Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Under the International League of Associations for Rheumatology (ILAR) classification of juvenile idiopathic arthritis (JIA), , enthesitis-related arthritis (ERA) is defined according to the inclusion and exclusion criteria shown in Table 20.1 . Children with ERA have inflammation predominantly affecting joints and entheses of the lower extremities, which may also eventually affect the sacroiliac (SI) joints and spine. It is characterized by a strong association with human leukocyte antigen (HLA)-B27 and absence of rheumatoid factor (RF). In many instances, the disease evolves to closely resemble ankylosing spondylitis (AS), although SI joint and spine involvement at disease onset is uncommon in childhood or adolescence.
| Inclusion Criteria | Exclusion Criteria |
|---|---|
|
|
Patients with this condition have been described using several different terms, many of which are included in the ERA category of JIA: juvenile spondyloarthritis (JSpA), seronegative enthesitis and arthritis (SEA) syndrome, and/or juvenile ankylosing spondylitis (JAS). The term JSpA refers to conditions of children who have arthritis presenting in late childhood and adolescence that have a strong association with HLA-B27 and potential axial involvement. SEA syndrome is a condition of children with arthritis and enthesitis and who are RF- and antinuclear antibody (ANA)-negative. Many have involvement of the axial spine and are HLA-B27 positive. The term JAS applies to a condition of children and adolescents who have peripheral and axial arthritis that conforms to the criteria for classification of adult AS. The ILAR criteria define ERA as a distinct population separate from the other JIA subtypes, especially psoriatic arthritis, which would be included under the JSpA definition. The majority of children and adolescents with ERA have peripheral disease at presentation; axial disease is uncommon at disease onset, and when it occurs, it is often preceded by peripheral disease. Consequently, the ERA criteria capture the most common presentation of pediatric patients. Progression to JAS is unpredictable, but it does occur, and early identification of patients with axial disease is important so that appropriate therapeutic interventions can be initiated. Note that most children and adolescents do not meet the modified New York criteria for AS ( Table 20.2 ).
| Clinical Criteria |
|
| Definite AS |
|
| OR |
|
| Probable AS |
|
It is important to be familiar with the Assessment of SpondyloArthritis international Society (ASAS) criteria for adults with spondyloarthritis (SpA), which categorize patients into axial and predominantly peripheral forms of SpA ( Table 20.3 ). , The ASAS classification system along with its treatment guidelines have sometimes been applied to the pediatric population, especially to patients who do not fulfill ILAR criteria for ERA but are included under the axial forms of SpA. The main advantage of the ASAS classification is for earlier diagnosis of axial disease and implementation of appropriate therapeutics because it includes the full spectrum of SpA and all imaging modalities (sacroiliitis on radiographs or magnetic resonance imaging [MRI]). The main limitation of the ASAS criteria is that many pediatric patients with axial involvement may not appreciate or report symptoms of back pain for 3 months or longer, which is required for inclusion in the axial SpA category.
| Axial SpA | Peripheral SpA | ||
|---|---|---|---|
| In patients with >3 months back pain and age at onset <45 years | In patients with peripheral symptoms ONLY | ||
| Sacroiliitis on imaging∗ plus >1 SpA feature | OR | HLA-B27 plus >2 other SpA features | Arthritis or enthesitis or dactylitis plus |
SpA features
|
>1 SpA feature
OR
|
||
|
|||
ERA and JAS are often referred to as the undifferentiated and differentiated forms of JSpA , respectively. The ILAR ERA classification criteria represents the majority of children and adolescents presenting with undifferentiated SpA, but not all patients with JAS are included in the ERA category.
The data examining the epidemiology of ERA are emerging. For this reason, the discussion that follows relies heavily on the study of patients who fulfilled traditional criteria for AS. Where there is sufficient evidence, ERA-specific information is included.
Among children with JIA, the proportion with ERA ranged from 8.6% to 18.9%. Increasing awareness of ERA, and its clinical and laboratory differentiation from other chronic arthritides of childhood, will probably result in an increase in the proportion of children in this category. In contrast, children with JAS accounted for 1% to 7% of subjects in national pediatric rheumatic disease registries in the United States, Canada, and the United Kingdom and from studies in Sweden, Finland, and Croatia.
