Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Extremity compartment syndrome occurs whenever the tissue pressure within a limited space of the body reaches the point where the circulation, nerve function, and muscle function of that space are compromised. For compartment syndrome to occur, the body compartment should be enveloped by fascia that prevents inner tissue expansion, and there should be at least one cause of increased tissue pressure present, either externally or internally.
The German physician Dr. Richard Von Volkmann first described the late sequelae of compartment syndrome in 1881. He wrote: “The paralyses and contractures that follow tightly applied bandages, chiefly in the forearm and hand, and less often in the lower extremity, are to be viewed as ischemic. They arise from the arterial supply being interrupted for too long.” Volkmann’s ischemic muscle contractures of untreated forearm compartment syndrome are named after him ( eFig. 151.1 ). Later, in 1912, Wilson first described exertional compartment syndrome, and Mavor, in 1956, first reported chronic exertional compartment syndrome in a football player. Since then, various cases of compartment syndrome have been reported in the literature, and the pathophysiology and treatment options have been discussed. Compartment syndrome has been reported in a wide variety of clinical conditions, including tetanus, meningococcemia, malignant hyperthermia, frostbite, horseback riding, and childbirth. Typically, it occurs after traumatic events, most commonly those involving fractures or vascular trauma with subsequent ischemia-reperfusion injuries. The most commonly affected body compartments are lower extremity compartments. The literature describes an increasing incidence of around 2% in severely injured patients. This occurs with developments in shock resuscitation in a population of patients with otherwise noninjured extremities. The incidence of compartment syndrome varies depending on the patient population studied and the etiology of the syndrome. In a group of patients with leg pain, according to Qvarfordt and colleagues, 14% were noted to have anterior compartment syndrome. Compartment syndrome was also seen in 1%–9% of leg fractures.

Common locations of compartment syndromes are in the upper and lower limbs. The most commonly affected are the four compartments of the lower extremity (anterior, lateral, superficial posterior, and deep posterior), followed by two compartments of the forearm (volar and dorsal). Other compartments that may be affected are the deltoid and biceps compartments of the arm; interosseous compartments of the hand; gluteal compartment of the buttock; quadriceps compartment of the thigh; and interosseous, medial, central, and lateral compartments of the foot.
The etiology of compartment syndrome varies and can be divided into three major groups: decreased compartmental volume, increased compartmental content and therefore pressure, and externally applied pressure. There are probably more than 40 causes that fit into one of these groups. Common causes in critical care surgical cases are presented in Table 151.1 .
| Decreased compartmental volume |
|
| Increased compartmental content |
|
| Externally applied pressure |
|
The most common causes of extremity compartment syndrome are categorized based on increased compartmental content. The pathophysiologic mechanism of bleeding is easy to understand. Most commonly, bleeding is caused by trauma, but recently, more reports of atraumatic bleeding caused by wide use of warfarin, low-molecular-weight heparins, or recombinant tissue plasminogen activator (rtPA) have been reported.
Increasing volume of blood in a space limited by noncompliant fascia results in an exponential rise in intracompartmental pressure. Postischemic swelling or reperfusion injury is more complex because it causes the so-called double ischemic insult. Initial ischemic insult from any cause leads to abnormal function of all tissues, including nerves, muscles, and capillaries. This results in abnormalities of neuromuscular function; this is the first insult. Increased permeability after the relief of initial ischemia leads to postischemic swelling and subsequently increased compartmental volume and pressure. This leads to the development of compartment syndrome, which causes additional injury to neuromuscular function; this is the second insult. This process has clinical consequences. The classic physical examination in patients with reperfusion injury can be unreliable because of loss of motor and nerve function. , ,
Most extremity compartment syndromes in patients who are able to cooperate can be diagnosed by a clinical examination. The most common clinical signs found in the literature are the classic 5 Ps: pain (out of proportion), pulselessness (or weak pulse), pallor, paralysis (numbness and loss of motor function), and pressure (swelling and tenseness of compartment). Occasionally, poikilothermy (cold extremity) is also included ( Fig. 151.1 A). These signs are a consequence of increased intracompartmental pressure and loss of various tissue functions. Other signs include skin edema and blisters, swelling, and subcutaneous blood suffusions (see Fig. 151.1 B).
