Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The spectrum of Endometrial Glandular Neoplasia is wide and includes different types of etiologically, morphologically, and/or molecularly distinct entities. In this chapter, the rationale for the new classification of precursors of endometrial carcinoma and the main types of endometrial carcinoma are discussed. Common problems in the differential diagnosis, and the role of immunohistochemistry and molecular categorization of endometrial carcinoma, are also covered.
Endometrioid Carcinoma Precursors
Endometrial hyperplasia refers to an abundance of endometrial glands. The term has served historically to identify a spectrum of endometrial changes, some of them related to cancer. In 1975, the World Health Organization (WHO) proposed a classification scheme which recognized three main categories of endometrial hyperplasia: cystic, adenomatous, and atypical. A new classification was proposed in 1994 and accepted in 2003 dividing endometrial hyperplasia into four groups based on architectural complexity and cytologic atypia: (1) simple hyperplasia without atypia, (2) complex hyperplasia without atypia, (3) simple hyperplasia with atypia, and (4) complex hyperplasia with atypia. This scheme was since challenged due to its lack of reproducibility, particularly concerning criteria for nuclear atypia. Consequently, to improve interobserver reproducibility, it was proposed to collapse the four-tier scheme into two categories: nonatypical and atypical. Approximately 15%–20% of patients with a biopsy diagnosis of atypical hyperplasia have a well-differentiated endometrioid carcinoma upon hysterectomy; likewise, 25%–30% of patients with atypical endometrial hyperplasia progress to carcinoma if untreated. In contrast, nonatypical endometrial hyperplasia has a much lower (1.6%) risk of carcinoma progression.
The study of endometrial hyperplasia using morphometry and molecular approaches led to the characterization of a lesion characterized by gland crowding and expansion within the endometrial compartment with underlying genetic alterations akin to endometrial carcinoma (namely PTEN and PAX2 loss, microsatellite instability, and chromosome X inactivation). As a result, an alternative terminology, endometrial intraepithelial neoplasia (EIN) and criteria for its recognition was developed. Since its conception, the EIN approach has shown good interobserver reproducibility and good cancer outcome prediction. A Gynecologic Oncology Group trial treating women diagnosed with EIN on endometrial biopsy or curettage with immediate hysterectomy found concurrent endometrioid adenocarcinoma in 38% of subjects. Furthermore, the risk of developing endometrial carcinoma over time was 45 times higher in women with EIN compared to those without. Finally, a case control conditional logistic regression study found 8.95- and 9.2-fold increased risk of cancer using atypical endometrial hyperplasia and EIN definitions, respectively. In 2015, EIN was endorsed by the American College of Obstetricians and Gynecologists (ACOG) and the Society of Gynecologic Oncology (SOG).
The 2014 WHO classification proposal combined EIN criteria with former WHO terminology. The resulting categories are: nonatypical endometrial hyperplasia and atypical endometrial hyperplasia/endometrioid intraepithelial neoplasia (AEH/EIN) ( Box 10.1 ). The WHO 2014 approach has been shown to be prognostically relevant.
Nonatypical endometrial hyperplasia
Atypical endometrial hyperplasia/endometrial intraepithelial neoplasia (AEH/EIN)
Nonatypical endometrial hyperplasia is relatively common and approximately 150,000–200,000 new cases are diagnosed every year in western countries. Although it typically occurs in perimenopause, it can be also seen in reproductive-age and postmenopausal women. It is part of the spectrum of endometrial changes secondary to continuous unopposed estrogen stimulation, which may result from (1) successive prolonged periods of anovulation, (2) estrogen administration, (3) peripheral conversion of androgens to estrone in adipose tissue in obese women or patients with polycystic ovarian syndrome, or (4) estrogen-secreting ovarian neoplasms. Patients typically present with abnormal uterine bleeding (hypermenorrhea, dysmenorrhea, intermenstrual bleeding).
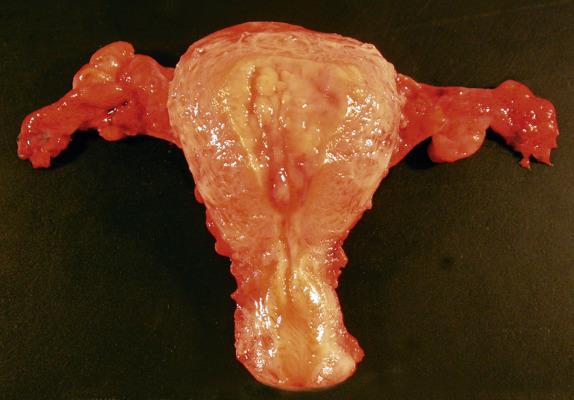
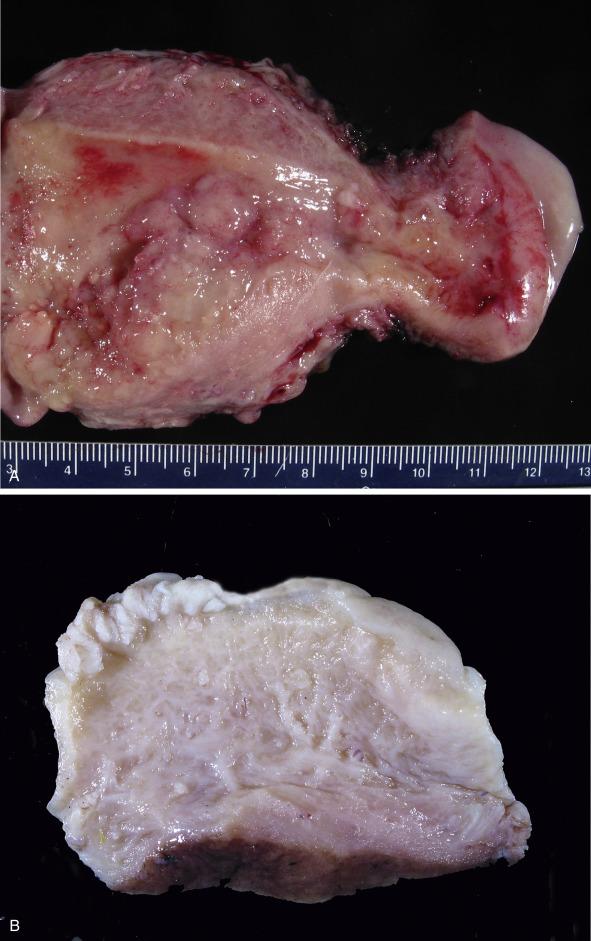
Macroscopically, hyperplastic endometrium is often characterized by abundant white to tan tissue that may have a diffusely thickened or polypoid appearance ( Fig. 10.1 ).
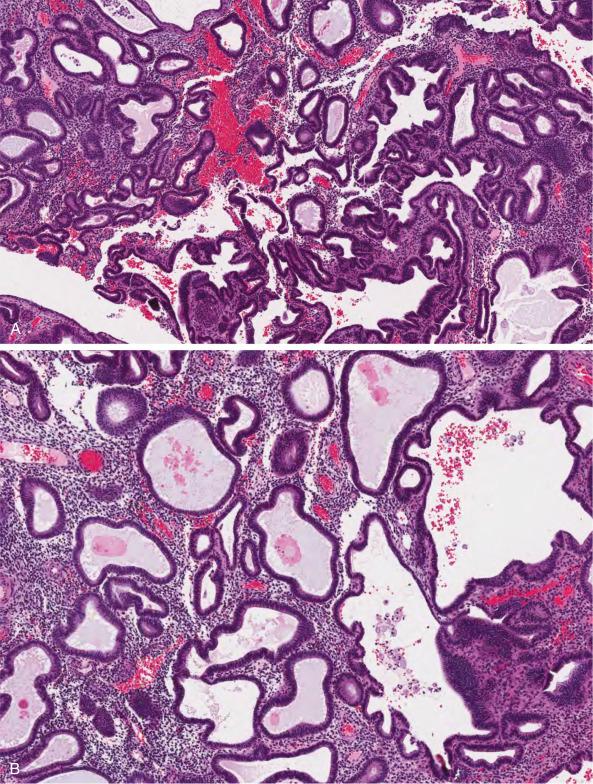
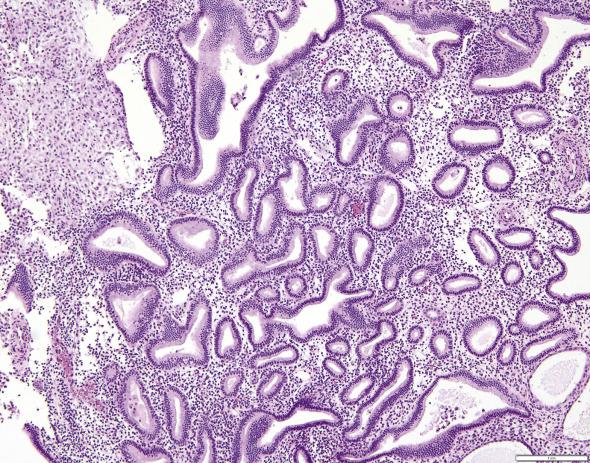
Nonatypical endometrial hyperplasia is characterized by an increase in the amount of endometrial glands, seen at scanning magnification as multifocal to diffuse crowding, in which the glands occupy more than 50% of the surface area (thus leading to a gland-to-stroma ratio greater than 1). The crowded glands may be uniformly round and tubular ( Fig. 10.2A–C ) or display irregular outlines ( Fig. 10.2D ). Cell stratification and cellular polarity are generally maintained. The epithelial lining is pseudostratified, with cells being similar to those seen in mid to late proliferative phase. Tubal metaplasia and ciliated cells are frequently seen. The cells display elongated nuclei with evenly dispersed chromatin and inconspicuous nucleoli. Mitotic figures and apoptotic bodies are common. Importantly, crowded (hyperplastic) glands imperceptibly blend with noncrowded glands, and there are no significant cytologic differences between the two populations.
