Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In contemporary clinical practice, multiple modalities of cancer treatment allow therapy to be tailored according to the continuum of disease severity. As trends in prostate cancer (PCa) presentation change, existing treatment options must be adapted to adjust risk and efficacy accordingly, or in some cases, new treatment modalities must be developed.
In recent years, PCa presentation and diagnosis has changed significantly with the identification of prostate-specific antigen (PSA) as a biomarker for prostate disease screening. In the “PSA era,” a significant downward stage migration in detected PCa has been observed. This trend has led to increased detection of early stage, localized cancers in the prostate, which invites the opportunity to manage the disease at an earlier point in its natural history.
The rise in overall PCa detection includes diagnosis of many indolent cancers that are unlikely to cause harm. The consequent “overdiagnosis” of PCa has prompted the need for alternate diagnostic and treatment strategies to avoid side effects of conventional therapy. With such shifts in clinical characteristics of PCa at presentation, novel strategies must be explored to more accurately risk-stratify patients and provide alternatives to highly morbid whole-gland therapy.
One strategy that has evolved is active surveillance (AS) of low-risk PCa, a protocol utilized by clinicians to prevent overtreatment of potentially indolent disease. However, up to one-third of men under AS require definitive treatment, and nearly 30% of patients who fail AS are found to have locally advanced after radical prostatectomy. Additionally, in its current form, AS requires frequent monitoring by serum testing, office visits, imaging, and biopsy. This results in anxiety and a reduction in quality of life for many patients. Therefore, there exists a need for management options that are more aggressive than surveillance but provide similar preservation of functional outcomes and quality of life.
Focal therapy may provide the capability to treat disease at an early stage while minimizing the risks associated with whole-gland therapy, thereby satisfying a middle-ground between AS strategies and radical therapy for men with localized PCa. In this chapter, we aim to describe the present state of focal therapy, including the goals of therapy, challenges of successful implementation, development of multiple treatment modalities, and contemporary clinical trials that have explored the potential of focal therapy of PCa.
The primary advantage of focal therapy is the selective eradication of PCa and preservation of healthy prostate tissue. The intent of this approach is to mitigate the sexual and urinary side effects that are frequently associated with radical therapy. In this regard, focal therapy holds promise in multiple clinical settings in which the goals of therapy might differ.
In men with low-risk, unifocal disease, focal therapy may provide complete eradication of isolated PCa, leaving the remaining prostate gland disease-free with reduced morbidity compared to whole-gland therapy. Although the incidence of such disease states has risen, recent analysis of 1400 men undergoing radical prostatectomy showed that only 11% had unilateral, low-risk disease (PSA <10 ng/mL, Gleason score <7, <10% tumor involvement). A similar study showed that unifocal and unilateral disease in men with low to intermediate preoperative risk features (PSA ≤20 ng/mL, Gleason score ≤7, ≤66% cores positive) existed in only 14 and 13%, respectively. In addition, identification of men with truly unifocal cancer is difficult with current biopsy techniques. As such, the use of focal therapy in this setting alone would result in relatively few candidates.
Broader applications of focal therapy may be envisioned in light of other observations on the natural history of PCa. The concept of an index lesion, or a single, high-risk focus as the nidus of most lethal PCa, opens the potential for effective treatment of multifocal disease by targeting only the dominant focus of cancer. In a study of radical prostatectomy specimens, Ohori et al. found that 80% of total tumor volume and 92% of extracapsular extension arose from one dominant focus of cancer within the prostate. Additional studies suggest that nondominant cancer foci typically demonstrate features consistent with low-risk disease. Ohori et al. also concluded that the Gleason score was rarely higher in a secondary lesion than the primary or index lesion, while Villers et al. observed that 80% of secondary tumors are less than 0.5 cm 3 . Rukstalis et al. similarly reported that the median volume of secondary lesions was 0.3 cm 3 and suggested that 79% of men would likely have insignificant residual cancer after ablation of the dominant tumor focus. Subsequent investigations have determined that sites of metastatic PCa, when present, are typically derived from one genotypic clonal cell population from the prostate, which supports the potential efficacy of focal therapy in preventing disease spread if directed to the correct tumor focus.
Accordingly, in men with multifocal, intermediate-risk disease, it may be reasonable to treat the dominant cancer focus while allowing residual “indolent” cancer to be monitored on AS, thereby expanding the pool of eligible men who may benefit from focal therapy by aiming for disease control rather than cure. Such an approach would extend the acceptable period of AS, prolong the natural history of the disease to avoid cancer-specific mortality within the patient’s natural longevity, minimize the need for radical therapy, and reduce patient anxiety due to surveillance in the absence of therapy.
“Subtotal” strategies of PCa treatment, whereby ablative techniques are applied to nearly the entire gland while preserving the posterolateral capsule on one side to avoid damage to one neurovascular bundle (NVB) and preserve potency, has been put forth as an alternative to prostatectomy in medium- to high-risk patients. Initially captured under the umbrella of focal therapy, a recent consensus panel determined that such treatment strategies should not be considered “focal,” and thus will not be in focus in this chapter. Finally, focal therapy presents utility in multimodal approaches to PCa, as either an adjunct to primary therapy or use in salvage therapy for recurrent disease. There are limited data in this regard, and as such, they will not be presented in this chapter.
In light of the specific goals of treatment, considerations for focal therapy include candidate selection, disease mapping, choice of treatment modality, and methods for clinical follow-up.