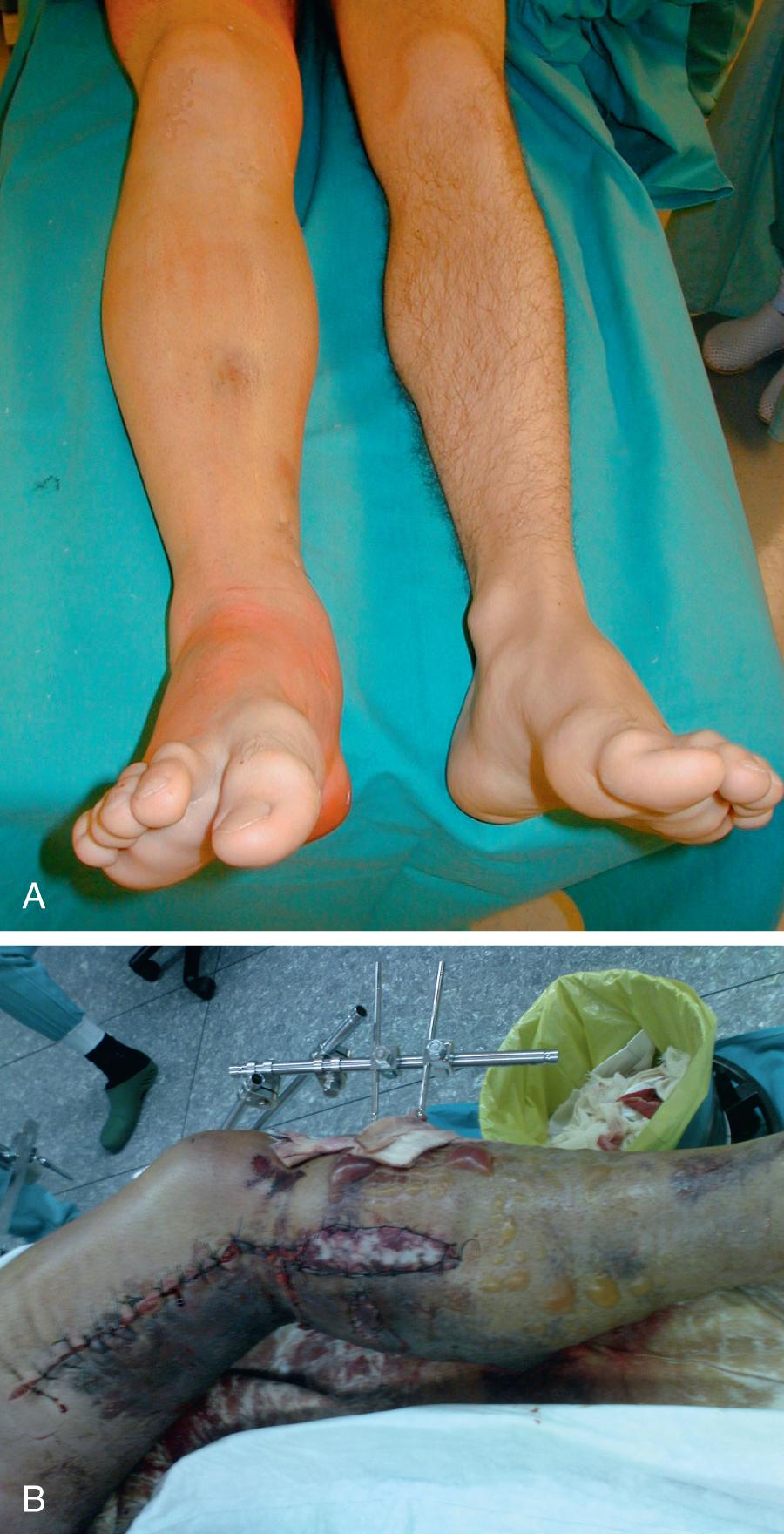
To make the diagnosis, there must first be evidence of increased intracompartmental pressure. If so, these signs do not occur simultaneously but develop with time. One of the first signs is a swollen or tight compartment in combination with severe pain that is out of proportion to the injury and is not relieved by typical analgesia. Other signs present late and often, when present, represent irreversible damage to soft tissues. Numerous other pathophysiologic events can cause a similar clinical picture. In fact, a large meta-analysis of studies comparing clinical signs with the development of acute lower extremity compartment syndrome showed a sensitivity of 13%–19%, specificity of 97%, positive predictive value of 11%–15%, and negative predictive value of 98%. Thus the absence of this sign rules out compartment syndrome, but the presence rarely confirms the correct diagnosis.
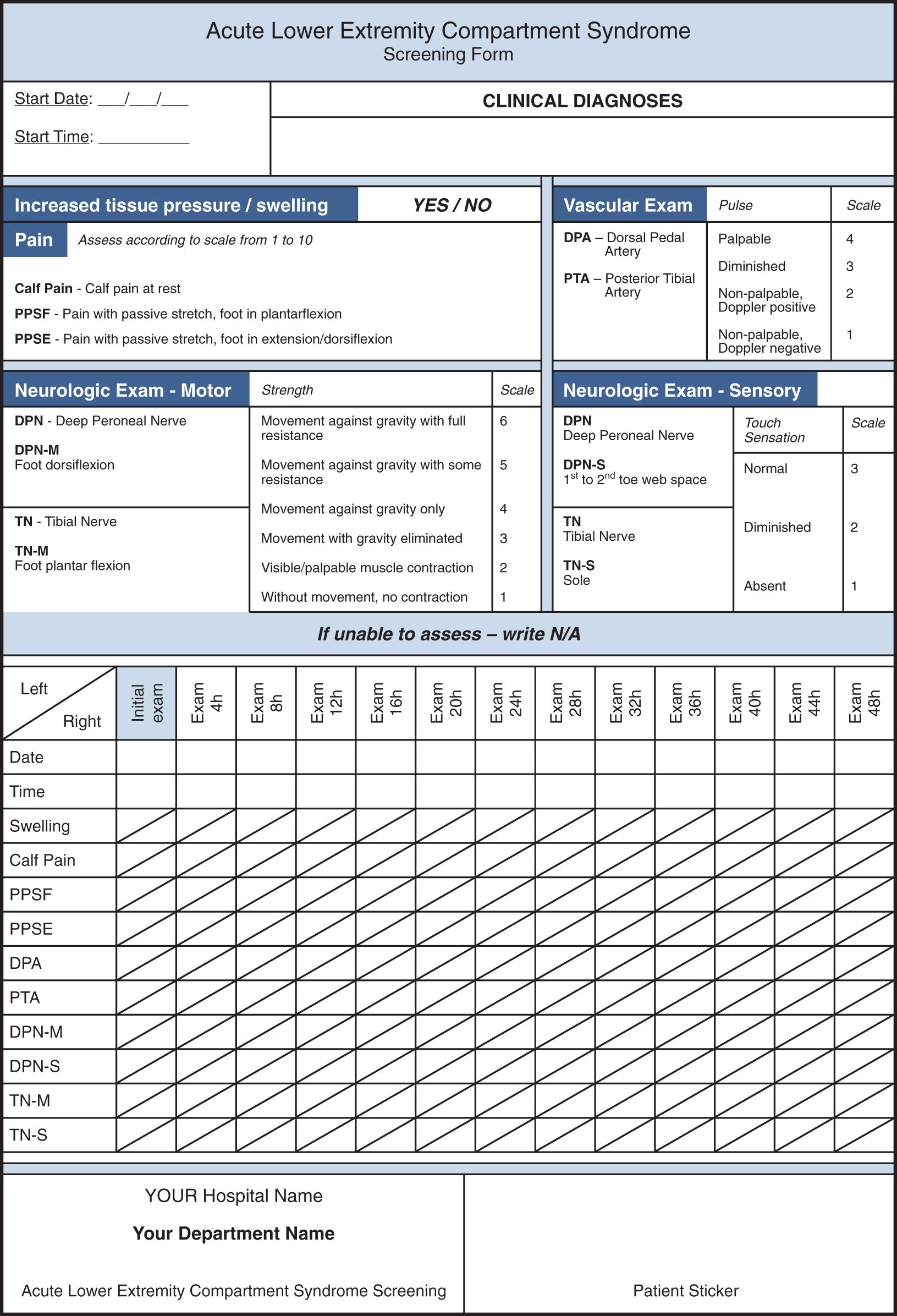
Nevertheless, clinical observation of the suspected compartment is what is recommended because the progress of the symptoms over hours is what should be noted first before making a definitive diagnosis and initiating definitive treatment. In traumatic cases, careful observation is required in open fractures as well. Although early surgery is usually indicated in this group of patients, they can still present with compartment syndrome. One animal study has confirmed the necessity of careful monitoring of both open and closed tibial fractures. A useful tool for such monitoring could be a simple sheet with notes about date, time, location, pain level, and motor and sensory testing. To do this, one must know the anatomic position of various compartments and their vascular and nerve content. A simple screening form of the most commonly observed acute lower extremity compartments is a useful tool for monitoring of the patient ( eFig. 151.2 ). To focus on this relatively rare syndrome and prevent missed diagnoses, more complex systems for monitoring and recognition have been developed in some institutions, with better recognition and outcomes. In literature, special groups of patients with various etiology are described, where this condition occurs more often. Screening in these patients is highly recommended. In a large review study, stepwise logistic regression identified the presence of vascular injury, need for packed red blood cell (PRBC) transfusion, male gender, open fracture, elbow or knee dislocation, Gunshot Wounds (GSW), Injury Severity Score (ISS) greater than or equal to 16, and age less than 55 years as independent predictors for the need for extremity fasciotomy. Several statistically significant predictors of relevant compartment syndromes after surgical reperfusion were found, including lactate, uric acid, transcutaneous oxygen pressure, bilirubin, intrafascial pressure, and serum myoglobin.
To make a diagnosis of compartment syndrome, there must be evidence of increased tissue pressure, inadequate tissue perfusion, and loss of tissue function. When all three factors are present, the diagnosis may be made with assurance; when one or more of these factors are absent, the diagnosis is less accurate. Evidence of increased tissue pressure may include patient complaints of tightness or pressure in the involved area. By palpation, a physician may perceive tenseness of the compartmental envelope.