In the event of superimposed ovulation or exogenous progestin administration, superimposed secretory changes will be observed; in this scenario, changes will be similar to the normal secretory endometrium, but glands remain somewhat disorganized and lack the perpendicular arrangement of the normal secretory phase.
Endometrial hyperplasias are frequently misdiagnosed as variations of the normal cycling endometrium, anovulatory cycles including disordered proliferative endometrium, postmenopausal cystic atrophy, polyps, and metaplasias. Of note, endometrial hyperplasia usually generates abundant material in curettage specimens, which is a helpful clue in the distinction from atrophy. Nonatypical hyperplasia is usually a diffuse process whereas polyps and metaplastic changes are discrete lesions.
Physiologic proliferative endometrium can display artificial changes including telescoping and pseudocrowding (multiple separate glands artificially apposed together within the sample), which can mimic endometrial hyperplasia. However, with practice these artifacts are easy to recognize, and importantly, the diagnosis of hyperplasia must be made in intact pieces of endometrium without significant fragmentation. The same principle applies to secretory endometrium . During the secretory phase, glands increase in volume, exceeding 50% of the surface area. However, unlike nonatypical hyperplasia, secretory endometrium retains organization and orderly glandular distribution perpendicular to the endometrial surface.
The distinction between nonatypical hyperplasia and disordered proliferative endometrium is often difficult but not clinically relevant as they represent a continuum in the spectrum of estrogen-related endometrial changes. Disordered proliferative endometrium commonly occurs in perimenopausal women, frequently in the setting of anovulatory cycles. It usually shows apoptosis and stromal breakdown. The defining feature of hyperplasia is the increased glandular density; although disordered proliferative endometrium has gland irregularity, the glands do not exceed 50% of the surface area, unlike hyperplasia. Frequently, both conditions coexist in the same sample/patient.
Cystic atrophy occasionally may be confused with nonatypical hyperplasia that shows prominent gland dilation; however, the glands in the former are lined by a single nonstratified layer of low cuboidal cells with no mitotic activity in contrast to the pseudostratified appearance of the nuclei associated with mitoses seen in nonatypical hyperplasia. Moreover, the stroma is compact or densely collagenous.
Endometrial polyp differs from nonatypical hyperplasia in its polypoid configuration, presence of dilated thick-walled blood vessels, stromal fibrosis or compact appearance of the stroma, and glandular irregularity with arrangement parallel to surface endometrial epithelium. In this distinction, it is important to keep in mind that polyps can have glandular crowding; in other words, polyps can contain areas of “hyperplasia.” The latter scenario is frequently overdiagnosed as hyperplasia because the cytomorphology of the glands within the polyp may differ from the native endometrium, thus comparison to the noncrowded glands within the polyp is essential. If the crowded areas are not significantly different from the background polyp glands, then it is nonatypical, and the lesion should be classified as endometrial polyp.
Finally, the absence of plasma cells, granulomata, and reactive changes in the stroma helps in the distinction between endometrial hyperplasia and chronic endometritis .
Any type of endometrial metaplasia (ciliated eosinophilic, mucinous) may occur as part of endometrial hyperplasia. Attention should be paid to the architectural arrangement of the glands. If the low-power architecture is that of a normal endometrium, the diagnosis of hyperplasia can be excluded. The distinction between nonatypical hyperplasia and AEH/EIN is discussed in the following section.
Nonatypical endometrial hyperplasia is usually treated with progestins, with response rates varying from less than 40% to 100%. This form of hyperplasia is now regarded as benign with a high chance of regression and a low risk of progression to endometrioid carcinoma (2%–4%). However, the risk increases if the cause of hyperestrogenism persists untreated, likely via genetic mechanisms leading ultimately to AEH/EIN.
Endometrial glandular proliferation characterized by increased glandular density
Secondary to excess estrogen stimulation
Frequent (150,000–200,000 new cases diagnosed every year in western countries)
Low risk of progression to carcinoma (∼2%–4%)
Risk increases if underlying excess estrogen persists over time
Typically perimenopausal women
Young women and adolescents less common
Abnormal vaginal bleeding most frequent
Occasionally asymptomatic (diagnosed incidentally)
Hormonal therapy (progestins) and follow-up sampling
Irregular endometrial thickening
Increased endometrial glandular volume defined as glands exceeding 50% of the surface area (gland-to-stroma ratio >1)
Glands are round and tubular, some with irregular contours (angulated, cystic)
Nuclear pseudostratification with preserved polarity (similar to proliferative phase)
Scant but present stroma in between glands
Absence of atypia (similar cytomorphology between crowded and noncrowded areas)
Variations/artifacts of normal cycling endometrium
Anovulatory cycles/disordered proliferative endometrium
Cystic atrophy
Endometrial polyp
Endometrial metaplasias
Chronic endometritis
Atypical endometrial hyperplasia/endometrioid intraepithelial neoplasia
Well-differentiated endometrioid carcinoma
AEH/EIN is present in ∼1.4% of endometrial biopsies. Similar to nonatypical hyperplasia, AEH/EIN usually occurs around menopause; however, it is increasingly more commonly seen in young women and adolescents as a consequence of long-standing unopposed estrogen stimulation. Indeed, 15%–25% of women with AEH/EIN are premenopausal and ∼4% are younger than 40 years of age. Patients present with abnormal uterine bleeding (postmenopausal bleeding, hypermenorrhea, dysmenorrhea, intermenstrual bleeding); others may be found to have an abnormal endometrium on ultrasound during work-up for infertility, pain, or other complaints. A minority is identified after an abnormal Papanicolaou smear showing atypical glandular cells or endometrial cells after the age of 45 years prompting endometrial sampling.
Macroscopically, AEH/EIN may appear as abundant tan tissue with a polypoid or indistinct appearance. Focal lesions are not grossly visible.
