Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Hemolysis is a reduction in the normal red cell survival of 120 days. A general approach to diagnosing hemolytic anemia in the context of other causes of anemia is described in Chapter 3 , Classification and Diagnosis of Anemia in Children and Neonates. It may result from corpuscular abnormalities such as membrane, cytoskeleton, enzyme, or hemoglobin (Hb) defects, or from extracorpuscular abnormalities involving immune or nonimmune mechanisms (see Chapter 8 : Extracorpuscular Hemolytic Anemia).
The following clinical features suggest a hemolytic process in a child with anemia:
history of anemia, jaundice, or gallstones in family
persistent or recurrent anemia associated with reticulocytosis
anemia unresponsive to hematinics
intermittent bouts or persistent indirect hyperbilirubinemia/jaundice
splenomegaly
hemoglobinuria
presence of multiple gallstones
chronic leg ulcers
development of anemia or hemoglobinuria after exposure to certain drugs
cyanosis without cardiorespiratory distress
polycythemia (2,3-diphosphoglycerate mutase deficiency)
dark urine due to dipyroluria (unstable hemoglobins, thalassemia, and ineffective erythropoiesis)
ethnic factors:
Incidence of sickle gene carrier in the African-American population (8%)
High incidence of thalassemia trait in people of Mediterranean ancestry
High incidence of glucose-6-phosphate dehydrogenase (G6PD) deficiency among those with ethnic origins arising in territories near the Mediterranean Sea, Africa, and Southeast Asia
High incidence of hereditary ovalocytosis in Southeast Asian populations
age factors: anemia and jaundice in a Rh-positive infant born to a mother who is Rh negative or a group A or group B infant born to a group O mother (setting for a hemolytic anemia).
Laboratory findings of hemolytic anemia consist of:
evidence of accelerated hemoglobin catabolism due to reduced red cell survival and
evidence of increased erythropoiesis.
Accelerated hemoglobin catabolism varies with the type of hemolysis as follows:
extravascular hemoglobin catabolism (see Fig. 7.1 )
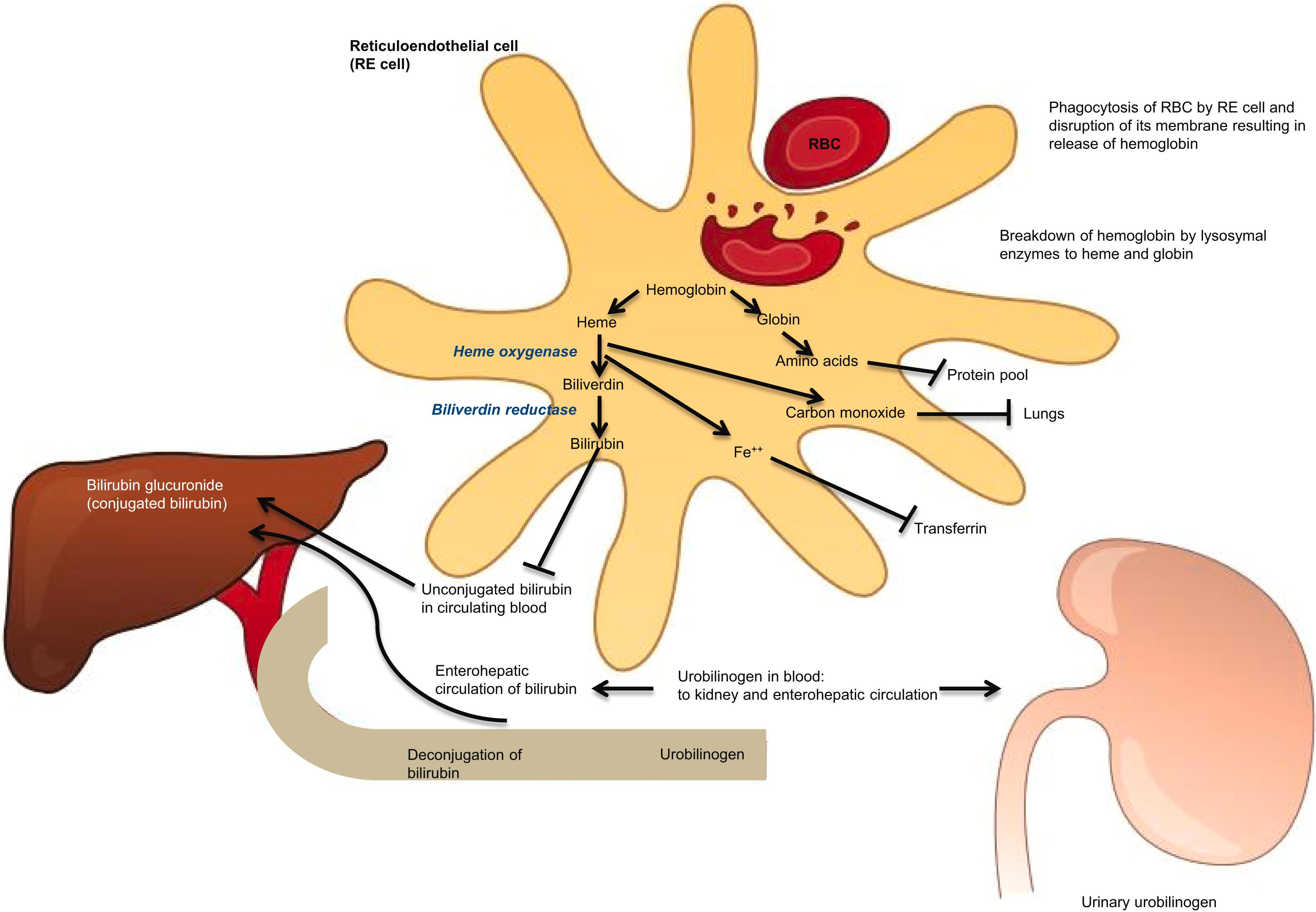
intravascular hemoglobin catabolism (see Fig. 7.2 ).
