the axial skeleton and in the proximal metaphyses of the femurs and humerus ( eTable 71-2 ).
| Green Sign | Red Sign | |
|---|---|---|
| Children and adolescents | Homogeneous distribution | Heterogeneous distribution |
| Adults and elderly | Red marrow in the metadiaphyseal region | Red marrow in the epiphyses |
Yellow marrow may reconvert into red marrow if there is demand for more hematopoietic cells, which can be seen with benign conditions, such as in heavy smokers, marathon runners, obesity, or middle-aged women, and also with chronic disorders associated with anemia. The reconversion of yellow to red marrow occurs in the opposite sequence of the initial conversion, that is, the expansion of red marrow occurs from the central to the peripheral skeleton. In the long bones, it takes place first in the proximal metaphyses, then in the distal metaphyses, and finally in the diaphyses.
The T1-weighted spin-echo (SE) sequence is the most important sequence for bone marrow MRI. It is able to depict the wide changes in normal red marrow distribution and composition that occur with aging because the marrow signal intensity is directly proportional to the amount of fat-laden adipocytes in the marrow cavity, whereas bony trabeculae contribute little to bone marrow signal intensity.
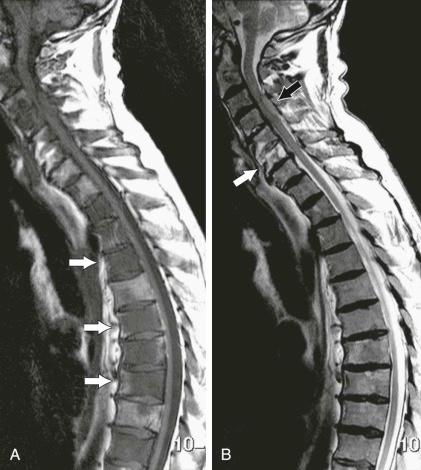
The rationale for its use as the main sequence for marrow imaging is that it is able to demonstrate changes in the numbers of fat cells that occur in almost any abnormal marrow condition ( eFig. 71-3 ). Consequently, this sequence lacks specificity because it depicts the disappearance of fat and not the concomitant appearance of abnormal cells. In addition, the T1-weighted SE sequence is widely available, and it is reproducible over time and in different imaging centers.
Normal adult red marrow presents an intermediate signal intensity that is higher than the intervertebral disk or muscle, which is related to complex interactions between almost equal quantities of fat and water and a smaller contribution from a protein component. Any signal intensity lower than that of muscle or intervertebral disk is abnormal.
The intermediate-weighted SE sequence without fat saturation has no role to play in marrow imaging because many marrow constituents show a similar intermediate signal intensity.
The T2-weighted fast SE sequence has a limited value for lesion detection but can contribute to lesion characterization. However, it is generally obtained mainly for the assessment of adjacent structures (see eFig. 71-3 ).
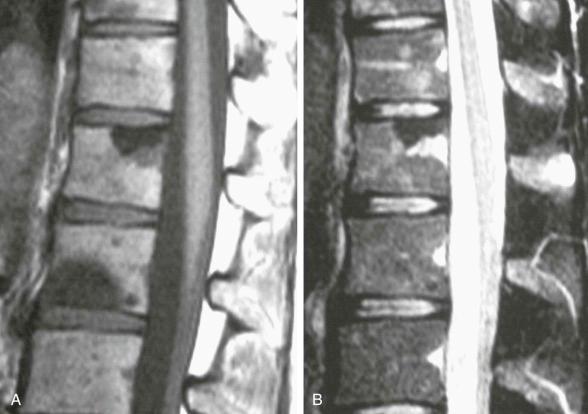
Many marrow lesions do not significantly alter the amount of marrow water, and the lesion's signal intensity widely varies according to poorly understood parameters ( eFig. 71-4 ).