Evidence of inadequate perfusion of local tissue pressure may include the symptom of pain out of proportion to what would be anticipated from the clinical situation. Increasing analgesia requirements in a properly immobilized leg should raise suspicion. Pain on a passive stretch of the intracompartmental muscles is another useful indication of increased pressure, especially if the muscles have not been injured. Reduced peripheral pulses are very late signs of compartment syndrome; in fact, studies have shown normal pulses with Doppler signals in otherwise severely elevated intracompartmental pressures. Arterial flow is rarely compromised in elevated tissue compartment pressures. On the other hand, diminished pulses could be a result of other causes (e.g., vascular lesions) and, in combination with reperfusion injury, could also lead to the development of compartment syndrome.
Evidence of abnormal tissue function includes weakness of the intracompartmental muscles and nerves, including sensory branches, leading to hypoesthesia. Both nerve and muscle function may be altered by direct injury; therefore evidence of progressive loss of function over time may be a more reliable sign.
To summarize, in awake and cooperative patients who can be reexamined frequently, the diagnosis of compartment syndrome is associated with the following findings:
Pain out of proportion to what is anticipated from the clinical situation
Weakness of the muscles in the compartment
Pain on a passive stretch of the muscles in the compartment
Hypoesthesia in the distribution of the nerves coursing through the compartment
Tenseness of the compartmental envelope
Because some clinical signs progress over time, the clinical decision making can be challenging. , In addition, clinical symptoms and signs have well-documented poor sensitivity. , Especially in critically ill patients who are unable to cooperate because of head trauma, sedation, or even neuromuscular-blocking drugs, the diagnosis cannot be made based on the clinical examination alone. In the pediatric population, compartment syndrome does not always present classically, making clinical diagnosis uniquely challenging.
Although the clinical examination should be a cornerstone of the diagnosis of compartment syndrome, it has the disadvantage of being subjective and requiring patient cooperation. , , Therefore tissue pressure measurement should be performed to assist in establishing a diagnosis so that immediate treatment can be initiated. The normal compartmental interstitial tissue pressure is around 5 mm Hg. Capillary blood flow becomes compromised at 20 mm Hg, and pain develops at pressures between 20 and 30 mm Hg. A tissue pressure of more than 45 mm Hg has been reported to be usually associated with compartment syndrome, and a pressure of more than 60 mm Hg can confirm the diagnosis. However, the tolerance of tissues for increased pressure may be reduced by other factors, such as arterial occlusion, limb elevation, and shock. , In these conditions, compartment syndrome may occur at significantly lower interstitial pressures. According to the arteriovenous gradient theory, the local blood flow (LBF) depends on the pressure gradient between arteries (Pa) and veins (Pv) and local vascular resistance to flow (R). This is described by the following formula :
LBF should be maintained to deliver enough oxygen to the tissues. According to the previous relationship, increased resistance that correlates with interstitial pressure is not the only factor that reduces LBF. The arterial pressure is also important, whereas venous pressure is somehow related to interstitial pressure. Increasing interstitial pressure also increases venous pressure and furthermore decreases blood flow.
