Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Hydrops fetalis is a pathological condition of excessive fluid accumulation in at least two extravascular compartments, including fetal soft tissues and body cavities. Hydrops fetalis is a clinical finding and not a final diagnosis.
There are two main pathophysiologies for hydrops fetalis, immune and nonimmune. For nonimmune, the diagnostic categories are placental, cardiovascular, chromosomal, haematologic, lymphatic dysphasia, infection, thoracic malformations, genetic syndromes, inborn errors of metabolism, extrathoracic tumours and genitourinary tract and gastrointestinal malformations. Undiagnosed causes are still common.
Hydrops fetalis should be considered as an urgent finding with timely evaluation, which requires a directed ‘stepwise approach’.
Fetal hydrops can cause a maternal physiological disturbance, mirror syndrome, with possible severe maternal consequences.
Fetal therapy is an option for certain causes of hydrops but requires complete evaluation and informed consent before its use.
Prognosis and recurrence risk counselling is dependent on the underlying cause, but overall the mortality rate remains high. If no specific diagnosis is made prenatally, autopsy should be offered for all perinatal losses because a significant proportion of cases have a genetic cause with risk of recurrence in future pregnancies.
Hydrops fetalis (fetal hydrops) is a pathologic condition of excessive accumulation of fluid in at least two extravascular compartments, including fetal soft tissues and body cavities. It is the physiological end-stage process in a number of fetal conditions and placental pathologies. The underlying causes are classified under two major categories, immune and nonimmune. In countries that have introduced immunoglobulin prophylaxis for Rhesus-D alloimmunisation, the incidence of immune fetal hydrops has significantly decreased, and in these countries, represents only 10% to 15% of fetal hydrops cases. Nonimmune fetal hydrops has a reported incidence of 1 in 2000 to 3000 pregnancies but accounts for a disproportionate share (3%) of overall mortality in the perinatal period. There is a broad spectrum of well-recognised causes, and with the use of next-generation sequencing (NGS) technology, an increasing number of single-gene disorders are being diagnosed in cases of fetal hydrops. Determining the cause of the fetal hydrops is important because some conditions are treatable. Furthermore, knowing the underlying cause provides additional information that may be useful to predict the prognosis and is a key element to determine recurrence risk in future pregnancies.
The diagnosis of immune (IHF) or nonimmune hydrops fetalis (NIHF) requires an abnormal fluid accumulation in two or more fetal body compartments. Body compartments (and the fluid collections) are designated as subcutaneous space (skin oedema or cystic hygroma [CH]), pleural space (pleural effusion), pericardial space (pericardial effusion) and abdomen (ascites).
Subcutaneous skin or scalp oedema requires an ultrasound measurement or thickness of at least 5 mm ( Fig. 36.1 ). The fluid collection over the scalp or forehead can be identified with the use of both the transverse and sagittal planes. Skin thickening of at least 5 mm is required because some macrosomic fetuses may have thickened skin secondary to fat deposition measuring up to 5 mm.
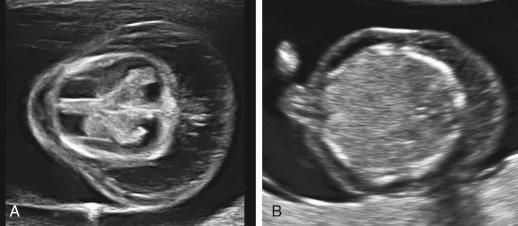
Cystic hygroma is a cystic lymphatic lesion. It can be a single or multiple congenital cysts, most commonly found within the soft tissues of the neck. Generally, the CH is bilateral, asymmetric, with thin-walled, multiseptate cystic lesions, and often located posterior and lateral to the high cervical vertebrae. Nuchal CH is the clinical consequence of a delay in development or absence of the communications that normally develop between the jugular lymph sacs and the internal jugular veins at approximately 40 days of gestation.
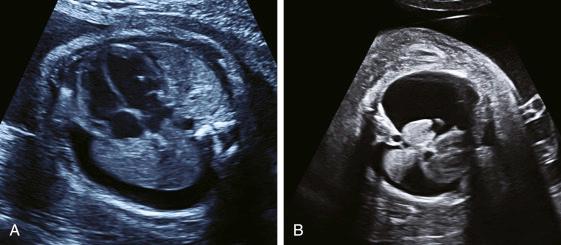
Pleural effusion is an accumulation of fluid in the pleural space. Effusion composition can be either chylous or clear (hydrothorax) with most primary congenital effusions being chylous and occurring on the right. Ultrasound identifies an anechoic space peripherally around the compressed lung. If the effusion is unilateral and large, there may be additional mediastinal shift; bilateral effusions can be either symmetric with no shift or asymmetric with shift ( Fig. 36.2 ).