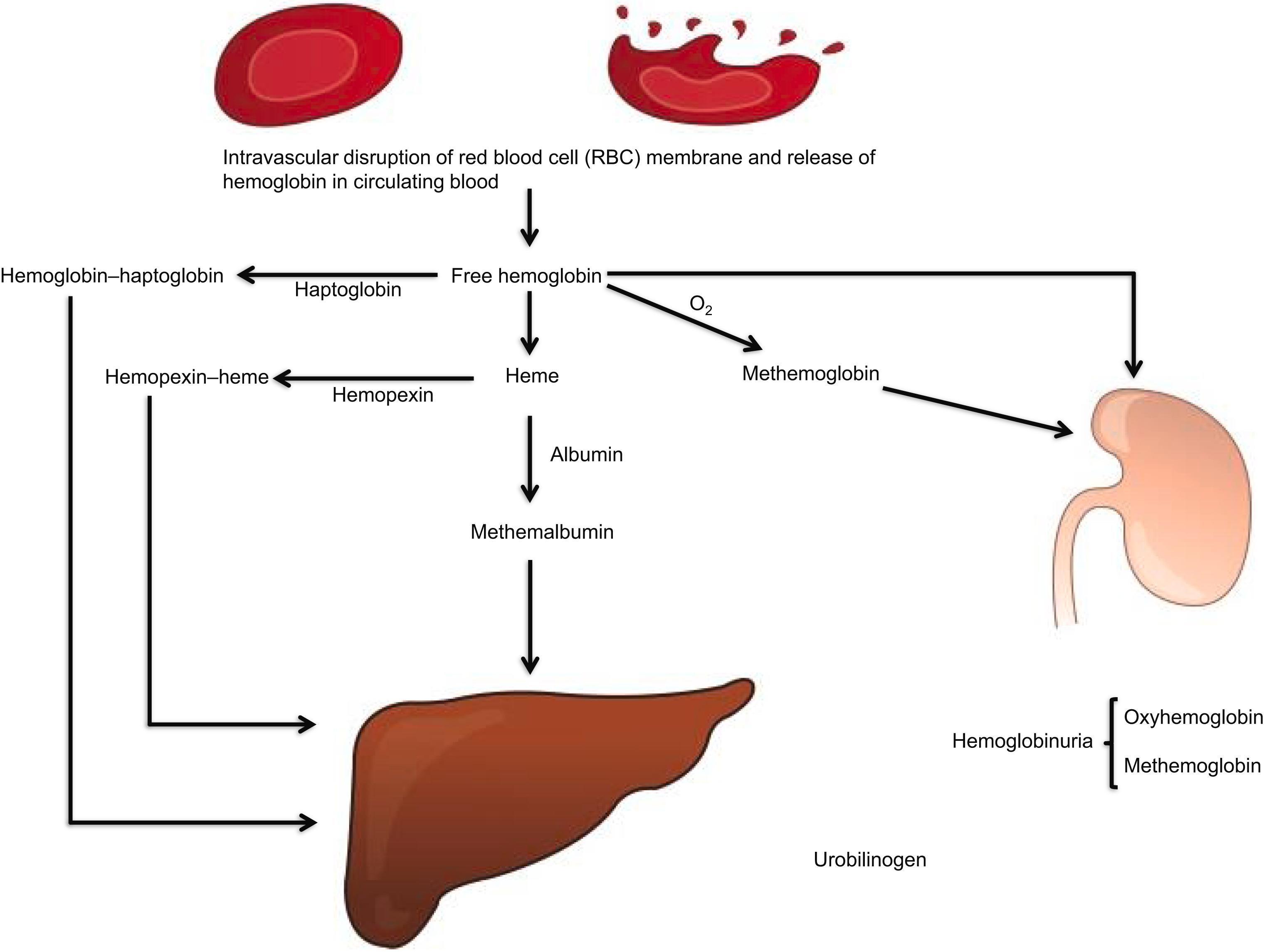
The two may not be easily distinguished if the cause for hemolysis is not obvious, hence the long lists of markers of testing indicated next. The presence of hemoglobinuria and hemosiderinuria and the absence of haptoglobin are the major markers of intravascular hemolysis in practice.
