Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Enlargement of the vestibular aqueduct is a commonly detected inner ear anomaly.
The hearing loss of enlargement of the vestibular aqueduct (EVA) characteristically shows fluctuation, progression, or both.
Patients with EVA often have a low-frequency conductive hearing loss in the presence of a normal middle ear. The otolaryngologist should have a high index of suspicion to avoid unnecessary placement of tympanostomy tubes in such children.
EVA is often caused by mutations of SLC26A4, which encodes a transmembrane anion exchanger called pendrin.
The Caucasian enlarged vestibular aqueduct (CEVA) haplotype defines the most common pathogenic allele associated with hereditary hearing loss. SLC26A4 can be identified as the gene responsible for EVA when CEVA occurs in trans to mutations affecting the coding regions or splice sites of SLC26A4.
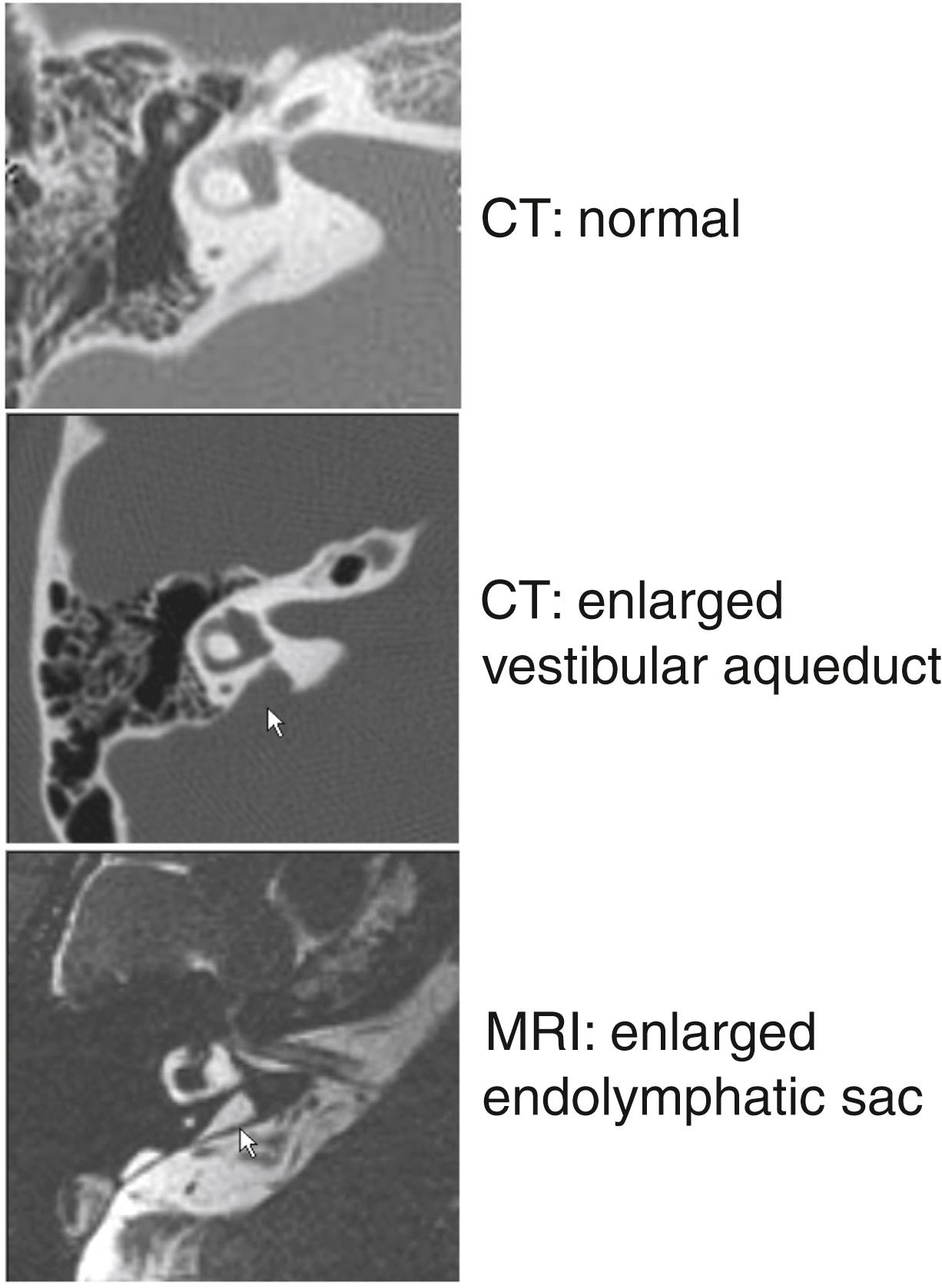
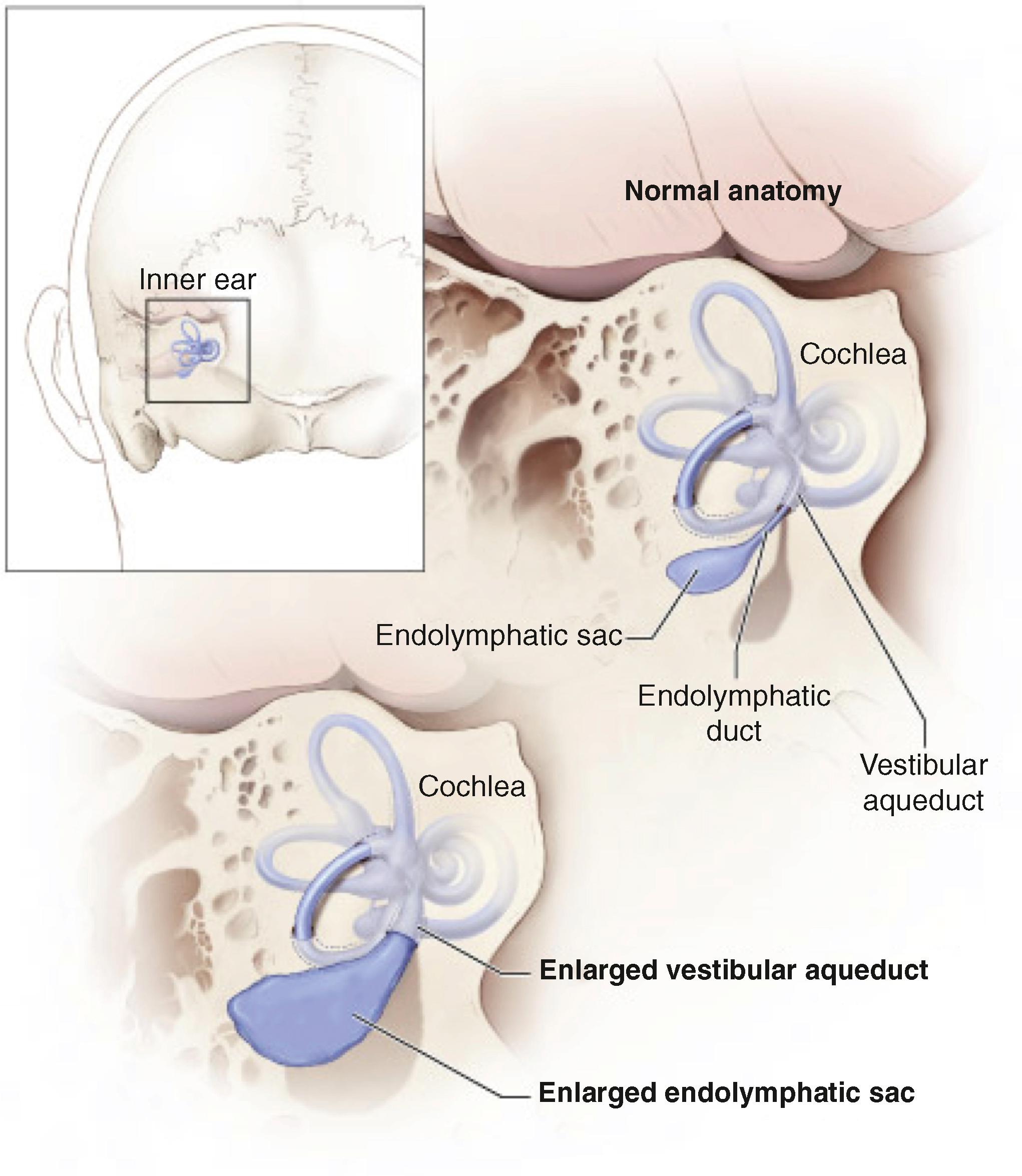
The vestibular aqueduct is an osseous canal containing the endolymphatic duct and a portion of the endolymphatic sac, which extends into the posterior fossa. Enlargement of the vestibular aqueduct (EVA), which is also referred to as a dilated or large vestibular aqueduct (DVA or LVA), is the most commonly detected osseous malformation of the inner ear in children with sensorineural hearing loss (SNHL). The radiologic definition of EVA was first established as a midpoint diameter of >1.5 mm or a grossly malformed overall morphology of the vestibular aqueduct. Newer criteria, based on computed tomography (CT) studies of children with and without SNHL, define EVA as ≥1.0 mm at the midpoint, ≥2.0 mm at the operculum, or both ( Fig. 14.1 ). , Magnetic resonance imaging (MRI) of patients with EVA shows enlargement of the endolymphatic duct and sac (see Fig. 14.1 ). , The relationship of the vestibular aqueduct with the endolymphatic duct and sac is shown in Fig. 14.2 .