It is estimated that the prevalence of AS in adults is from 0.5% to 1.9%, , with 8.6% to 11% having onset in childhood. However, the occurrence of AS varies among different racial and ethnic groups as demonstrated by population-based studies, with reported prevalence of AS in adults to be 0.24% in Europe, 0.17% in Asia, 0.10% in Latin America, 0.32% in North America, and 0.07% in Africa. On the basis of the prevalence of HLA-B27 and the frequency of sacroiliitis in the HLA-B27–positive population, the prevalence of AS was estimated to be 0.86% to 1% , and was highest in HLA-B27–positive individuals. Although this estimate includes asymptomatic persons, it also excludes the 8% to 10% of the AS population who do not have HLA-B27, and it may be a more accurate reflection of the prevalence of the entire spectrum of AS.
In cohorts of ERA patients around the world, the mean age at diagnosis has been reported to be around 10 to 13 years (range 2.8 to 17.6 years), , , , similar to that for JAS. The age distribution appears to be homogeneous and presumably is continuous with that described in adult populations, suggesting that, at least on this basis, the disease as seen in adults is the same or very similar to that in children.
In ERA, there is a predominance of boys but variations in the male-to-female ratio can be seen among cohorts from Canada (3.4:1 to 4.1:1), , the United States (1.4:1), Taiwan (3.3:1), Spain (8:1), and India (9:1). JAS has a much higher frequency in boys than in girls: of 247 children with this disorder, 216 were boys, for a male-to-female ratio of 7:1. This disproportionate representation of boys may not accurately represent the actual occurrence of the disease in girls. The strong correlation of JAS and ERA with HLA-B27 and the equal distribution of this allele in males and females suggest that JAS and ERA could be as common in girls as in boys. The use of sex as a possible criterion in the ILAR classification of JIA may also affect the apparent male-to-female ratio of ERA. Furthermore, in radiographic surveys of HLA-B27–positive adult blood donors, SI arthritis was as common in women as in men. In a questionnaire survey of members of the National Ankylosing Spondylitis Society in the United Kingdom, the male-to-female ratio was 2.7:1. However, in women, manifestations of the disease may occur later and be less severe, and they may have more peripheral and less axial disease. It is possible that these observations contribute to the relative infrequency of the diagnosis in women.
Few data are specifically related to geographical and racial differences in the frequency of ERA or JAS. The low incidence of AS in African Americans and in Japanese, and the high frequency in the Haida Indians of Pacific Canada reflect, in part, the frequency of HLA-B27 in these populations. Other factors may be significant, however, because this allele occurs in only 50% of African Americans with AS and in 65% to 90% of Japanese people with the disease. In a multiethnic Canadian population, ERA was twice as common in children of European ancestry as in those of non-European origin. However, among children of non-European ancestry, children of Asian origin were more likely to develop ERA than any of the other categories of JIA. , ,
AS is a complex genetic disease with overall heritability estimated to be greater than 90%. In addition to the major histocompatibility complex (MHC) class I HLA-B allele (B27), more than 100 non-MHC genomic regions that confer risk have been identified through genome-wide association studies. HLA-B27 is responsible for about 20% of heritability, whereas the non-MHC regions combined account for only another 8%, with over 70% of heritability undefined. Despite an incomplete genetic landscape, these discoveries, together with translational and functional genomics studies, have led to a better understanding of pathogenesis and identified new targets for therapeutic intervention. Because the principle clinical features of ERA are similar to JAS and AS, the genetic underpinnings are expected to overlap. However, ERA is more heterogeneous in its clinical features and outcome, suggesting there may be important differences.
The major genetic risk factor for ERA is HLA-B27, similar to JAS/AS and other forms of SpA. The frequency of HLA-B27 in JAS has been reported to be close to 90%, , , , which is similar to adult-onset AS. Precise quantification of the effect of HLA-B27 on the risk of developing ERA is not possible because HLA-B27 is used to classify ERA using the ILAR criteria. Nevertheless, some studies have reported that HLA-B27 is present in 60% to 90% of ERA.
