Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
For 2019, the predicted incidence of esophageal and gastric cancers in the United States is projected to be 17,650 and 27,510 cases, respectively. The predicted number of deaths is 16,080 and 11,140, respectively. The site of origin is shifting to tumors localized in the distal esophagus, gastroesophageal junction (GEJ), and proximal stomach.
The main prognostic factors relate to locoregional tumor extent, nodal involvement, and distant metastasis. However, significant advances have been made in understanding the molecular biology of esophagogastric cancers.
Initial evaluation should include history and physical examination, complete blood count, liver chemistries, endoscopy with biopsy and ultrasonography (to determine the degree of direct tumor extension), and computed tomography (CT) of the chest and abdomen (and pelvis for gastric cancers). A positron emission tomography (PET) scan is also a standard staging examination for both esophageal and gastric tumors when obvious metastatic disease is not detected by CT. Additional studies that may help define extent of disease include bronchoscopy for esophageal tumors located at or above the carina and laparoscopy for high-risk gastric cancer to rule out peritoneal seeding or early liver metastasis. Nutritional status should be assessed in all patients.
Surgical resection is the primary therapy for resectable esophagogastric cancers. The earliest tumor stages may be amenable to endoscopic resection. Cure rates of 80% or higher are achieved with early lesions (nodes negative, confined to mucosa or submucosa), which are uncommon in the United States. Minimally invasive surgical techniques are acceptable for esophagectomy. For gastric cancers, D2 node dissection has not improved survival in randomized trials, but extended nodal dissection yielding at least 15 lymph nodes while avoiding elective splenectomy and pancreatectomy has become the accepted standard of care.
Adjuvant therapy is indicated on the basis of patterns of relapse and survival results achieved with surgery alone (high incidence of locoregional relapse and distant metastases). However, treatment sequence has diverged based on tumor location: definitive chemoradiotherapy (CRT) is preferred for proximal esophageal tumors; neoadjuvant CRT is preferred for most cancers of the distal esophagus and GEJ; and perioperative chemotherapy is preferred for true gastric cancers. Definitive CRT leads to cure in approximately 30% of patients with esophageal cancer, but local recurrence rates are high (30%-55%). Preoperative CRT improved survival in the CROSS (ChemoRadiotherapy for Oesophageal cancer followed by Surgery Study) Phase III trial compared with surgery alone for 366 analyzable patients with squamous cell carcinoma (SCC) or adenocarcinoma (ACA) of the esophagus or GEJ. Median overall survival (OS) was 49.4 months versus 24 months, 3-year OS 58% versus 44%, and 5-year OS 47% versus 34% ( p = 0.003). The POET study (PreOperative therapy in Esophagogastric adenocarcinoma Trial) of 120 patients with GEJ lesions tested preoperative chemotherapy versus CRT; outcome trends favored preoperative CRT over chemotherapy alone for both OS ( p = 0.07) and local control ( p = 0.06). A US Intergroup Phase III trial of 556 patients with ACA of stomach or GEJ demonstrated a survival benefit for combined-modality postoperative CRT compared with surgery alone (3-year relapse-free survival [RFS] 48% vs. 31%, p = 0.001; 3-year OS 50% vs. 41%, p = 0.005). The Phase III ARTIST trial (Adjuvant Chemoradiotherapy in Stomach Tumors, 458 patients) compared postoperative chemotherapy with postoperative CRT after D2 resection for gastric ACA. In a subset analysis of patients with positive nodes (396 patients), 3-year disease-free survival (DFS) was improved with CRT (76% vs. 72%, p = 0.04). The British Phase III MAGIC trial of 503 patients with gastroesophageal ACA demonstrated a survival advantage for perioperative epirubicin, cisplatin, 5-fluorouracil ([5-FU]; epirubicin, cisplatin, and 5-FU [ECF]) chemotherapy when compared with surgery alone (5-year OS 36% vs. 23%, p = 0.009). In addition, a French Phase III trial of 224 patients found a survival advantage for perioperative cisplatin and 5-FU (CF) compared with surgery alone (5-year OS 38% vs. 24%, p = 0.02). A recently reported study of 716 patients achieved further improvements in survival (3-year OS 57% vs. 48%, p = 0.012) for perioperative 5-FU, leucovorin, oxaliplatin, and docetaxel (FLOT) compared with ECF/ECX (epirubicin, cisplatin, and capecitabine—substituting capecitabine for 5-FU). Postoperative or preoperative CRT and perioperative CF or FLOT are all appropriate current standards of care.
For patients with locally advanced disease that appears unresectable for cure, several treatment options appear to favorably impact disease control and survival. External beam radiotherapy (EBRT) plus concurrent chemotherapy followed by an attempt at maximal resection plus intraoperative radiotherapy (IORT) result in long-term survival in 10% to 20% of patients in randomized and nonrandomized trials. Neoadjuvant chemotherapy may increase resection rates, but the incidence of locoregional relapse is still high. Pathological complete response (pCR) in surgical specimens is uncommon (0%–9%).
