Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The later part of the first trimester is an opportune time to assess the fetus for chromosomal aneuploidies and structural anomalies.
One approach to screening is utilizing sonographic markers such as measurement of the nuchal translucency (NT) and appearance of the fetal nasal bone in combination with serum analytes to provide a pregnancy-specific risk of the common aneuploidies. An enlarged NT is associated with an increased risk of common aneuploidies and is also associated with other genetic abnormalities. Similarly, fetuses with an enlarged NT have an increased risk of structural malformations and adverse perinatal outcomes.
Recent advances in aneuploidy screening strategies by analysis of cell-free DNA has revolutionized the detection of the common autosomal trisomies by providing high sensitivity and low false-positive rates. , This type of screening is optimally performed in conjunction with a sonographic assessment of the fetus to ensure appropriate counseling and utilization of this technology as well as to provide a correct interpretation of the results.
The sonographic evaluation of the structural integrity of the fetus in the later first trimester results in the detection of many major malformations. Contour abnormalities including acrania, abdominal wall defects, and limb–body wall anomalies will almost always be identifiable; other anomalies such as cardiac defects and craniofacial abnormalities are potentially identifiable; and other abnormalities such as microcephaly and agenesis of the corpus callosum are not identifiable during the first trimester due to the timing of development.
A standardized imaging protocol and the use of transvaginal sonography maximize the detection of structural malformations. A thorough understanding of embryologic development and an appreciation of the natural history of anomalies are necessary for accurate interpretation of sonographic findings. Anatomic imaging in the first trimester does not replace the standard or detailed second-trimester anatomic scan. Abnormalities detected in this gestational age window should prompt genetic counseling and consideration of diagnostic genetic testing.
Many normal fetal anatomic features can by identified by sonography between 11 0/7 and 13 6/7 weeks’ gestation. , Fetal structural anomalies can often be identified between 11 0/7 and 13 6/7 weeks’ gestation. While some anomalies are identifiable in the earlier part of this gestational age window, optimal detection is at approximately 13 weeks’ gestation. , Knowledge of normal embryologic development is vital to making a correct diagnosis, avoiding false-positive diagnoses, and providing appropriate prenatal counseling. For brevity, and in keeping with common usage, we will refer to such scans as “11–14 week scans.”
Approximately 40%–50% of major structural anomalies in singletons can be diagnosed in the latter part of the first trimester. , , , Detection rate increases between 11 and 14 weeks’ gestation.
Using a standardized protocol, 28% of anomalies in singleton fetuses without a known chromosomal abnormality can be detected in the first trimester.
Identification of structural anomalies in twin gestations without a known chromosomal abnormality occurs at a higher rate among monochorionic twins than dichorionic twins (53% versus 27%). Monochorionic twins have a higher incidence of anomalies (2.8%) compared with dichorionic twins (1.3%). The higher detection rate in monochorionic twins is related to the distribution of anomalies that are more easily identified by ultrasound (e.g., acrania/anencephaly), as well as the presence of anomalies specific to this type of twinning (e.g., conjoined twins and twin reversed arterial perfusion sequence).
Detection rates vary based on structural anomaly.
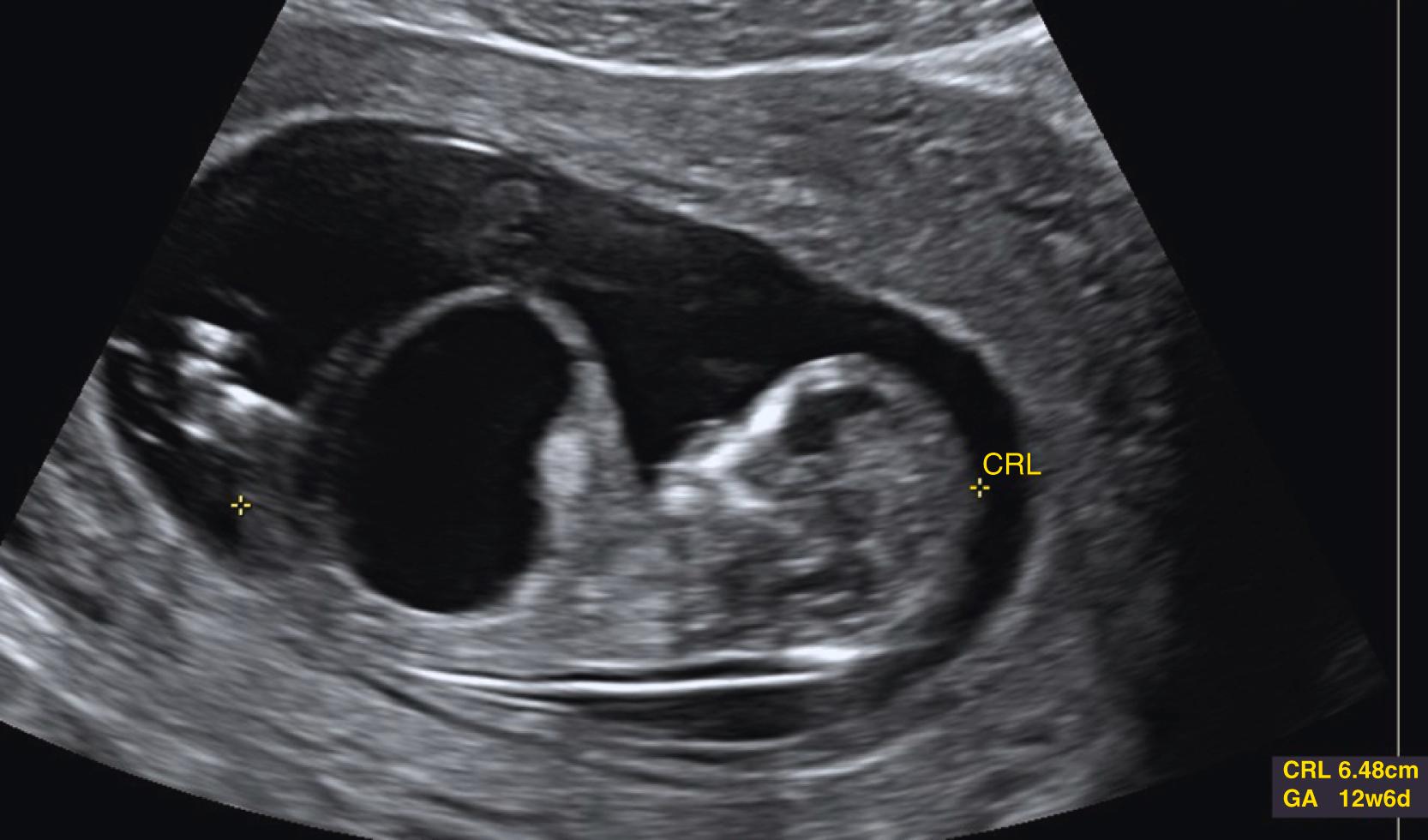
Some anomalies should almost always be detectable before 14 weeks’ gestation. These include major abnormalities in fetal contour such as acrania, anencephaly, encephalocele, ectopia cordis, and body stalk anomaly and abdominal wall defects including exomphalos, gastroschisis, and pentalogy of Cantrell. Anomalies such as alobar holoprosencephaly, sirenomelia, and megacystis should be apparent in most cases ( Figs. 29.1 through 29.5 ).