HLA-B27 is polymorphic with over 150 genetic variants (subtypes) reported to date that are predicted to alter its amino acid sequence. The most common subtype and the ancestral allele is HLA-B∗2705, which is strongly associated with disease in multiple populations around the world. Other subtypes that clearly confer disease risk include HLA-B∗2702, HLA-B∗2704, and HLA-B∗2707. Two subtypes, HLA-B∗2709 and HLA-B∗2706, appear to be less strongly or not associated with SpA, respectively. , HLA-B∗2706 is common in Southeast Asia, whereas HLA-B∗2709 is found almost exclusively in Sardinia. There is very little information on HLA-B27 subtypes in ERA. In a study of 56 Latvian children with JIA who were HLA-B27 positive, eight HLA-B27 subtypes were identified in the subjects classified as ERA, with B∗2705 being the most common. Many of the HLA-B27 subtypes are very rare and some represent single reports. Thus their relationship with AS and other forms of SpA remains unknown.
Homozygosity for HLA-B27, which would be expected to occur by chance in about 0.5% of a population where the prevalence of HLA-B27 is 7%, has been reported to increase the risk of developing AS and contribute to an earlier age of onset. However, there is no evidence that JAS is associated with HLA-B27 homozygosity.
HLA class II associations have also been described for JAS and ERA. A higher frequency of HLA-DRB1∗08 has been reported in a Mexican population with JAS, and in ERA associations with HLA-DRB1∗01, DQA1∗01, and DQB1∗05 have been observed, whereas DRB1∗07 and DPB1∗02 were protective. HLA class II effects are small in comparison with risk conferred by HLA-B27.
The risk of developing some form of SpA in an HLA-B27–positive individual in the general population is estimated to be around 5%. However, in certain families AS is inherited with HLA-B27 in an autosomal dominant pattern at a penetrance of about 20%, and in small studies monozygotic twin concordance rates as high as 75% have been reported. Thus, the development of AS depends on genetic factors beyond HLA-B27. The increased incidence of AS in certain families underscores the importance of obtaining a complete family history.
Of the more than 100 genomic regions implicated in AS susceptibility, those of particular significance include IL-23R, CARD9, IL-12B, PTGER4, IL-6R, TYK2, STAT3, and ERAP1. The potential role of these genes will be further considered in the context of pathogenic mechanisms in the next section. One of the IL-23R single nucleotide polymorphisms (SNPs) that is strongly associated with AS (rs11209026) was also associated with juvenile psoriatic arthritis (JPsA), but not with ERA. In contrast, in the same study one ERAP1 SNP (rs30187) that is strongly associated with AS was associated with ERA but not JPsA. Larger studies are needed to determine the extent to which AS risk alleles contribute to the development of ERA.
Clinical and epidemiological similarities between ERA, JAS/AS, and other forms of SpA, particularly inflammatory bowel disease (IBD)-associated and reactive arthritis, have long suggested a role for microbes in disease. In addition, HLA-B27 transgenic rats that develop SpA-like disease in conventional animal facilities are completely protected from inflammation in a germ-free environment. , The introduction of normal gut flora is sufficient to trigger colitis and arthritis. The SpA-IBD overlap in human disease is well recognized. In IBD, sacroiliitis is common, particularly in individuals with HLA-B27, and in both adults and children with SpA there is a high frequency of subclinical gut inflammation. , Although certain gastrointestinal (e.g., Salmonella, Shigella, Yersinia ) and genitourinary infections (e.g. Chlamydia ) are clear triggers for reactive arthritis, which can evolve into more chronic SpA, there is little evidence that these or other pathogens are a cause of AS. However, there is accumulating evidence that “normal” gut microbial communities are altered in SpA including AS, PsA, reactive arthritis, and ERA, , and that HLA-B27 may play a role. However, effects of HLA-B27 are complicated and studies so far suggest an “ecological” model of dysbiosis where the pattern of changes in microbes is highly dependent on other genetic and environmental factors. , Two studies have associated certain microbes with disease activity in adults with SpA. , However, cause and effect relationships between specific gut microbiota and SpA remain to be established.