Multidrug chemotherapy regimens have response rates of 30% to 50%, but survival improvements are modest. Phase III trials demonstrate improvement in quality and duration of life with palliative chemotherapy versus supportive care. First-line treatment usually consists of a combination of fluoropyrimidine and platinum agents. In tumors with HER2 overexpression (primarily gastric cancer), the addition of trastuzumab to chemotherapy produces a survival benefit. Inhibition of vascular endothelial growth factor (VEGF) with the monoclonal antibody ramucirumab has shown a survival advantage. More recently, immunotherapy checkpoint inhibitors are being investigated for efficacy in subsets of gastric cancer patients. Radiotherapy is a useful modality for palliation of local symptoms, including obstruction, bleeding, and pain.
Reflecting the ongoing shift in gastroesophageal cancers, these topics will be discussed in a single integrated chapter. Clinicians recognize that in the western world, adenocarcinoma (ACA) of the distal esophagus and gastroesophageal junction (GEJ) represents the most common form of this disease in daily practice; thus, it is a major focus of this chapter. Squamous cell carcinoma (SCC), typically arising in the proximal two-thirds of the esophagus, is also addressed and differences from ACA noted in biology and clinical management. Finally, in each section, distinctions are made for cancers of the true stomach, where biological and anatomic differences have led to a separate treatment algorithm.
The epidemiology of gastric and esophageal cancers has undergone major changes over the past 4 or 5 decades. For many years, esophageal cancers were primarily SCC in the upper portion of the esophagus, and gastric cancers were ACA in the body and distal stomach. Those patterns still exist in some patients in the developed world, but there has been a broad shift in esophageal cancers occurring as ACA in the region of the GEJ and gastric cancers occurring proximally in the cardia extending to the GEJ. For both GEJ and gastric cardia, the rate of increase of incidence during the 1970s and 1980s surpassed that of any other cancer. Similar trends of an increased incidence of gastric cardia cancer have been reported in Europe, where rates were highest among those in higher socioeconomic classes.
In the United States, esophageal and gastric cancer incidence is projected to be 17,650 and 27,510 respectively, with death rates of 16,080 and 11,140 in 2019. This is a major change from the historical incidence. At the turn of the 20th century, gastric cancer was the leading cause of cancer death in the United States and its incidence has been decreasing since that time ( eFig. 54.1 ). In the United States, from 1930 to 1980, the incidence decreased from 38 to 10 per 100,000 for men and from 30 to 5 per 100,000 for women. The disease rarely occurs before the age of 40 years, but its incidence increases steadily thereafter and peaks in the seventh decade. African Americans, Hispanic Americans, and Native Americans are twice as likely to develop gastric cancer as are whites. There has been a substantial improvement in overall relative 5-year survival among US patients over the last 4 decades for both diseases.
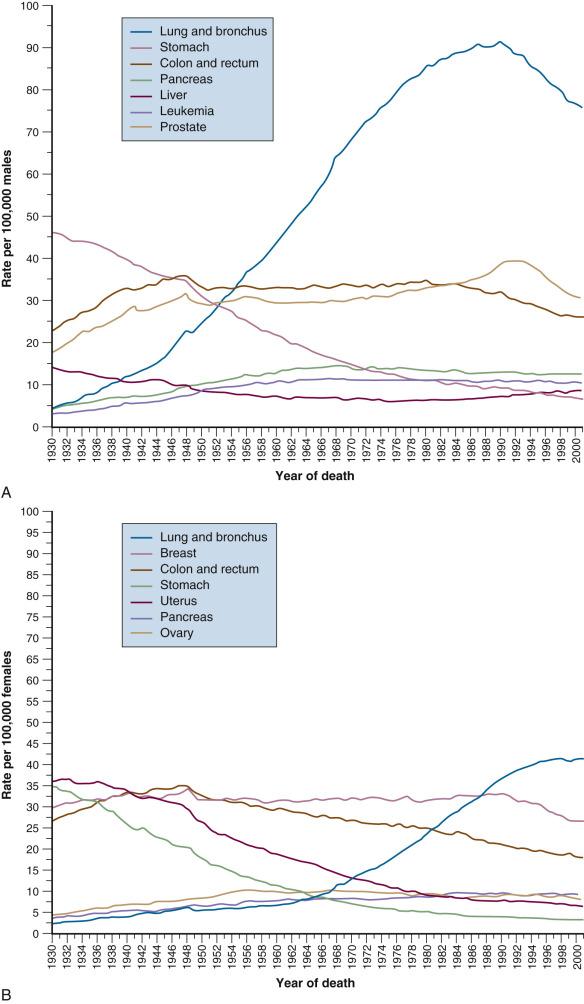
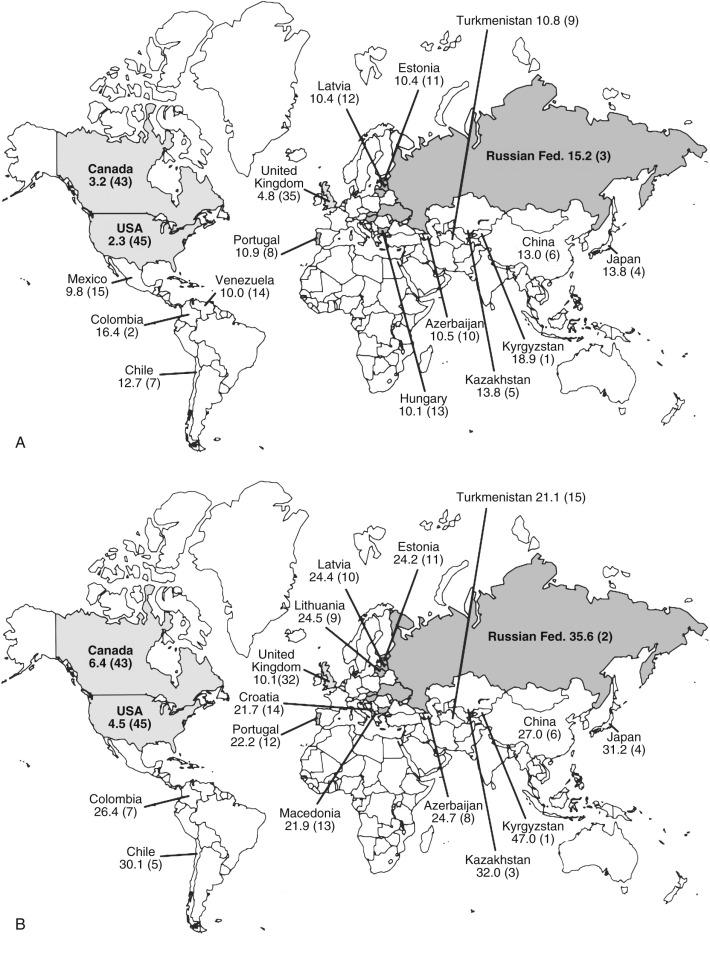
Although gastric cancer incidence has decreased significantly in the United States, on a worldwide scale, gastric cancer has a high incidence and is still a leading cause of cancer death. Of the 45 countries in which age-adjusted death rates for gastric cancer were compared for 2000 ( eFig. 54.2 ), the United States ranked 45th for both males and females. Kyrgyzstan ranked first for both males (47.0 in 100,000) and females (18.9 in 100,000).
As mentioned earlier, the site of origin within the stomach has changed in frequency in the United States over recent decades, with proximal lesions now being diagnosed much more commonly than previously. The largest percentage of gastric cancers still arises within the antrum or distal stomach (≈ 40%); are least common in the body of the stomach (≈ 25%); and are of intermediate frequency in the fundus, cardia, and esophagogastric junction (≈ 35%). In 1930, most cases of gastric carcinoma originated in the distal stomach (body and antrum). The reduction in incidence of gastric cancer from 1930 to 1980 is attributable primarily to a decline in distal lesions.