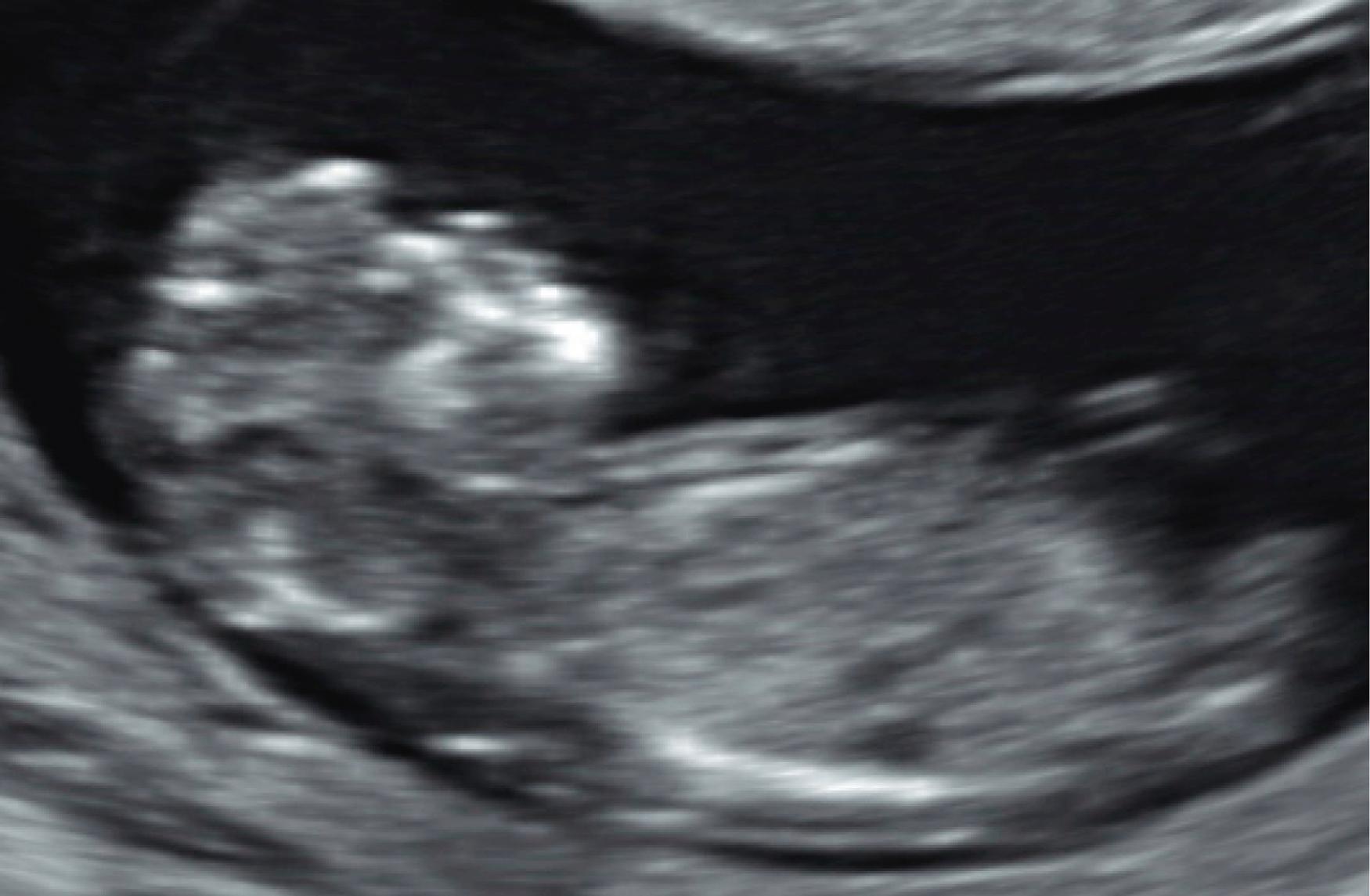
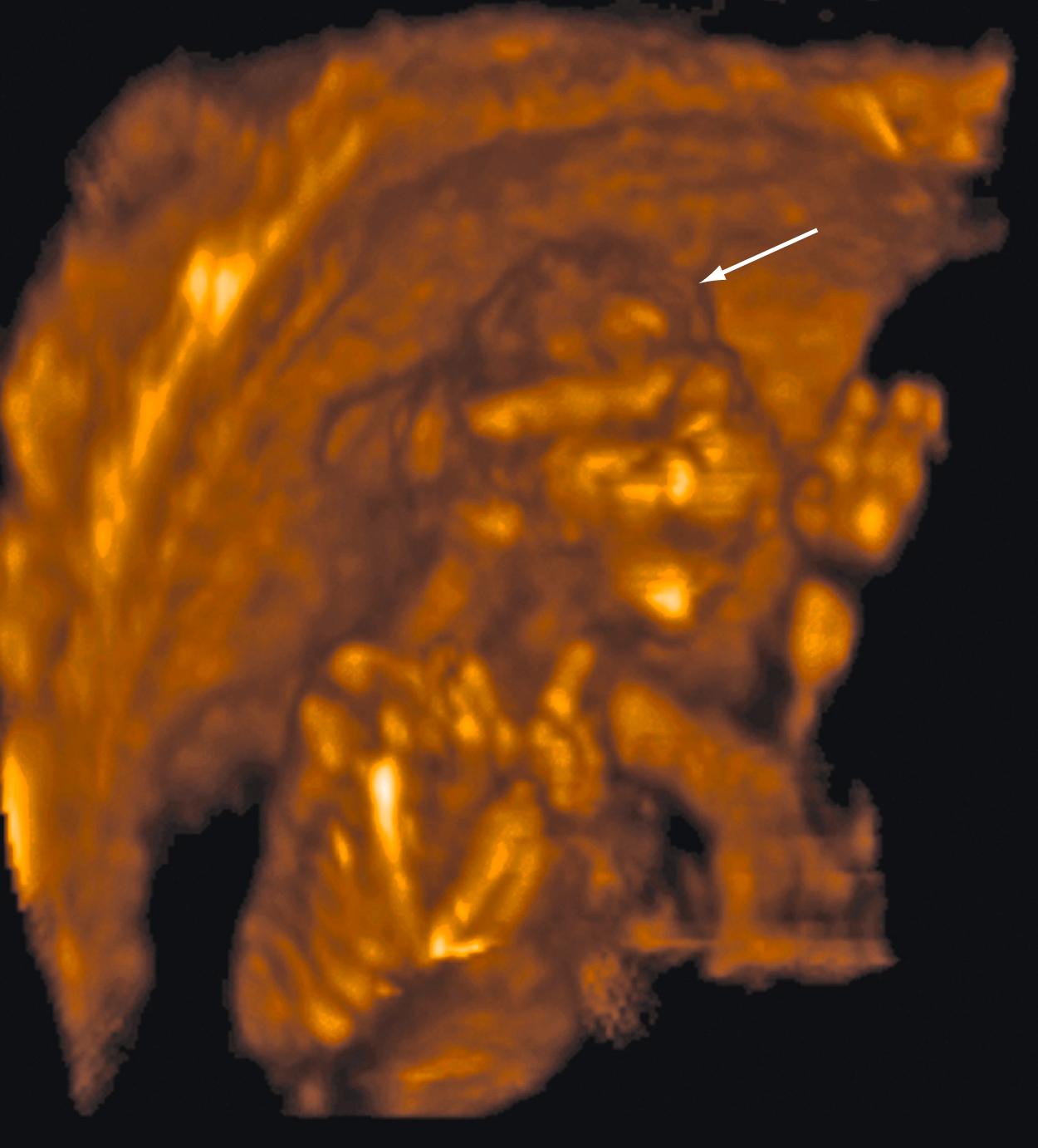
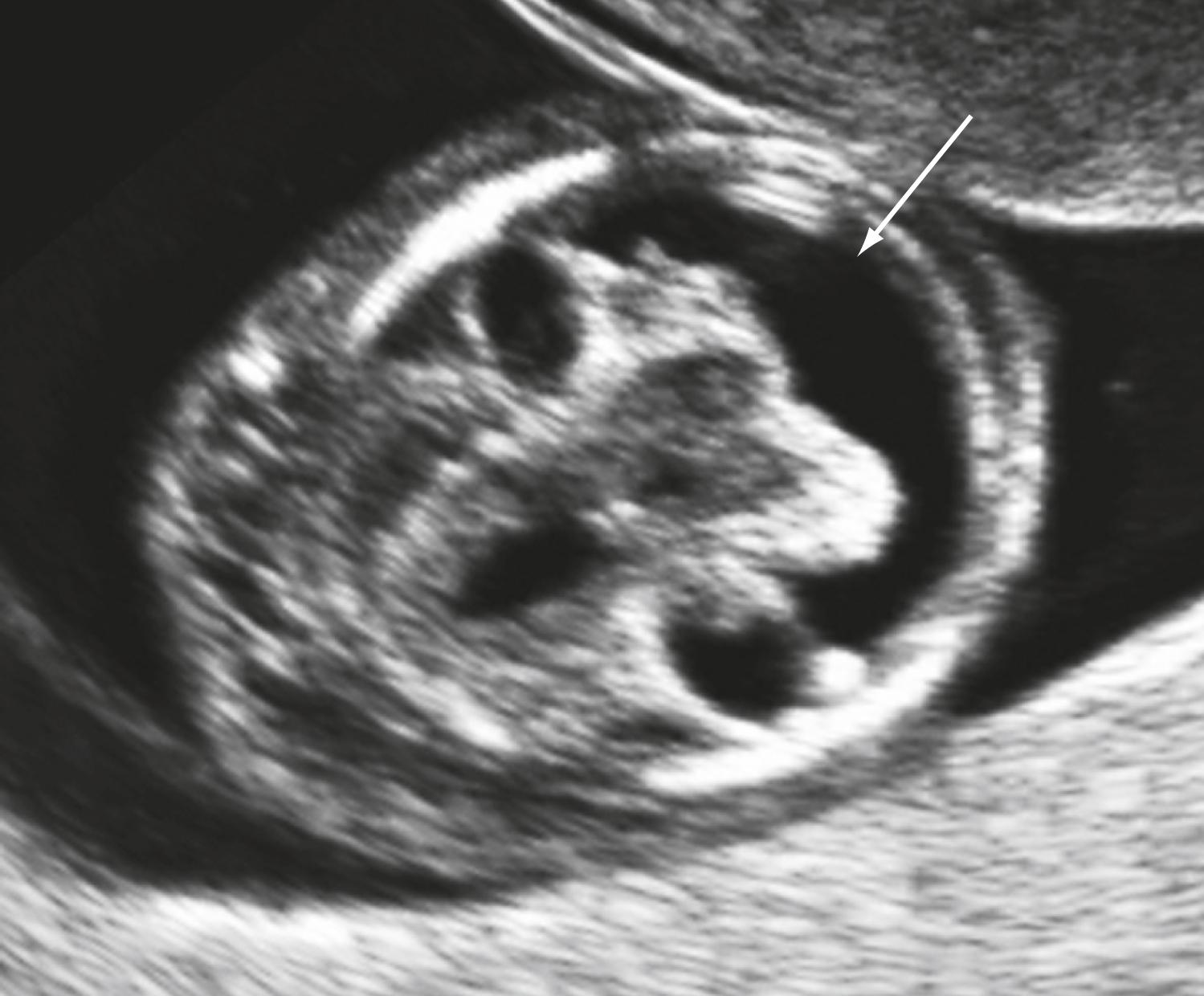
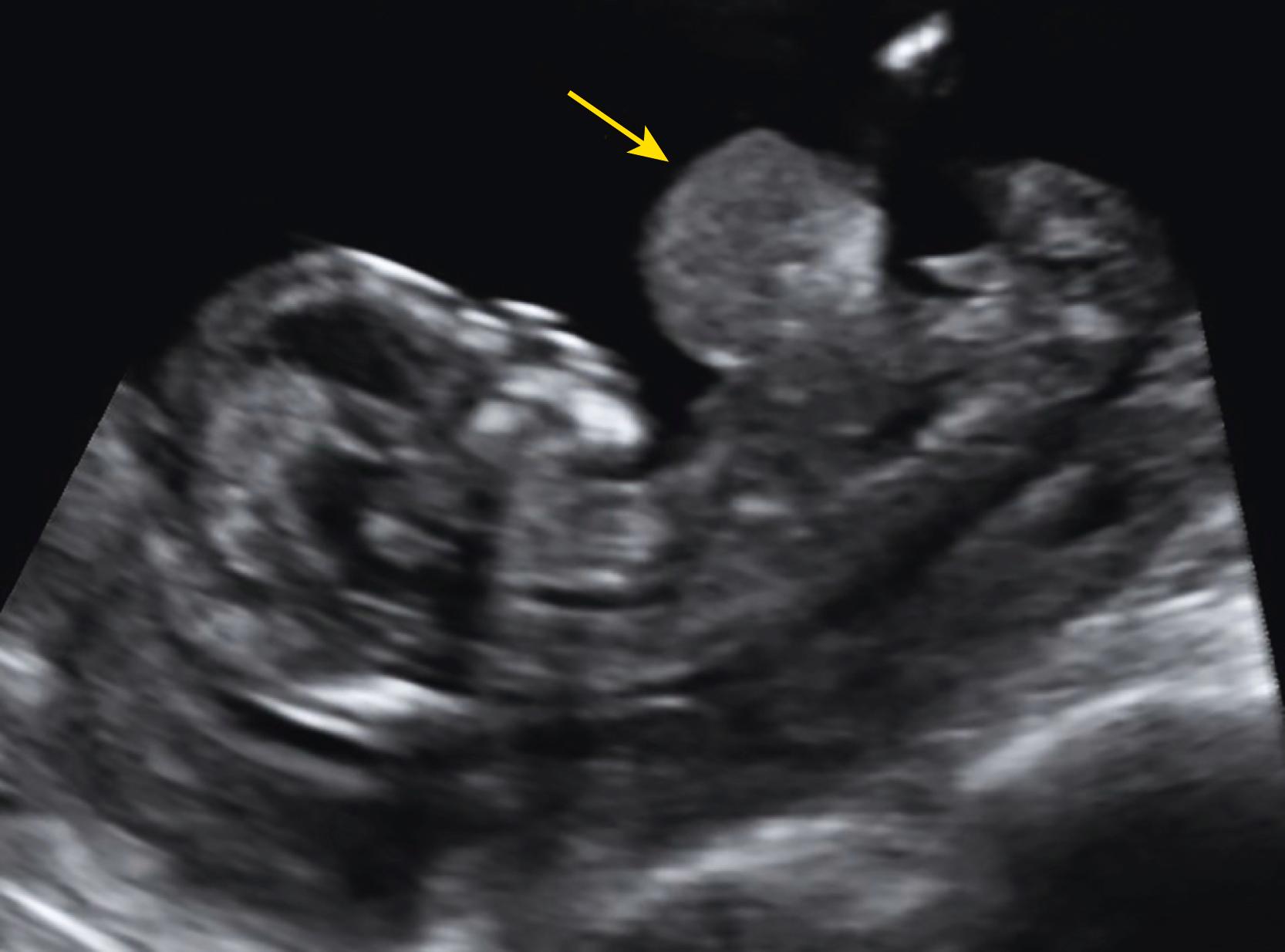
Approximately 40%–50 % of major cardiac defects can be identified at 14 weeks’ gestation or less. , , , The detection rate is higher with dedicated fetal echocardiography. ,
Many other anomalies may potentially be detected. Limb anomalies may be seen in the majority of cases, while detection of spina bifida, facial cleft, and gastrointestinal anomalies is more challenging , , , ( Figs. 29.6 through 29.8 ).