The mechanism(s) whereby HLA-B27 promotes disease pathogenesis remain unresolved. Three main concepts have evolved. The first is that HLA-B27 is targeted by autoreactive CD8 + T lymphocytes because it presents self-peptides that resemble peptides derived from microorganisms (molecular mimicry). Known as the arthritogenic peptide hypothesis, this concept would suggest the tissue specificity of inflammation is the result of local presentation of autoreactive self-peptides. To date, no arthritogenic peptides have been identified, and this hypothesis is inconsistent with results from HLA-B27 transgenic rats where CD8 + T cells are dispensable. , HLA-B27 also exhibits aberrant features, including a tendency to misfold and form disulfide-linked dimers in the endoplasmic reticulum before assembling into complexes containing peptide and β 2 m. , In addition, cell surface HLA-B27 complexes that are endocytosed after losing peptide and β 2 m tend to dimerize during recycling, which results in expression of aberrant molecules on the cell surface.
Misfolding of the HLA-B27 heavy chain in the endoplasmic reticulum can result in its intracellular accumulation and can provoke an inflammatory response through overproduction of certain cytokines including IL-23, which promotes CD4 + T helper (Th)17 T lymphocyte survival and IL-17 production. One consequence of protein misfolding, activation of the cellular unfolded protein response (UPR), has been linked to overproduction of IL-23. , In addition, HLA-B27 expression can render cells defective in killing intracellular bacteria that are implicated in triggering reactive arthritis, consistent with reports that bacterial antigens or DNA can be found in synoviocytes from patients with reactive arthritis. The UPR associated with HLA-B27 has been shown to enhance intracellular replication of Salmonella, a common trigger for reactive arthritis. HLA-B27 homodimers that form at the cell surface can be recognized by immunoreceptors in the killer immunoglobulin receptor (KIR) and leukocyte immunoglobulin-like receptor (LILR) families expressed on natural killer (NK) cells and other lymphocytes, including the CD4 + Th17 subset of CD3+ T lymphocytes, and trigger IL-17 production.
These potential mechanisms linking HLA-B27 to disease are not mutually exclusive, and no single mechanism is consistent with all literature on the subject. In addition, HLA-B27 expression has effects on dendritic cells (DCs) that lack a molecular explanation at this juncture but may be important in SpA pathogenesis. ,
Several genes implicated in susceptibility to AS (e.g., IL-23R, CARD9, IL-12B, PTGER4, IL-6R, TYK2, and STAT3 ) are of particular interest because of their potential to influence the development, activation, and cytokine production of CD4 + Th17 T lymphocytes and other IL-17–producing innate lymphoid cells. For example, CARD9, IL-12B, and PTGER4 variants could influence IL-23 production downstream of pattern recognition receptors. Other genes that can influence IL-23 responsiveness include IL-23R, TYK2 (a Jak gene), and STAT3. Variants in IL-6R, which encodes a subunit of the IL-6 receptor, and IL-27, a subunit of the IL-27 cytokine, might influence the number and frequency of IL-23–responsive cells. PTGER4 encodes the prostaglandin E 2 (PGE 2 ) receptor (EP4), and PGE 2 can regulate bone formation, as well as the development of Th17 T cells. Expression of the PGE 2 receptor EP4 by Th17 T cells in AS is associated with high disease activity.
Enthesitis, a key pathological feature of ERA, JAS/AS, and other forms of SpA, can be induced in mice by systemic expression of IL-23. The enthesitis affects both axial and peripheral entheses and is mediated by an unusual variety of CD3 + T lymphocytes that lack CD4 and CD8. These T cells express the IL-23 receptor and a transcription factor (retinoic acid receptor–related orphan receptor-γt [ROR-γt]) that drives a Th17-like program of cytokines and chemokines (e.g., IL-6, IL-17, IL-22, CXCL1) in response to IL-23. In another animal model where autoreactive CD4 + Th17 T cells accumulate because of a mutation in the ZAP-70 subunit of the T-cell receptor (“SKG mice”), the SpA phenotype can be induced by microbial β-glucans (e.g., curdlan, zymosan), which induces IL-23 production. These models demonstrate clear evidence that IL-23 can generate the unique entheseal inflammation phenotype of SpA through entheseal-resident T cells without invoking tissue-restricted CD8 + T-cell mediated HLA-B27 peptide presentation.