Because tolerance of tissues to increased intracompartmental pressure varies among different individuals, and as there are more factors that influence LBF, only one isolated measurement of interstitial pressure may not be enough to diagnose compartment syndrome. For example, higher compartment pressures may be necessary before injury occurs to peripheral nerves in patients with systemic hypertension, whereas compartment syndrome may develop at lower pressures in those with hypotension and/or peripheral vascular disease. , It has been proposed that the difference between diastolic pressure and intracompartmental pressure is a better marker for compartment syndrome. ΔP is calculated as follows: ΔP = DBP (diastolic blood pressure) − IP (interstitial pressure), and values greater than 30–35 mm Hg suggest compartment syndrome, but a specific threshold does not exist.
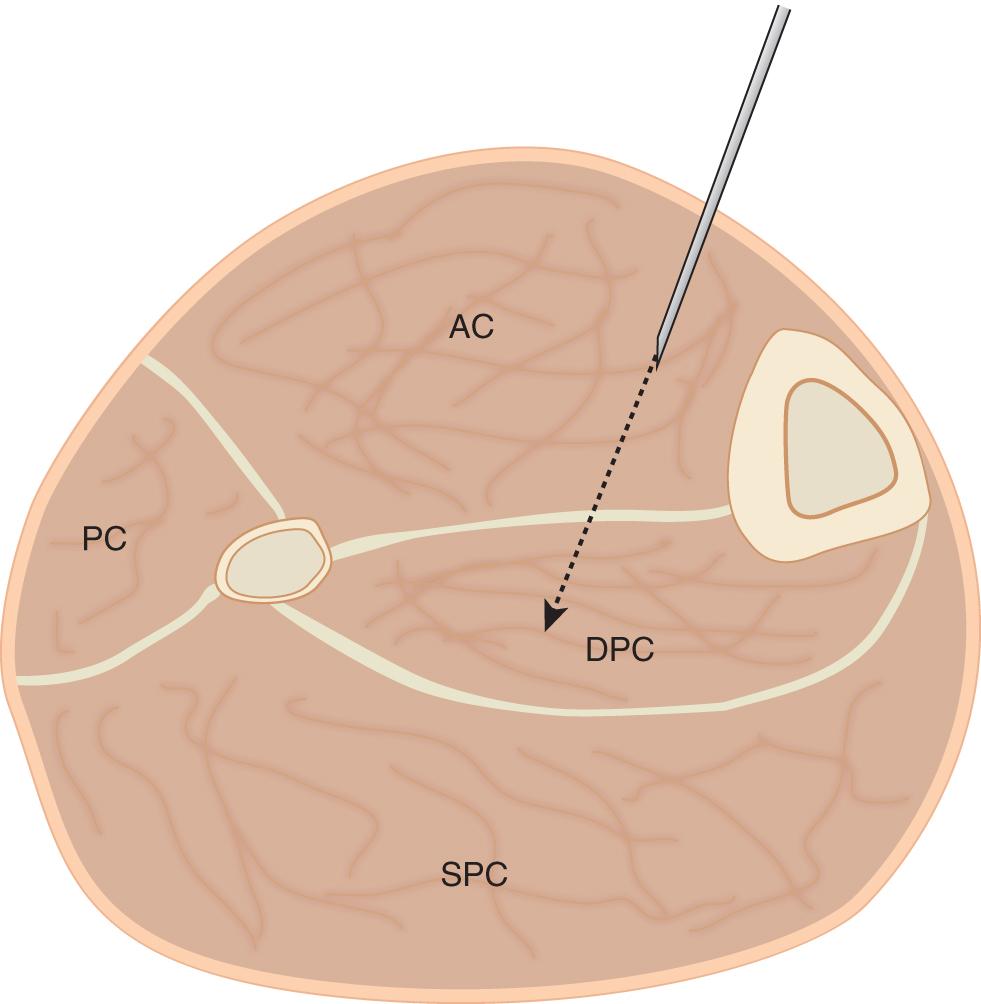
There are numerous methods of tissue pressure measurement. The most commonly used are commercial handheld pressure monitors (e.g., the Stryker device); a simple needle manometer system (Whitesides technique); and the needle, wick, or slit catheter techniques. The question is accuracy because there are reports that an arterial line manometer is the most accurate device. The slit catheter, side-ported bevel-tipped needle, or an 18-gauge needle, when appropriately used with current electronic transducer monitoring, may be used clinically with confidence. The arterial line manometer device has an additional advantage of being able to monitor pressure continuously. This is reported to be useful in tibial diaphyseal fractures, where the estimated sensitivity and specificity of continuous pressure monitoring are high and continuous monitoring should be considered. , Independent of the method used to measure compartment pressures, accuracy depends on proper calibration of the measuring device and placement of the needle or pressure sensor at the level of the injured compartment. The principle of tissue pressure measurement in the case of acute lower extremity compartment syndrome is shown in Fig. 151.2 .