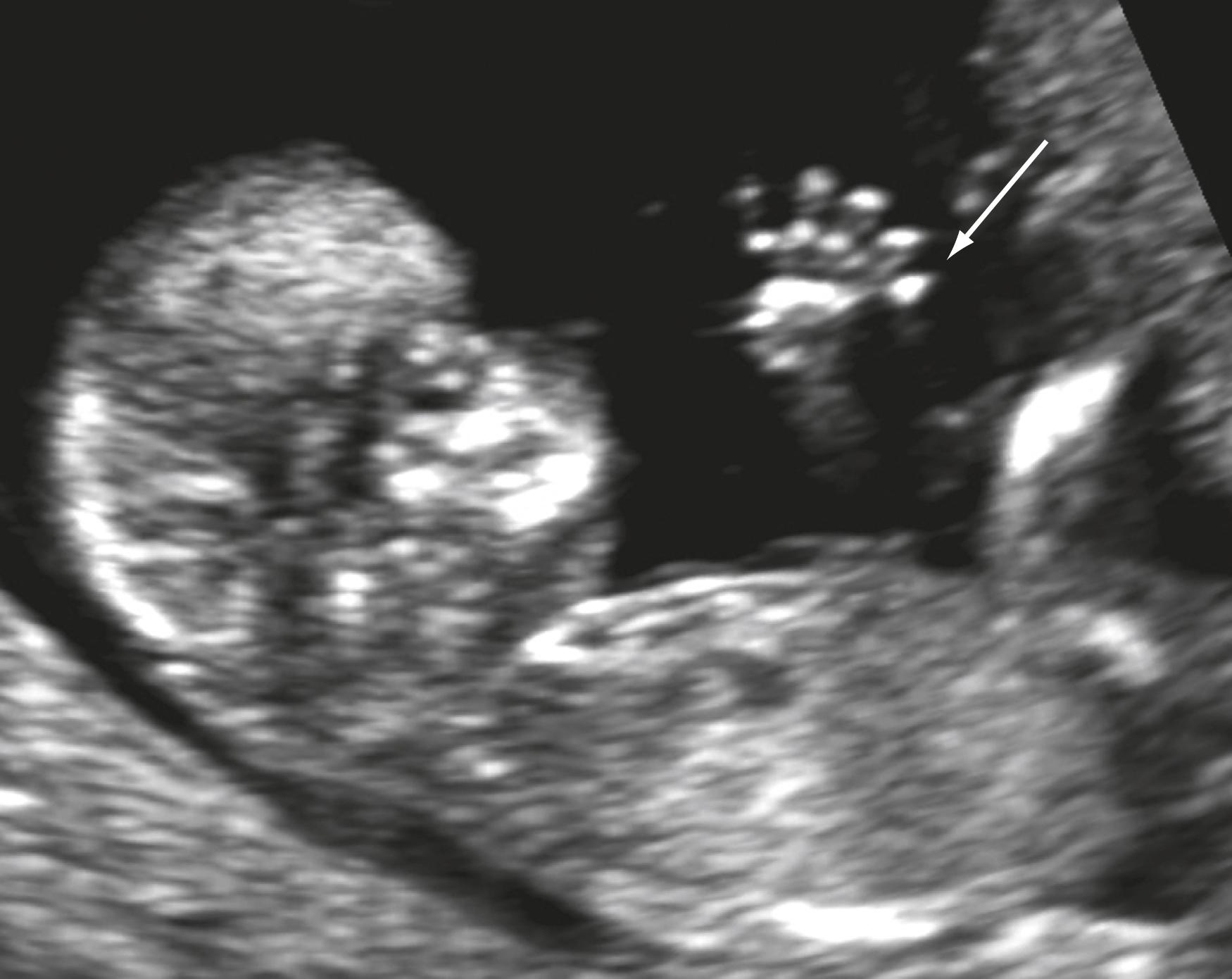
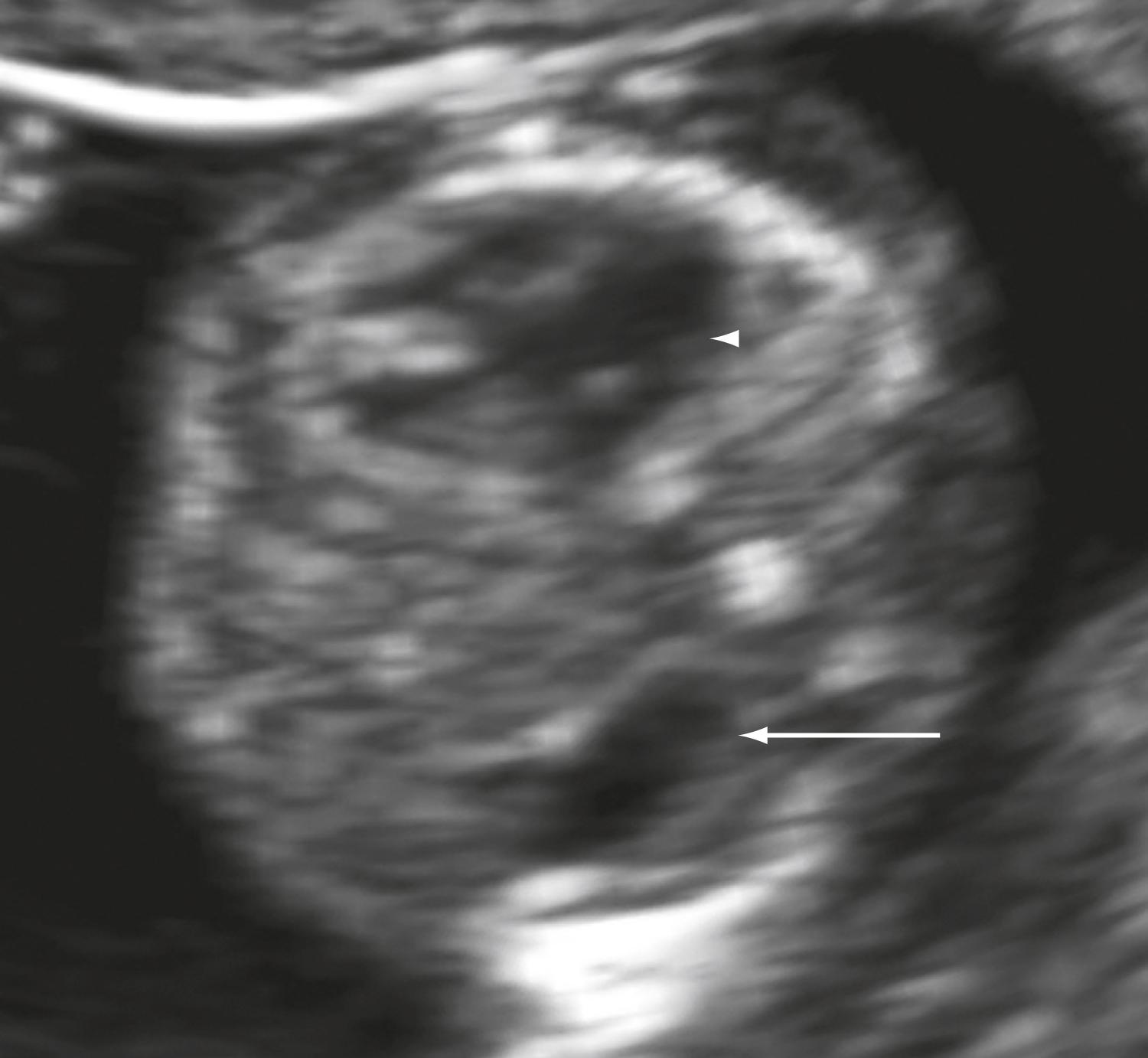
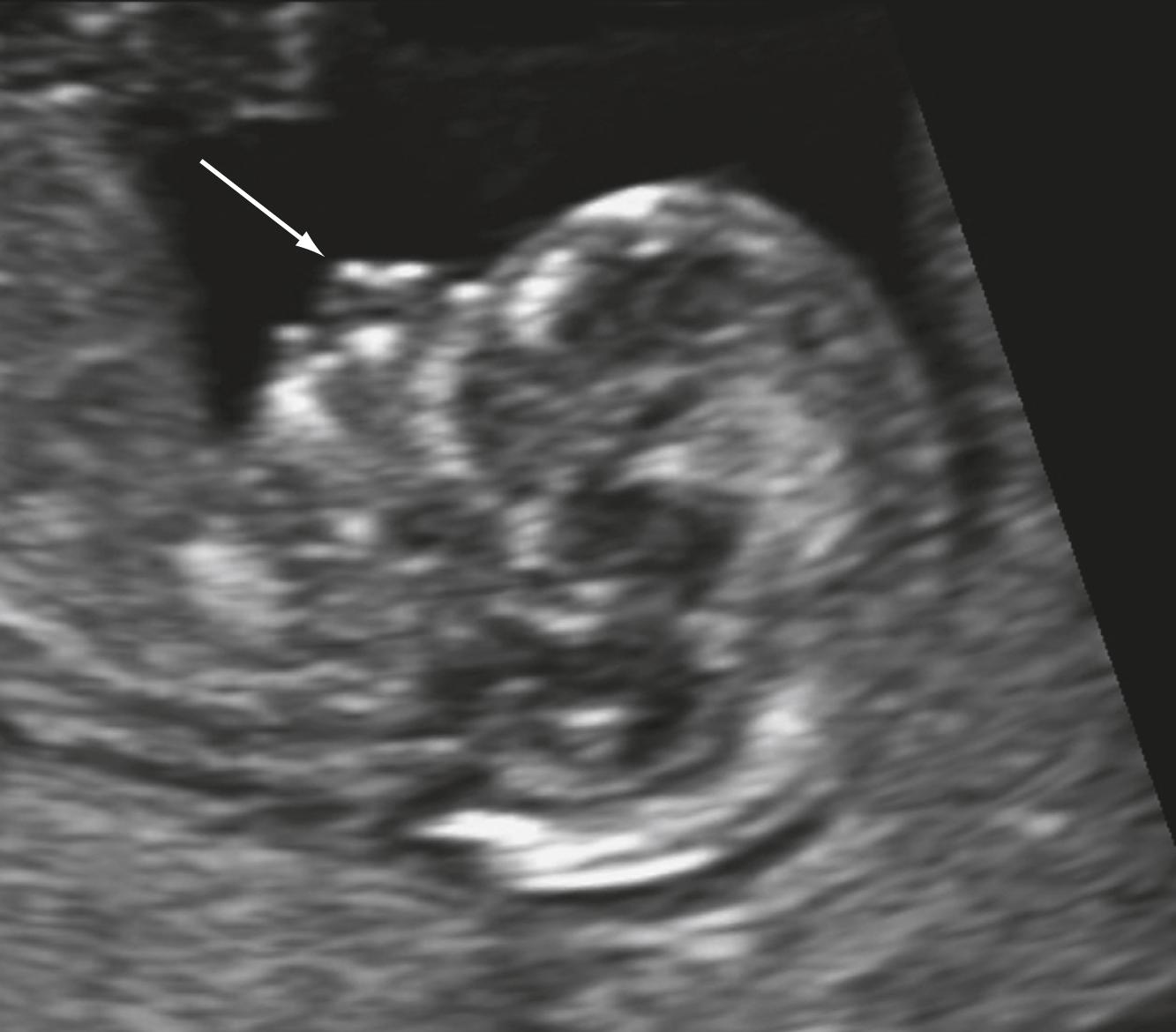
Attention to findings such as a maxillary gap, absence of a mandibular gap, position of the aqueduct of Sylvius, an abnormal intracranial lucency, and posterior fossa may be helpful in detecting specific anomalies ( Figs. 29.9 and 29.10 ).
