Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Fungi are eukaryotic, unicellular (yeasts) and multicellular (filamentous) microorganisms, ubiquitous in nature. They acquire food and nutrition by feeding on biotic (living tissue) or abiotic (dead tissue, plant debris).
Few, if any, fungi can penetrate intact corneal tissue. Risk factors for invasion and infection include breach of epithelium (overt, minor trauma), chronic use of antimicrobials, topical steroids, and increasingly contact lens wear.
Spectrum and diversity of fungal pathogens vary by climate, occupational status, ocular surface health, and healthcare exposure (contact lens, medical, surgical interventions). In tropical and subtropical regions, filamentous fungi ( Aspergillus species, Fusarium species) are the predominant pathogens, while in cooler climates, Candida species are the most common.
Delays in clinical diagnosis and laboratory confirmation contribute to the protracted clinical course, characteristic of the disease.
Medical management remains problematic due to poor penetration and limited availability and efficacy of current antifungal preparations. No single antifungal provides adequate coverage for common ocular fungal pathogens.
Surgical interventions are often required for control and management of the disease.
Fungal or mycotic keratitis is a leading cause of ocular morbidity, opacification, and preventable blindness. The true incidence and/or prevalence of fungal keratitis is unknown. Worldwide, fungal keratitis constitutes up to 50% of all microbial keratitis. Historically, bacterial keratitis was reported to be more common in children than fungal keratitis, but a recent study out of India found that fungus as opposed to bacteria was more commonly the infectious agent in children with keratitis (54% vs. 41%). Overall, fungal keratitis is much more common in tropical and subtropical regions as compared to other climates. In the United States, fungi are recovered in 6%−20% of cornea infections.
Filamentous fungi ( Aspergillus , Fusarium , Bipolaris, and Curvularia ) are the predominant pathogens in tropical climates, while Candida species ( C. albicans, C. parapsilosis ) are the most common in temperate regions. Common risk factors for the disease include traumatic implantation (foreign body, vegetable/plant materials), contact lens wear, ocular surface disorders, and chronic use and abuse of topical medications (anesthetics, antibiotics, and corticosteroids). , The increasing use of broad-spectrum topical antibiotics is thought to reduce the normal ocular surface microbiome and provide a noncompetitive environment for fungi to proliferate. , In addition, the use of topical corticosteroids enhances the growth of fungi while suppressing host immune response. , , There is also an increasing recognition of fungal keratitis related to the use of soft contact lenses.
Fungal keratitis represents one of the most difficult forms of microbial keratitis for the ophthalmologist to diagnose and manage. , , Difficulties arise in clinical recognition and diagnosis, lack of rapid laboratory identification and confirmation, and limited selection and efficacy of current antifungal medication. Advanced cases involving the stroma can be quite challenging to eradicate, often requiring prolonged and intensive topical and systemic antifungal therapy. Failure with medical management alone is common and surgical intervention is often required. Limitations of currently available antifungal therapies include poor bioavailability and limited penetrance of the eye. , Surgical interventions (therapeutic keratoplasty [TKP], penetrating keratoplasty [PK], conjunctival flap, lamellar keratoplasty, cryotherapy, crosslinking) are thus often required for visual rehabilitation and control. , , , Despite these measures, both medical and surgical measures are sometimes not effective and more definitive management (e.g., enucleation) is needed to control infection.
Fungi are ubiquitous, eukaryotic, unicellular (yeast-nonfilamentous) and multicellular (molds-filamentous) organisms. They are unique in that they contain chitin and glucan, rather than cellulose in their cell walls. Yeasts are single-celled fungi widely distributed in nature and can be recovered from human skin and intestines. , Molds are multicellular fungi characterized by hyphal growth in filaments and can be recovered intermittently from human skin and mucosa. Fungi acquire their nutrients by absorption and enzymatic digestion of living tissue or decaying organic matter from soil, building materials and food. They reproduce both sexually and asexually by budding (yeast) and/or the formation of asexual and sexual spores (filamentous). ,
Fungi gain access into the corneal stroma through a defect in the epithelial barrier. This defect may be due to traumatic implantation with contaminated soil or plant materials, insertion and removal or improper contact lens wear, a compromised ocular surface, local immunosuppression (e.g., topical chronic corticosteroid use), or previous surgery.