The incidence of SCC of the esophagus is declining in North America and Western Europe as the incidence of ACA of the distal esophagus and GEJ is increasing, the latter currently representing approximately 70% of all esophageal carcinomas in the United States. SCC remains dominant in endemic regions and underdeveloped parts of the world. Data from the Surveillance Epidemiology and End Results (SEER) program of the National Cancer Institute (NCI) demonstrated that the incidence of esophageal cancer in white males steadily increased by 45% in the United States between 1975 and 2004, with the incidence of ACA increasing by 460% and the incidence of SCC declining by 50% during the same time period.
The incidence of SCC has shown a significant decline in black males beginning in 1992, but it remains the most common cell type among blacks in the United States despite the twofold to threefold increase in incidence of ACA in black males and females. Interestingly, a review of the New England tumor registries demonstrated that the incidence of ACA has increased even when tumors located at the esophagogastric junction have been excluded, and data from the SEER database (1973-2001) found no evidence of histological reclassification of esophageal SCC or anatomic reclassification of ACA of the gastric cardia to explain this trend.
Racial disparities have been observed in the esophageal cancer population. Black males are more likely to present with advanced and/or metastatic disease, resulting in a survival rate that is 60% of that for white males. SCC remains the most common histological type among black males and may be associated with worse outcomes, facts that could explain some of the observed racial disparities in survival rates.
The use of tobacco is an established risk factor for SCC of the esophagus as well as for ACA, but to a lesser extent for the latter. Alcohol consumption is a risk factor for SCC but not ACA. Other environmental factors associated with the development of SCC include thermal injury and exposure to nitrates and potentially carcinogenic nitrosamines, asbestos fibers, or water contaminated with petroleum products. In addition, the presence of human papillomaviruses (HPV-16 and HPV-18) may be risk factors for the development of cancers of the esophagus.
Obesity and high body mass index (BMI) are strong risk factors for esophageal ACA and tumors of the GEJ but not SCC. Individuals who are in the 75% highest BMI range have a 7.6-fold increased risk of developing ACA of the esophagus compared with those with lower BMIs. Obesity-related comorbid illnesses that are associated with the development of esophageal cancer include gastroesophageal reflux disease (GERD) and type 2 diabetes. Diet affects both types of esophageal carcinoma, with increased intake of fruits and vegetables associated with a reduced incidence of cancer.
GERD predisposes to Barrett esophagus. Both are associated with an increased risk of ACA of the esophagus with severe long-standing disease and are thought to be the cause of the major increase in incidence. The incidence of GERD appears to be increasing in the United States. Barrett esophagus is associated with GERD and is a precursor lesion for esophageal ACA, although most Barrett lesions do not proceed to carcinoma. The normal squamous mucosa is damaged by chronic GERD and is replaced by columnar metaplasia in Barrett esophagus. A 30% to 60% increased risk of esophageal ACA has been demonstrated in patients with Barrett esophagus. Long-standing GERD, length of Barrett esophagus, male gender, age, and hiatal hernia size are strongly associated with high-grade dysplasia. The risk of esophageal malignancy increases from a very low level with low-grade dysplasia to much higher levels in patients with high-grade dysplasia and other risk factors. Elective esophagectomy is not usually performed in these patients because of the morbidity of the procedure and the low rate of conversion to malignancy with careful medical and local ablative management.