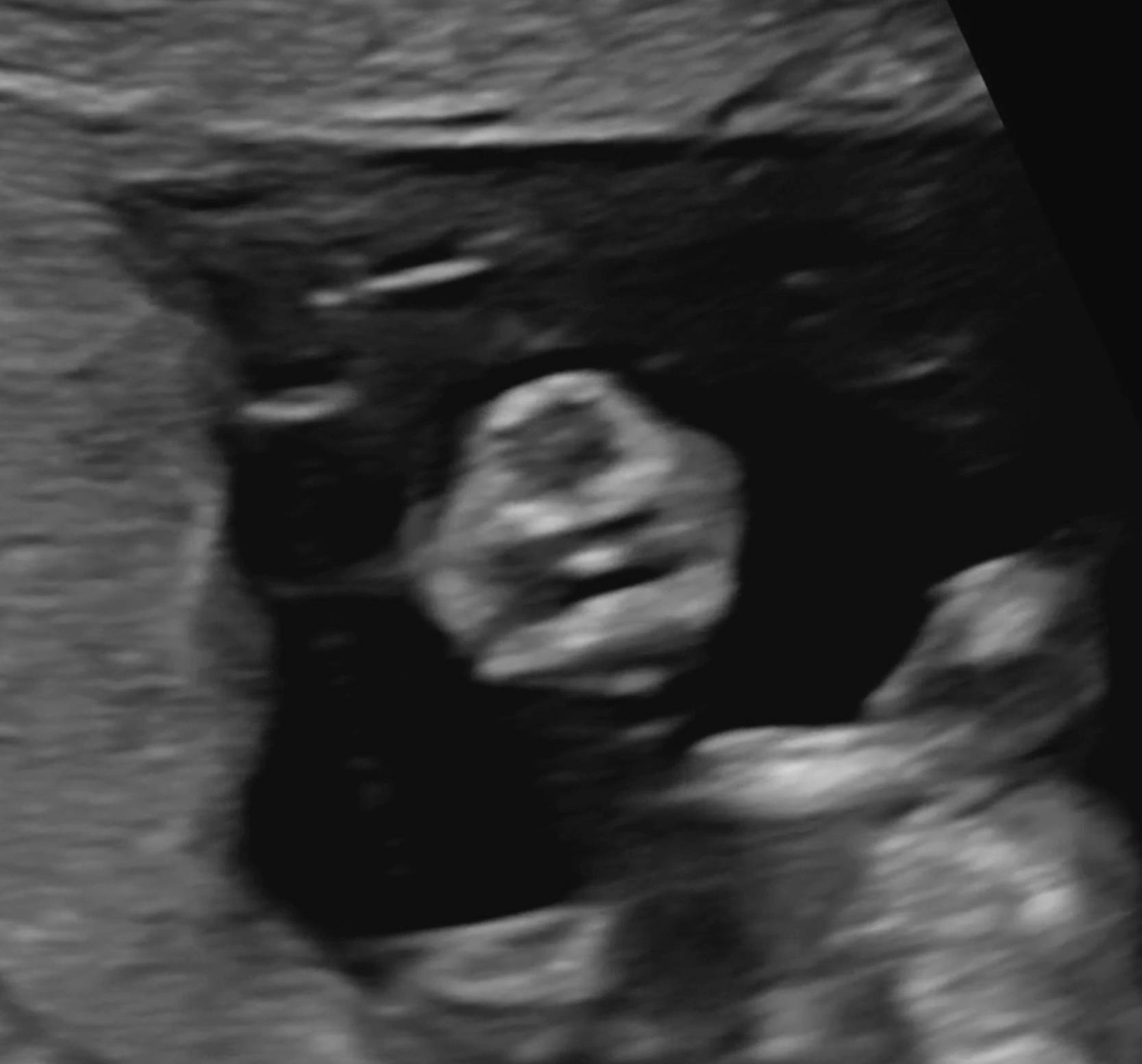
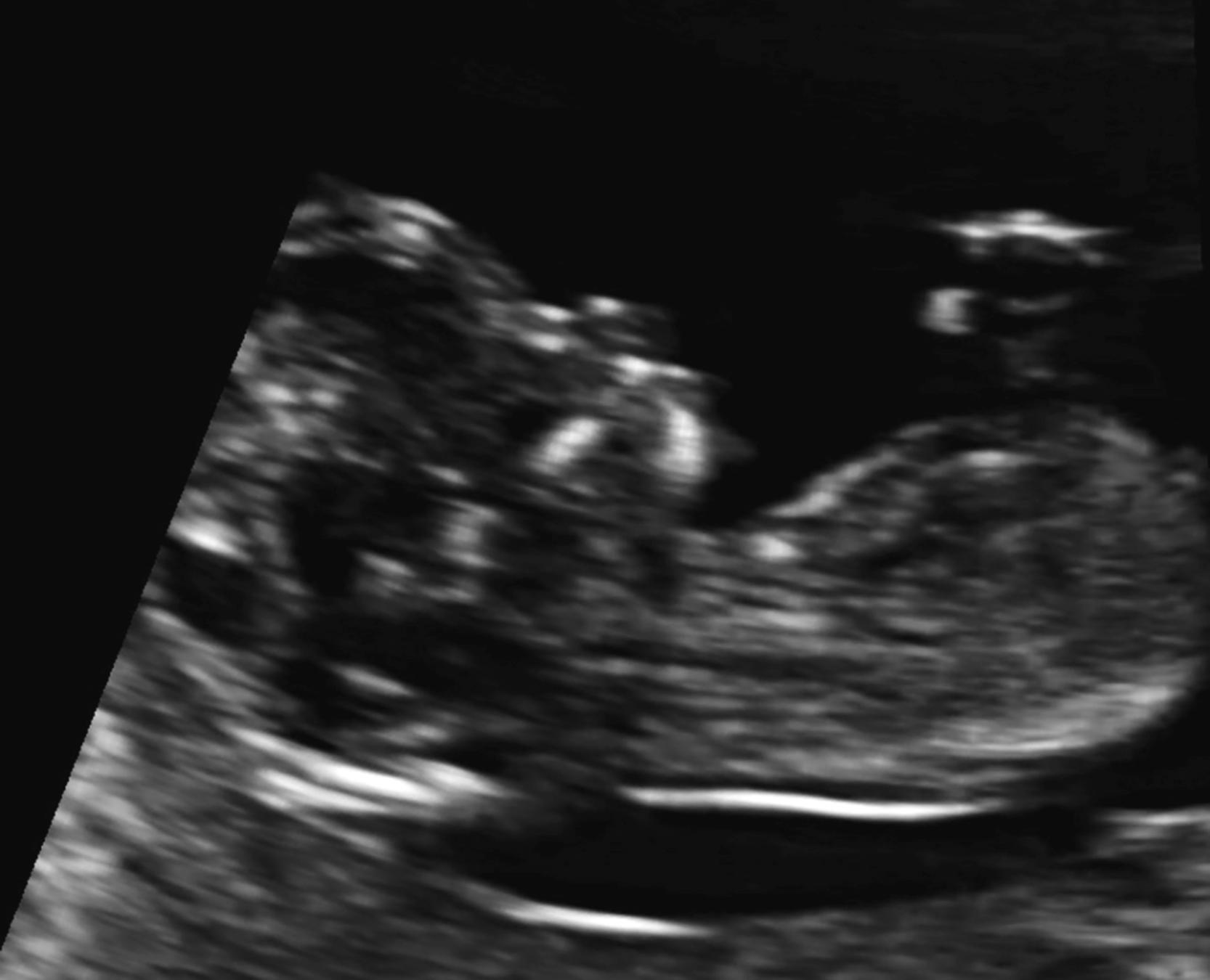
Some malformations are not detectable in the first trimester, including microcephaly and agenesis of the corpus callosum.