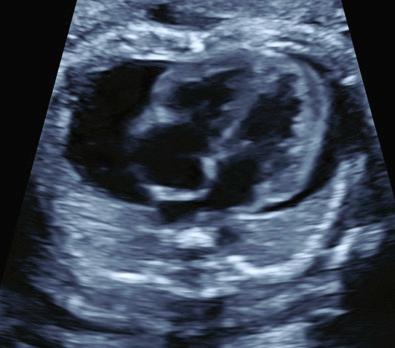
Pericardial effusion is identified by the appearance of an echolucent rim greater than 2 mm around both cardiac ventricles ( Fig. 36.3 ). Pericardial effusions of 1 to 2 mm are considered a physiological fluid collection.
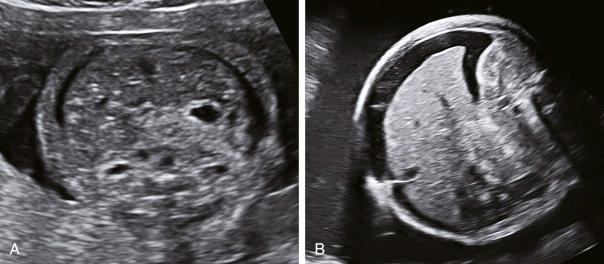
Fetal ascites is identified by ultrasound by visualisation of an echolucent fluid rim encompassing the entire fetal abdomen in the transverse view, usually at the level of the umbilical cord insertion or liver. The echolucent collection of fluid outlines the visceral contents ( Fig. 36.4 ).
Fetal hydrops is the result of abnormal fluid movement between the plasma and tissues that leads to excess fluid in both the tissues and serous cavities (skin oedema, ascites, pleural and pericardial effusions). Four main mechanisms have been postulated to explain this abnormal distribution of body fluids: (i) an increase in hydrostatic capillary pressure (resulting for primary or secondary heart failure or from obstruction of venous return), (ii) a reduction in plasma osmotic pressure (from decreased albumin production or increased albumin loss), (iii) obstruction or reduction of lymphatic flow and (iv) damage to peripheral capillary integrity. Table 36.1 provides examples of conditions linked to each mechanism. In some conditions, more than one mechanism may be involved.
| Mechanism | Hydrops fetalis conditions |
|---|---|
| Increased hydrostatic capillary pressure | Fetal congestive heart failure caused by fetal arrhythmias, congenital heart disease, obstructive intracardiac tumour, fetal anaemia Elevated central venous pressure secondary to high intrathoracic pressure caused by intrathoracic masses or heart failure |
| Reduced intravascular osmotic pressure | Fetal hypoalbuminaemia secondary to haepatic failure seen in metabolic conditions or increased albumin loss seen in chylothorax or nephrotic syndrome |
| Obstructed or reduced lymphatic flow | Disorders associated with abnormal lymphatic development (e.g., Down syndrome, Noonan syndrome, primary lymphangiectasia) or with decreased lymph flow associated with reduced fetal movement (e.g., fetal akinesia sequence, multiple pterygium syndrome) |
| Damaged peripheral capillary integrity | Conditions of fetal hypoxemia caused by fetal anaemia or placental insufficiency Conditions of fetal inflammation caused by fetal infection |
Maternal sensitisation to a fetal red blood cell (RBC) antigen (usually from a previous pregnancy) is followed in the next pregnancy by transplacental passage of the circulating maternal IgG antibody against that red blood cell antigen resulting in fetal haemolytic anaemia, and if severe, fetal hydrops. The severity of the haemolytic process is dependent on the antigen–antibody involved and the maternal antibody load. Chapter 40 details the diagnosis and management of RBC alloimmunisation. This aetiology for hydrops will not be further discussed in this chapter. However, in the approach to a fetus with fetal hydrops, immune fetal hydrops is an important condition to consider because it is one of the treatable causes with strong evidence-based approaches.
Nonimmune hydrops fetalis has a variety of aetiologies. A systematic review of the aetiology of NIHF divided the affected cases into 14 classification groups ( Table 36.2 ). This section reviews the different NIHF aetiologies according to the same classification. Some of the aetiologies of NIHF are the focus of other chapters in this book. These aetiologies will only be mentioned briefly in this chapter.