Increased unconjugated bilirubin
Increased lactic acid dehydrogenase in serum
Decreased plasma haptoglobin (normal level, 36–195 mg/dL)
Increased fecal and urinary urobilinogen
Increased rate of carbon monoxide production.
Increased unconjugated bilirubin (although often less than extravascular hemolysis as urinary losses leave less hemoglobin to be scavenged and processed to bilirubin)
Increased lactic acid dehydrogenase in serum
Hemoglobinuria ( Fig. 7.3 lists the causes of hemoglobinuria)

Low or absent plasma haptoglobin
Hemosiderinuria (due to sloughing of iron-laden tubular cells into urine)
Raised plasma hemoglobin level (normal value <1-mg hemoglobin/dL plasma, visibly red plasma contains greater than 50-mg hemoglobin/dL plasma)
Raised plasma methemalbumin (albumin bound to heme; unlike haptoglobin, albumin does not bind intact hemoglobin)
Raised plasma met hemoglobin (oxidized free plasma hemoglobin) and raised levels of hemopexin–heme complex in plasma
Erythropoiesis increases in response to a reduction in hemoglobin and is manifested by:
Reticulocytosis : frequently up to 10–20%; rarely, as high as 80%
Increased mean corpuscular volume (MCV) due to the presence of reticulocytosis
Increased red cell distribution width (RDW) as the hemoglobin level falls
Normoblasts in the peripheral blood
Specific morphologic abnormalities : sickle cells, target cells, basophilic stippling, irregularly contracted cells or fragments (schistocytes), elliptocytes, acanthocytes, and spherocytes
Erythroid hyperplasia of the bone marrow : erythroid:myeloid ratio in the marrow increasing from 1:5 to 1:1
Expansion of marrow space in chronic hemolysis resulting in:
Prominence of the frontal bones and broadened cheekbones
Widened intratrabecular spaces with hair-on-end appearance of skull radiographs
Biconcave vertebrae with fish-mouth intervertebral spaces
Decreased red cell survival demonstrated by 51 Cr red cell labeling
Red cell creatine levels increased
Fig. 7.4 lists the tests used to establish the cause of the hemolytic anemia.
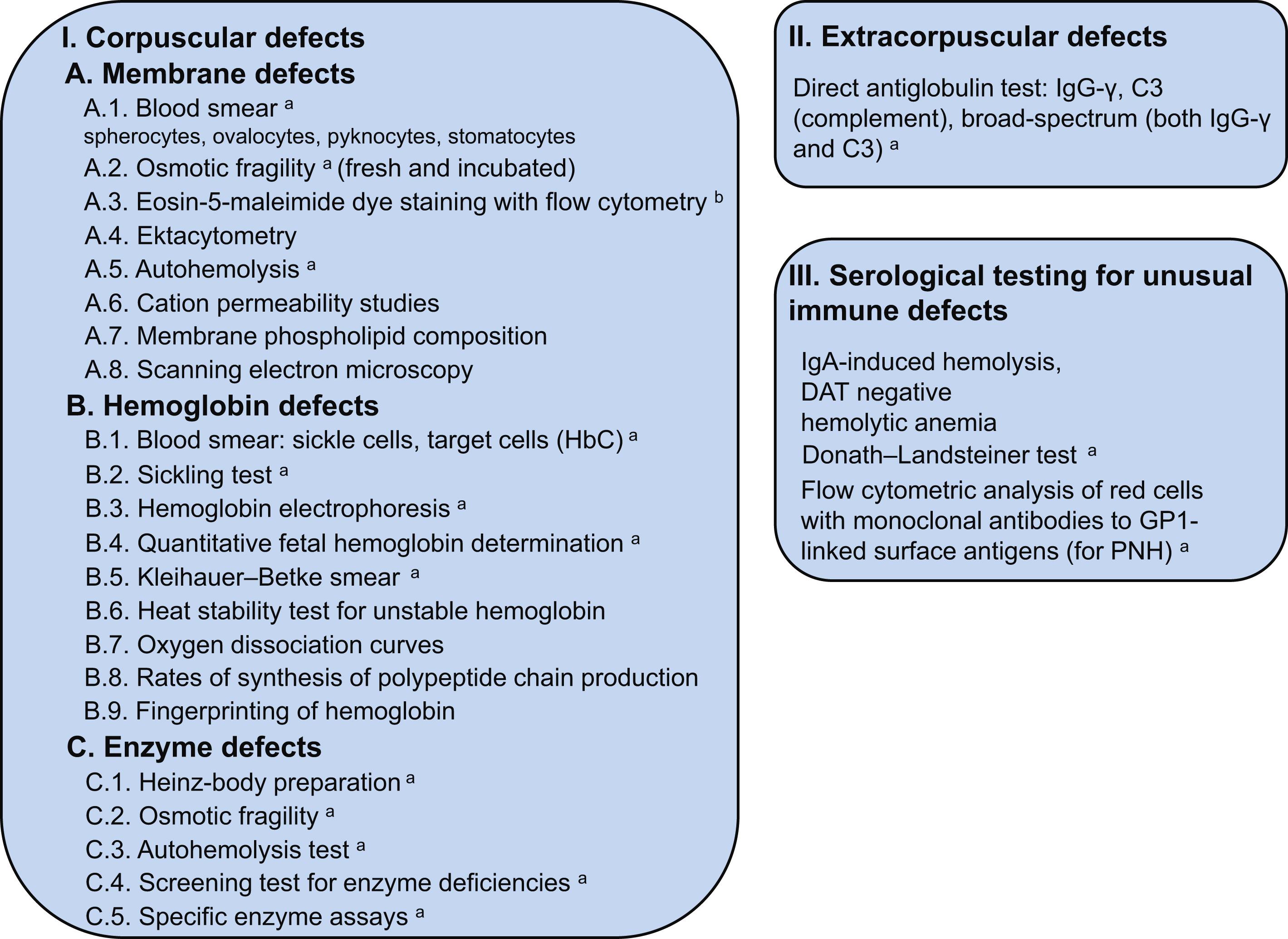
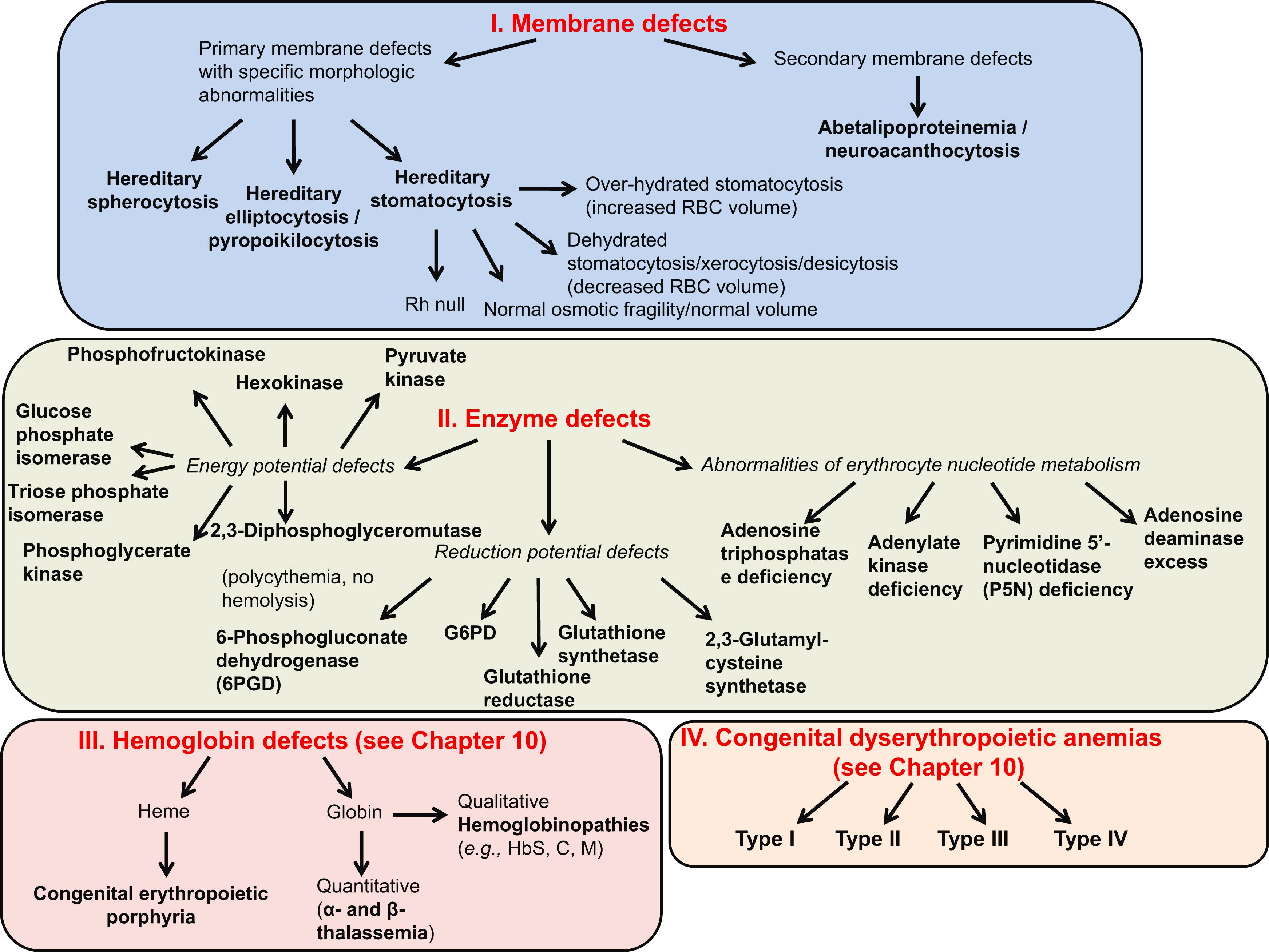
Fig. 7.5 lists causes of hemolytic anemia due to corpuscular defects.