For gastric cancer, there is a major role for Helicobacter pylori infection in the etiology of the disease. Infection with H. pylori is very common in patients with gastric cancer, although the vast majority of patients who have an infection do not develop a malignancy. The association of H. pylori with gastric cancer is only for tumors of the body and distal stomach and not for tumors of the cardia and GEJ, where there may even be a mild protective effect. There have been many other factors that have been speculated to be associated with gastric cancer and its changing etiology—including smoked and salted foods, change in refrigeration, and low intake of fruits and vegetables—but none of these have been established as being causative. Chronic H. pylori infection results in an atrophic mucosa and decreased acid production that is opposite to the high acid production usually seen in esophageal ACA.
For esophageal cancer, decreasing the exposure to the etiological factors mentioned earlier, including tobacco and alcohol, could help decrease the incidence of the disease. Because of the strong association of obesity and high BMI with esophageal ACA, likely related to chronic GERD, addressing those underlying problems could be of benefit. Patients with Barrett esophagus with high-grade dysplasia should undergo surgical resection or other local strategies, such as endoscopic mucosal resection (EMR), cryoablation, or radiofrequency ablation (RFA), although is not clear the extent to which these strategies will decrease the incidence of esophageal and GEJ cancer formation. The American College of Gastroenterology recommends screening of high-risk patients for Barrett esophagus. Barrett esophagus patients without dysplasia should have routine endoscopic surveillance every 3 to 5 years. Endoscopic ablative therapy is recommended for patients with Barrett esophagus and high-grade dysplasia or T1a ACA. Patients who have ablation still need routine follow-up, because the risk of recurrence is high.
The use of chemopreventive agents is under investigation. A meta-analysis demonstrated that the use of nonsteroidal antiinflammatory drugs (NSAIDs) decreases the risk of both SCC and ACA, with odds ratios of 0.58 (95% confidence interval [CI], 0.43–0.78) and 0.67 (95% CI, 0.51–0.87), respectively. There is a large multicenter, randomized controlled clinical trial (AspECT) evaluating the long-term chemopreventive effect of esomeprazole with or without aspirin, the results of which should be reported shortly.
Early detection would markedly improve the prognosis of gastric cancer because surgical resection has a high cure rate for lesions limited to the mucosa or submucosa. In the United States, however, the incidence of early gastric cancer is generally less than 5%. In Japan, where the incidence of carcinoma confined to the mucosa or submucosa was only 3.8% from 1955 to 1956, by 1966 the incidence of early lesions had increased to 34.5% because of vigorous screening (5-year survival rates of 90.9% in this cohort of patients). The value of screening is evidenced by the 5-year survival rate of 53% for all gastric cancers in Japan versus 21% for the rest of the world.
Although mass screening has been useful in Japan to detect early cancers, defined high-risk populations do not exist in the United States to justify the expense of widespread screening. Individual practitioners could use endoscopy to screen patients who have occupational or precursor risk factors or patients with persistent dyspepsia or GERD symptoms, but this strategy is not appropriate for populations as the conversion rate to cancer per year is low. It is not yet known whether screening of patients with H. pylori infection will be of value in achieving diagnosis of early gastric cancer, although treating H. pylori infection could reduce incidence.
Germline mutations in the CDH-1 gene, which encodes the E-cadherin protein, have been recognized in families with hereditary diffuse gastric ACA. Carriers of these mutations have a 70% lifetime risk of developing gastric cancer. Early total gastrectomy has been recommended for this small patient population because of the lack of effective early tumor detection. Microscopic evaluation of the proximal and distal resection margins for complete removal of the gastric mucosa is necessary because residual gastric mucosa can degenerate and result in a gastric cancer.
Cancers of the stomach and esophagus are recognized as a highly heterogeneous disease among individuals; strategies for subclassification into more biologically rational homogenous subtypes may improve patient outcomes by allowing subtype-specific treatment regimens. An overview is provided here of the key biological characteristics and molecular biology of these cancers, along with current potential molecular alterations relevant to clinical therapy.
Advances in next-generation sequencing (NGS) technologies and improved understanding of cancer biology have unlocked opportunities to characterize the genomic landscape of cancer. Using multiplatform analyses, The Cancer Genome Atlas (TCGA) Research Network developed a robust molecular classification of gastroesophageal cancers, which may serve as a valuable diagnostic companion to the conventional system of subtyping based on histopathology. Dysregulated pathways and candidate drivers may be identified and targeted for therapy and offer new opportunities to identify biomarkers that may be predictive of tumor response to treatment.
Several attempts to classify gastric cancer have been made over the past decades. The classification by Lauren using microscopic morphology alone is most widely used. The key subtypes include diffuse types, which encompass the signet ring–type gastric cancer, with a higher propensity for intraperitoneal metastasis and silencing of CDH1 ; intestinal-type distal gastric cancer, arising from precursor lesions in the setting of atrophic gastritis and chronic inflammation due to H. pylori ; and gastric cardia and GEJ cancer associated with inflammation resulting from gastric acid reflux and lifestyle factors, such as obesity and smoking.