Reported diagnostic performance characteristics of clinical symptoms and signs of acute compartment syndrome (ACS) and intracompartmental pressure monitoring are shown in Table 151.2 .
| Symptom or Sign | Sensitivity (%) | Specificity (%) | Positive Predictive Value (%) | Negative Predictive Value (%) |
|---|---|---|---|---|
| Pain | 19 | 97 | 14 | 98 |
| Pain on passive stretch | 19 | 97 | 14 | 98 |
| Paralysis and motor changes | 13 | 97 | 11 | 98 |
| Paresthesia and sensory changes | 13 | 98 | 15 | 98 |
| Swelling | 54 | 76 | 70 | 63 |
| Intracompartmental pressure monitoring | 94 | 98 | 93 | 99 |
Use of near-infrared spectroscopy (NIRS) for detection of low tissue oxygenation, and therefore the development of compartment syndrome, is controversial. It has been reported as a useful noninvasive tool in diagnosing compartment syndrome after surgical revascularization of lower limb ischemia and in traumatically injured patients. But some subsequent studies did not prove its utility in injuries because of severe edema of the soft tissues, and it cannot measure tissue hemoglobin oxygen saturation (StO 2 ) inside the muscle compartment. , ,
In a large review of studies, several noninvasive diagnostics for extremity compartment syndrome after traumatic injury have been analyzed, including tissue hardness, ultrasound for fascial displacement, magnetic resonance imaging (MRI) and microwave tomography for tissue imaging, B-mode ultrasound for displacement, pulse oximetry for peripheral SpO 2 , infrared imaging for surface temperature, contrast-enhanced ultrasound for perfusion, Doppler ultrasound in brachial arterial flow, and NIRS for StO 2 detection. Although NIRS has shown the most promise, whether such technologies will be of diagnostic benefit await the completion of ongoing clinical trials.
In animal research, the role of intramuscular glucose and partial pressure of oxygen for detecting compartment syndrome has been evaluated. Intramuscular glucose concentration and partial pressure of oxygen measured with commercially available probes rapidly identified muscle ischemia with a high sensitivity and specificity after an experimentally induced compartment syndrome in an animal model.
Ultrasound shear wave elastography for measuring intracompartmental pressure of compartment syndrome has been used in another recent animal study, and this noninvasive technology can potentially help surgeons with early detection, monitoring, and prognosis of intracompartmental pressure.
Another recent study described a noninvasive ultrasound compartment fascia flattening pressure measurement method in a cadaver model of anterior lower leg compartment syndrome. In this study ultrasound is coupled with pressure transducer and fascia flattening is visualized and pressure is recorded. This method is reported to be as an adjunct to clinical examination and includes evaluating injured and noninjured extremities, is noninvasive, is easily reproducible, and can be performed quickly without patient discomfort.
In establishing a diagnosis of compartment syndrome, other causes of pain-producing symptoms have to be ruled out or confirmed or a cause of elevated intracompartmental pressure should be determined. When facing traumatic injuries, a workup for rhabdomyolysis (creatinine phosphokinase [CPK], renal function, urinalysis, and urine myoglobin) should be considered. In a group of patients with tibial fractures with or without compartment syndrome, a model combining maximal CPK level greater than 4000 U/L, maximal chloride level greater than 104 mg/dL, and minimal blood urea nitrogen (BUN) level less than 10 mg/dL had a 100% association with compartment syndrome.
Moderate evidence supports that in patients with acute vascular ischemia, femoral vein lactate concentration samples during surgical embolectomy may assist in the diagnosis of ACS. On the other hand, there is little evidence supporting that serum troponin may assist in diagnosing ACS in patients with traumatic lower extremity injury.
Extremity x-ray or computed tomography (CT) scans can confirm the presence of a fracture. MRI features of ACS include increased fascial and intramuscular T2 signal reflective of edema, and bulging of the fascia may also be present postcontrast, and fat-suppressed T1-weighted images demonstrate diffuse enhancement corresponding to the edema identified on the T2 sequences. Ultrasonography can show muscle or any other tissue tears. Doppler ultrasonography or arteriography can detect vascular abnormalities.
The objective of treatment of compartment syndrome is to minimize deficits in muscular and neurologic function by promptly restoring LBF. Certain nonoperative measures may be effective, such as eliminating external pressure and maintaining local arterial pressure. When there is external pressure that causes compartment syndrome, such as tight casts, it is essential to release the envelope immediately (remove and exchange for a noncircular splint) when only one symptom or sign is present. Usually, this is pain and is most often observed in patients with fracture splints several hours after treatment. Restoration of normal limb perfusion has priority over closed fracture treatment, and this can be postponed until perfusion returns to normal. Before resorting to operative methods for reducing tissue pressure, it is important to consider improvement of LBF if it has been reduced by shock, peripheral vascular disease, or elevation of the limb above the heart. All causes of systemic hypotension should be treated. Limb elevation should be avoided because it lowers local arterial pressure and does not help in reducing swelling. Use of vasodilating drugs or sympathetic blockade appears to be ineffective because, in this condition, local maximal vasodilatation is already present. The use of phosphodiesterase inhibitors in experimental animal models caused modulation of compartmental pressures. In a large study on trauma patients with isolated arterial injury, early anticoagulation with heparin was found to reduce the incidence of compartment syndrome without significant bleeding as a consequence. Carbon monoxide–releasing molecule-3 displays a potent protective/antiinflammatory action in an experimental model of compartment syndrome, suggesting a potential therapeutic application for patients at risk of developing compartment syndrome.
The primary goal of treating compartment syndrome is to decrease intracompartmental pressures. Surgical decompression of all limiting envelopes is the gold standard of treatment, indicated in the presence of a characteristic clinical picture of compartment syndrome in a cooperative patient. When the clinical examination is unreliable or difficult to obtain, pressure measurement should be obtained, where either pressure should not exceed 45 mm Hg or ΔP should not be below 30 mm Hg.
Standard treatment involves a long skin incision and fasciotomy of all involved compartments and debridement of obvious nonviable tissue. Usually, this procedure is performed under general or spinal anesthesia. Bedside fasciotomy under local anesthesia has been described in select cases to be feasible and reliable. Fasciotomy should be performed without a tourniquet to avoid prolonging ischemia and to permit the surgeon to assess the degree of viability and restoration of blood flow. The skin is incised through the entire length of the involved compartment. Obvious muscle bulging is observed in true compartment syndrome ( Fig. 151.3 ). Only obvious necrotic muscle should be removed because the tissue may have the potential for reperfusion and recovery. The sign of contractility with electrostimulation should not be used initially. After fascial release, postischemic swelling should be anticipated; therefore the skin should be left open and the wound temporarily closed with a patch of compliant artificial temporary skin closures. If release of the compartment is not complete, “rebound” compartment syndrome may occur.