Once in the stroma, fungal pathogens multiply and proliferate via production of several proteases and mycotoxins, resulting in severe tissue necrosis. These mycotoxins, which are produced by fungi in vitro, such as in the case of Fusarium obtained from mycotic keratitis cultures, are directly toxic to human epithelial cells. , The most common mycotoxins isolated from Fusarium mycotic keratitis were nivalenol (NIV), deoxynivalenol (DON), and trichothecene (T2). Studies have shown a direct linear relationship between increased concentrations of mycotoxins and human epithelial cell death, thus increasing the pathogenicity of Fusarium in mycotic keratitis. Clinically, patients with Fumonisin B mycotoxin-producing Fusarium strains were found to have a worse initial visual acuity and a higher risk of progression to PK.
Once in the stroma, fungi can penetrate deep into the stroma through an intact Descemet membrane into the anterior chamber and other intraocular structures. Filamentous fungal pathogens may also extend from the cornea into the sclera. Fungal dissemination to the anterior chamber and/or sclera is associated with a more virulent/aggressive pathogen, longer disease course, and poorer outcomes. , ,
Frequency and distribution of fungal pathogens vary by geography, climate, occupation, and healthcare exposure. , , The true incidence and/or prevalence of fungal keratitis is difficult to ascertain. Studies from Asia and other developing countries indicate that fungi may be recovered in up to 50% of cases in South India. , This is in contrast to a disease prevalence of 37% in Ghana and 17% in Nepal. , In the United States, prevalence ranges from 6% in northern latitudes to 20% in southern states. ,
Among the more than 250,000 fungal species that have been identified, fewer than 100 have been associated with mycotic keratitis. , , , Filamentous fungi are the most frequently recovered pathogens in fungal keratitis worldwide. The hyaline hypomycetes ( Fusarium species, Aspergillus species, Paecilomyces species) are the most common, but the dematiaceous hypomycetes ( Curvularia species, Colletotricum species, Alternaria species) may be recovered in up to 20% of the cases. Yeast and yeast-like organisms ( Candida species, Cryptococcus species, and Trichosporum species) are more likely to be recovered from patients with underlying ocular surface disease and chronic use and abuse of ocular anesthetic and antibiotics, , as well as after surgery either acutely or delayed. Candida , for example, is a common pathogen after PK and Descemet stripping automated endothelial keratoplasty (DSAEK) ( Fig. 81.1 ). Common genera and species that have been recovered from fungal keratitis are outlined in Box 81.1 .
| Genus | Species | Filamentous Fungi | Reservoirs |
|---|---|---|---|
| Alternaria | A. alternata, A. infectoria , Alternaria spp. | Dematiaceous | Soil, air, water, plant debrs |
| Aureobasidium | A. pullulans | Dematiaceous | Soil, air, water, limestone |
| Bipolaris | B. hawaiiensis, B. spicifera (formerly Drechslera) | Dematiaceous | Soil, plant debris |
| Chaetomium | C. atrobrunneum, Chaetomium species | Dematiaceous | Soil, air, water, plant debrs |
| Cladosporium | C. cladosporioides | Dematiaceous | Soil, organic material, air, dust |
| Colletotrichum | C. capsici, C. coccodes , C. dematium, C. graminicola-(Glomerella graminicola-telemorph, C. gloeosporioides-Glomerulla cingulata-telemorph, C. siamense | Dematiaceous | Soil, grass, grain, plant pathogens, soil, cereals |
| Curvularia | C. aeria, C. brachyspora, C. geniculata, C. lunata-Cochliobolus lunatus-telemorph, C. pallescens, C. senegalensis, C. turberculata, C. verruculosa | Dematiaceous | Soil, plants, mainly in tropical areas can occur in temperate zones |
| Dichotomophthoropsis | D. nymphearum, D. portulacae | Dematiaceous | Soil, aquatic plants |
