Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Dysmorphology is the study of differences in human form and the mechanisms that cause them. It has been estimated that 1 in 40 newborns, or 2.5%, have a recognizable birth defect or pattern of malformations at birth; approximately half these newborns have a single, isolated malformation, whereas in the other half, multiple malformations are present. From 20–30% of infant deaths and 30–50% of deaths after the neonatal period are caused by congenital abnormalities ( http://www.marchofdimes.com/peristats/ ). In 2001, birth defects accounted for 1 in 5 infant deaths in the United States, with a rate of 137.6 deaths per 100,000 live births, which was higher than other causes of mortality, such as preterm/low birthweight (109.5/100,000), sudden infant death syndrome (55.5/100,000), maternal complications of pregnancy (37.3/100,000), and respiratory distress syndrome (25.3/100,000).
Birth defects can be subdivided into isolated (single) defects or multiple congenital anomalies (multiple defects) in one individual. An isolated primary defect can be classified, according to the nature of the presumed cause of the defect, as a malformation, dysplasia, deformation, or disruption ( Table 128.1 and Fig. 128.1 ). Most birth defects are malformations. A malformation is a structural defect arising from a localized error in morphogenesis that results in the abnormal formation of a tissue or organ. Dysplasia refers to the abnormal organization of cells into tissues. Malformations and dysplasias can both affect intrinsic structure. In contrast, a deformation is an alteration in shape or structure of a structure or organ that has developed, or differentiated, normally. A disruption is a defect resulting from the destruction of a structure that had formed normally before the insult.
| TERMINOLOGY | DEFINITION | EXAMPLE |
|---|---|---|
| Sequence | Single error in morphogenesis that results in a series of subsequent defects | Pierre-Robin sequence, in which a small jaw results in glossoptosis and cleft palate DiGeorge sequence of primary 4th brachial arch and 3rd and 4th pharyngeal pouch defects, leading to aplasia or hypoplasia of the thymus and parathyroid glands, aortic arch anomalies, and micrognathia |
| Deformation sequence | Mechanical (uterine) force that alters structure of intrinsically normal tissue | Oligohydramnios produces deformations by in utero compression of limbs (e.g., dislocated hips, equinovarus foot deformity), crumpled ears, or small thorax |
| Disruption sequence | In utero tissue destruction after a period of normal morphogenesis | Amnionic membrane rupture sequence, leading to amputation of fingers/toes, tissue fibrosis, and tissue bands |
| Dysplasia sequence | Atypical organization of cells into tissues or organs | Neurocutaneous melanosis sequence, with atypical migration of melanocyte precursor cells from the neural crest to the periphery, manifesting as melanocytic hamartomas of skin and meninges |
| Malformation syndrome | Appearance of multiple malformations in unrelated tissues that have a known, unifying cause | Trisomy 21 Teratogens Numerous multiple congenital anomaly syndromes as described above |
Most inherited human disorders with altered morphogenesis display multiple malformations rather than isolated birth defects. When several malformations coexist in a single individual, they can be classified as a syndrome, sequence, or an association. A syndrome is defined as a pattern of multiple abnormalities that are related by pathophysiology, resulting from a single, defined etiology. Sequences consist of multiple malformations that are caused by a single event, although the sequence itself can have different etiologies. An association refers to a nonrandom grouping of malformations in which there is an unclear, or unknown, relationship among the malformations, such that they do not fit the criteria for a syndrome or sequence.
Human malformations and dysplasias can be caused by gene mutations, chromosome aberrations and copy number variants, environmental factors, or interactions between genetic and environmental factors ( Table 128.2 ). Some malformations are caused by deleterious sequence variants in single genes, whereas other malformations arise because of deleterious sequence variants in multiple genes acting in combination ( digenic or oligogenic inheritance). In 1996 it was thought that malformations were caused by monogenic defects in 7.5% of patients; chromosomal anomalies in 6%; multigenic defects in 20%; and known environmental factors, such as maternal diseases, infections, and teratogens, in 6–7% ( Table 128.3 ). In the remaining 60–70% of patients, malformations were classified as caused by unknown etiologies. Currently, the percentages have increased for all categories of known causes of malformations, the result of improved cytogenetic and molecular genetic methods for detecting small chromosomal abnormalities and next-generation sequencing studies that can screen multiple genes simultaneously and identify novel genes and deleterious sequence variants.