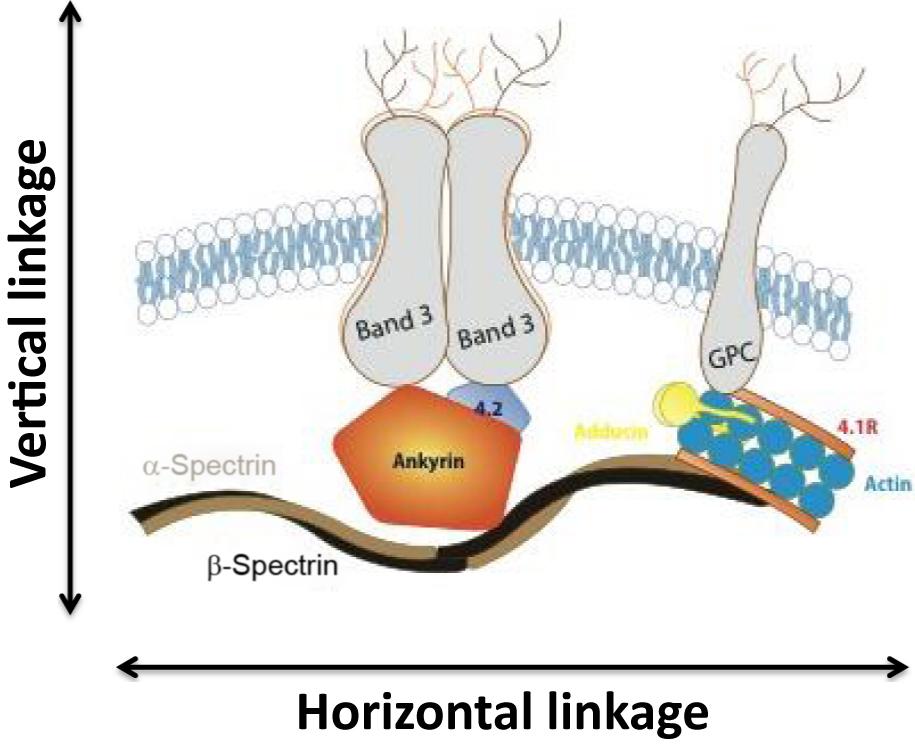
Spectrin, the major red cell membrane protein, is largely responsible for maintaining the normal red cell shape and overall morphology. It is composed of two large subunits, α- and β-spectrin, which are encoded by separate genes and are structurally distinct. Spectrin is integrated vertically into the lipid bilayer of the red cell membrane through the intercession of smaller proteins (ankyrin, 4.2) to integral membrane-spanning proteins (band 3, Rh antigen, and glycophorin A). These vertical interactions maintain red cell membrane cohesion. Spectrin associates with itself head to head, while the tail associates with actin and other members of the junctional complex (4.1R, adducin). These horizontal interactions maintain membrane stability. Fig. 7.6 summarizes the structure of the normal red cell membrane, highlighting the vertical and horizontal interactions.
The red cell membrane is semipermeable and must maintain its volume in order for the erythrocyte to negotiate the narrower spaces in the circulatory system. Red cell volume is maintained by a number of passive, gradient-driven cation and anion channels as well as active transporters.
Hereditary spherocytosis (HS), elliptocytosis, stomatocytosis, acanthocytosis, xerocytosis, and pyropoikilocytosis can be diagnosed on the basis of their characteristic morphologic abnormalities.