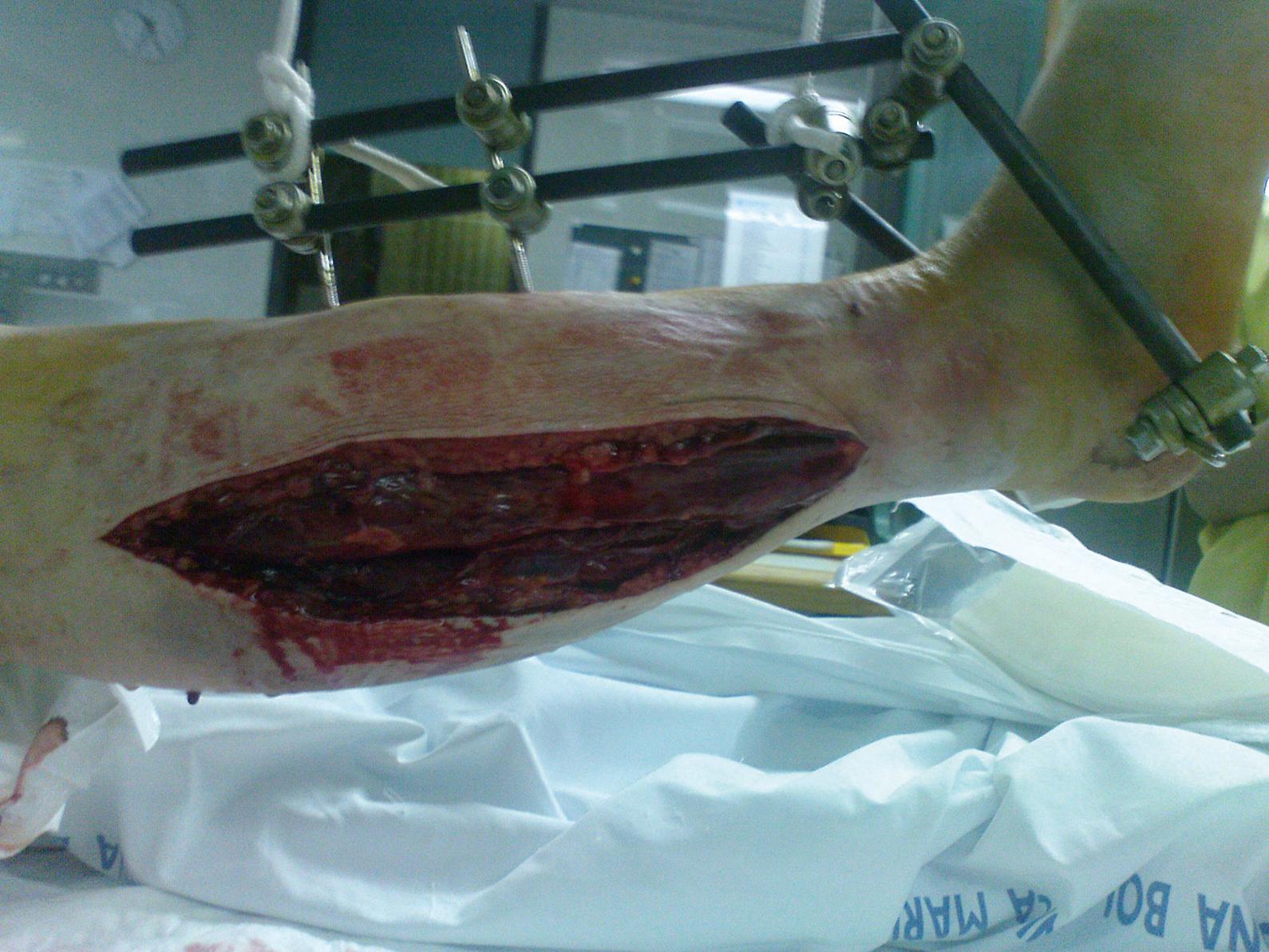
After surgical decompression and temporary skin closure, sterile dressings are applied and the extremity is usually splinted in a functional position. In the presence of fractures, one should consider fixation with external fixators, rarely with plates or intramedullary nails. This stabilization is performed immediately after fascial decompression and greatly facilitates later care of the wound, limb, and fracture. Passive stretching exercises are performed to maintain range of joint motion. Skin closure may usually be performed 3–5 days after surgical decompression, usually by a mesh graft and rarely by direct suturing ( Fig. 151.4 ). At that time, additional debridement of nonviable tissue can be performed. Fascial closure is not recommended because this requires closure under tension and can lead to redevelopment of compartment syndrome. Muscle hernia is left behind and should be large enough to not cause additional late problems. When an optimal cosmetic result is desired, one may progressively approximate the wound edges over 7–14 days with sutures or commercial devices to achieve direct skin closure.