| Doratomyces | D. stemonitis | Dematiaceous | Soil, dung, rotting plants, dead wood |
| Exserohilum | E. rostratum, E. longirostratum | Dematiaceous | Soil, contaminated medications |
| Fonsecaea | F. pedrosoi | Dematiaceous | Soil, plants |
| Lasiodiplodia | L. theobromae | Dematiaceous | Soil, decaying wood |
| Lecythophora | L. mutabilis | Dematiaceous | Soil, raw milk, cheeses |
| Microsphaeropsis | M. olivacea | Dematiaceous | Soil, plant pathogen |
| Nigrospora | N. osmanthi | Dematiaceous | Soil, air, plants |
| Ochroconis | Ochroconis species | Dematiaceous | Soil, decaying vegetation, hot tubs, immunocompromised patients |
| Periconia | Periconia species | Dematiaceous | Soil, decaying plant material |
| Phialemoniopsis | P. curvata | Dematiaceous/hyaline | Soil, contaminated water, sewage |
| Phaeoisaria | P. clematitidis | Dematiaceous | Soil, sediment |
| Phaeotrichoconis | P. crotalariae | Dematiaceous | Soil, plant pathogen |
| Phialophora | P. bubakii, P. verrucosa | Dematiaceous | Soil, decaying wood |
| Phoma | P. eupyrena, P. oculo-hominis, Phoma species | Dematiaceous | Soil, dung, living and dead plants, wallpaper, caulk, paint |
| Pyrenochaeta | Pyrenochaeta species | Dematiaceous | Soil, plant debris, mainly tropical areas |
| Schizophyllum | S. radicatum | Dematiaceous | Mushroom, emerging human pathogen, soil |
| Scytalidium | Scytalidium sp. | Dematiaceous | Soil |
| Stachybotrys | Stachybotrys | Dematiaceous | Soil, wet building materials, air |
| Tetraploa | T. aristata | Dematiaceous | Soil, plants |
| Trichoderma, previously Gliocardium | Trichoderma species | Dematiaceous | Soil, plant debris |
| Ulocladium | U. atrum | Dematiaceous | Soils, walls, plants |
| Epidermophyton | Epidermophyton species | Dermatophyte | Soil, humans |
| Microsporum | Microsporum species, M. canis | Dermatophyte | Soil, humans |
| Genus | Species | Dimorphic Fungi | Reservoirs |
|---|---|---|---|
| Blastomyces | B. dermatitidis | Dimorphic | Dust |
| Coccidioides | C. immitis | Dimorphic | Soil |
| Paracoccidioides | P. brasiliensis | Dimorphic | Soil |
| Sporothrix | S. schenckii | Dimorphic | Soil, living and decaying plant materia1, peat moss |
| Ustilago | Ustilago species | Dimorphic | Smut fungi, soil, crops. |
| Genus | Species | Hyaline Fungi | Reservoirs |
|---|---|---|---|
| Acremonium | A. atrogriseum, A. curvum, A. kiliense, A. potronii, A. recifei, Acremonium species | Hyaline | Soil, dead plant material |
| Absidia | Absidia species, A. corymbifera | Hyaline (zygomycete) | Soil, decaying plant material, compost |
| Acrostalagmus | A. cinnabarensis | Hyaline | Soil, plants, fruits |
| Apophysomyces | Apophysomyces species | Hyaline | Soil, tropical, subtropical |
| Arthrographis | A. kalrae | Hyaline | Soil and compost |
| Aspergillus | A. brasilensis, A. clavatus, A. fischerianus, A. flavipes, A. flavus, A. glaucus, A. fumigatus, A. janus, A. lignicola, A. nidulans, A. niger, A. nomius, A. oryzae, A. pseudotamarii, Aspergillus species, A. tamarii, A. terreus. A. tubingensis, A. viridinutans, A. wentii | Hyaline | Soil, air, damp walls, soil, foods |
| Beauveria | B. bassiana | Hyaline | Soil, insects |
| Cephaliophora | C. irregularis | Hyaline | Soil, plants |
| Chlamydoabsidia | C. padenii | Hyaline (zycomycete) | Soil, decaying plant material, compost |
| Chrysonilia | C. sitophila (formerly Neurospora sitophila) | Hyaline | Soil, bead |
| Chrysosporium | C. parvum | Hyaline | Soil, skin, nails |
| Cylindrocarpon | C. lichenicola (C. tonkinense) | Hyaline | Soil, plant pathogen |
| Diplodia | Diplodia species | Hyaline | Soil, pine trees |
| Diplosporium | Diplosporium species | Hyaline | Soil, plants |
| Engyodontium | E. alba (formerly Beauveria alba) | Hyaline | Soil, plant debris, painted walls, textiles, paper |