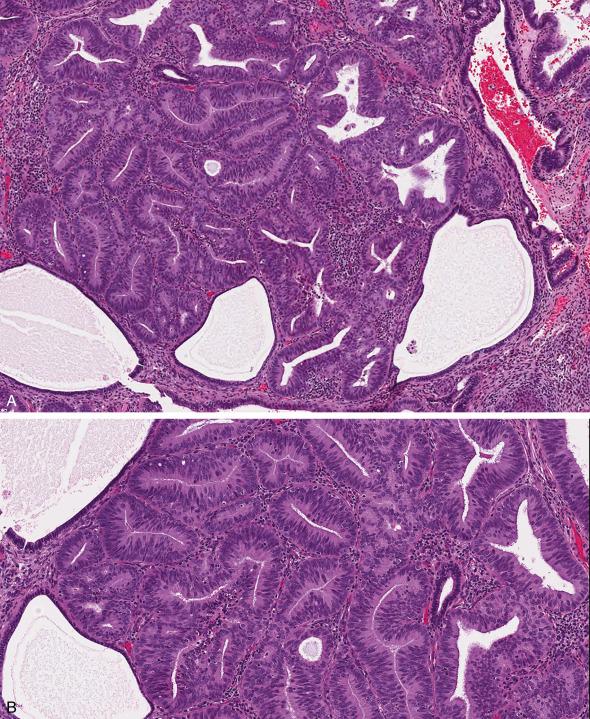
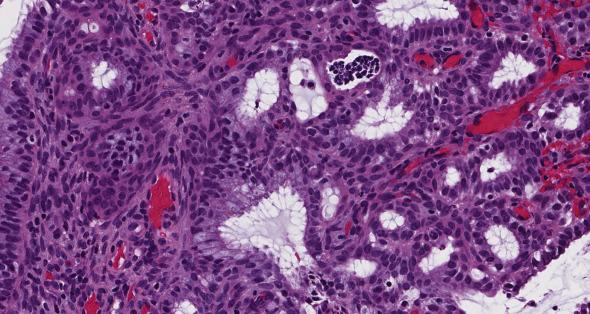
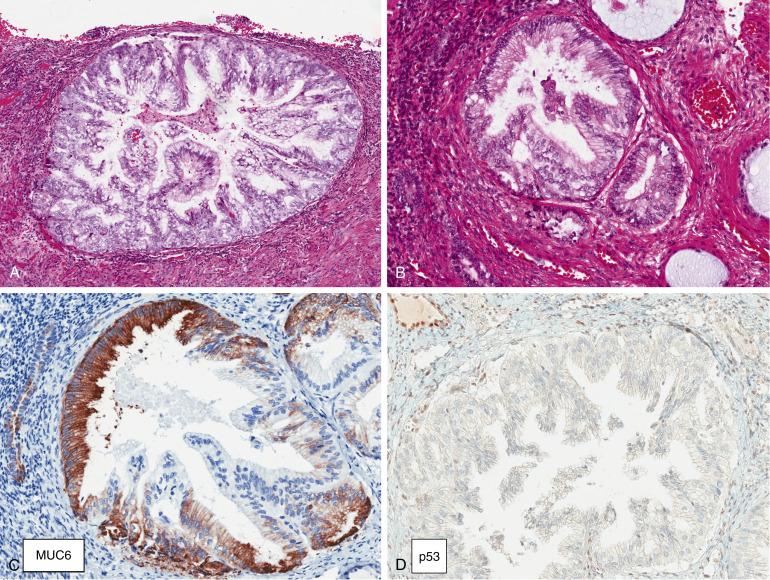
AEH/EIN is characterized by crowded glands lined by cytologically altered cells with little intervening stroma, and typically seen as a discrete expansile focus that is distinct from the surrounding endometrium ( Fig. 10.3 A–C ). The histologic criteria for EIN, as originally established, are:
Maximum linear dimension exceeding 1 mm in greatest dimension (usually encompassing >5–10 glands) ( Fig. 10.3A ).
Glandular crowding with little intervening stroma (gland-to-stroma ratio greater than 1) ( Fig. 10.3B ).
Cytologically altered tubular or branching glands with different cytology between architecturally crowded focus and background ( Fig. 10.3C ). Nuclear atypia is variable both qualitatively and quantitatively.
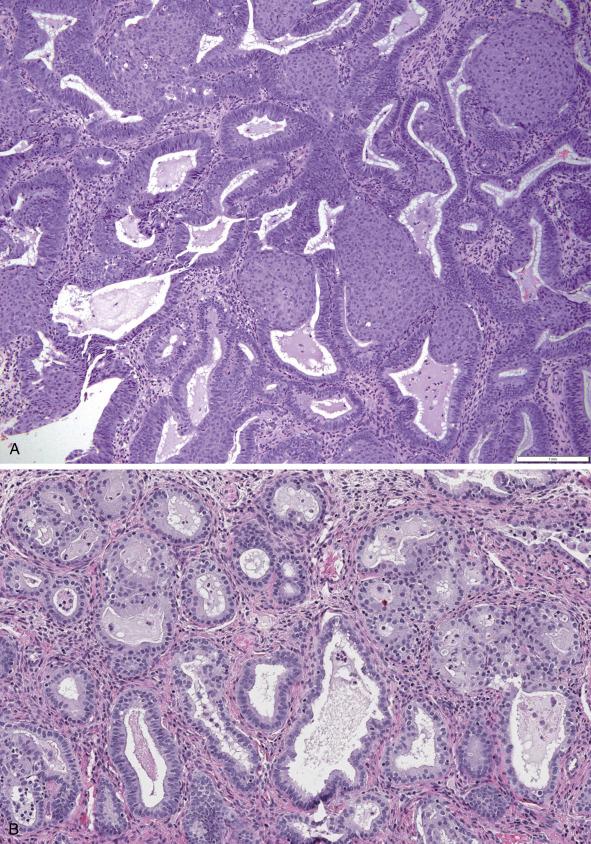
In most cases, the altered cytomorphology of the crowded area is seen as large round nuclei with inconspicuous nucleoli, increased nuclear-to-cytoplasmic ratio and loss of axial polarity. The mitotic rate varies. It is important to be aware that cytologic alterations are restricted to the architecturally crowded focus, and they should not be seen in the background noncrowded endometrial glands. The neoplastic epithelium can display squamous morular, tubal, secretory, eosinophilic, mucinous, or papillary metaplasia ( Fig. 10.4A–D ).
AEH/EIN subjected to treatment with high-dose progesterone levels may display a variety of changes. Pathologic assessment in this setting requires adequate clinical information, including time since initial diagnosis and duration of progestin treatment. Ideally, the first diagnostic sample should be compared with the follow-up specimen. In general, AEH/EIN lesions responding to progestin display cytologic and architectural changes; the former usually appear first. Architectural changes include decrease in volume of disease and number of involved fragments, diminution of the glandular crowding, effacement of the nuclear stratification, and decreased cellularity. Cytologic changes include lower nuclear-to-cytoplasmic ratio due to decreased nuclear size, nuclear rounding, cytoplasmic secretory change, intraluminal secretions, and glandular metaplasia (squamous, mucinous, and eosinophilic) ( Fig. 10.5 ). For diagnostic purposes, the criteria for the diagnosis of AEH/EIN and carcinoma should also be applied in this setting. In other words, even if there are morphologic features or response, if there is glandular crowding and features diagnostic of AEH/EIN or carcinoma, the biopsy should be reported as such with a comment stating that changes associated with progestin treatment are noted.
AEH/EIN is positive for ER and PR, usually in a diffuse fashion. Loss of PTEN and PAX2 staining is seen in approximately 44% and 71% of EIN lesions, respectively.
AEH/EIN requires distinction from benign conditions including nonatypical endometrial hyperplasia, normal endometrium, endometrial polyp, endometrial metaplasia, and certain endocervical reactive proliferations. Of note, areas of gland crowding suspicious but insufficient for the diagnosis of AEH/EIN can be encountered in all these situations. If there is clinical and/or pathologic concern for neoplasia, it is advisable to report this finding; we render the diagnosis of “endometrium with focal gland crowding,” accompanied by a note recommending follow-up and repeated sampling in 3–6 months. In our experience, focal gland crowding is followed by AEH/EIN in ∼20% of cases.
Common to AEH/EIN and nonatypical hyperplasia is the presence of glandular crowding. AEH/EIN, on one hand, represents a clonal process with potential to progress towards malignancy, whereas nonatypical hyperplasia is a field-effect due to excess estrogen and is not by itself prone to malignant transformation. They have, however, related pathogenesis, and it is possible to find areas of AEH/EIN in a background of nonatypical hyperplasia. The distinction relies on the presence of altered cellular differentiation in the crowded glands that is significantly different from the background endometrium. A helpful clue is the diffuse nature of nonatypical hyperplasia, in contrast to the (usually) focal or multifocal expansile distribution of AEH/EIN. Although PTEN and PAX2 are lost in most AEH/EIN lesions, foci with absent expression can also be encountered in bona fide indolent benign endometria and nonatypical hyperplasia, and the use of these markers for diagnostic purposes is not advocated.
Similar to nonatypical hyperplasia, benign endometrium during the proliferative and secretory phases can mimic AEH/EIN. Attention to the presence of artifacts (e.g., agglutinated individual glands) and awareness of the pattern of normal physiologic changes is helpful in reaching the correct diagnosis.
As explained previously, endometrial polyps can have areas of increased glandular density which can be misdiagnosed as AEH/EIN involving a polyp. As in the nonpolypoid endometrium, comparison between crowded and noncrowded glands within the polyp is imperative. A distinct cytomorphology, particularly in the presence of nuclear enlargement and pseudostratification, is in keeping with AEH/EIN. This exercise may be challenging in curettage specimens, in which it may be difficult to determine whether the fragments involved by gland crowding represent a polyp or not. Comparative analysis of the gland populations within the same tissue fragment suffices in most cases.
Epithelial metaplasia can involve neoplastic and normal endometrium. It is therefore recommended that the spectrum of metaplasia across the endometrial sample is established first, and to only consider appreciable differences in nuclear morphology between crowded and noncrowded glands as diagnostic of AEH/EIN. It is also recommended that assessment of cytologic alteration is performed by comparing with nonmetaplastic areas, as metaplasia will usually be cytologically distinct from the background endometrium and can be confused with AEH/EIN.