Genetic discoveries, results from animal models, and effects of HLA-B27 misfolding and dimerization implicating the IL-23/IL-17 axis in SpA pathogenesis are strongly supported by patient-derived translational studies. IL-17 and IL-23 overexpression has been documented in serum and synovial fluid and at sites of inflammation in SpA patients, including JIA (although not specifically ERA). This has culminated in clinical trials demonstrating efficacy of IL-17 blockade in AS, , although it should be noted that IL-23 and IL-12/23 blockade has not been effective in ameliorating symptoms of disease. ,
Studies of ERA have also documented increased levels of IL-6, interferon (IFN)-γ, and tumor necrosis factor (TNF) in synovial fluid compared with either polyarticular JIA or RA. ERA also exhibits increases in “intermediate” monocytes that express CD14 and CD16, as well as NK and γδ T cells in the peripheral blood. ,
One of the genes associated with AS and ERA, endoplasmic reticulum (ER) aminopeptidase-1 (ERAP1) , trims N-terminal amino acids from peptides, which optimizes their length for binding to most MHC class I molecules. , The genetic effect of ERAP1 variants in AS is limited to HLA-B∗27– and HLA-B∗40–positive individuals, suggesting a gene–gene or epistatic interaction. ERAP1 variants that confer loss of function or reduce expression are protective for AS. , It is unclear why inefficient generation of optimal HLA-B27 binding peptides would be “protective” in this disease. The epistatic interaction between HLA-B27 and ERAP1 should yield more clues to SpA. Although ERAP1 clearly implicates peptides in pathogenesis, peptides not only serve as antigens presented by HLA molecules but also greatly affect aberrant properties such as misfolding and dimerization of HLA-B27 heavy chains.
The onset of ERA may be insidious and characterized by intermittent musculoskeletal pain and stiffness or objective inflammation of peripheral joints, particularly those of the lower extremities, together with enthesitis at one or more sites around the knee or foot. Occasionally, the disease may have an abrupt onset. Systemic signs are often minimal, but fatigue, sleep disturbances, and low-grade fever may be present. Symptoms related to the back are usually absent at onset but become increasingly evident during the disease course in adolescents. Differing modes of presentation and course may characterize specific population groups. ,
Entheses, where ligaments, tendons, fascia, or capsules attach to bone, are characteristic sites of inflammation. Enthesitis occurs in 60% to 80% of ERA and JAS patients and is more frequent in the lower limbs, especially insertions of the infrapatellar tendon on the tibial tuberosity, quadriceps insertion to the patella, and Achilles and plantar fascia insertion into the calcaneus. Consequently, many patients with ERA complain of knee, foot, or heel pain. Weiss et al. noted that in 32 newly diagnosed patients with ERA, 66% had at least one tender entheseal site whereas 44% had more than two sites involved. The presence of an increasing number of tender entheses at presentation was associated with persistence at 6 months (odds ratio [OR] 2.18). Although the presence of exquisite, well-localized tenderness at characteristic entheses strongly suggests ERA, it must be noted that enthesitis occurs occasionally in other types of JIA, other rheumatic diseases, and occasionally in normal children. Osgood–Schlatter disease should also be excluded before making a diagnosis of enthesitis affecting the tibial tuberosities. The presence of enthesitis is the most helpful clinical feature in differentiating ERA from other types of JIA. Peripheral enthesitis is a characteristic early manifestation of JAS and occurs with greater frequency in JAS than in adult-onset AS. It frequently produces severe pain and resultant disability, which may be the child’s most important complaints.
A careful history and a thorough but gentle palpation of entheses may document evidence of past or present inflammation. The entheseal exam can include any of the anatomical sites shown in Fig. 20.1 and Fig. 20.2 . Enthesitis is diagnosed clinically as marked localized tenderness and/or swelling at the enthesis. A study in 30 ERA and 30 control subjects, in which tenderness detected by a standardized dolorimeter examination correlated poorly with ultrasound (US)-confirmed enthesitis, supports the need for improved clinical accuracy and reliability in determining enthesitis based solely on the physical examination finding of localized tenderness. Fibromyalgia tender points may mimic enthesitis. In fibromyalgia there is usually evidence of multiple nonentheseal tender points. However, ERA and secondary fibromyalgia can coexist.