| Fusarium | F. aquaeductum, F. dimerum, F. falciforme, F. keratoplasticum, F. oxysporum, F. solani, F. verticilloides (F. moniliforme), F. nivale, F. subglutinans, F. ventricosum | Hyaline | Soil, plant debris, turtles, bananas |
| Glenospora | G. graphii | Hyaline | Soil, plants |
| Gongromeriza | Gongromeriza species | Hyaline | Soil, mushroom pathogen |
| Graphium | G. basitruncatum | Hyaline | Soil, plant debris, woody materials, manure, polluted waters |
| Metarhizium | M. anisopliae | Hyaline | Soil, insect pathogens |
| Mucor | M. cornealis, M. racemosus, Muor species | Hyaline (zygomycetes) | Soil, decaying plant material, compost |
| Myrathecum | Myrathecum species | Hyaline | Soil |
| Ovadendron | O. sulphureo-ochraceum | Hyaline | Soil, plants |
| Paecilomyces | P. farcinosus, P. variotii | Hyaline | Soil, decaying plants, food products |
| Penicillium | P. citrinum, P. expansum | Hyaline | Soil, organic material, air, dust |
| Purpureocillium | P. lilacinus (previously Paecilomyces lilacinus) | Hyaline | Soil, decaying vegetation, insects |
| Rhizopus | Rhizopus species | Hyaline (zygomycetes) | Soil, decaying plant material, compost |
| Rhizoctonia | Rhizoctonia species | Hyaline | Soil, crops |
| Sarcopodium | S. oculorum | Hyaline | Soil, plants |
| Scedosporium | S. apiospermum, S.boydii- previously Pseudallescheria boydii) | Hyaline | Soil, polluted waters |
| Scopulariopsis | S. brevicaulis | Hyaline | Soil, food, paper |
| Tritirachium | T. oryzae | Hyaline | Soil, food, paper, decaying plants, crops |
| Verticillium | V. searrae, Verticillium species | Hyaline | Soil, crops |
| Genus | Species | Yeast | Reservoirs |
|---|---|---|---|
| Candida | C. albicans, C. famata, Candida fermentati, C. glabrata, C. guilliermondii, C. krusei, C. parapsilosis, C. tropicalis | Yeast | Humans (skin, gut, urogenital tract) |
| Geotrichum | G. candidum | Yeast | Soil, cereal, water, air, sewage, plants |
| Malassezia | M. furfur | Yeast | Human skin, animal skin |
| Rhodosporidum | R. toruloides | Yeast | Wood pulp |
| Rhodotorula | R. glutinis, R. rubra, Rhodotorula species | Yeast | Humans, animals, plants |
| Torula | Torula species | Yeast | Skin, olives |
| Genus | Species | Mixed Morphology Fungi | Resevoirs |
|---|---|---|---|
| Cryptococcus | C. laurentii, C. neoformans | Yeast-like, filobasidiella-telemorhph is filamentous | Soil, bird droppings, air |
| Exophiala | E. jeanselmei var. dermatitidis, E. jeanselmei var. jeanselmei | Yeast-like, dematiaceous | Soil, decaying plant, organic matter, decaying wood |
| Guehomyces | Guehomyces pullulans | Yeast-like fungi | Skin, soil |
| Pythium | P. insidiosum | Fungus-like | Soil, plants |
In developing countries, fungal keratitis after trauma is more common than associated with contact lens wear. In Ghana, India, and China, trauma accounted for more than 90% of cases of fungal keratitis, with trauma with soil and plant materials the most frequent inciting factors. , , , In developed countries, the opposite is true. In a study of 733 confirmed cases of confirmed fungal keratitis in the United States, 37% of fungal keratitis cases were attributed to contact lens wear, 29% of cases were attributed to ocular surface disease, and 25% of cases were attributed to trauma. No identifiable risk factor was documented in 10% ( n = 76). Of note, this survey occurred during the outbreak of fungal keratitis linked to the contact lens solution ReNu with MoistureLoc. Since the removal of ReNu with Moisture Loc, contact lens-associated fungal keratitis has returned to earlier baseline frequency of less than 25%. In the United States and other similar countries, fungal keratitis due to contact lens wear was most often specifically due to Fusarium . , , Common risk factors for filamentous and nonfilamentous fungal keratitis can be found in Box 81.2 .