| DISORDER | CAUSE/INHERITANCE | SELECTED CLINICAL FEATURES | PATHOGENESIS |
|---|---|---|---|
| Spondylocostal dysostosis syndrome | Mendelian; autosomal recessive | Abnormal vertebral and rib segmentation | Deleterious sequence variants in DLL3 and other genes |
| Rubinstein-Taybi syndrome | Autosomal dominant | Intellectual disability Broad thumbs and halluces; valgus deviation of these digits Hypoplastic maxillae Prominent nose and columella Congenital heart disease |
Deleterious sequence variants in CBP and EP300 |
| X-linked lissencephaly | X-linked | Male: severe intellectual disability, seizures Female: variable |
Deleterious sequence variants in DCX |
| Aniridia | Autosomal dominant | Absent iris or iris/foveal hypoplasia | Deleterious sequence variants in PAX6 |
| Waardenburg syndrome, type I | Autosomal dominant | Deafness White forelock Wide-spaced eyes Iris heterochromia and/or pale skin pigmentation |
Deleterious sequence variants in PAX3 |
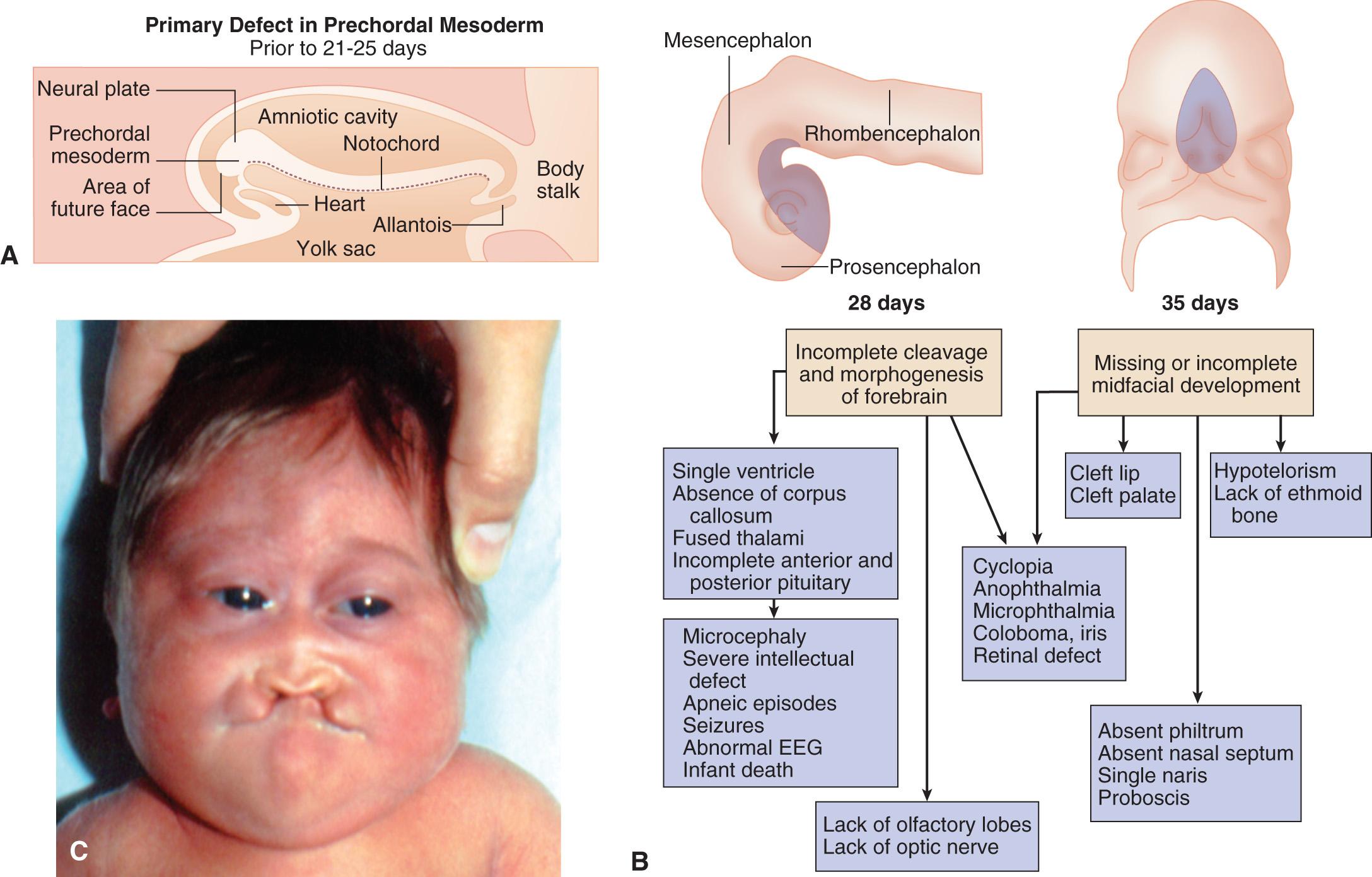
| Holoprosencephaly | Loss of function or heterozygosity for multiple genes | Microcephaly | SHH, multiple other genes |
| Cyclopia | |||
| Single central incisor | |||
| Velocardiofacial syndrome | Microdeletion 22q11.2 |
Congenital heart disease, including conotruncal defects | TBX1 haploinsufficiency/mutations; haploinsufficiency for other genes in the deleted interval also contributes to the phenotype. |
| Cleft palate | |||
| T-cell defects | |||
| Facial anomalies | |||
| Down syndrome | Additional copy of chromosome 21 (trisomy 21) | Intellectual disability | Increase in dosage of an estimated 250 genes on chromosome 21 |