A comprehensive molecular characterization of gastric cancer was published by TCGA. Historically, this disease has been viewed as a single entity, but data from TCGA redefined the disease into four major genomic subtypes that could more readily be applied in clinical care: Epstein-Barr virus (EBV)-infected tumors (9%), microsatellite instability (MSI) tumors (22%), genomically stable (GS) tumors (20%), and tumors with chromosomal instability (CIN, 50%). Each subtype was found throughout the stomach, but CIN tumors showed elevated frequency in the GEJ/cardia (65%, p = 0.012), whereas most EBV-positive tumors were present in the gastric fundus or body (62%). GS tumors were diagnosed at an earlier age (median age, 59 years), whereas MSI tumors were diagnosed at relatively older ages (median age, 72 years). MSI patients tended to be female (56%), but most EBV-positive cases were male (81%). No systematic differences in distribution of subtypes between patients of East Asian and Western origin were observed.
The EBV-positive tumors exhibited high prevalence of DNA promoter hypermethylation. All EBV-positive tumors displayed CDKN2A ( p16 INK4A ) promoter hypermethylation. In addition, a strong predilection for nonsilence PIK3CA mutations was observed in 80% of the subgroup. Notably, a novel recurrent amplification at 9p24.1 at the locus containing JAK2 , CD274 , and PDCD1LG2 was enriched in the EBV subgroup (15% of tumors). CD274 and PDCD1LG2 encode PD-L1 and PD-L2, immunosuppressant proteins currently being evaluated as targets to augment the endogenous antitumor immune response. This gene amplification is consistent with studies showing elevated PD-L1 expression in EBV-positive lymphoid cancers.
The MSI tumors, characterized by genomic instability due to a deficient DNA mismatch repair system, were enriched for MSI and hypermethylation, with patterns distinct from that observed of EBV-positive tumors, including hypermethylation at the MLH1 promoter region, leading to MLH1 silencing. Elevated somatic mutation rates were identified, including highly recurrent mutations of PIK3CA (42%) and ERBB3 (26%), and 12% of tumors having alterations of both genes. Of note, the BRAF V600E mutation commonly seen in MSI colorectal cancer was absent. However, gastric MSI tumors had a high rate of PD-L1 expression.
The CIN tumors were characterized by the presence of extensive somatic copy-number aberrations. They were noted to have marked aneuploidy and, although they lacked common mutations in PIK3CA and/or ERBB3 , the frequency of TP53 mutations (71%) and recurrent amplifications of receptor tyrosine kinases (RTKs) and cell cycle mediators were high, most notably HER2 (24%). Recurrent amplification of the gene-encoding ligand VEGFA was notable, as was the presence of frequent amplifications of cell cycle mediators ( CCNE , CCND , and CDK6 ).
The GS tumors were characterized by the elevated expression of molecules in the cell adhesion and angiogenesis-related pathways, mainly represented in the diffuse histological subtype (73%), with an absence of extensive somatic copy-number aberrations or elevated rates of mutation or hypermethylation. Loss of the tumor-suppressor gene CDH1 , encoding the cell adhesion molecule E-cadherin, was observed in the GS subtype (37%). The analysis by TCGA identified that 30% of GS tumors harbored novel alterations in components of the Rho signaling pathway, particularly somatic mutations of RHOA or fusion genes involving Rho-GTPase activating proteins CLDN18-ARHGAP6 or 26 . The CLDN18–ARHGAP fusions were mutually exclusive with RHOA mutations and were enriched in GS tumors. Given the role of RHOA in cell motility, alterations in either RHOA or CLDN18-ARHGAP6 might contribute to the disparate growth patterns and lack of cellular cohesion that are hallmarks of diffuse tumors.
Although esophageal ACA and SCC share some recurring genomic alterations, substantial differences have been identified in the patterns of alterations between these two diseases. TCGA has confirmed the molecular distinctiveness of esophageal SCC and ACA. Strikingly, this study found that SCC arising in the esophagus were more similar to SCC of other organ types than to esophageal ACA, which more strongly resembled the CIN subtype of gastric cancer.
Genomic alterations in cell cycle mediators are frequent in both esophageal SCC and ACA. Inactivation of CDKN2A and amplification of CCND1 or CDK6 , or loss of RB1 were present in a majority of squamous tumors. Patterns of cell-cycle dysregulation differed in ACA, where CCND1 was amplified in only 15% of tumors and amplification of CCNE1 was more common. CDKN2A was inactivated in 76% of ACA by mutation, deletion, or epigenetic silencing. Esophageal ACA had a wider range of potentially oncogenic amplifications than SCC, most commonly ERBB2 , which was altered in 32% of ACA. Additional analysis identified dysregulation of the transforming growth factor-β (TGF-β) pathway and less frequent CTNNB1 (β-catenin) activation both to be more common in esophageal ACA than SCC. Therefore, although many of the same pathways were somatically altered in ACA and SCC, the specific genes affected were dissimilar, probably reflecting distinct pathophysiology and suggesting different therapeutic approaches. These data caution against performing clinical trials in mixed populations of esophageal ACA and SCC.