Alterations in the quality and/or quantity of the proteins involved in the maintenance of the unique properties of the red cell membrane (deformability and stability) lead to the red cell membrane disorders:
HS: perturbations in the vertical linkage
Hereditary elliptocytosis (HE): perturbations in the horizontal linkage
Stomatocytosis: perturbations in the function of the ion transporters.
Note : Enzyme defects and many hemoglobinopathies have nonspecific morphologic abnormalities related to secondary effects on red cell membrane proteins and pumps [e.g., adenosine triphosphate (ATP) depletion].
Autosomal dominant inheritance (75% of cases). The severity of anemia and the degree of spherocytosis may not be uniform within an affected family
No family history in 25% of cases. Some show minor laboratory abnormalities suggesting a carrier (recessive) state. Others are due to a de novo mutation
Most common in people of northern European heritage, with an incidence of 1 in 5000.
The primary defect is membrane instability due to dysfunction or deficiency of a red cell skeletal or membrane protein, including:
Ankyrin mutations account for 50–60% of HS. In many patients, both spectrin and ankyrin proteins are deficient. Mutations of ankyrin occur in both dominant and recessive forms of HS. The clinical course varies from mild to severe. Red cells are typically spherocytes
α-Spectrin mutations occur in recessive HS and account for less than 5% of HS. The clinical course is severe. Contracted cells, poikilocytes, and spherocytes are seen
β-Spectrin mutations occur in dominant HS and account for 15–20% of HS. The clinical course is mild to moderate. Acanthocytes, spherocytic elliptocytes, and spherocytes are seen
Protein 4.2 mutations occur in the recessive form of HS and account for less than 5% of HS. The clinical course is mild to moderate. Spherocytes, acanthocytes, and ovalocytes are seen
Band 3 mutations occur in the dominant form of HS and account for 15–20% of HS. The clinical course is mild to moderate. Spherocytes are occasionally mushroom-shaped or pincered cells. The deficiency of these proteins in HS results in a vertical defect, which causes progressive loss of membrane lipid and surface area. The loss of surface area results in characteristic microspherocytic morphology of HS red cells. The sequelae are as follows:
Depletion of membrane lipid
Decrease in membrane surface area relative to volume, resulting in a decrease in surface-area-to-volume ratio
Tendency to spherocytosis
Influx and efflux of sodium increased; cell dehydration
Sequestration of red cells in the spleen due to reduced erythrocyte deformability
Rapid ATP utilization and increased glycolysis leading to increased loss of surface area under ATP-depleted conditions. This leads to the observation of splenic conditioning where the changes in glucose utilization, as well as cell volume control, are dramatically exacerbated with each circulatory passage through the spleen.
Premature red cell destruction.
Anemia : mild to moderate when there is a compensated hemolytic anemia. In erythroblastopenic (aplastic or hypoplastic) crisis, hemoglobin may drop to 2–3 g/dL
MCV usually decreased, mean corpuscular hemoglobin concentration (MCHC) raised, and RDW elevated.
The MCHC is raised in HS, hereditary xerocytosis, hereditary pyropoikilocytosis (HPP), pyruvate kinase (PK) deficiency (which has acquired xerocytosis), and cold agglutinin disease. The presence of elevated RDW and MCHC (performed by aperture impedance instruments, e.g., Coulter) makes the likelihood of HS very high, because these two tests used together are very specific for HS
Reticulocytosis (3–15%)
Blood smear : spherocytes, microspherocytes (vary in number); hyperdense cells with or without polychromasia. The percentage of microspherocytes is the best indicator of the severity of the disease but not a good discriminator of the HS genotype. Hyperdense cells are seen in HbSC disease, HbCC disease, and xerocytosis. In HS, hyperdense cells are a poor indicator of disease severity but an effective discriminating feature of the HS phenotype
Direct antiglobulin test (DAT) negative
Increased red cell osmotic fragility (spherocytes lyse in higher concentrations of saline than normal red cells) occasionally only demonstrated after incubation of blood sample at 37°C for 24 h (therefore always do this test incubated). In spite of normal osmotic fragility, increased MCHC or an increase in hyperdense red cells is highly suggestive of HS
Autohemolysis at 24 and 48 h increased, corrected by the addition of glucose
Survival of 51 Cr-labeled cells reduced with increased splenic sequestration
Marrow: normoblastic hyperplasia; increased iron
Eosin-5-maleimide dye staining of red cells and analysis by flow cytometry is the test of choice to diagnose HS but is only available in special reference laboratories
Genetic analysis for the α- and β-spectrin, ankyrin, and band 3 mutations is available, but rarely necessary to be performed for diagnosis.
Indirect hyperbilirubinemia
Obstructive jaundice with increased direct bilirubin; may develop due to gallstones, a consequence of increased pigment excretion.