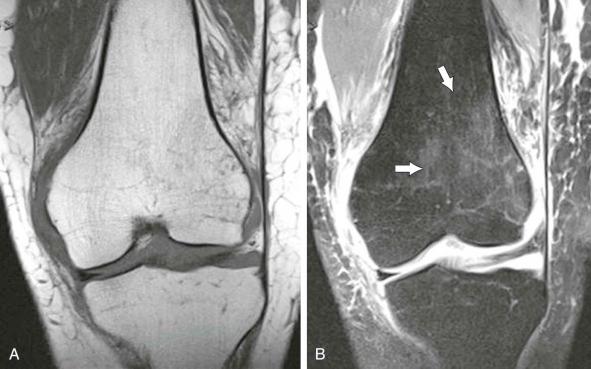
Fat saturation plays an important role in bone marrow imaging. Actually, fat protons contribute so heavily to the medullary signal on T1- and T2-weighted SE images that they occasionally decrease MR sensitivity for the detection of abnormal marrow components. Therefore, the application of techniques that decrease the influence of fat protons on signal intensity is likely to facilitate lesion detection ( eFig. 71-5 ).
The short tau inversion recovery (STIR) sequence, in which the inversion time is selected to suppress fat contribution, provides critical information because the signal of fat is suppressed, and T1 and T2 contrast are additive. Fat saturation can also be achieved by selective presaturation of hydrogen protons from fat molecules and can be followed by T1-, intermediate-, or T2-weighted sequences. STIR sequences and fat-saturated intermediate-weighted sequences give similar results in the detection of subtle marrow alterations.
As a significant drawback, the specificity of fat-saturated sequences is lower than that of T1- and T2-weighted SE sequences, partly because several tissue components that can be recognized on T1- or T2-weighted SE images will present a less specific signal intensity after fat saturation ( eFig. 71-6 ).
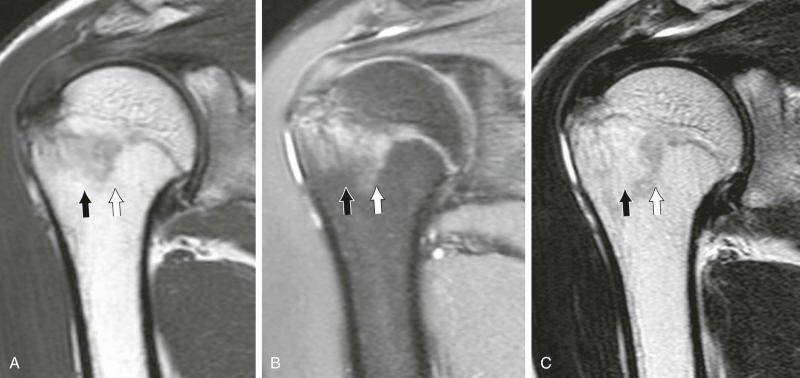
Gradient-echo sequences play a limited role in marrow imaging. The network of cancellous bone is responsible for local field inhomogeneities that account for a marked decrease in signal intensity of cancellous bone–containing areas on T2 * -weighted gradient-echo sequences. These sequences are occasionally used for the detection of purely lytic lesions and also for the selective evaluation of the cancellous bone network, such as in osteoporosis.
Normal adult red and yellow marrow displays no significant enhancement after intravenous gadolinium injection that can be appreciated by the naked eye on T1-weighted images. A measurable signal intensity difference of 15% can be detected when the interstices of the adult bone marrow are measured. Significant enhancement of highly cellular marrow can be found in infants and children in the first few years of life.
In addition, bone marrow disease tends to enhance markedly, so gadolinium-enhanced sequences may be used to differentiate normal signal enhancement in questionable red marrow areas or abnormal enhancement for lesion characterization.
Gadolinium-enhanced T1-weighted SE sequence is also recommended for intradural lesion detection, but it is generally not necessary for bone marrow lesion detection, at least without fat saturation, because many lesions become invisible on this sequence. If fat saturation is applied, marrow lesions become more conspicuous.