Fetuses with multiple anomalies (60%) are identified more often than those with a single anomaly (44%).
Fetal anomaly detection rate is higher in women at high risk (61%–65%) than in an unselected population (32%–50%). ,
Using a standardized anomaly screening protocol results in a higher sensitivity for the detection of anomalies. ,
Evaluation of anatomic structures should follow an established imaging protocol. The brain, spine, face, lungs, four-chamber heart, abdomen, gastrointestinal tract, kidneys, and extremities can be completed at 12–13 weeks in the majority of fetuses. ,
The falx cerebri and choroid plexus are visible by 10 weeks’ gestation. Lack of lateralization is abnormal and suggests holoprosencephaly.
Cranial bone ossification should be seen by 11 weeks’ gestation. Abnormalities in cephalic contour suggest cranial defects such as anencephaly or acrania. Acrania and anencephaly are differentiated based on the presence of cerebral tissues. Encephaloceles may be seen as a protrusion of brain tissue outside the cranium.
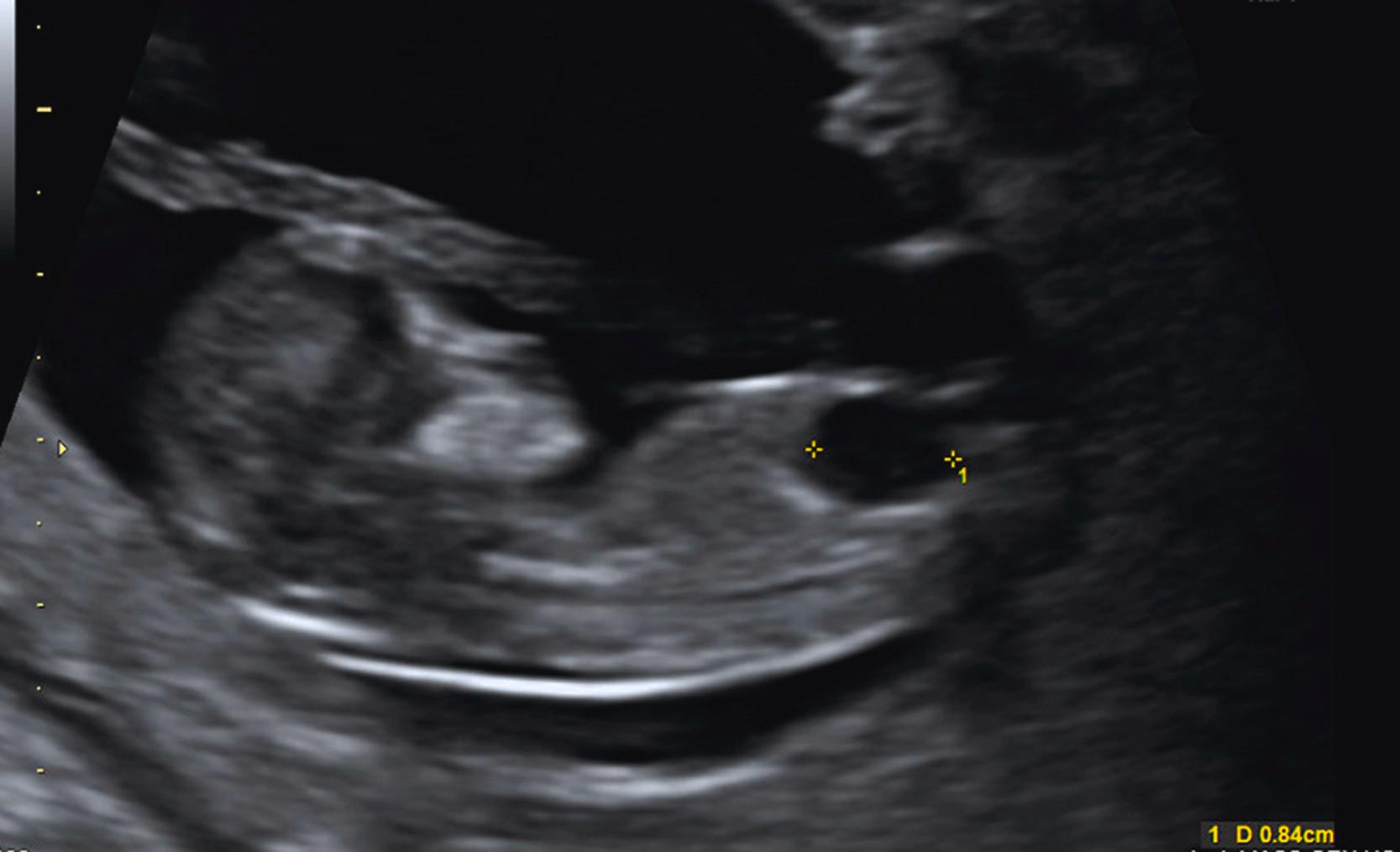
The fetal bladder is seen by 12 weeks’ gestation. Megacystis is identified when the longitudinal bladder diameter is ≥7 mm and may be associated with aneuploidy, lower urinary tract obstruction (LUTO), or other syndromes , (see Fig. 29.5 ). Resolution with normal outcome is also possible (see Fig. 29.15 , later). ,
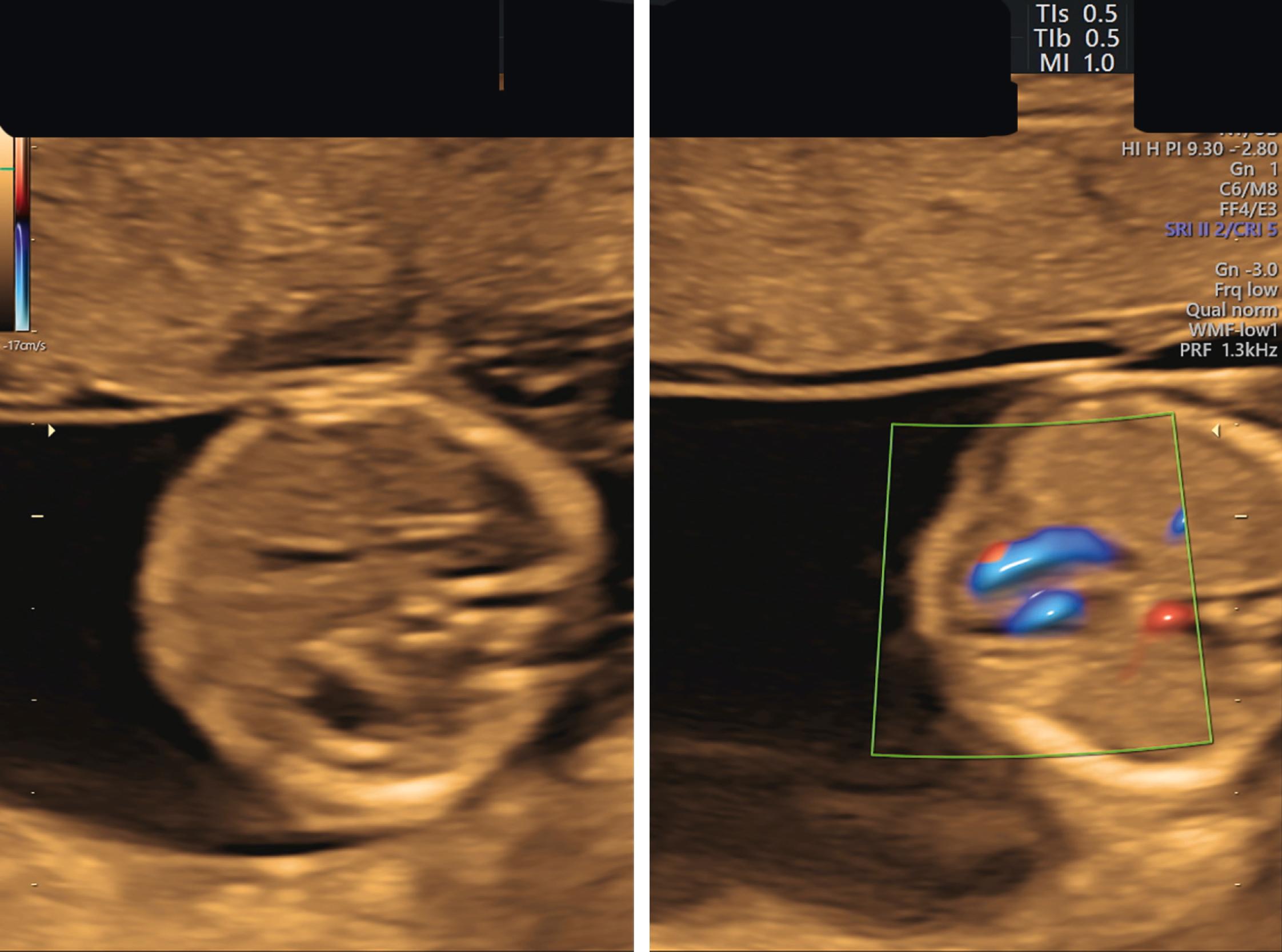
The atria, ventricles, atrioventricular valves, veins, and outflow tracts, including the three-vessel trachea view, are fully formed and may be identifiable during the 11–14 week scan. , , , A recent meta-analysis and systematic review of fetal echocardiography in the first trimester showed high sensitivity and specificity for diagnosing congenital heart disease, especially major congenital heart disease, where the pooled sensitivity was 84% and specificity was 100% ( Figs. 29.11 and 29.12 ; ![]() ).