Endocervical tissue with reactive changes, particularly florid squamous metaplasia and microglandular hyperplasia may be observed in endometrial samplings. Since these lesions have increased glandular density, they can simulate AEH/EIN. It is important to recognize the endocervical origin of the fragment(s) usually evident by the presence of significant acute and chronic inflammatory cells and a distinct basal layer of reserve cells underneath immature metaplastic squamous and endocervical epithelium ( Fig. 10.6 ).
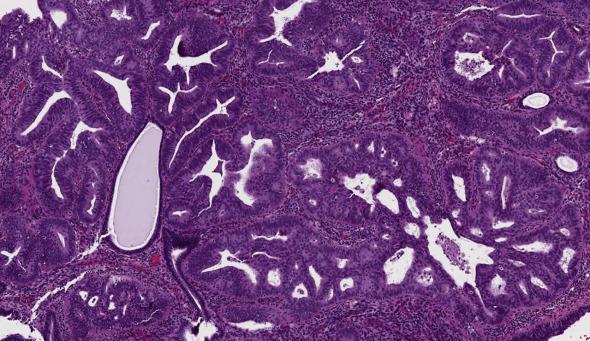
The differential diagnosis of AEH/EIN also includes malignant lesions (endometrial endometrioid adenocarcinoma and endocervical adenocarcinoma involving the endometrium). The distinction between AEH/EIN and well-differentiated endometrial endometrioid carcinoma may be very difficult, especially in endometrial biopsies with scant and/or very fragmented diagnostic material. In the continuum of endometrioid neoplasia, carcinoma is defined by the presence of invasion and/or effacement of the endometrial stroma by the glandular proliferation . This is seen in the form of:
Desmoplastic stromal response in the vicinity of infiltrating glands
Cribriform or microacinar formations that exceed that which is expected for intraglandular growth of AEH/EIN in both size and shape
Extensive papillary and villoglandular architecture with absence of stroma in between
The definition of the minimum amount (size of span, surface area, or number of fragments) of these patterns needed to diagnose low-grade endometrioid adenocarcinoma is not resolved and remains a subjective interpretation by the individual pathologist. However, some authors recommend that the last three findings are required to occupy at least half of a low power field, roughly corresponding to an area >2 mm in maximum dimension. In practice the most useful feature to distinguish AEH/EIN from carcinoma is the absence of stroma between adjacent glands, seen as confluent, back to back, villoglandular, or cribriform glandular growths. Marked cellular atypia, beyond that usually seen in AEH/EIN, is also a useful feature. Additional findings suggestive of carcinoma include necrosis and foamy macrophages. Despite this, uncertainty in this differential persists in some cases, which for reporting purposes can be described as “at least AEH/EIN,” or “AEH/EIN bordering on well-differentiated endometrial endometrioid carcinoma,” or “AEH/EIN, cannot rule out well-differentiated endometrial endometrioid carcinoma” because both conditions will, in general, receive the same treatment ( Fig. 10.7 ).
Lastly, endocervical adenocarcinoma of both human papillomavirus (HPV)-related and HPV-unrelated types can colonize the endometrium and manifest clinically with bleeding, thus simulating AEH/EIN. AEH/EIN will lack the more significant nuclear atypia seen in these cases; HPV-related endocervical adenocarcinoma will show hyperchromasia and abundant apical mitotic figures (floating mitosis) and apoptosis, whereas gastric-type endocervical adenocarcinoma will show abundant pale to eosinophilic mucinous cytoplasm and markedly enlarged nuclei with prominent nucleoli ( Fig. 10.8 ). Immunohistochemistry for ER, PR, and vimentin can be of value, as these markers are negative in endocervical adenocarcinoma and positive in AEH/EIN.
Given its frequent association with concurrent and subsequent endometrial carcinoma, the standard of treatment for AEH/EIN is surgical (simple hysterectomy and bilateral salpingo-oophorectomy). This can be safely recommended to peri- and postmenopausal patients and those who do not want to preserve fertility. As outlined earlier in this chapter, a significant subset of patients with this condition will have endometrial carcinoma on hysterectomy. Most, but not all, will be well differentiated (International Federation of Gynecology and Obstetrics [FIGO] grade 1) and noninvasive.
An increasingly larger number of patients are young and desire to retain their uterus for fertility purposes. In these situations, hormonal treatment with high-dose progestins is a valid temporary alternative. Progestin is administered either orally or via intrauterine device (IUD). Progestin treatment is recommended for at least 6 continuous months, at which point a follow-up biopsy is required; biopsies earlier in the treatment may be indicated if new symptoms or complications occur. Depending on the diagnosis on follow-up sampling, the clinical outcome is classified as: (1) Resolution (benign endometrium), (2) Persistence (AEH/EIN), or (3) Progression (endometrial carcinoma).
Hormonal therapy has variable response: ∼75% show resolution (complete response); however, of these, ∼35% ultimately have AEH/EIN recurrence after treatment cessation. Persistence of AEH/EIN or progression to carcinoma is almost invariable, seen in ∼25% patients within first 3 years, 51% after 3 years, and 72% after 7 years of initial diagnosis. In terms of pregnancy-related outcomes, the live birth rate after progestin treatment is up to 73% for those who actively attempted pregnancy.
Endometrial glandular proliferation characterized by increased glandular density and abnormal cytomorphology that is usually clonal in nature, and harboring malignant potential
Present in 1.2%–1.4% of endometrial biopsies
∼38% risk of concurrent carcinoma
45-fold increased risk of developing carcinoma over time
Typically peri- and postmenopausal
Reproductive-age women less common
Abnormal vaginal bleeding and postmenopausal bleeding most frequent
Abnormal endometrium on ultrasound (thickened or polypoid)
Hysterectomy and bilateral salpingo-oophorectomy are standard of treatment and curative if no associated malignancy
High-dose progestins (oral or levonorgestrel-releasing IUD) if fertility preservation desired
Management includes continuous hormonal treatment for at least 6 months, and follow-up with repeated endometrial sampling
Persistence of AEH/EIN or progression eventually seen in most women (25% in first 3 years, 72% after 7 years)
Most (77%) patients achieve pregnancy after resolution of AEH/EIN with hormonal therapy
May appear polypoid or thickened endometrial lining
Crowded glands (exceeding 50% of the surface area; gland-to-stroma ratio >1)
Glands are round to tubular, or increasingly irregular with branching, stellate, or dilated forms
Stroma, while scant, separates individual glands
Cytomorphology distinct from background (noncrowded) endometrium
Restricted to architecturally crowded area
Typically pseudostratification, nuclear rounding, and enlargement; however features vary greatly
Variations/artifacts of normal cycling endometrium
Nonatypical endometrial hyperplasia
Endometrial polyp
Endometrial metaplasias
Endocervical mucosa with reactive changes (squamous metaplasia, microglandular hyperplasia)
Well-differentiated endometrioid carcinoma
Endocervical adenocarcinoma
Endometrial carcinoma (EC) is the most common malignant tumor of the female genital tract in western countries, accounting for 10–20 per 100,000 cases per year. From the clinical point of view, endometrial carcinoma has been traditionally divided into two main categories: type I and type II. However, this is not an ideal pathologic classification, as there is morphologic overlap between the two types, particularly in the setting of a high-grade tumor. Furthermore, recently the potential importance of incorporating the molecular profile in the classification of endometrial cancer has been shown.
Macroscopic findings are similar in all tumor types. In biopsy and curettage specimens, the tissue may appear normal or quite abundant, depending on tumor volume. In the latter case, tumoral tissue is usually soft, fleshy, and tan with variable hemorrhage and necrosis.