In esophageal ACA, TCGA analysis identified significant mutations in TP53 , CDKN2A , ARID1A , SMAD4 , and ERBB2 . These findings are consistent with the prominence of CDKN2A and TP53 mutations in dysplastic Barrett esophagus, a precursor to esophageal ACA. Of these mutations, amplifications containing VEGFA , ERBB2 , KRAS , EGFR , IGF1R , VEGFA , or GATA6 and deletion of SMAD4 were commonly detected only in ACA but absent in SCC.
TCGA network demonstrated that esophageal ACAs more closely resemble gastric cancer than other esophageal SCCs. When evaluated jointly with gastric cancers, esophageal ACA and CIN gastric cancer formed a group distinct from EBV, MSI, or GS tumors. Evaluating all gastroesophageal ACA, there was increasing prevalence of CIN moving proximally, with 71 of 72 esophageal ACAs classified as CIN. When further compared, TCGA found clear similarity between chromosomal aberrations in gastric CIN tumors and esophageal ACA. However, differences between esophageal ACA and gastric CIN tumors were also present. Esophageal ACA tumors harbor significantly higher DNA hypermethylation than among gastric CIN cancers. Moreover, esophageal ACA had higher rates of mutations of SMARCA4 and deletion of tumor suppressor RUNX1 but lower APC mutation rates relative to gastric tumors, suggesting a less prominent role for Wnt/beta-catenin. Overall, while some distinctions are apparent, the data suggests that gastric and esophageal CIN tumors lack absolute dichotomizing features and do not appear to be distinct tumor types.
In esophageal SCC, the TCGA study identified significant mutation rates in genes TP53 , NFE2L2 , MLL2 , ZNF750 , NOTCH1 , and TGFBR2 . Amplifications of TERT , FGFR1 , MDM2 , NKX2-1 , and deletion of RB1 are specifically found in esophageal SCC. These tumors also had amplifications of chromosome 3q, focused on the SOX2 locus. Genes that encode SOX2 or squamous transcription factor p63, also on chromosome 3p and essential for squamous epithelial cell differentiation, were amplified in 48% of esophageal SCC. These data suggest the presence of lineage-specific alterations that drive progression in esophageal SCC. Moreover, mutations in ZNF750 and NOTCH1 in esophageal SCCs may similarly modulate squamous cell maturation.
Despite the clinical heterogeneity and molecular complexity of gastric and esophageal cancers, targeted therapeutics directed against the genetic mutations and signaling pathways that drive tumor growth and invasion have been developed and clinically investigated. Biological or targeted agents are not typically beneficial for esophageal SCC. Targeted therapies currently in clinical use for gastroesophageal ACA include trastuzumab, ramucirumab, and pembrolizumab. For advanced or metastatic esophagogastric adenocarcinoma overexpressing HER2, trastuzumab should be added to first-line chemotherapy. Ramucirumab (an anti-VEGFR2 antibody) is considered the standard-of-care second-line treatment either as monotherapy or in combination with paclitaxel for advanced gastric or gastroesophageal junction ACA. Pembrolizumab has recently been approved as third-line treatment for gastric cancer expressing PD-L1. Other targeted therapeutics directed against the signaling pathways of mitogenesis, angiogenesis, and immune checkpoints are under clinical investigation for treatment of gastric cancer.
Human epidermal growth factor receptor (HER2, also known as ErbB2) is a transmembrane tyrosine kinase receptor encoded by proto-oncogene HER2 ( ERBB2 ), located on chromosome 17. Current clinical guidelines define tumor HER2 overexpression as 3+ immunohistochemical staining or fluorescence in situ hybridization (FISH) positivity. Cancers located at the GEJ have a higher rate of HER2 positivity than distal gastric cancers. Well and moderately differentiated tumors have a higher rate of HER2 positivity than poorly differentiated tumors. Although HER2 amplification seemed to be associated with a worse prognosis in one study, large investigations could not confirm HER2 protein expression or gene amplification as an independent prognostic factor. Trastuzumab is a humanized immunoglobulin G1 (IgG1) monoclonal antibody (mAb) directed against the extracellular domain of HER2, which prevents dimerization of the HER2 receptors. In a cohort of 3665 patient samples, the Trastuzumab for Gastric Cancer (ToGA) study revealed that 810 (22%) were HER2 positive. The ToGA trial showed a significant overall survival (OS) benefit for patients with HER2-positive advanced-stage gastric cancer treated with trastuzumab and cisplatin–fluoropyrimidine (fluorouracil or capecitabine) chemotherapy versus cisplatin-fluoropyrimidine alone. The development of tumor resistance against HER2-directed treatment is also an important question. Loss of HER2 expression occurs in approximately one-third of patients with HER2-positive gastric cancer treated with trastuzumab and presents a possible mechanism for trastuzumab resistance. Upon tumor progression, molecular alterations have been observed in EGFR (13%), TP53 (92%), cell cycle mediators, such as cyclin-dependent kinases (42%), and in the PI3K/AKT/mTOR axis (21%). These data suggest the need for repeat biopsies to accurately determine the appropriate use of HER2-directed therapy upon tumor progression.