).
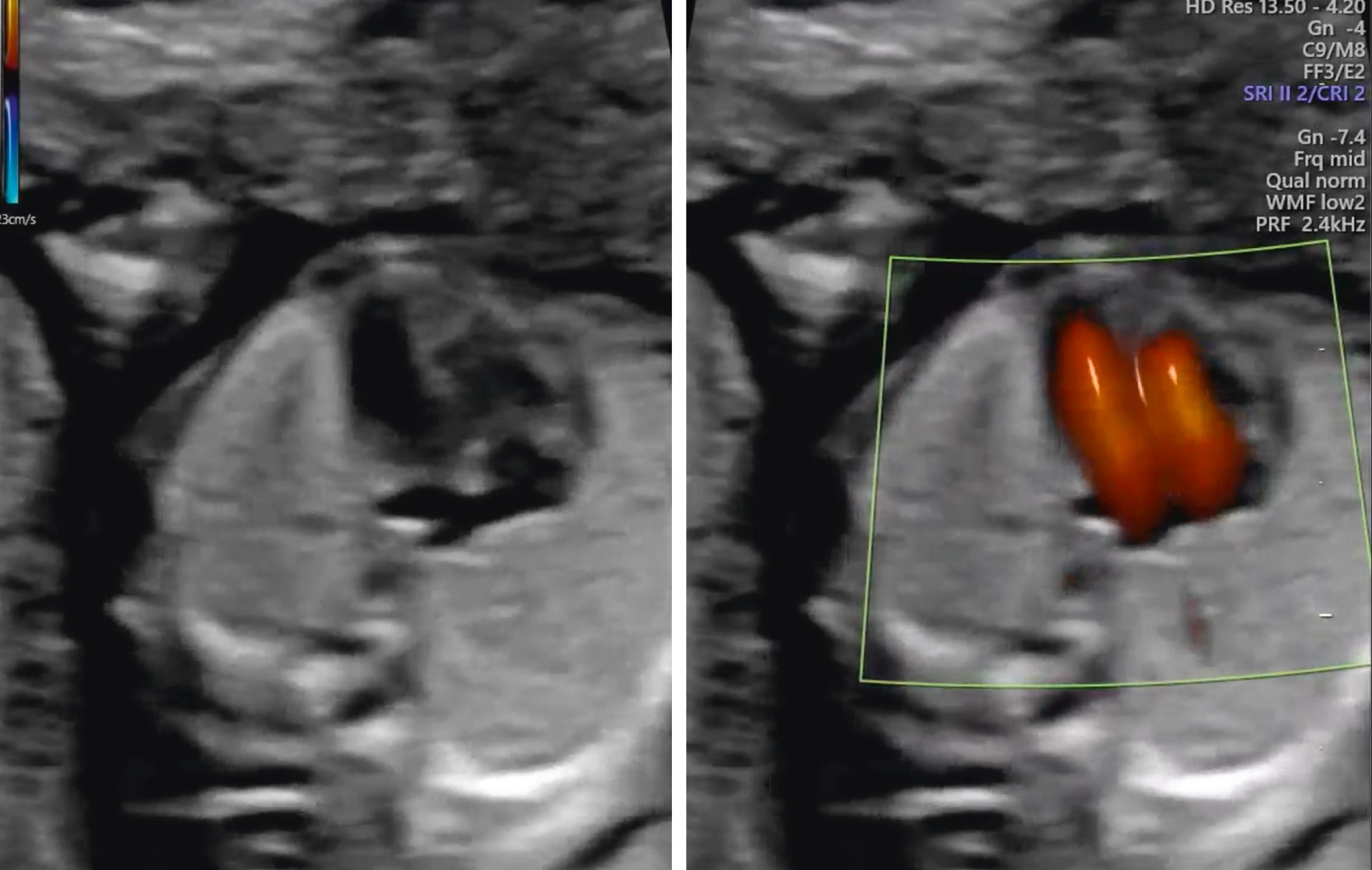
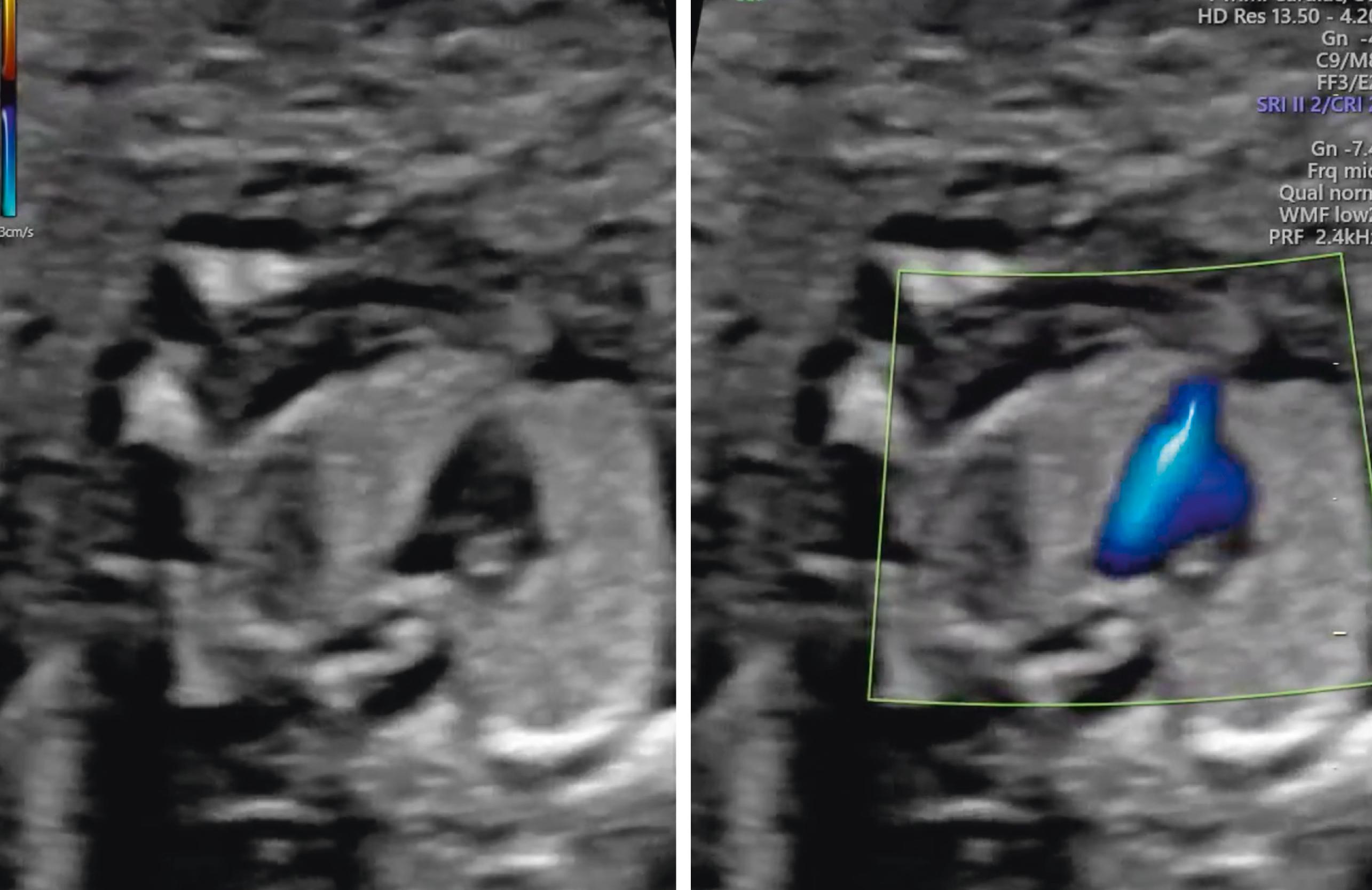
A high rate of detection of acrania or anencephaly, alobar holoprosencephaly, omphalocele, gastroschisis, megacystis, body stalk anomalies, ectopia cordis, and major limb abnormalities is expected in the first trimester. These anomalies should be specifically excluded.