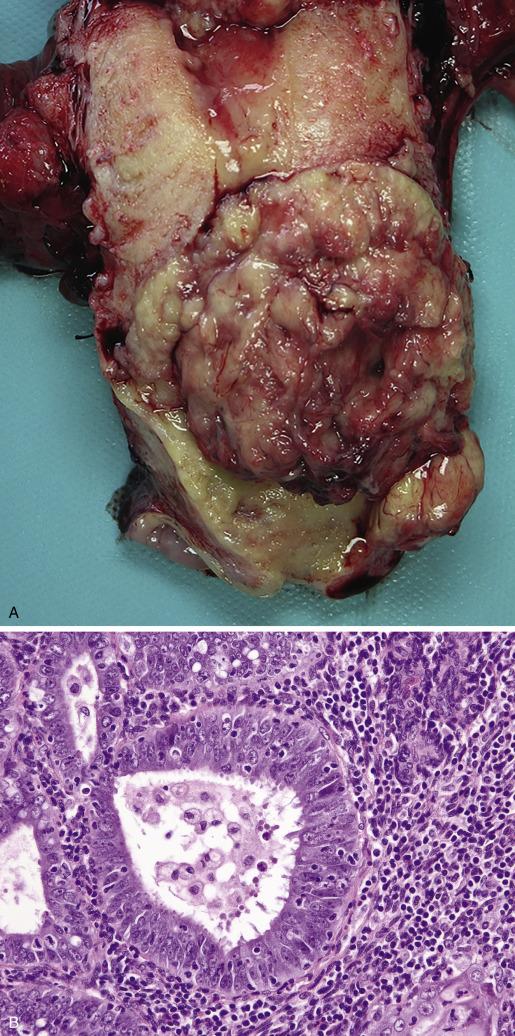
In hysterectomy specimens, the uterus may be normal in size or enlarged (due to massive cavitary growth and/or extensive myometrial invasion). Upon opening the uterus, the tumor may appear as localized polypoid masses (most commonly involving the posterior wall) or completely coating and filling the endometrial cavity ( Fig. 10.9 A–F ). It also may be seen as discrete nodules or variably thickened plaques with a white to pale tan and soft cut surface. Tumor may not be grossly evident or may only involve an endometrial polyp (a finding more commonly seen in serous carcinoma). In younger women, the lower uterine segment is more frequently involved. Rarely, carcinomas may arise in the cornual region or within intramural adenomyotic foci; in these situations, proper visualization of the tumor and proper sectioning in relationship to the uterine wall thickness may be difficult. Adenomyosis involvement by the tumor appears as islands of fleshy and soft tan tissue dispersed throughout the wall. In contrast, myometrial invasion often appears as a firm confluent mass extending into the wall with a pushing or infiltrative border. There is, however, macroscopic overlap between adenomyosis and myoinvasive carcinoma, and in some instances, invasion may not even be evident on gross examination. Extension to the cervix may be seen with the naked eye and should be noted as it affects stage and prognosis. If complicated by cervical stenosis, endometrial carcinoma may be associated with pyometra or hematometra. Carcinomas with exuberant squamous differentiation may appear flaky, whereas mucinous carcinomas may display a gelatinous or mucoid appearance. Necrosis and hemorrhage are usually seen in the context of high-grade or large, deeply myoinvasive tumors.
Specimen sectioning and processing is critical for diagnostic and management purposes. Whenever possible, sections of the tumor and underlying uterine wall should be full-thickness (from endometrial cavity to uterine serosa); if the uterine wall is too thick to fit in a single block, a composite section should be provided. The deepest point of gross penetration into the wall should always be sampled as a full-thickness section. In addition, adjacent normal-appearing endometrium (if present) should be included. If the tumor grossly involves the cervix or is close to it, a composite section from the distal aspect of the tumor to the ectocervix is required.
Several factors have been implicated in endometrial carcinoma prognosis including:
The FIGO staging system is the strongest prognostic parameter in patients with endometrial carcinoma. The current American Joint Committee on Cancer (AJCC) (2017) and FIGO (2018) are harmonized so each TNM category has a matching FIGO stage ( Box 10.2 ). Patients’ 5-year disease-free survival is 90% for stage I, 83% for stage II, and 43% for stage III tumors.
I (T1): Tumor confined to the uterus
IA (T1a): Tumor confined to the endometrium or invades less than 50% of the myometrial wall
IB (T1b): Tumor invades more than 50% of the myometrial wall
II (T2): Tumor infiltrates cervical stroma
III (T3, N1): Tumor locally/regionally extends outside the uterus or to lymph nodes
IIIa (T3a): Tumor involves serosa and/or adnexa (direct extension or metastases)
IIIB (T3b): Tumor involves vagina or parametria (direct extension or metastases)
IIIC (N1): Tumor with pelvic and/or paraaortic lymph node metastasis
IIIC (N1 mi): Micrometastases (>0.2 mm or 200 cells, but ≤2 mm)
IIIC (N1a): Macrometastases (>2 mm in size)
IV (T4, M1): Tumor invades bladder or bowel mucosa, and/or metastasizes to distant sites
IVa (T4): Tumor invades bladder mucosa and/or bowel mucosa.
IVb (M1): Distant metastases (excluding metastases to vagina, pelvic serosa, or adnexa)
Pathologic staging requires careful macroscopic and microscopic assessment of the tumor extent. Challenging situations in the staging exercise are discussed in the following paragraphs.
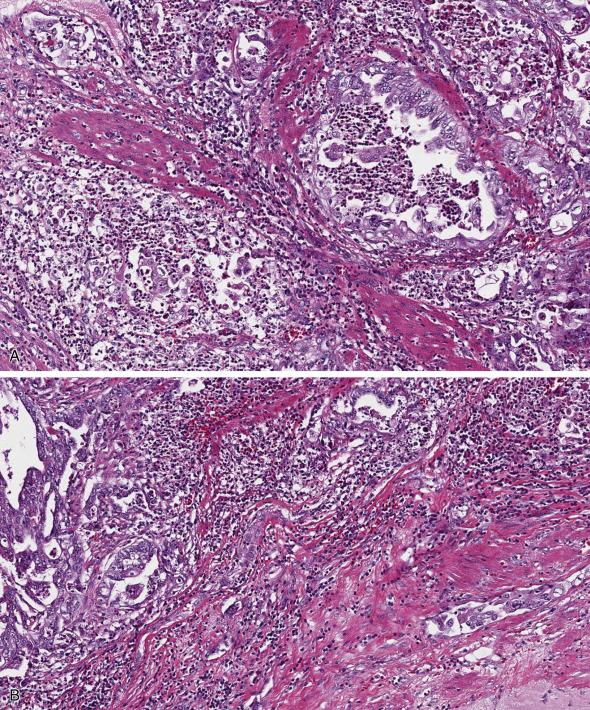
Myometrial invasion is an independent predictor of outcome in low-stage endometrial carcinomas as it correlates with risk of extrauterine extension and metastases to pelvic and paraaortic lymph nodes. The current FIGO staging system subdivides stage I endometrial carcinoma into two categories: 1) tumor limited to the endometrium, or invading less than half of myometrial wall; and 2) invasion of equal to or more than half of myometrial wall without involvement of the uterine serosa. Assessment of myometrial invasion is straightforward if the tumor has an infiltrative border composed of irregular glands associated with desmoplastic stroma and/or an inflammatory response. However, there is a myriad of situations that can complicate this assessment. For instance, myometrial infiltration can be subtle and easily missed on low power examination. That can be the case in tumors with a microcystic, elongated, and fragmented (MELF) pattern of invasion which is typically associated with prominent fibromyxoid stroma, histiocytes, and neutrophils. The glands are completely or partially lined by attenuated epithelium (resembling vascular spaces), display fragmentation, and microcystic dilation, and contain luminal neutrophilic infiltrates ( Fig. 10.10A,B ). The associated inflammatory response may be exuberant and mask the neoplastic cells, thus potentially underestimating myometrial invasion. In addition, lymphovascular involvement is commonly associated with this pattern of invasion and may be subtle as tumor cells often have a histiocytic appearance.
Another not uncommon pitfall is overdiagnosing invasion in polypoid tumors in which smooth muscle bundles are “pulled” into the bulk of the mass simulating myoinvasion. Evaluation of the endomyometrial junction in adjacent sections and evaluating the gross appearance of the tumor may be helpful.
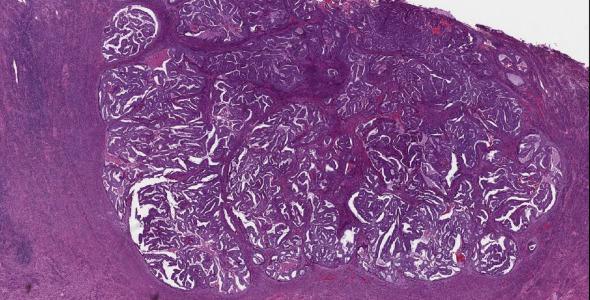
Tumors with an expansile (pushing) invasive front, which does not elicit desmoplasia and simulates a normal endomyometrial junction, may appear noninvasive ( Fig. 10.11 ). Comparison of these areas to the uterine wall thickness and the endomyometrial interface in uninvolved parts of the uterus is quite helpful; expansile tumors will extend beyond the expected confines and will lack preexisting nonneoplastic glands at the base.