Among contemporary cohorts undergoing surgery, approximately 20% of men demonstrate unilateral disease on final pathology. Conceivably a significant number of these men may have been candidates for focal therapy prior to surgery. Additional candidates include the nearly 50% of men who develop unfavorable disease features while on AS, and up to one-third who ultimately undergo radical therapy, presumably having missed the temporal window for localized disease control with focal treatment. Traditionally, focal therapy trials have including men with unilateral disease as a surrogate for unifocal or limited focality cancer. Recently, the emergence of MRI assessment of the prostate has opened the door for men with bilateral, limited focality disease as potential candidates for targeted approaches.
There are no broadly accepted criteria for identifying appropriate candidates for focal therapy. However, while the boundaries of acceptable risk are not strictly defined, it is clear that focal therapy is inappropriate for low-risk disease, which may do well on AS, as well as for the treatment of high-risk disease in which the window of opportunity for local control may be missed if focal therapy fails ( Figure 60.1 ). Initial general consensus accordingly held that investigational efforts to implement focal therapy protocols should be limited to men with low- to low-intermediate-risk disease in order to limit the likelihood of early systemic failure. In part this was also due to suboptimal methods of disease mapping, which meant that accurate selection of men with early localized intermediate- to high-risk disease was limited, and therefore inclusion of such men would risk early treatment failure. Recently, however, there has been a proposed shift to focus on intermediate risk patients, in light of growing clinical data with medium-term follow-up as well as consideration for overtreatment of low-risk disease.
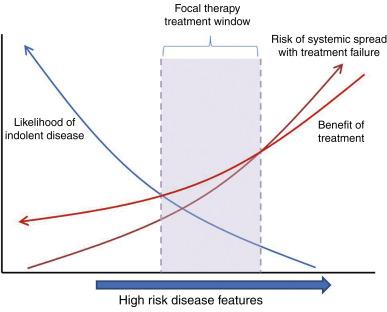
Pretreatment cancer risk is often defined using features such as Gleason score, clinical stage, and PSA (among others), as in the risk classification schemes proposed by D’Amico and Kattan. Additional risk of treatment failure in focal therapy may be added by the presence of multifocal disease if either (1) the dominant lesion is not correctly identified or (2) ancillary lesions carry malignant potential. Few trials have explored the efficacy of focal therapy in multifocal disease. Ahmed et al. demonstrated 85% biochemical disease-free survival (BDFS) and 77% negative biopsies at 6 months after focal therapy in a mixed cohort of men with unilateral unifocal, unilateral multifocal, and bilateral multifocal disease, in whom all detected cancer foci were targeted for therapy. Furthermore, potency and continence were preserved in these men ( Table 60.1 ). Their results suggest that multifocal disease may be adequately treated with focal therapy and still provide superior functional outcomes to whole-gland therapy. In this study, seven of nine (89%) men with positive biopsies at 6 months had residual cancer evident on MRI in previously treated areas of the prostate, suggesting that improvements in lesion targeting and/or estimation of tissue ablation margin may improve the ability for focal therapy to treat multifocal cancer. Critically important in this assertion is the documentation of longer-term follow-up. In the study of Ahmed et al., men were staged prior to therapy with a transperineal template biopsy, but efficacy was assessed with a targeted biopsy of the treated area. The validity of this approach in the demonstration of disease control can only be assessed in longer-term follow-up.
| Authors | Pts | Clinical stage (%) | Ablation extent | Follow-up (months) | BDFS | Negative f/u biopsy | Urinary function | Potency preservation | Adverse events | Secondary conclusions |
|---|---|---|---|---|---|---|---|---|---|---|
| Barret et al. | 21 | ≤T2a | Hemi | 12 | NR | NR | No incontinence | ↓IIEF-5 20–14 | 5 (24%) Retention | |
| Ahmed et al. | 41 | T1c (90) T2a (10) | Focal(3–5 mm margin); 49% multifocal | 12 | 85% (ASTRO) | 30/39 (77%) at 6 months | No incontinence | No difference in IIEF-5 at 12 months from baseline | 7 (17%) UTI; 1 (2%) acute urinary retention; 1 (2%) urethral stricture | mpMRI has a high-sensitivity for residual disease |
| Ahmed et al. | 20 | ≤T2b | Hemi | 12 | NR | 17/19 (89%) at 6 months | 2 men (10%) using pads | 100% Potent at 12 months | 1 (5%) Urethral stricture | mpMRI was 100% sensitive and specific for identifying residual cancer |
| El Fegoun et al. | 12 | ≤T2a | Hemi | 127 | NR | 91% at 1 year 90% at 5 years 38% at 10 years | NR | NR | 2 (17%) UTI; 1 (8%) epididimo-orchitis; 1 (8%) acute urinary retention |
100% Cancer free survival at 10 years, though five underwent salvage therapy (mean 8.6 years later) |
| Muto et al. | 29 | T1c (78) T2a (10) T2b (12) | All PZ 1 lobe TZ | 32 | 83% (low risk) 54% (med. risk) 0% (high-risk) | 25/28 (89%) at 6 months; 13/17 (77%) at 12 months | Minimal incontinence (not specified) | NR | 1 (4%) UTI; 1 (4%) urethral stricture | Focal HIFU provided comparable cancer control to whole gland HIFU |
Regardless of the specific combination of features chosen, a primary concern for reliable risk stratification is accurate PCa disease mapping, strategies and limitations of which are discussed next.
The appropriateness and efficacy of focal therapy is heavily dependent on reliable determination of PCa risk, distribution, and extent, which underpins the ability to selectively target the foci of cancer. Two broad methods have been developed for determination of these factors: (1) prostate biopsy and (2) prostate imaging.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here