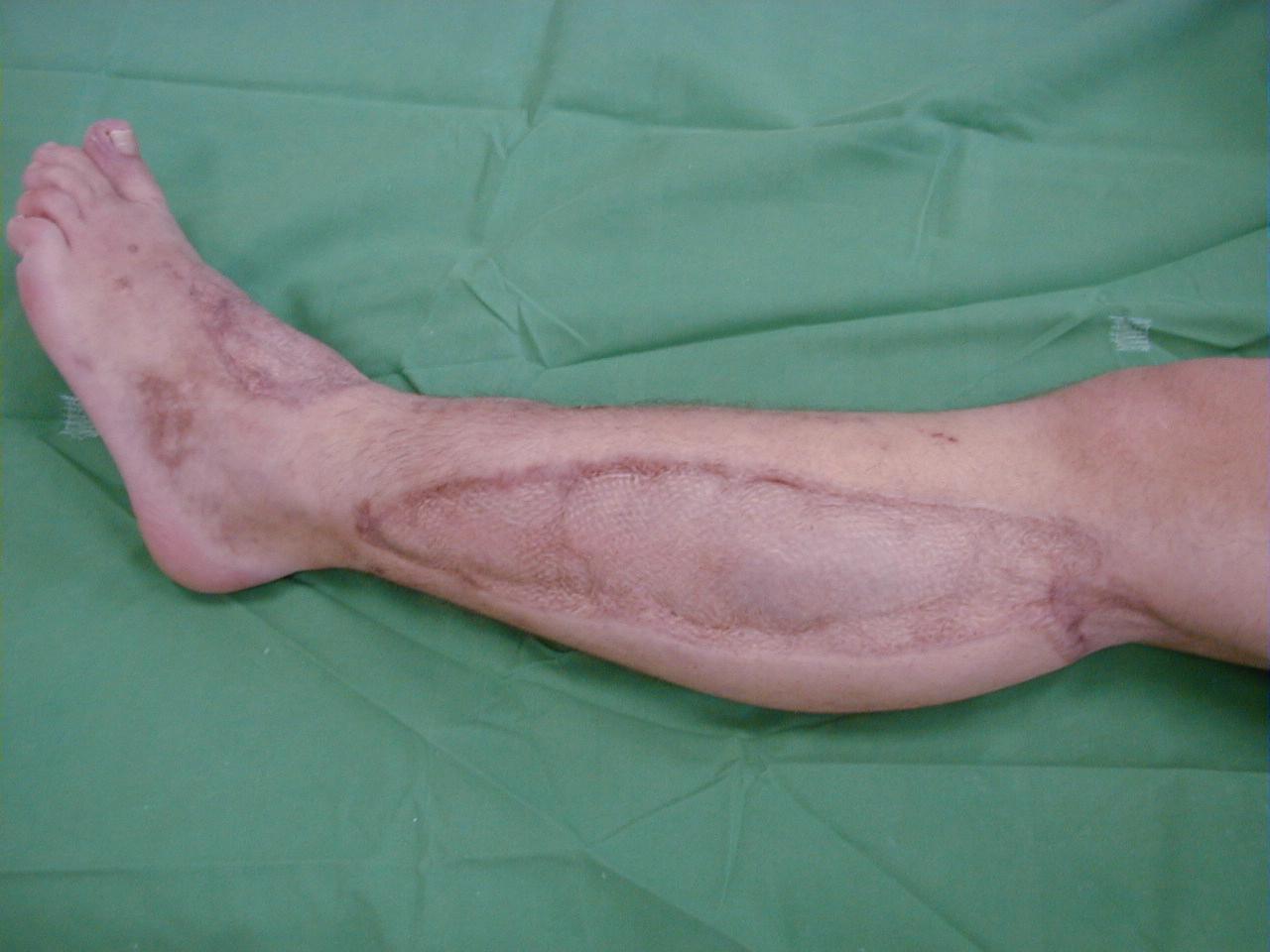
Negative-pressure wound care closure devices can be useful in the management of fasciotomy wounds. Negative pressure decreases wound edema, facilitates approximation of the skin edges, enhances LBF, promotes granulation tissue, and decreases bacterial colonization. Conversely, in one animal study, negative pressure may be harmful to skeletal muscle after compartment syndrome. One randomized study compared vacuum-assisted closure (VAC) with the shoelace technique. Both techniques were safe, reliable, and effective methods for closure of leg fasciotomy wounds. VAC required a longer time for definite wound closure and was reported to be far more expensive than the shoelace technique, especially when additional skin grafting was required. In retrospective analyses, negative pressure led to significantly higher rates of complete skin closure and decreased time to skin closure. , In a recent large meta-analysis the highest success rate for closure of the wounds without skin grafting was observed with dynamic dermatotraction and gradual suture approximation, whereas VAC had the lowest complication rate. Hyperbaric oxygen as an adjunct to management after fasciotomy is reported in some case reports and animal studies, but there is a lack of evidence suggesting that this is advantageous over current practices.
Fasciotomy depends on the underlying condition or mechanism that caused compartment syndrome. The length of the lower extremity skin incisions has been debated for a long time. Minimal skin incisions with more extensive fascial incisions could place the patient at risk of recurrent compartment syndrome. The degree of muscle swelling after reperfusion cannot be predicted, and peak edema occurs several hours after surgery.