Targeting other RTKs with monoclonal antibodies or small molecule inhibitors has not led to compelling efficacy thus far in patients with gastroesophageal cancers. Anti-EGFR directed treatment with the mAbs cetuximab and panitumumab did not provide survival benefit in two large randomized Phase III studies, despite promising data from Phase II trials. However, retrospective biomarker analysis suggests that a subpopulation of patients with tumors harboring EGFR amplifications or copy-number gains might benefit from this therapy.
Negative results have also been reported regarding targeting the HGF/MET axis in two Phase III studies. Alternative biomarkers to guide patient selection other than immunohistochemistry (IHC) to determine MET status might be required for the promise of anti-HGF/MET treatments in gastric cancer to be realized. One such option is MET amplification, which might be a more appropriate biomarker to select for tumors that will respond to MET inhibitors. Preliminary data showed that some patients with MET amplification (which occurs in < 5% of gastric cancers) can achieve complete responses with MET inhibitors, such as crizotinib or AMG337.
In the TCGA gastric-cancer dataset, 9% of CIN tumors and 8% of GS tumors harbored FGFR amplification. In addition, activation of the FGFR2 pathway was found to be required to drive growth and survival of gastric cancers carrying FGFR2 amplifications, both in vitro and in vivo. Targeting FGFR in a Phase II trial showed no statistically significant difference in progression-free survival (PFS) in patients with FGFR2 -amplified or FGFR2-polysomy tumors selected by FISH. Reasons for the lack of efficacy could be the marked intratumor heterogeneity of FGFR2 amplification and the low concordance with elevated FGFR2 protein expression, as exploratory biomarker analysis revealed. Different biomarker-based patient selection strategies, such as FGFR2 copy-number in cell-free plasma DNA, should be tested in clinical practice.
Vascular endothelial growth factor (VEGF) and its receptors are amplified and overexpressed in approximately 7% of all GEJ and gastric cancers. Ramucirumab is a recombinant humanized IgG1 mAb targeting VEGFR2. It was investigated in patients with advanced-stage gastric cancer following disease progression during or after first-9line chemotherapy. The combination of ramucirumab and paclitaxel seems to be the most effective current treatment regimen for patients with advanced-stage gastric cancer in whom disease progresses following first-line therapy.
The presence of viral antigens such as EBV (a hallmark of 9% of gastric cancers) has been shown to result in increased neoepitope presentation, which might contribute to an antitumor immune response. Moreover, the strength of the interleukin-12 (IL-12)–mediated signaling signature suggests a robust immune cell presence and provides support for targeted immunotherapy when coupled with evidence of PD-L1/2 overexpression. PD-L1 may therefore represent promising targets in these tumors. Pembrolizumab and nivolumab are humanized mAbs directed against PD-1. They enhance the ability of the immune system to detect and destroy cancer cells by blocking the interaction between PD-1 and PD-L1/L2 and countering the tumor's immune-escaping tactic.
Frequent amplifications of cell cycle mediators ( CCNE1 , CCND1 , and CDK6 ) suggest the potential for therapeutic inhibition of cyclin-dependent kinases. These data reveal a potential role for inhibitors of cell cycle kinases for treatment, especially in esophageal SCC. The discovery of mutations in the RHOA and CLDN18 gene of the GS gastric ACA subtype also could be exploited to develop new therapeutic strategies.
In recent years, the characterization of esophageal and gastric cancers into molecular subtypes has evolved and provides a roadmap for the next generation of potential targets. Through the development of new drugs and combinations, this emerging molecular understanding can be translated into therapeutic biomarkers and targets for clinical benefit. The TCGA classification will help identify the opportunities and challenges of developing strategies toward the goal of precision medicine in gastric and esophageal ACA. Large-scale international studies are ongoing, and more results will be reported soon. Hopefully, further insights into the molecular characteristics will stimulate the role of biomarker-driven targeted therapy in gastroesophageal cancer.
As mentioned earlier, there has been a shift in location of esophageal cancers from a mid- or high esophageal location to tumors occurring in the lower third of the esophagus, with the higher tumors being more commonly SCC and the distal tumors being ACA. There are sporadic reports of other histologies in the esophagus, but none is common enough to describe in detail. Adenocarcinomas accounts for 90% to 95% of all gastric malignancies but lymphoma, leiomyosarcoma, carcinoid, adenoacanthoma, and SCC occur sporadically. Gastrointestinal stromal tumors (GISTs) are a distinct entity; their management (surgery alone or plus imatinib) is distinctly different from ACA and they are rarely treated with radiotherapy.
Gastric carcinomas have been pathologically categorized with regard to both microscopic and gross pathological features. The Lauren classification system includes an intestinal type with improved prognosis (predominates in regions with high prevalence of gastric cancer) and a diffuse histological type with poor prognosis (occurs more commonly in countries with low prevalence). Diffuse carcinomas occur more often in young patients and develop throughout the stomach but especially in the cardia. Intestinal-type lesions are frequently ulcerative and occur in the distal stomach more commonly than the diffuse type. Grossly, gastric cancers can be categorized according to Bormann's five types: 1, polypoid or fungating; 2, ulcerating lesions surrounded by elevated borders; 3, ulceration with invasion of the gastric wall; 4, diffusely infiltrating (linitis plastica); and 5, not classifiable. The Japanese Research Society for Gastric Cancer has a classification system that divides lesions into protruded (1), superficial (2, with elevated [2a], flat [2b], and depressed [2c] subtypes), and excavated (3) types.