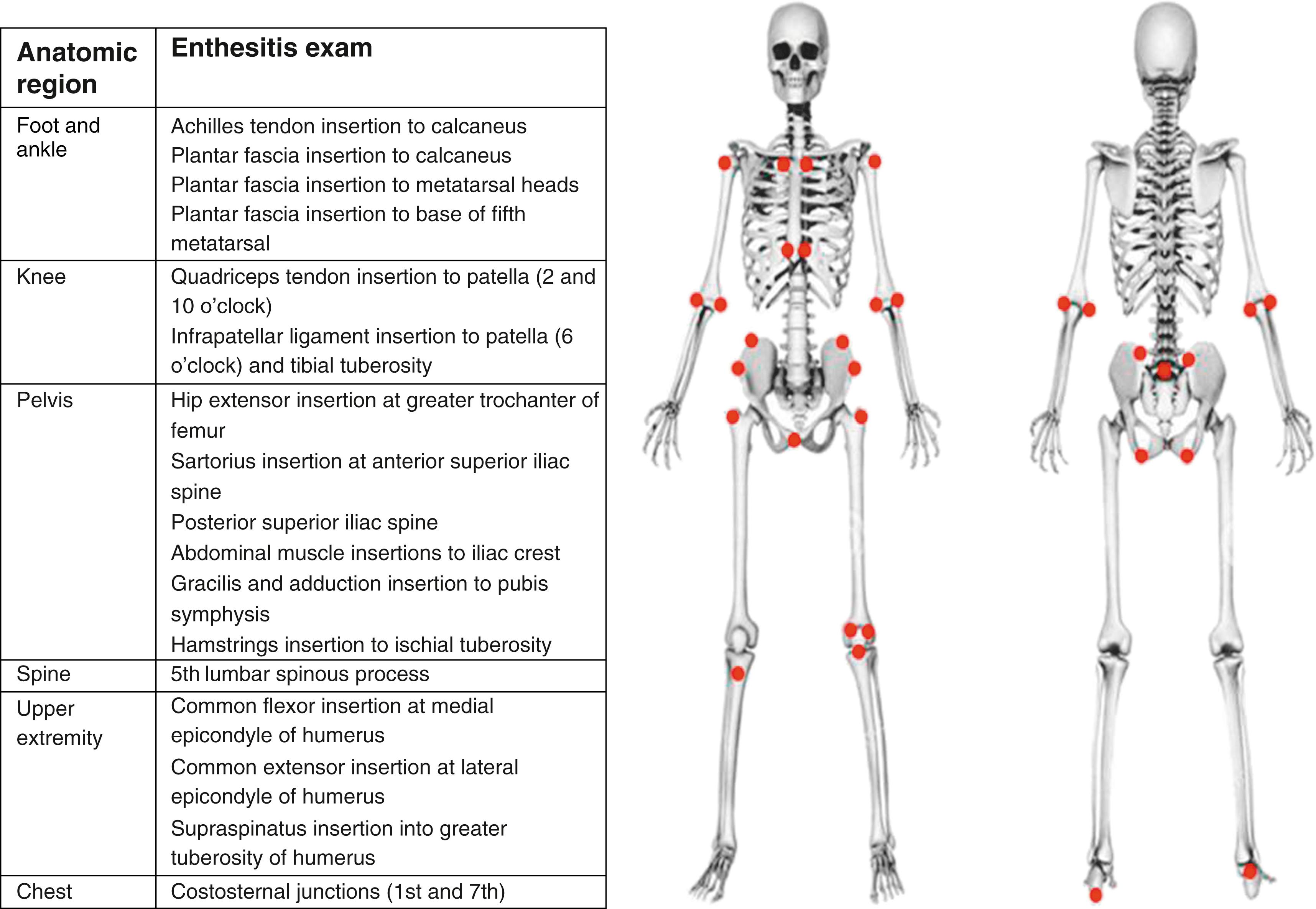
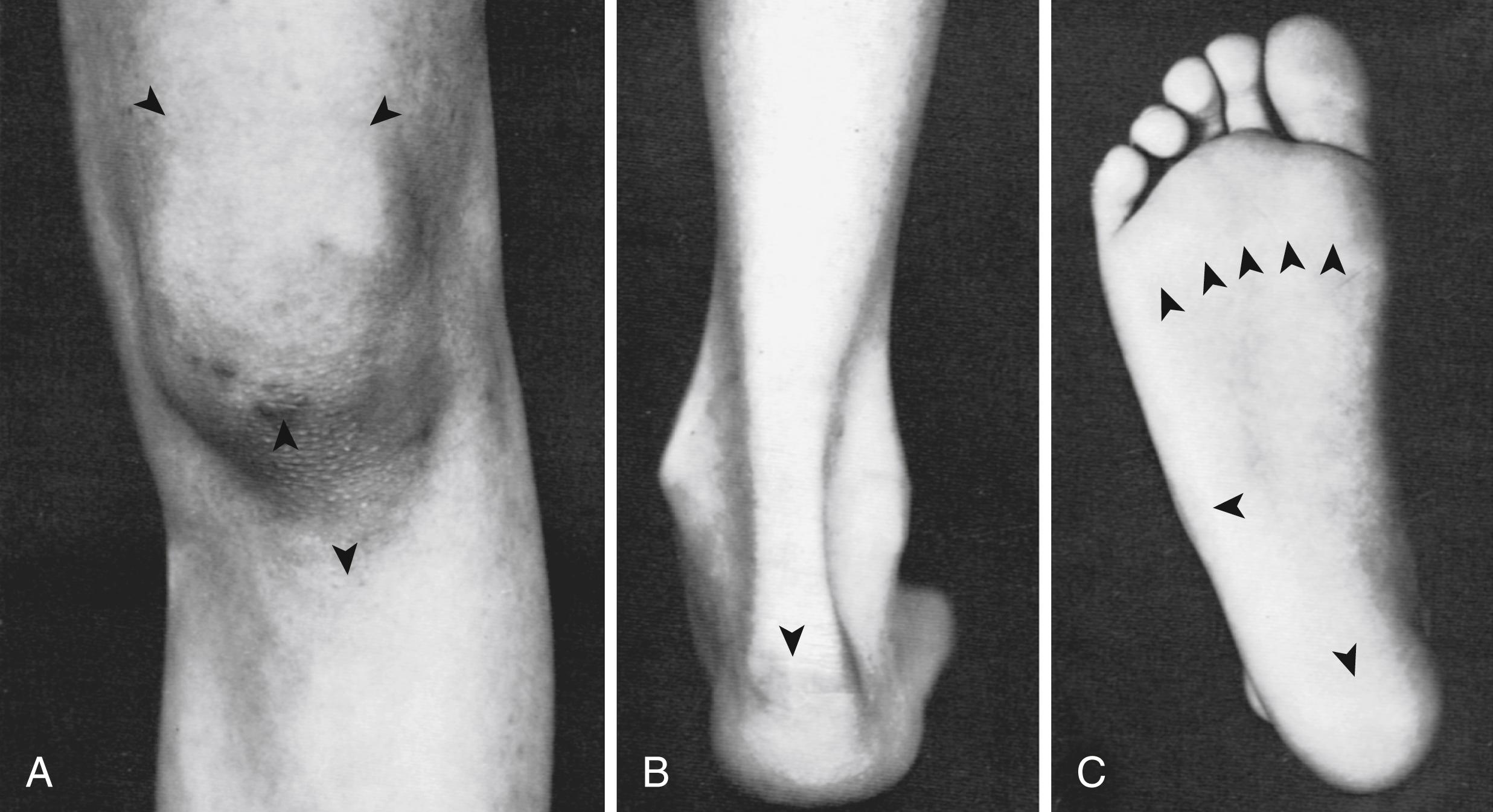
A diagnosis of ERA is strongly supported by enthesitis in the lower extremities especially at insertional sites around the patella (10, 2, and 6 o’clock positions especially), heel, and plantar fascial attachments to the heads of the metatarsals ( Fig. 20.2 ). Less commonly, tenderness can also be demonstrated at insertional sites around the pelvis, over the spinous processes, and at the entheses of the upper extremities. Observation of stance and gait (including walking on the toes and heels) may reveal altered weight-bearing as the child avoids pressure on inflamed entheses.
The arthritis in ERA has a predilection especially for joints in the lower extremities but is less commonly in axial joints (SI and spine) at disease onset and during the disease course. Numerous ERA cohorts have reported that the most frequently affected joints at disease onset are knees (45% to 65%), midfoot (58%), and ankles (36% to 48%), , whereas during the disease course knees (52%), hips (42%), ankles (38%), midfoot, and lumbosacral spine (48%) are involved. Upper extremity involvement was infrequent but may include the wrist and shoulder.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here