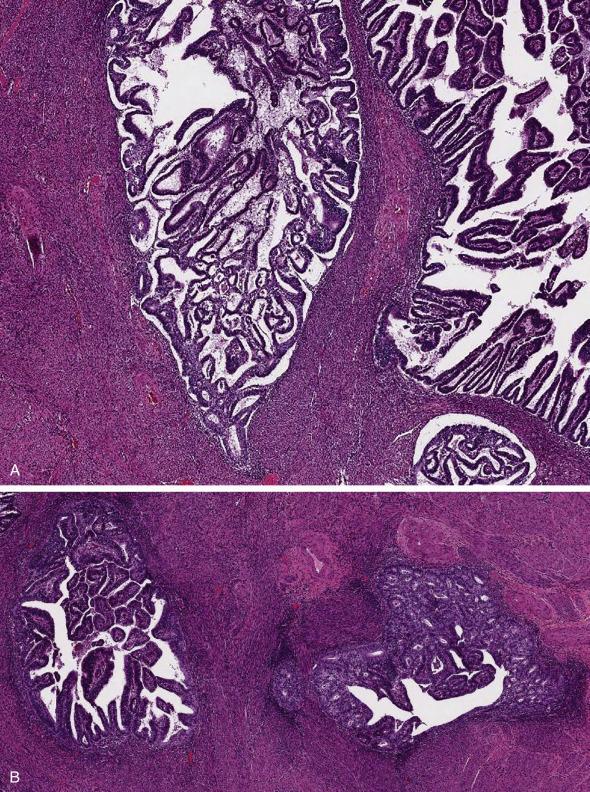
In approximately 25% of cases, carcinoma involves foci of adenomyosis. Regardless of the depth within the wall, extension into adenomyosis does not carry an adverse prognosis. It is, therefore, important to distinguish this scenario from a myoinvasive carcinoma ( Fig. 10.12A,B ). Useful features in this distinction include (1) smooth rounded contours of the myometrial tumor deposits, (2) absence of desmoplasia or an inflammatory response, (3) finding of associated nonneoplastic endometrial glands or endometrial stroma, and (4) uninvolved adenomyosis with a similar distribution throughout the wall as the neoplastic foci. It is important to keep in mind that CD10 (a marker of endometrial stroma) is not helpful in this distinction since cells around foci of myoinvasion frequently show aberrant CD10 expression; lack of expression, however, favors myoinvasion. If myometrial invasion occurs from carcinoma within adenomyosis, the deepest myoinvasive point should be reported according to where this is located in the myometrium, and regardless of whether or not it arises from adenomyosis.
Finally, it is important to be aware that rare endometrioid carcinomas diffusely infiltrate the myometrium in a deceptive manner with minimal or no associated stromal response. These tumors are typically low grade and have been designated “adenoma malignum”; as seen in minimal deviation carcinomas of the endocervix, these lesions are identified by virtue of their haphazard and deep distribution in the wall ( Fig. 10.13 ).
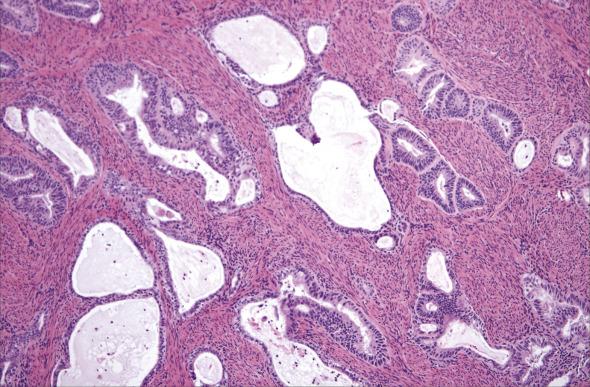
Invasion into the cervical stroma corresponds to a FIGO stage II; it may occur either by direct extension or secondary to lymphatic spread. The diagnosis of cervical stromal invasion can be difficult, which is reflected by its poor reproducibility among gynecologic pathologists. Importantly, there is no statistical difference in survival for patients with Stage II disease when compared to patients with Stage I tumors matched for other prognostic factors (e.g., deep myometrial invasion, high histologic grade). Nonetheless, the pathologist is expected to determine the presence and extent of cervical involvement for radiation purposes.
It is sometimes difficult to identify the boundary between the lower uterine segment and the upper endocervix. The presence of endocervical-type mucinous glands and an inked or cauterized outer edge (indicating absence of serosa) are helpful anatomic landmarks of the uterine cervix. If a tumor is seen in close apposition or surrounded by endocervical mucinous glands, it should be interpreted as involving the cervix ( Fig. 10.14 ). Another complicating issue is the implantation of endometrial carcinoma in the cervical mucosa as a consequence of fractional dilatation and curettage, which occurs infrequently (∼5%). In this situation, invasion can be excluded if the tumor foci are superficial and surrounded by hemorrhage, reactive fibrosis, inflammation and/or giant cell reaction. Lastly, the distinction between endocervical gland involvement versus cervical stromal invasion may be challenging, as the gland contour may be irregular, and a desmoplastic response is not always seen with invasion imparting a “burrowing” or pseudo–mesonephric hyperplasia appearance. In the absence of unequivocal signs of invasion (desmoplasia, angulated glands, tumor cell clusters, and single cells), attention to the overall endocervical gland distribution is usually helpful as invasive carcinoma exceeds that expected for glandular involvement in terms of glandular density and spacing.
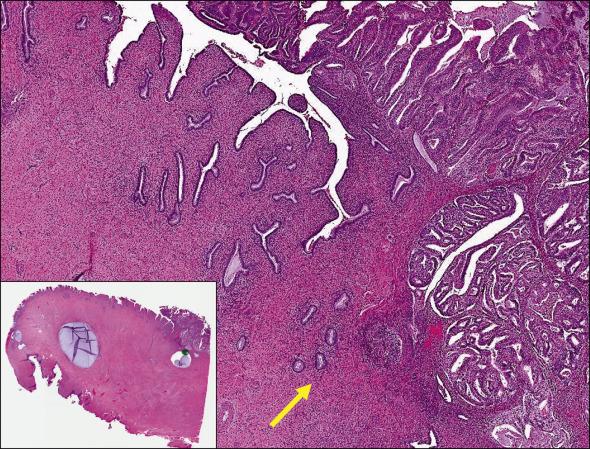
When present, the depth of invasion into the cervical wall should be estimated and reported for radiation planning purposes, ideally as a percentage of the cervical wall or as separate measurements of the invasive tumor depth and the cervical wall.
A synchronous ovarian carcinoma is present in ∼5% of women undergoing surgery for endometrial carcinoma (up to 30% in reproductive-age women). In most cases, the ovarian and endometrial neoplasms are of endometrioid type. The outcome of patients with simultaneous endometrial and ovarian endometrioid carcinoma is similar to that of those with endometrial-only or ovarian-only carcinoma, leading to the belief that they are independent primary tumors occurring synchronously. It may be difficult to distinguish between synchronous ovarian and endometrial primaries (both stage I) and endometrial carcinoma metastatic to the ovary (by definition stage III), but several clinicopathologic features can be used in this distinction. The following would favor an endometrial carcinoma with secondary ovarian spread: large uterine tumor size with deep myometrial invasion and lymphovascular space invasion, bilateral ovarian involvement, surface ovarian involvement, similar morphologic appearance and grade of both tumors, and fallopian tube involvement. Conversely, features in keeping with synchronous primaries include: endometrial tumor confined to the endometrium or with superficial invasion only, absence of lymphovascular space invasion, unilateral and large ovarian tumor, association with ovarian endometriosis and different morphologies, and tumor grade between endometrial and ovarian tumors. Immunohistochemistry for mismatch repair (MMR) protein may help in the scenario with endometrioid carcinomas (different MMR expression profiles would be in keeping with synchronicity), and WT1 in the scenario of serous carcinomas (WT1-negative endometrial and WT1-positive ovarian, supporting synchronicity). Using a DNA sequencing approach, it has been recently demonstrated that in most cases the endometrial and the ovarian neoplasms are clonally related, even if by standard criteria they were classified as synchronous. Thus, most of these instances seem to represent metastatic ovarian involvement by a slow-progressing and otherwise indolent endometrial carcinoma. However, the previously mentioned criteria to distinguish between “independent” and “metastatic” tumors should be used, regardless of clonality results, because they have shown to be relevant to assess prognosis.