There is general agreement that fat-saturated gadolinium-enhanced T1-weighted SE images and fat-saturated intermediate-weighted SE images are equivalent for the detection of subtle marrow changes that are not obvious on T1- and T2-weighted SE images.
Diffusion-weighted imaging (DWI) is a noninvasive imaging technique that permits study of the physical structure of a biologic tissue at a microscopic level, below the typical millimeter-scale resolution of standard MRI clinical sequences. This sequence takes advantage of the random translational motion of water protons in biologic tissue, reflecting the specific tissue capacity, which can be used for tissue characterization. The signal loss with diffusion-weighted imaging is proportional to the diffusion strength used and the mobility of the water protons, which, in turn, is proportional to the temperature and diffusion capacity of the tissue under examination.
The degree of restriction to water diffusion in biologic tissues is inversely proportional to the tissue cellularity and the integrity of cell membranes. In rapidly growing tissues with high turnover, such as within tumoral processes, barriers to diffusion are increased, resulting in restricted diffusion and in a decline in the diffusion capacity.
The sensitivity of diffusion-weighted sequences to water motion can be varied by changing the gradient amplitude, the duration of the applied gradient, and the time interval between the gradients. The diffusion sensitivity is changed by modifying the “b-value.” In general, the larger the b-value, the greater the degree of signal attenuation from water molecules.
This sequence can also provide a qualitative and quantitative evaluation. A qualitative analysis may be performed by observing the relative attenuation of signal intensity on the images obtained at different b-values, whereas the quantitative analysis can be carried out by calculating automatically, on a workstation, the apparent diffusion coefficient (ADC). The ADC is calculated for each pixel of the image and displayed as a parametric map. Then, following regions of interest (ROI) drawn on these maps, the ADCs of different tissues can be obtained. Areas of restricted diffusion in highly cellular areas present high signal intensity on diffusion-weighted images, but low ADC values. On the other hand, less cellular areas will appear with low signal intensity on diffusion-weighted imaging, yet with higher ADC values.
It is essential to remember as well that normal structures, such as the spleen, prostate, testes, ovaries, endometrium, and spinal cord remain visible with this technique and will be depicted together with tumoral areas, cellular lymph nodes, edema, and infectious lesions that present restricted diffusion. Besides, although lytic lesions are well depicted with diffusion-weighted images, commonly, complete osteoblastic lesions are not identified. Consequently, DWI is not suitable for evaluating vertebral fractures associated with sclerotic metastases or to examine the appearance of sclerotic bone after treatment with chemotherapy, radiotherapy, or bisphosphonates. Another pitfall that must be remembered is that a false-positive diffusion-weighted hyperintensity may be present in benign fractures, which can be attributed to extensive edema and/or hemorrhage. Therefore, it is paramount to compare DWI with standard MRI sequences, to accurately depict pathology or rule out artifacts or a normal finding. Thus, it should be combined with routine MR sequences, initially using DWI to screen the whole body for a suspect lesion, increasing sensitivity. Localized T1-, T2-weighted, STIR, and contrast-enhanced T1-weighted sequences should then be added for better specificity ( eFig. 71-7 ).
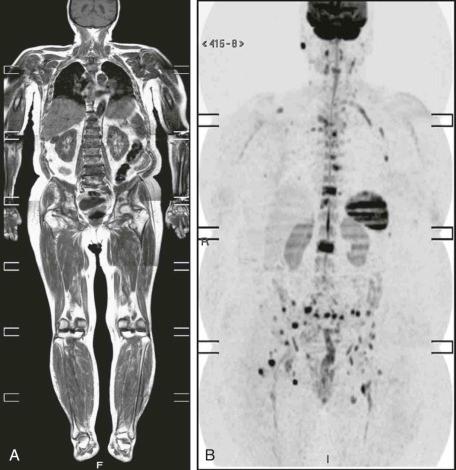
The main drawbacks of this technique are its greater sensitivity to motion, such as cardiac and respiratory movements; the lower anatomic detail of the sequence; and the higher imaging time for acquisition.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here