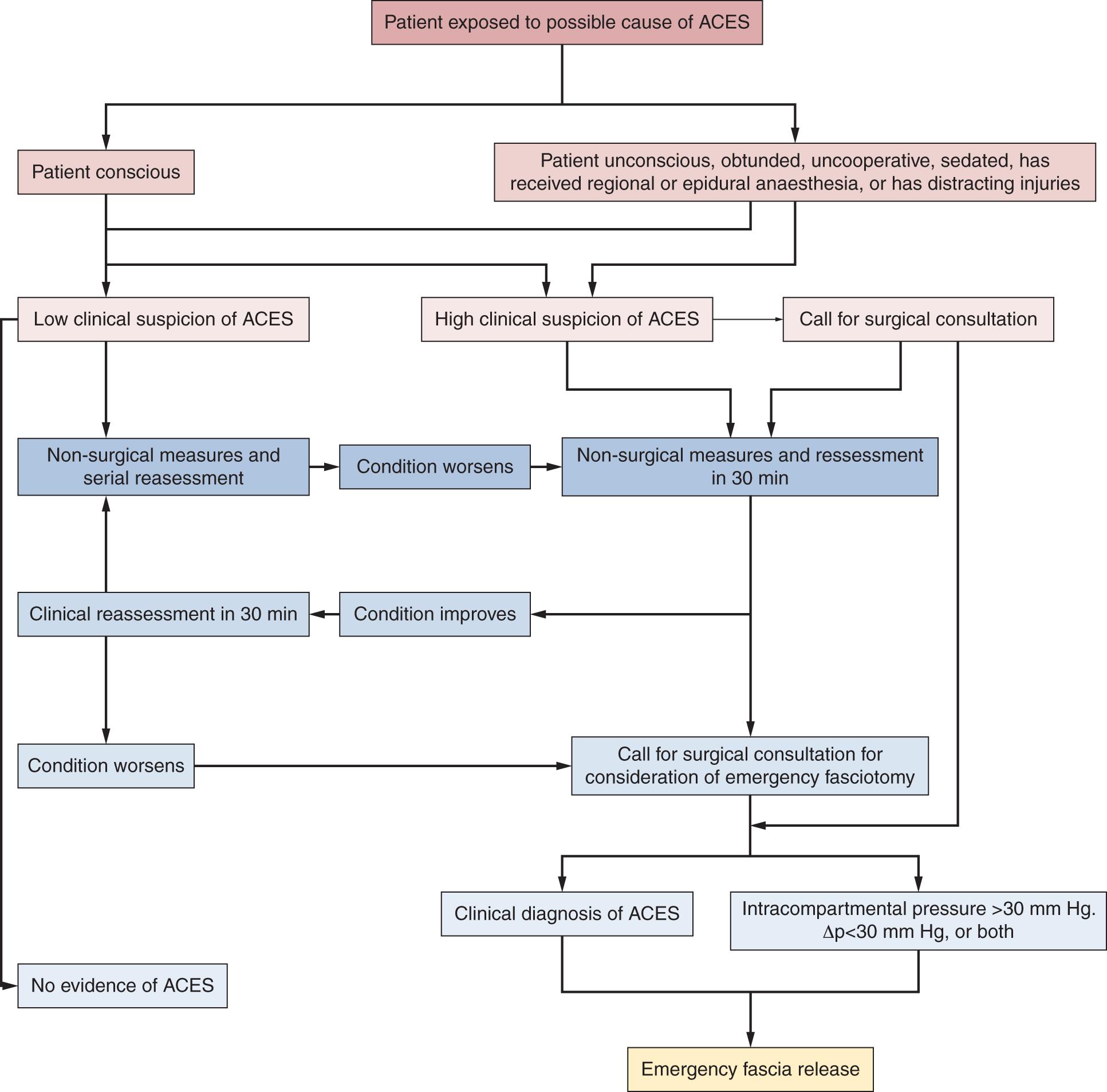
An example of a treatment algorithm that includes both conscious and unconscious groups of patients helping in making a decision when to perform emergency fascia release is shown in Fig. 151.5 .
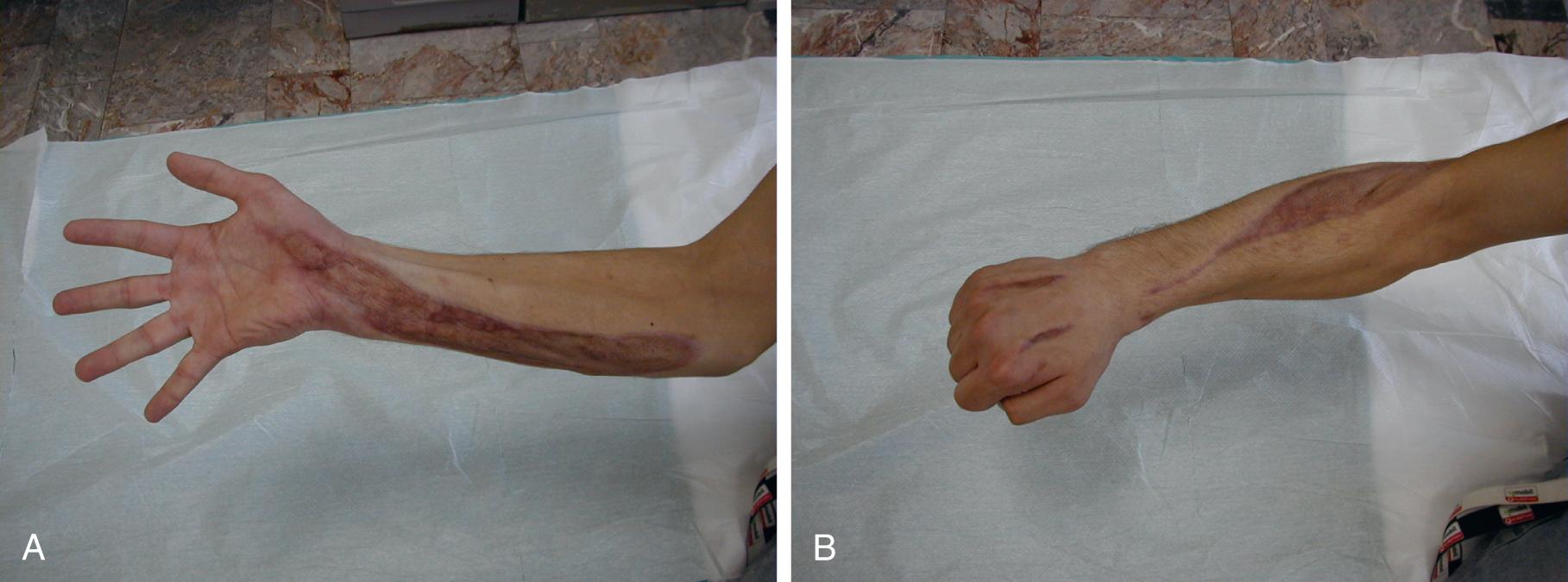
The upper extremity is anatomically divided into the brachium, antebrachium, and hand. Each of the anatomic segments has a different number of compartments with various muscle functions. Techniques for release of these compartments have to be discussed separately, and combined fasciotomy is shown in Fig. 151.6 .
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here