Lymphatic vascular invasion (LVI) is an independent predictor of tumor recurrence and outcome and is associated with increased risk of lymph node metastases. It is common in endometrioid carcinomas with MELF invasion, and in high-grade types, particularly serous carcinoma. The prognostic value of LVI is related to its extent, as it was demonstrated in a study derived from the PORTEC trial, which showed that substantial LVI is a strong and independent prognostic factor for pelvic regional recurrence, distant metastasis, and overall survival, in contrast to focal or no LVI. The distinction between focal and substantial LVI is not well defined and represents a spectrum, making application in daily clinical practice difficult; however, LVI is substantial in the setting of multiple (>1) intravascular foci in separate tissue sections or within the same section but widespread. Some authors recommend that LVI should be reported semiquantified as “focal” (one or two foci) or “substantial/extensive” (three or more).
Lymphovascular space invasion is often seen along the infiltrative border of the tumor as cohesive clusters of tumor cells with smooth borders conforming to the shape of a lymphovascular space ( Fig. 10.15A ). There is often a change in the quality of the cytoplasm which may become either more eosinophilic or vacuolated, mimicking histiocytes. A lymphocytic infiltrate is often seen at the periphery of the involved vessel ( Fig. 10.15B ). Occasionally, endometrial carcinomas confined to the inner half of the myometrium are associated with tumor emboli in vessels in the outer half of the myometrium, or within vascular channels in the cervix, parametria, or adnexa. However, these findings should not result in upstaging the tumor.
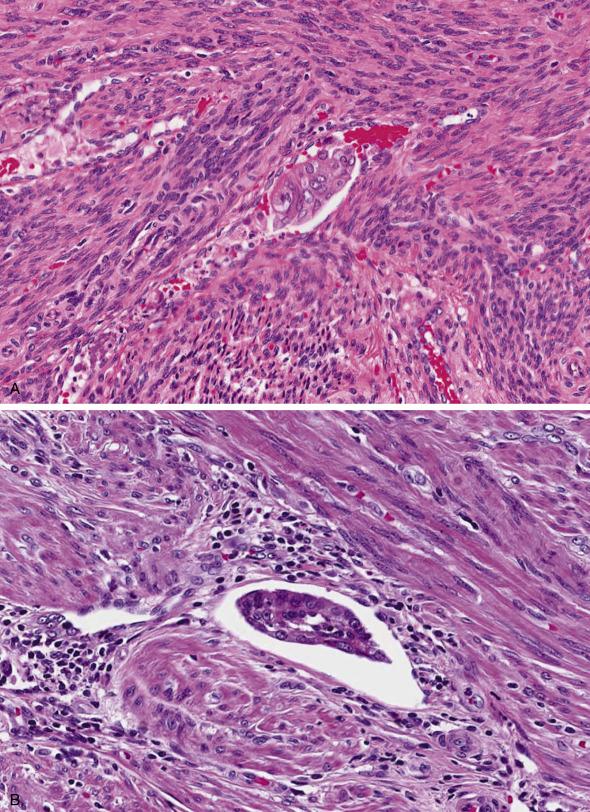
Importantly, laparoscopic robot-assisted hysterectomies use an intrauterine manipulator which can dislodge tumor fragments and artificially introduce them into vascular spaces of the myometrium, adnexa, and cervix. Artifact is favored if intravascular tumor cells are disaggregated and associated with inflammatory debris, if they differ in shape and size from the harboring vessel, and appear “intact” (sometimes with gland lumen formation) ( Fig. 10.16A ). In the setting of artificial displacement, nontumoral endometrium sometimes can also be found inside vessels.
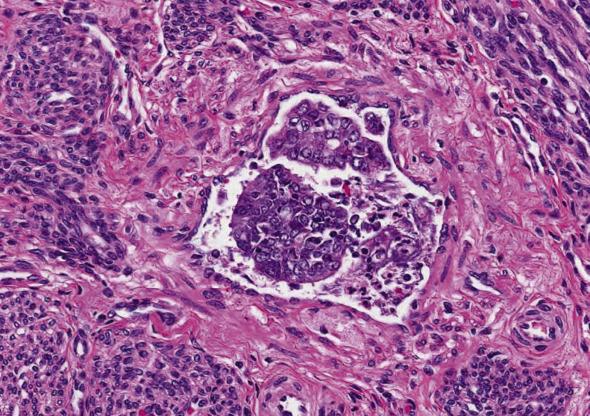
The frequency of lymph node metastases in clinical stage I patients is related to the depth of myometrial invasion and the tumor grade. One-third of patients with metastatic pelvic lymph nodes also have positive paraaortic lymph nodes. Prognosis of patients with metastatic tumor to pelvic lymph nodes is better than that of patients with positive paraaortic lymph nodes.
Lymph node metastases in the context of gynecologic cancer are currently divided by size using the same criteria established for breast cancer: macrometastases (contiguous tumor deposit >2 mm), micrometastases (>0.2 but ≤2 mm, or >200 scattered individual tumor cells) and isolated tumor cells (ITCs, contiguous tumor deposit ≤0.2 mm or ≤200 scattered individual tumor cells). ITCs are usually detected by immunohistochemistry (performed in some institutions as part of sentinel lymph node protocols). A few studies have shown that patients with only ITCs or micrometastases in sentinel lymph nodes are associated with excellent disease-free survival comparable to patients without nodal disease. Conversely, patients with macrometastases have significantly worse recurrence-free survival. Although further evidence is required, it is recommended to routinely report the size of nodal metastatic disease and categorize them as macrometastasis, micrometastasis, or ITCs. Although there is not strong scientific evidence-based data, it is recommended to report the presence of extracapsular lymph node spread.
Positive peritoneal cytology has been associated with high histologic grade, deep myometrial invasion, and extrauterine spread. This feature has been eliminated from the most recent FIGO staging.
Histologic type has been recognized as an important prognostic factor in endometrial carcinoma ( Box 10.3 ). Low-grade endometrioid and mucinous adenocarcinoma are associated with a favorable prognosis. In contrast, serous, clear cell, high-grade endometrioid, and undifferentiated carcinoma are associated with an unfavorable outcome, with overall 5-year survival rates ranging from 30% to 70%. The presence of signet-ring cells, trophoblastic or hepatoid differentiation is considered to impart a worse prognosis. Tumors confined to an endometrial polyp, including high-grade carcinomas such as serous or neuroendocrine carcinoma, may be associated with an overall good outcome.
Endometrioid carcinoma a.Squamous differentiation
Villoglandular
Secretory
Mucinous adenocarcinoma
Serous carcinoma
Clear cell carcinoma
Neuroendocrine tumors a.Low-grade neuroendocrine tumor (carcinoid)
High-grade neuroendocrine carcinoma
b.1. Small cell neuroendocrine carcinoma
b.2. Large cell neuroendocrine carcinoma
Mixed adenocarcinoma
Undifferentiated and Dedifferentiated carcinoma
The grade of endometrial carcinomas has close correlation with the tumor type, as serous, clear cell, undifferentiated, and dedifferentiated carcinomas and carcinosarcoma are all, by definition, high-grade. Endometrioid and mucinous carcinomas, on the other hand, are divided into low- and high-grade categories. Separation of endometrial carcinomas according to grade has important diagnostic and prognostic implications. From the diagnostic point of view, low-grade endometrioid and mucinous carcinomas (FIGO grade 1 and 2) are usually straightforward and can be easily distinguished from other endometrial carcinoma types. Conversely, high-grade endometrioid carcinomas (FIGO grade 3) may show significant morphologic overlap with other high-grade types. Indeed, the interobserver reproducibility of histotype assignation in high-grade endometrial carcinoma is poor. Immunohistochemistry helps, but only to a certain degree. As a result, a subset of endometrial carcinomas requires categorization as “high-grade endometrial carcinoma with ambiguous features.” A molecular-based classification is an emerging and promising alternative to histologic type, particularly in the subset of high-grade tumors.
The Cancer Genome Atlas Research Network (TCGA) comprehensively characterized the genomic profile of endometrioid and serous carcinomas, and categorized them in four groups of tumors with different prognoses:
Ultramutated carcinomas harboring mutations in the exonuclease domain of the DNA polymerase ɛ ( POLE ) gene. Most tumors in this category are endometrioid. Ultramutated carcinomas account for 6% of low-grade and 17% of high-grade endometrioid endometrial carcinomas. Despite their high-grade morphology, patients with these lesions had an exceptionally good prognosis with conventional therapy (which depended on grade, stage, and other parameters).
Hypermutated carcinomas with defective MMR protein function and microsatellite instability (MSI). They exhibit MLH1 promoter methylation and reduced MLH1 gene expression, or somatic or germline mutations in any of the MMR genes ( MLH1, MSH2, MSH6, PMS2 ). These tumors are typically endometrioid. This group accounts for 29% of low-grade and 54% of high-grade endometrioid endometrial carcinomas. They have an intermediate survival.