Physiologic midgut herniation of bowel into the base of the umbilical cord begins in the embryonic period and is usually complete by the time the crown-rump length reaches 45–54 mm. In 5% of fetuses, midgut herniation may still be seen at 12 weeks’ gestation. If present at this stage, it should be followed up as it may represent an omphalocele or it may resolve spontaneously later in gestation ( Fig. 29.13 ).
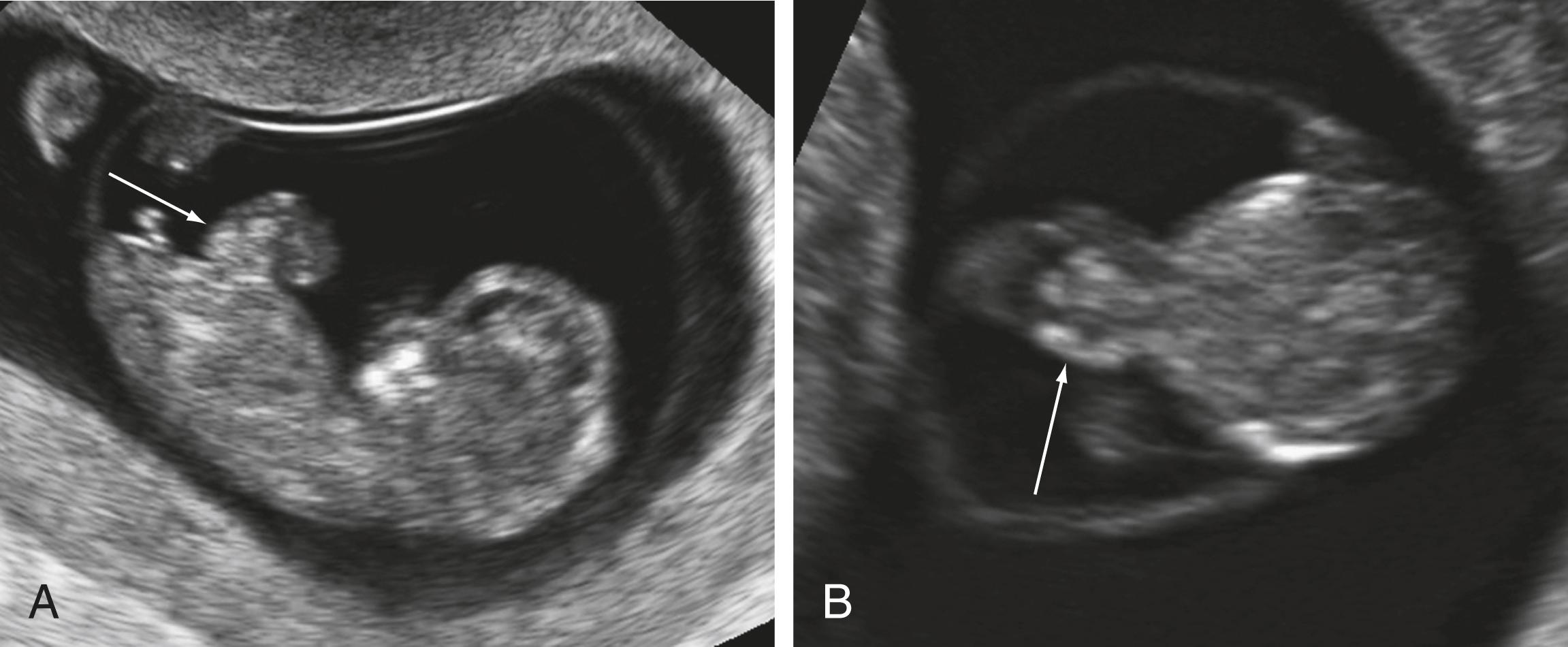
Liver identified in the base of the umbilical cord is always abnormal and consistent with an abdominal wall defect (see Fig. 29.4 ).
Normal appearance of the fetal kidneys in the first trimester does not exclude the development of a major renal abnormality such as polycystic kidneys, because these typically present later in gestation. Care must be taken not to mistake the fetal adrenal for the kidney.
The amniotic fluid is primarily a transudate across the fetal skin and is not dependent on fetal renal function in this gestational age window.
Practitioners should be familiar with the As Low As Reasonably Achievable Principle (ALARA). , Scans should be performed with attention to the output display standard (ODS), which is an on-screen display of the thermal index (TI). This should be monitored using the formula for bone, as ossification is present. The recommended ratio is 0.7 or less. Color Doppler may be used and is necessary for a detailed evaluation of the first-trimester fetal heart. Spectral Doppler should be reserved for specific clinical situations where information that is potentially obtained is clinically relevant ( Fig. 29.14 ).
The International Society of Ultrasound in Obstetrics and Gynecology (ISUOG) has published practice guidelines for the performance of a first-trimester fetal ultrasound scan that includes a suggested anatomic assessment at the time of the 11–14 week scan.
The American Institute of Ultrasound in Medicine has published a practice parameter for a detailed anatomic and placental assessment of the fetus in the first trimester that includes the evaluation of the fetal heart using color Doppler.
Many first-trimester imaging studies incorporate transvaginal sonography to increase the detection of structural defects. The use of both transabdominal and transvaginal imaging is associated with a higher detection rate of anomalies (62%) than either transabdominal (51%) or transvaginal (34%) imaging alone. The decreased field of view with transvaginal sonography may limit its application in some situations.
The natural history of antenatally diagnosed anomalies varies based on the specific defect. Some findings may improve or resolve over gestation, whereas others worsen. ,
Euploid fetuses with bowel-only omphalocele or megacystis ≤15 mm may resolve during the course of gestation with normal outcome ( Fig. 29.15 ).
Some CNS abnormalities may not be identified in the first trimester, including agenesis of the corpus callosum and cerebellar vermian abnormalities. The timeline for their normal development extends beyond the first trimester.
Certain cardiac abnormalities, such as coarctation of the aorta or hypoplastic left heart syndrome, may not be identified in the first trimester, because the pathologic process may occur later. There may be significant worsening or improvement of some anomalies over the course of gestation. , ,
If one defect is identified, a diligent search for other defects is essential.
The first-trimester anatomic scan does not replace a second-trimester anatomic scan, as some anomalies may not be apparent or detected until later in gestation and some anomalies require confirmation in the second trimester. Additionally, some anomalies may not be detected until the third trimester or even after birth. ,
In the presence of a structural defect, genetic counseling should be considered, as well as diagnostic testing. Microarray testing should be considered as part of the workup of those fetuses with structural defects, because cell-free fetal DNA may be falsely reassuring. , In the setting of genetic counseling by an experienced provider, whole exome sequencing may provide additional diagnostic yield in a fetus with hydrops or structural anomalies. ,
The prognosis depends on the specific structural anomaly, the severity of the lesion, and associated defects and potential genetic syndromes.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here