Anemia and jaundice: severity depends on rate of hemolysis, degree of compensation of anemia by reticulocytosis, and ability of liver to conjugate and excrete indirect hyperbilirubinemia
Splenomegaly
Presents in newborn period in 50% of cases with hyperbilirubinemia, reticulocytosis, normoblastosis, spherocytosis, negative DAT, and splenomegaly. Patients may present with a requirement for transfusion in the first 8 weeks of life that may not be reflective of their ultimate clinical severity
Presents before puberty in most patients
Diagnosis sometimes made much later in life, often after the birth of an infant with neonatal jaundice caused by HS
Coinheritance of HS with HbSC disease may increase the risk of splenic sequestration crisis
Coinheritance of α- or β-thalassemia trait and HS has been reported to have variable effects on hemolysis
Iron deficiency may correct the laboratory values (artificially reducing the MCHC, etc.) but not the red cell life span in HS patients
HS with other system involvement:
interstitial deletion of chromosome 8p11.1–8p21.1 causes ankyrin deficiency, psychomotor retardation, and hypogonadism,
HS may be associated with neurologic abnormalities such as cerebellar disturbances, muscle atrophy, and a tabes-like syndrome,
in patients presenting with common bile duct obstruction associated with gallstones, the increased cholesterol and triglyceride load from the induced dyslipidemia can correct the membrane defect and the resulting spherocyte morphology, MCHC, and osmotic fragility results, hence masking the diagnostic features of the disease. The removal of bile duct obstruction leads to a reappearance of the disease phenotype.
Table 7.1 lists a classification of HS in accordance with clinical severity and indications for splenectomy.
| Classification | Trait | Mild spherocytosis | Moderate spherocytosis | Severe spherocytosis a |
|---|---|---|---|---|
| Hemoglobin (g/dL) | Normal | 11–15 | 8–12 | 6–8 |
| Reticulocyte count (%) | ≤3 | 3.1–6 | ≥6 | ≥10 |
| Bilirubin (mg/dL) | ≤1.0 | 1.0–2.0 | ≥2.0 | ≥3.0 |
| Reticulocyte production index | <1.8 | 1.8–3 | >3 | |
| Spectrin per erythrocyte b (percentage of normal) | 100 | 80–100 | 50–80 | 40–60 |
| Osmotic fragility | ||||
| Fresh blood | Normal | Normal to slightly increased | Distinctly increased | Distinctly increased |
| Incubated blood | Slightly increased | Distinctly increased | Distinctly increased | Distinctly increased |
| Autohemolysis | ||||
| Without glucose (%) | >60 | >60 | 0–80 | 50 |
| With glucose (%) | <10 | ≥10 | ≥10 | ≥10 |
| Splenectomy | Not necessary | Usually not necessary during childhood and adolescence | Decision based on quality of life | Necessary, not before 5 years of age |
| Symptoms | None | None | Pallor, erythroblastopenic crises, splenomegaly, gallstones | Pallor, erythroblastopenic crises, splenomegaly, gallstones |
Clinical features and family history
Hematologic features.
Hemolytic crisis : with more pronounced jaundice due to accelerated hemolysis (may be precipitated by viral infection)
Erythroblastopenic crisis ( hypoplastic crisis ): dramatic fall in hemoglobin level (and reticulocyte count); usually due to maturation arrest and often associated with giant pronormoblasts in the recovery phase; often associated with parvovirus B19 infection. Parvovirus B19 infects developing normoblasts, causing a transient cessation of production. The virus specifically infects Colony-Forming Unit-Erythroid and prevents their maturation. Giant pronormoblasts are seen in bone marrow. Diagnosis is made by increased IgM antibody titer against parvovirus and polymerase chain reaction for parvovirus on bone marrow
Folate deficiency : caused by increased red cell turnover; may lead to superimposed megaloblastic anemia. Megaloblastic anemia may mask HS morphology as well as its diagnosis by osmotic fragility
Gallstones (in approximately one-half of untreated patients): increased incidence with age, can occur as early as 4–5 years of age. Occasionally, HS may be masked or improved in obstructive jaundice due to increase in surface area of red cells and the coinheritance of Gilbert syndrome markedly increases the incidence of gallstones
Complications of chronic anemia : patients with more severe HS (see Table 7.1 ) may suffer growth retardation, anemic heart failure, and failure to thrive, necessitating intermittent or chronic transfusion
Hemochromatosis: rarely, this may occur more frequently when a restricted or partial splenectomy is carried out (see next)
Splenic rupture: the risk of splenic rupture in HS is similar to that of the normal population. Nonetheless, a patient with a large spleen below the costal margin should be cautioned against contact sports or other activities known to lead to blunt trauma to the abdomen
Folic acid supplement (1 mg/day).
Leukocyte-depleted packed red cell transfusion for severe erythroblastopenic crisis.