| Characteristic dysmorphic features | |||
| Congenital heart disease | |||
| Increased risk of leukemia | |||
| Alzheimer disease | |||
| Neural tube defects | Multifactorial | Meningomyelocele | Defects in folate sensitive enzymes or folic acid uptake |
| Fetal alcohol syndrome | Teratogenic | Microcephaly | Ethanol toxicity to developing brain |
| Developmental delay | |||
| Facial abnormalities | |||
| Behavioral abnormalities | |||
| Retinoic acid embryopathy | Teratogenic | Microtia | Isotretinoin effects on neural crest and branchial arch development |
| Congenital heart disease |
| MONOGENIC |
|
| CHROMOSOMAL ABERRATIONS and COPY NUMBER VARIANTS |
|
| MATERNAL INFECTION |
|
| MATERNAL ILLNESS |
|
| UTERINE ENVIRONMENT |
|
| ENVIRONMENTAL AGENTS |
|
| MEDICATIONS |
|
| UNKNOWN ETIOLOGIES |
|
| SPORADIC SEQUENCE COMPLEXES |
|
| NUTRITIONAL |
|
Many developmental abnormalities that are caused by deleterious sequence variants (mutations)in a single gene display characteristic, mendelian patterns of inheritance (autosomal dominant, autosomal recessive, and X-linked inheritance). Genes that cause birth defects or multiple congenital anomaly syndromes are often transcription factors, part of evolutionarily conserved signal transduction pathways, or regulatory proteins required for key developmental events ( Figs. 128.2 and 128.3 ). Examples include spondylocostal dysostosis syndromes, Smith-Lemli-Opitz syndrome, Rubinstein-Taybi syndrome, and X-linked lissencephaly (“smooth brain”) syndrome (see Table 128.2 ).