| Category | Organ or System | Most Frequent Pathologies | Fetal Therapy |
|---|---|---|---|
| Placental or umbilical cord | Placenta or umbilical cord (5.3%) | Vascular shunting, TTTS, TRAP, chorioangioma | Endoscopic laser, RFA |
| Fetal | |||
|---|---|---|---|
| Cardiovascular (21.4%) | Abnormal structural Arrhythmias Tumour |
Balloon dilatation for stenotic valves Systemic or fetal medication No prenatal option |
|
| Chromosomal (12.5%) | Trisomy 21, trisomy 18, Turner syndrome | No option | |
| Haematologic (10.1%) | α-Thalassemia other rare inherited forms of anaemia | IUT in selected cases | |
| Lymphatic dysplasia (7.5%) | Chylothorax Congenital lymphatic dysplasia Noonan syndrome |
Percutaneous shunt or pleurodesis | |
| Infection (6.8%) | Parvovirus B19, CMV, toxoplasmosis, syphilis, varicella, rubella | IUT for anaemia seen with parvovirus B19 only | |
| Thoracic malformations (5.3%) | Diaphragmatic hernia CPAM |
Experimental balloon tracheal occlusion Steroids, RFA, percutaneous shunt, pleurodesis |
|
| Syndromic (4.6%) | Multiple pterygium, fetal akinesia, skeletal dysplasias | No option | |
| Genitourinary tract (2.0%) | Finnish nephrosis Bladder outlet obstruction |
No option Shunt, percutaneous cystoscopy |
|
| Inborn errors of metabolism (1.1%) | Lysosomal storage disorders | No option | |
| Extrathoracic tumours (0.7%) | SCTs | Open fetal surgery | |
| Gastrointestinal (0.7%) | Meconium peritonitis, intestinal atresias | No option | |
| Miscellaneous (3.7%) | |||
| Idiopathic (18.2%) | Case by case |
Twin-to-twin transfusion syndrome (TTTS) is a complication associated with monochorionic twins caused by an imbalance of the vascular communications in the placenta between the two fetuses and represents approximately 5% of cases of NIHF. Haemodynamic and osmotic changes in the fetuses can lead to fetal hydrops in the recipient twin most commonly, but donor fetal hydrops has been identified as well. (See Chapter 44 for more details on TTTS.)
Placental chorioangiomas are rare benign placental tumours. Large chorioangiomas act as peripheral arteriovenous shunts, leading to increased cardiac output, cardiomegaly and finally heart failure and hydrops. Some cases are complicated by fetal anaemia, which further contributes to the development of the heart failure and hydrops. Umbilical cord haemangiomas have also been reported with NIHF. (See Chapter 9 for more on placental pathology.)
Cardiovascular conditions are the primary aetiology in 21% of NIHF cases and are identified as the most common group of disorder leading to NIHF. The cardiovascular aetiology can be divided into structural malformations, arrhythmias, cardiac tumours and cardiomyopathy ( Table 36.3 ). (See Chapter 29 for more details on cardiovascular conditions.)
| Cardiovascular Condition | Specific Diagnosis | Key Points |
|---|---|---|
| Congenital heart defect (CHD) | Hypoplastic left heart syndrome (HLHS) |
|
| Atrioventricular (AV) canal defect |
|
|
| Atrial septal defect |
|
|
| Ventricular septal defect |
|
|
| Tetralogy of Fallot |
|
|
| Hypoplastic right heart syndrome |
|
|
| Ebstein anomaly |
|
|
| Truncus arteriosis |
|
|
| Transposition of the great arteries (TGA) |
|
|
| Aortic stenosis or atresia |
|
|
| Cardiomyopathy |
|
|
| Endocardial fibroelastosis |
|
|
| Premature closure of ductus arteriosus |
|
|
| Premature closure of foramen ovale |
|
|
| Cardiac arrhythmia | Supraventricular tachycardia (SVT) |
|
| Atrial flutter |
|
|
| Bradyarrhythmia or congenital heart block (CHB) |
|
|
| Cardiac tumours | Rhabdomyoma |
|
| Intrapericardial teratoma, fibroma, myoma |
|
For structural cardiac malformations, fetal hydrops can result from lesions that place a burden on the right-sided cardiac chambers, resulting in increased right atrial pressure such as left-sided obstructive lesions, hypoplastic left heart syndrome or premature closure or restriction of the ductus venosus and right-sided obstructive lesions such as pulmonary and tricuspid atresia. Nonobstructive lesions that result in right atrial volume overload such atrioventricular canal defect are reported in fetal hydrops.
Fetal tachyarrhythmias are the second most common cardiovascular abnormality associated with fetal hydrops. This aetiology has evidenced-based treatment protocols with secondary reversal of the fetal hydrops. Fetal bradyarrhythmias are less commonly associated with fetal hydrops and are not easily treatable.
Intracardiac tumours represent a rare cause of fetal hydrops. The most common fetal cardiac tumour is a rhabdomyoma with fetal hydrops caused by tumour obstruction to cardiac flow or an associated arrhythmia. Cardiac rhabdomyomas are frequently a fetal manifestation of tuberous sclerosis (autosomal dominant condition) and when suspected prenatally should result in a careful family history and assessment of the couple. (Details on fetal tumours are provided in Chapter 37 .)
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here