Splenectomy for moderate-to-severe cases (see Table 7.1 ). Most patients with less than 80% of normal spectrin content require splenectomy. Splenectomy should be carried out early in severe cases but not before 5 years of age, if possible. The management of the splenectomized patient is detailed in Chapter 5 , Lymphadenopathy and Diseases of the Spleen. Although spherocytosis persists postsplenectomy, the red cell life span normalizes and complications are prevented, especially transient erythroblastopenia and hyperbilirubinemia. There may, however, be an increased risk of arterial and venous thrombosis in later life as well as an increased risk for idiopathic pulmonary hypertension. Patients are at risk of sepsis after splenectomy, especially for those under 5 years of age. In partial splenectomy, up to 90% of the splenic mass is removed with small studies suggesting that the technique leaves enough splenic tissue to protect against infection. The technique is not widely utilized but its use should certainly be entertained in transfusion-dependent patients who are under 5 years of age. It is being offered more frequently as an elective alternative to full splenectomy in patients in the moderate-to-severe category. There may be an increased risk for iron loading in patients with HS who have not undergone splenectomy. Since patients in this situation are unlikely to tolerate phlebotomy, iron overload may make the decision for splenectomy or even partial splenectomy even more complex. Laparoscopic splenectomy is safe in children. Although it requires more operative time than open splenectomy, it is superior with regard to postoperative analgesia, smaller abdominal wall scars, duration of hospital stay, and more rapid return to a regular diet and daily activities. It is not known whether accessory spleens are readily identified with the laparoscope although the magnification afforded by the laparoscope might be advantageous in some cases.
Ultrasound should be carried out before splenectomy to exclude the presence of gallstones. If present, cholecystectomy is also indicated.
HE is clinically and genetically a heterogeneous disorder.
HE is characterized by an autosomal dominant or codominant mode of inheritance with variable penetrance, affecting about 1 in 25,000 of the population. The prevalence of HE is much higher in regions where malaria is endemic. This could be explained by the resistance of elliptocytes to malarial invasion.
Occasionally, patients who are severely affected appear to be the offspring of a family with only a single affected parent. In this case a “silent carrier”–like mutation in an α-spectrin gene of the unaffected parent may be the cause.
HE is due to various defects not only in the skeletal proteins, spectrin, and protein 4.1, but also in the integral protein glycophorin C. The basic membrane defects consist of
defects of spectrin self-association involving the α-chains (65%)
defects of spectrin self-association involving the β-chains (30%)
deficiency of protein 4.1
deficiency of glycophorin C
“silent carrier” effect: α-spectrin mutant genes which produce less α-spectrin when paired with an α-spectrin structural mutant. They lead to more severe disease (see next).
The mechanically unstable membrane of HE leads to shape change from discocyte to elliptocyte as the membrane is buffeted by shear stress in the circulation.
Patients who are heterozygotes for these defects have milder disease while double heterozygotes and homozygotes for these mutants have progressively more severe syndromes.
Blood smear: 25–90% of cells have elongated oval elliptocytes
Osmotic fragility is normal or increased
Autohemolysis is usually normal but may be increased and usually corrected by the addition of glucose or ATP.
Varies from patients who are symptom-free to severe anemia requiring blood transfusions. The percentage of microcytes best reflects the severity of the disease
About 12% have symptoms indistinguishable from HS
The percentage of elliptocytes varies from 50% to 90%. No correlation has been established between the degree of elliptocytosis and the severity of the anemia.
HE has been classified into the following clinical subtypes:
Common HE, which is divided into several groups:
silent carrier state
mild HE
HE with infantile pyknocytosis
Common HE with chronic hemolysis, which is divided into two groups:
HE with dyserythropoiesis
homozygous common HE, which is clinically indistinguishable from HPP (see later discussion)
Spherocytic HE, which clinically resembles HS; however, a family member usually has evidence of HE
Southeast Asian ovalocytosis, in which the majority of cells are oval. Some red cells contain either a longitudinal or transverse ridge
Infantile hemolytic elliptocytosis of infancy: these patients present with hemolytic elliptocytosis (mimicking HPP) that changes over the first 2 years of life to a clinical picture of mild HE as fetal hemoglobin changes to adult hemoglobin. Usually, there is a single affected parent with HE.
The indications and considerations for transfusion, splenectomy, and prophylactic folic acid are the same as for HS.
Homozygous or doubly heterozygous for spectrin chain mutants (e.g., Sp-a 1/74 and Sp-a 1/76 ). The spectrin chain defects found in HPP are similar to those found in HE.
HPP is a congenital hemolytic anemia associated with in vivo red cell fragmentation and marked in vitro fragmentation of red cells at 45°C. Because of the similarities in the membrane defect in this condition and HE, it is viewed as a subtype of HE.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here