In 1791 Mondini described the eponymous inner ear deformity (Mondini dysplasia) consisting of (1) a cochlea with one-and-one-half turns instead of the normal two-and-one-half turns, comprising a normal basal turn and a cystic apex in place of the distal one-and-one-half turns; (2) an enlarged vestibule with normal semicircular canals; and (3) an enlarged vestibular aqueduct containing a dilated endolymphatic sac. , The cochlear deformity of Mondini dysplasia is also known as incomplete partition type II (IP-II). In 1978 Valvassori and Clemis described a group of patients with abnormally enlarged vestibular aqueducts as the predominant inner ear anomaly, and termed it the “large vestibular aqueduct syndrome.” They reported that 40% of their patients had isolated EVA, and the remainder showed EVA with additional inner ear abnormalities including an enlarged vestibule (28%), an enlarged vestibule and lateral semicircular canal (14%), an enlarged vestibule and hypoplastic cochlea (8%), or a hypoplastic cochlea (8%).
Two landmark studies published in 1989 described a distinctive auditory phenotype associated with isolated EVA. , They described the hearing loss as sensorineural or mixed, variable in severity, and asymmetric or unilateral, with a prelingual or perilingual onset. The SNHL can fluctuate or progress in a stepwise incremental fashion. The stepwise hearing loss is often sudden and can be precipitated by minor head trauma or barotrauma. Patients with EVA often have an ipsilateral low-frequency air-bone gap in the presence of a normal middle ear. Such “cochlear conductive hearing loss” is attributed to a “third window” effect of the EVA, in which the power associated with waves elicited by motion of the stapes is shunted away from the cochlea. It is important to assess for the presence of middle ear effusion or disease to prevent unnecessary tympanostomy tube placement into normal middle ears of children with EVA. The diagnosis of EVA is too often unsuspected until the postoperative audiogram shows no improvement.
A systematic review demonstrated that EVA was detected with a pooled frequency of 7% for CT and 12% for MRI in children with unilateral SNHL. Likewise, EVA is often unilateral. The reported prevalence of patients with unilateral EVA in all EVA patients ranges from 6% to 51%. , A 2013 study demonstrated that among patients with unilateral EVA defined by the newer radiologic criteria, 45% had hearing loss only in the ipsilateral ear, and 54% had hearing loss in both ears. The high prevalence of hearing loss among contralateral, radiologically normal ears led the authors to suggest that unilateral EVA is the manifestation of a bilateral process, despite the absence of imaging findings in contralateral ears. Their suggestion was underscored by the observation that in 1% of unilateral EVA cases, the ipsilateral (EVA) ear had normal hearing, but the contralateral radiologically normal ear had hearing loss. In contrast, another study reported that only 4.9% of children with unilateral EVA had bilateral hearing loss, and the rate of contralateral hearing loss in children with unilateral EVA is no different from that in children without EVA.
Although the original reports emphasized EVA as an isolated radiologic abnormality, the incidence of associated inner ear abnormalities ranges from 22.5% to 88%, including cochlear dysplasia (IP-II), , , common cavity, modiolar hypoplasia, , and dysplasia of the vestibule. , The presence of an associated cochlear anomaly does not appear to be independently associated with severity of hearing loss in ears with EVA. , The relationship between the size of EVA and hearing loss is controversial. Some studies have found no correlation of the size of the EVA with hearing loss. , In contrast, others have reported significant associations between vestibular aqueduct size and audiological findings. This discordance could be caused by limited sample size in some studies, variability of defining hearing loss related to fluctuation or progression, different radiographic size criteria, undetected genetic differences between patient cohorts, or ascertainment bias caused by the method of subject recruitment.
It has been debated whether EVA is the direct cause of hearing loss or simply a gross anatomic correlate of some other underlying molecular, cellular, or physiologic abnormality that cannot be identified by radiologic imaging. One early theory proposed that reflux of electrolytes or hyperosmolar contents from the endolymphatic sac to the scala media of the cochlea could exert a toxic effect on the neurosensory epithelium and thus cause hearing loss. This reflux theory was based on the observation and assumption, respectively, that head trauma or barotrauma precipitates hearing loss by an increase in intracranial pressure which, in turn, squeezes the contents of the endolymphatic sac into the cochlea. This conceptual framework led some surgeons to perform endolymphatic sac obliteration or endolymphatic sac shunt operations to stabilize hearing. However, the results were poor, and hearing loss was exacerbated in many cases. , ,
The fluctuating or progressive hearing loss in ears with EVA has some resemblance to hearing loss associated with endolymphatic hydrops in Meniere disease. A 2016 study using contrast-enhanced MRI demonstrated that all 12 ears of 6 EVA cases showed mild to severe endolymphatic hydrops. In contrast, histopathological studies of classic Mondini dysplasia reported the absence of endolymphatic hydrops of the cochlea. Therefore endolymphatic hydrops may not be a direct cause of hearing loss but a nonspecific marker for the underlying cellular or molecular pathogenetic defect. ,
Another theory proposed that hearing loss results from leakage of perilymph from an abnormal fistulous round window, which has not been corroborated by others but, nevertheless, remains a potential albeit rare etiology in some patients.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here