Carcinomas with low mutation rates and low frequency DNA copy-number abnormalities ( copy-number low ). Almost all (92%) of cancers in this group have somatically altered PI3K pathway. Conversely, they lack TP53 alterations. They comprise 60% of low-grade, 9% of high-grade endometrioid carcinomas, 2% of serous carcinomas, and 25% of mixed histology carcinomas. They have an intermediate survival.
Carcinomas with low mutation rates and high frequency DNA copy-number abnormalities ( copy-number high ). These lesions are characterized by somatic TP53 mutations and consequent abnormal p53 expression. They account for >95% of serous carcinomas and 75% of mixed carcinomas. Carcinomas in this group are aggressive and have the worst survival outcomes.
Although initially conceived in a cohort of endometrioid, serous, and mixed carcinomas, the TCGA classification has also been applied successfully to endometrial clear cell carcinoma. The incorporation of this molecular classification into routine practice is gaining momentum thanks to the validation of its prognostic value and the development of the Proactive Molecular Risk Classifier for Endometrial Cancer (ProMisE), which uses (mostly) routine tools as a surrogate of the genomic profile. In this algorithm, carcinoma is classified based on POLE mutational testing and immunohistochemistry for p53 and MMR proteins. Tumors with loss of expression of any MMR marker (MLH1, MSH2, MSH6 or PMS2) are classified as MMR-deficient (surrogate for the hypermutated group); tumors with pathogenic POLE mutations are assigned to the ultramutated group; and tumors with abnormal p53 expression (overexpression [strong nuclear staining in ≥80% of tumor cells] or complete absence of staining) are assigned to the copy-number high phenotype. Finally, tumors without MMR, p53 and POLE abnormalities are allocated to the copy-number low (p53 normal) group.
Molecular classification seems to be particularly valuable in high-grade endometrioid carcinomas, since this is a highly heterogeneous group of tumors with variable prognosis.
Regarding molecular classification, there are some unresolved issues. For example, POLE -mutated and some microsatellite unstable tumors show also strong p53 immunostaining and p53 mutation (“double positive”). These cases raise the differential diagnosis with serous-like/high copy-number endometrial carcinoma. p53 immunostaining is used to identify the serous-like or copy-number high group. However, correlation between p53 immunostaining and p53 mutation is not perfect.
We envision that such categorization will become routine in the pathology work-up of endometrial carcinoma, at least as a complement to the standard prognostic pathologic variables. It remains to be determined whether POLE -mutated tumors can be managed conservatively with surgery and no systemic therapy, and the need to identify better tools to stratify patients in the intermediate categories (MMR-deficient and copy-number low).
In this regard, in the subset of low-grade early-stage endometrial carcinoma, mutations of CTNNB1 (beta-catenin) gene have been associated with worse survival. Nuclear expression of beta-catenin by immunohistochemistry has 100% specificity and 85% sensitivity in detecting CTNNB1 mutations. Nonetheless, and unlike mutational status, nuclear beta-catenin expression has not shown statistical association with patient outcome and further investigation is needed.
Surgery (hysterectomy with bilateral salpingo-oophorectomy) is the mainstay of treatment of women with endometrial carcinoma. The surgical approach may include peritoneal cytologic sampling and abdominal exploration and biopsy of any suspicious lymph nodes or lesions. Omentectomy may be performed for serous carcinomas. Pelvic and paraaortic lymphadenectomy is appropriate when there is obvious involvement by tumor or known high-risk prognostic factors (high-grade, nonendometrioid morphology, deep myometrial invasion, or extension to the cervix or adnexa). The role of sentinel lymph node biopsy in endometrial carcinoma is still a matter of debate; however, it is practiced in many centers as a valid alternative to regional lymphadenectomy. The success of this technique depends on the surgeon’s experience and the adherence to an algorithm for sentinel node detection. Special processing protocols for sentinel lymph nodes, including “ultrastaging,” allow the identification of very small volume metastases (isolated tumor cells and micrometastases), the significance of which is still a matter of investigation.
Low-grade and early stage (FIGO stage IA) patients can be placed on close surveillance, with surgical or radiation treatment of local (vaginal) recurrences, which is the most common complication. Adjuvant radiation therapy will be recommended in patients with high-risk prognostic indicators (high-grade, nonendometrioid morphology, deep myometrial invasion, or extension to the cervix or adnexa) and consists of vaginal vault brachytherapy or external beam radiotherapy. For women with disseminated disease, systemic therapy with progestational hormones or cytotoxic chemotherapy can be considered.
A special consideration in the treatment of early stage and low-grade (FIGO grade 1 and selected FIGO grade 2 patients) endometrioid carcinoma is the alternative of primary hormonal therapy as a method to temporarily halt or slow tumor progression to achieve fertility goals. The risks of hormonal treatment in endometrial cancer include the possibility of an under-sampled high-grade endometrioid carcinoma or nonendometrioid carcinoma, advanced stage subclinically (10%–30% of cases are stages III–IV at presentation), and the potential for a subclinical synchronous ovarian carcinoma (seen in 5%–30% of premenopausal women). The diagnostic considerations presented in the section of AEH/EIN also apply to this setting: if possible, the original biopsy should be reviewed in conjunction with any follow-up material, and it is critical to know the time lapse since the diagnosis and the duration of the hormonal treatment. Depending on the diagnosis on follow-up sampling, the clinical outcome is classified as: (1) Resolution (benign endometrium, nonatypical hyperplasia), (2) Regression (AEH/EIN), (3) Persistence (endometrioid carcinoma FIGO grade 1), or (4) Progression (endometrioid carcinoma FIGO grade 2 or high-grade carcinoma).
Lynch syndrome (known also as hereditary nonpolyposis colorectal cancer) is an autosomal dominant hereditary condition that predisposes patients to develop cancers in various organs including colon (most frequent), endometrium (second most frequent), ovary, and upper gastrointestinal tract among others. Women with Lynch syndrome have a risk of developing endometrial carcinoma ranging from 40%–70%; importantly, endometrial neoplasia is often the first manifestation of the syndrome (“sentinel” event); thus, diagnosis of Lynch syndrome in the work-up of endometrial carcinoma can have important clinical implications for these patients, as they will be placed in close surveillance. Certain clinical and pathologic features have been associated with Lynch syndrome-related endometrial cancer including ( Fig. 10.17A–C ): (1) young women with a low body mass index, (2) localization within the lower uterine segment, (3) dense peritumoral and tumor infiltrating lymphocytes (>40/high power field), (4) intratumoral heterogeneity (e.g., dedifferentiated carcinoma), (5) endometrioid high-grade carcinomas in patients <50 years old, and (6) synchronous clear cell and/or endometrioid carcinomas of the ovary. Any of these features should raise suspicion for Lynch syndrome. Nonetheless, these are not always present, and it is now known that selecting endometrial cancer patients for Lynch syndrome testing based on age or other criteria will invariably miss a significant number of cases. For instance, testing women 50 years or younger will miss ∼10% of women with Lynch syndrome-related endometrial cancer. For this reason, universal testing of women with endometrial carcinoma has grown support in the literature.
Lynch syndrome testing usually starts with tumor tissue-based analysis, namely immunohistochemistry for MMR proteins and/or microsatellite instability. The former is, in most practices, the test of choice given its feasibility and high sensitivity. Testing includes the four major proteins of the MMR complex: MLH1, PMS2, MSH2, and MSH6 ( Fig. 10.17D ). Loss of MSH2/MSH6, MSH2 only, or PMS2 only is highly suggestive of Lynch syndrome. Conversely, loss of MLH1/PMS2 is more likely to be secondary to hypermethylation of the MLH1 promoter, which is a sporadic event, thus hypermethylation testing should be performed in order to exclude this possibility. A two-marker approach has been proposed as more cost-effective, with initial testing for MSH6 and PMS2, and subsequently for MLH1 and MSH2 if abnormal.
If tissue-based testing is abnormal (loss of expression of MMR proteins, or high MSI, and absence of MLH-1 promoter hypermethylation), the patient should be referred to genetic counseling and offered germline testing. Lynch syndrome is diagnosed in one of the following scenarios: (1) germ-line mutation of one of the DNA MMR genes ( MLH1, PMS2, MSH2, MSH6 ), (2) germ-line EPCAM mutation, or (3) epimutation (abnormal transcriptional repression of active genes and/or abnormal activation of usually repressed genes caused by errors in epigenetic gene repression). In patients with confirmed Lynch syndrome, prophylactic hysterectomy and salpingo-oophorectomy is performed to prevent development of endometrial carcinoma. In these instances, submission of the entire endometrium including corpus and lower uterine segment, tubes and ovaries is indicated.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here