| Nonfilamentous Fungi (Yeasts) | Filamentous Fungi (Molds) |
|---|---|
| Contact lens wear (therapeutic) | Contact lens wear (cosmetic, therapeutic) |
| Chronic ocular surface disease/allergic/vernal conjunctivitis | Corneal trauma with soil or organic matter |
| Chronic keratitis (herpes simplex, herpes zoster) | Prior ocular surgery (penetrating keratoplasty, Lasik) |
| Corneal trauma | Topical corticosteroids |
| Immunosuppression | Chronic use of broad-spectrum antibiotics |
| Corneal anesthetic abuse | |
| Chronic use of broad-spectrum antibiotics | |
| Prior corneal surgery |
The use of topical corticosteroids has also been associated with the development and worsening of fungal keratitis. , Corticosteroids appear to activate and increase the virulence of fungi. Topical anesthetic abuse has also been associated with mycotic keratitis , as has the chronic use of broad-spectrum antibiotics, which can reduce the presence of normal flora and increase fungal infections with Candida species. Other factors associated with the risk of fungal keratitis include hospitalized patients with underlying systemic fungal infections or underlying immunosuppression. , , ,
DSAEK has also been associated with an increase in fungal keratitis. DSAEK has become increasingly popular over the last decade due to several advantages over PK, including reduced risk of allograft rejection, shorter postoperative steroid use, increased global integrity, and earlier removal of sutures. However, one major side effect is the increased incidence of postoperative fungal keratopathy. The most common organism identified in these cases is Candida albicans.
Systemic diseases such as diabetes mellitus have also been reported as a major independent risk factor for fungal infection. While patients with diabetes were found to have a higher incidence of fungal keratitis compared to nondiabetic patients, there were no significant differences in the pathogen profile that affected diabetics versus nondiabetics. The association between diabetes and fungal keratitis is hypothesized to be due to a decreased host defense and a good microenvironment for fungi to grow due to high glucose levels in the diabetic patient.