The esophagus is a hollow tube approximately 25 cm in length that extends from the pharynx to the stomach and consists of three parts: the cervical, thoracic, and distal esophagus. The cervical esophagus lies just left of the midline behind the larynx and trachea. The upper portion of the thoracic esophagus passes behind the tracheal bifurcation and left main stem bronchus. The lower thoracic esophagus runs behind the left atrium. The distal esophagus is 6 to 8 cm in length and merges into the gastroesophageal junction. Adenocarcinomas of the GEJ have been classified into three types by Siewert based on anatomic location and the epicenter of the tumor. The current classification system identifies tumors 1 to 5 cm above the GEJ as distal esophageal tumors (Siewert type 1); those tumors with the epicenter of mass located within 1 cm proximal and 2 cm distal to the GEJ are classified as Siewert type 2; and subcardial tumors with the epicenter located 2 to 5 cm distal to the GEJ are classified as Siewert type 3. The American Joint Committee on Cancer (AJCC) staging system specifies that Siewert types 1 and 2 tumors are classified as ACA of the esophagus for the purposes of staging, and those meeting criteria for Siewert type 3 are classified as gastric cancers.
In the esophagus, mucosal lymphatics merge with a submucosal plexus, which then merges with lymphatic channels in the muscularis. These channels communicate with an extensive network of lymphatics that extend longitudinally along the esophagus and eventually drain into nodal groups as cephalad as the internal jugular lymph nodes and as caudal as the celiac lymph nodes. The nodal groups at risk for involvement in cancers of the cervical esophagus include the low cervical, supraclavicular, and upper paratracheal nodes. The nodes at risk for gastroesophageal tumors include the pulmonary ligament, paracardial, left gastric, common hepatic, splenic, and celiac nodes. The esophagus lacks a serosal surface and is separated from adjacent structures only by a loose connective tissue, the adventitia. The adventitia provides little barrier to local spread; as a consequence, a tracheoesophageal or esophagobronchial fistula can develop in patients with cancers of the mid- and high esophagus.
The stomach is surrounded by organs and structures that can be involved by direct tumor extension when a lesion has spread beyond the gastric wall. These include the omenta, pancreas, diaphragm, transverse colon or mesocolon, duodenum, jejunum, spleen, liver, superior mesenteric and celiac vessels, abdominal wall, left adrenal gland, and kidney. Although adherence from inflammatory conditions can mimic direct extension of tumor, all adhesions between a gastric carcinoma and adjacent structures should be regarded as malignant, and en bloc resection of involved structures is preferred, if feasible. Abundant lymphatic channels are present within the submucosal and subserosal layers of the gastric wall. Microscopic or subclinical spread beyond the visible gross lesions (intramural spread) occurs commonly via the lymphatic channels. Frozen sections of the gastric resection margins should therefore be obtained intraoperatively to ensure that margins of resection are uninvolved microscopically. The submucosal lymphatic plexus is prominent in the esophagus and the subserosal plexus in the duodenum, allowing both proximal and distal intramural tumor spread.
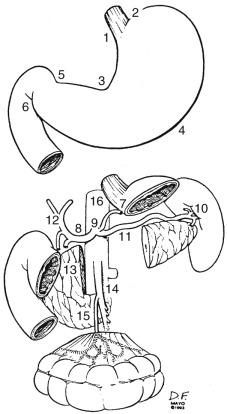
Because of the numerous pathways of lymphatic drainage in the stomach, it is difficult to perform a complete nodal dissection ( eFig. 54.3 ). Although initial drainage is usually to lymph nodes along the lesser and greater curvatures (perigastric or N1 nodes using the Japanese Research Society for Gastric Cancer designation), primary nodal drainage includes nodes along all three branches of the celiac (left gastric, common hepatic, splenic) and the celiac itself (Japanese N2 nodes). Node groups that are more distal include hepatoduodenal, peripancreatic, root of mesentery (Japan N3), periaortic, and middle colic (N4). When proximal gastric lesions extend into the distal esophagus, that nodal system is at risk.
For cancers that are limited to the gastric wall, venous drainage is primarily to the liver via the portal system, although spread can occur to virtually any organ. At initial evaluation, liver involvement is found in up to 30% of patients, predominantly resulting from hematogenous metastases but sometimes because of direct tumor extension. When cancers extend proximally to involve the esophagus or extend posteriorly, the risk of lung metastases increases.
Because the stomach is an intraperitoneal organ, peritoneal spread is possible when a lesion extends beyond the gastric wall to a free peritoneal (serosal) surface. Peritoneal involvement may initially be a localized process limited by surrounding organs and ligaments (gastrohepatic, gastrosplenic, and gastrocolic) but is most often a diffuse process.
More than 90% of patients with esophageal cancer present with progressive dysphagia and, often, significant weight loss. Other findings include odynophagia, chest pain, cough, and fever associated with possible respiratory fistulas, hoarseness associated with tumor involvement of the recurrent laryngeal nerve, and melena resulting from intraluminal bleeding. In contrast, for gastric cancer, the symptoms are nonspecific, including loss of appetite, abdominal discomfort, weight loss, weakness (as a result of anemia), nausea and vomiting, and melena. The duration of major symptoms is often fairly short before diagnosis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here