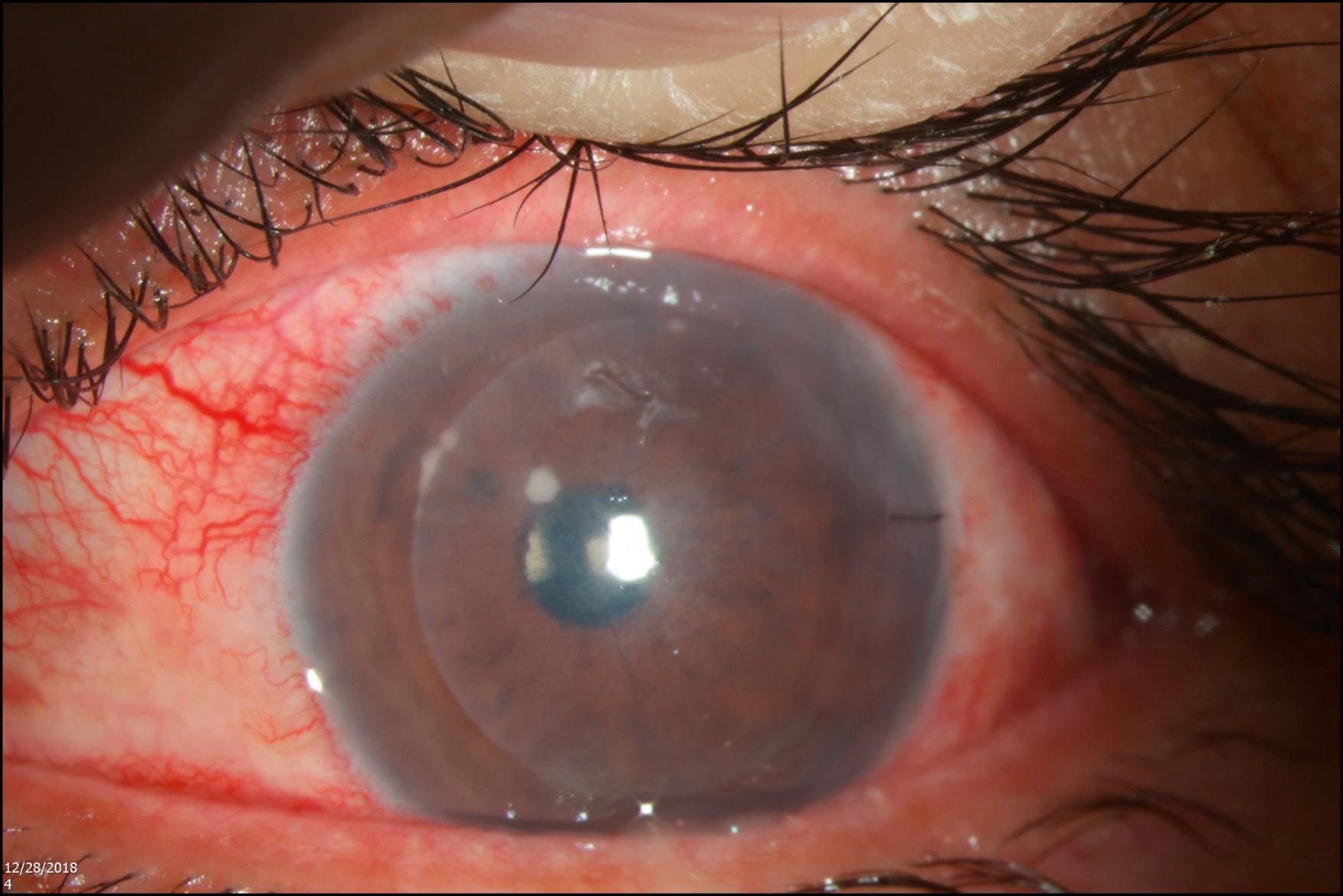
The clinical diagnosis of fungal keratitis can be difficult. , , , Diagnosis is made based on analysis of information from a combination of patient history, , perceived risk factors, and presenting signs and symptoms. Patients may report the initial symptom of a foreign body sensation for several days with a slow onset of increasing pain. , There is also an overlap between symptoms and signs of bacterial and fungal keratitis making it even harder to diagnose. These include suppuration, conjunctival injection, an epithelial defect, stromal infiltration, serrated margins, and an anterior chamber reaction or hypopyon ( Box 81.3 ). Clinical findings such as an elevated area, hyphate (branching) ulcers, irregular feathery margins, a dry rough texture, endothelial rings, and grey–white satellite lesions ( Figs. 81.2–81.6 ) are suggestive of filamentous fungal keratitis. , The presence of macroscopic brown pigmentation is commonly associated with the presence of a dematiaceous fungus ( Curvularia lunata ) ( Fig. 81.7 ). The presence of an intact epithelium with a deep stromal infiltrate may also be found in filamentous fungal keratitis ( Fig. 81.8 ). Despite these descriptive findings, studies to assess the clinical manifestations of suppurative (bacterial) versus fungal keratitis have demonstrated that it was not possible to differentiate clinically between bacterial and fungal keratitis, especially in cases where yeasts are the infecting fungi. , , , ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here