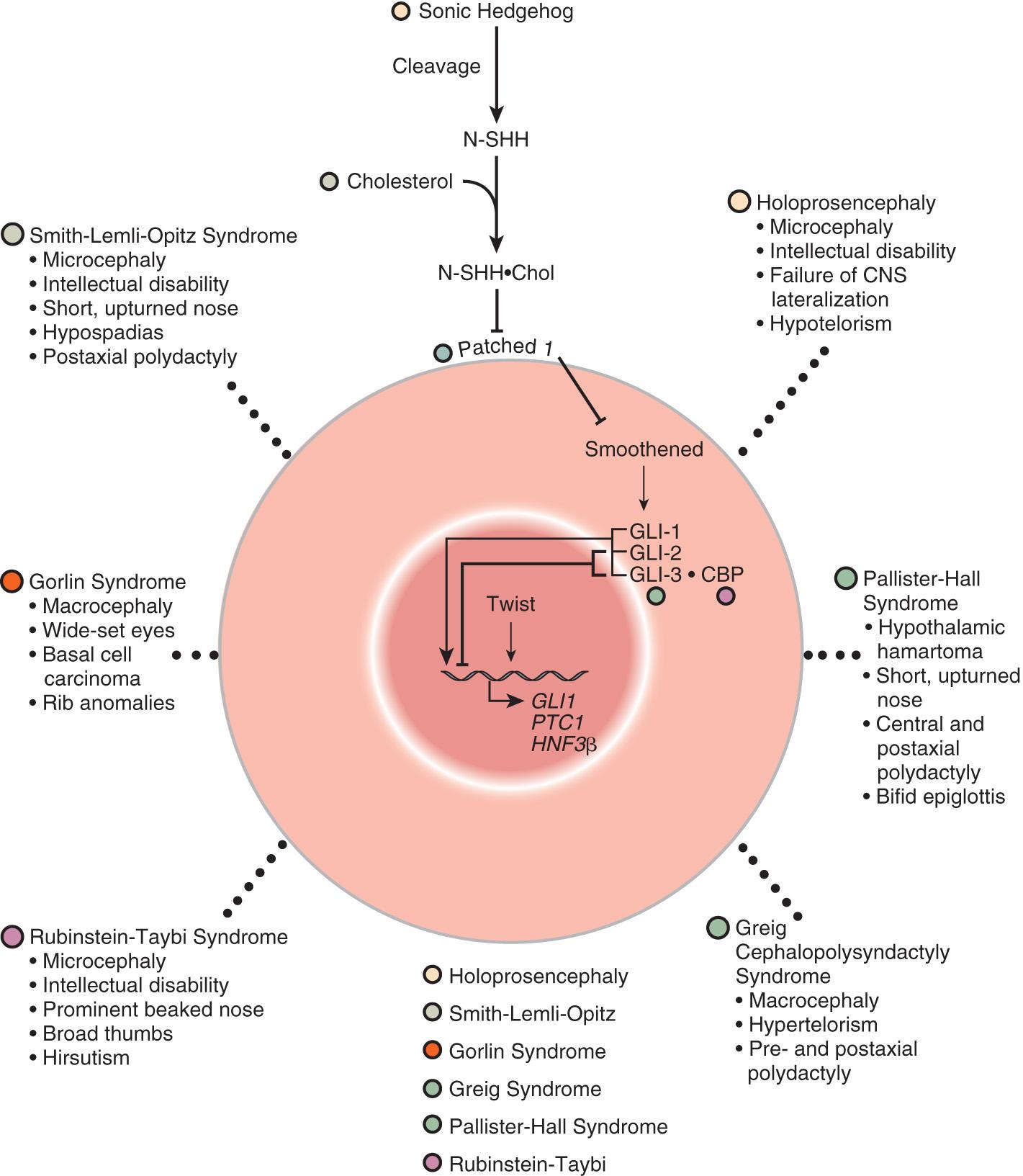
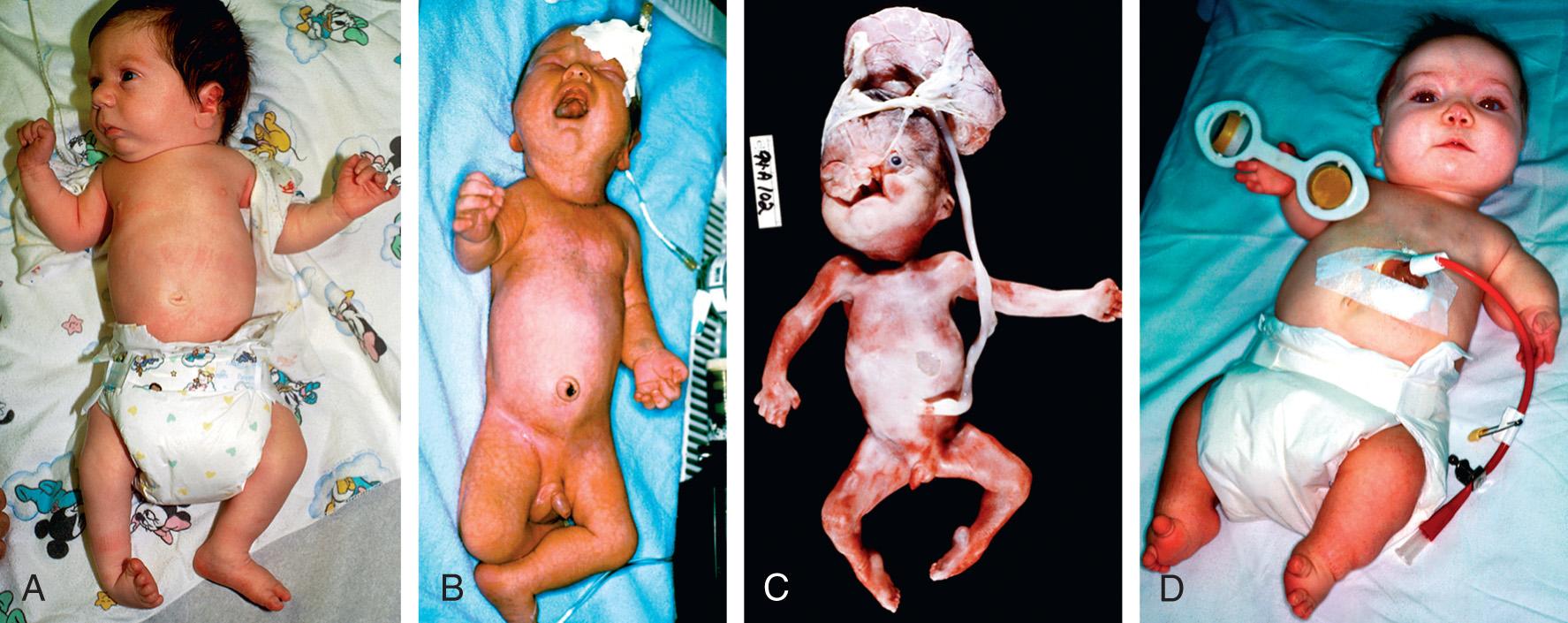
Patients with spondylocostal dysostosis (SCD) display a characteristic pattern of vertebral segmentation defects associated with a number of other malformations, such as neural tube defects. The SCD syndromes are etiologically heterogeneous and are often caused by mutations in the gene coding for delta-like 3 ( DLL3 ), a ligand of the Notch receptors. The Notch/delta pathway is conserved throughout evolution and regulates a number of developmental events. Smith-Lemli-Opitz syndrome (SLOS) results from mutations in the sterol delta-7-dehydrocholesterol reductase ( DHCR7 ) gene, an enzyme critical for normal cholesterol biosynthesis. Patients with SLOS (see Fig. 128.2 ) display syndactyly (fusion of the fingers and toes), in particular affecting the 2nd and 3rd toes; postaxial polydactyly (extra digits); anteverted (upturned) nose; ptosis; cryptorchidism; and holoprosencephaly (failure of separation of the 2 cerebral hemispheres). Many of the features in SLOS are shared with those arising from deleterious sequence variants in the SHH genes, and these mutations link cholesterol biosynthesis pathogenically to the sonic hedgehog (SHH) pathway, because SHH is posttranslationally modified by cholesterol (see Chapter 97 ). Rubinstein-Taybi syndrome (see Fig. 128.2 ) typically results from heterozygous, loss-of-function deleterious sequence variants in a gene coding for a broadly acting transcriptional coactivator called CREB-binding protein (CBP) and from deleterious sequence variants in the EP300 gene. The CBP coactivator regulates the transcription of a number of genes, which is why patients with deleterious sequence variants in CBP have a pleiotropic phenotype that includes developmental delays and intellectual disability, broad and angulated thumbs and halluces (1st toes), and congenital heart disease. One of the transcription factors that binds to CBP is GLI3, a member of the SHH pathway (see Fig. 128.2 ). X-linked lissencephaly is a severe neuronal migration defect that causes a smooth brain with reduction or absence of gyri and sulci in males and that gives rise to a variable pattern of intellectual disability and seizures in females. X-linked lissencephaly is caused by deleterious sequence variants in DCX . The DCX protein regulates the activity of dynein motors that contribute to movement of the cell nucleus during neuronal migration.
Malformation syndromes can also be caused by chromosomal aberrations or copy number variants and teratogens (see Tables 128.2 and 128.3 ). Down syndrome typically results from an extra copy of an entire chromosome 21 or, less frequently, an extra copy of the Down syndrome critical region on chromosome 21. Chromosome 21 is a small chromosome that contains an estimated 250 genes, and thus individuals with Down syndrome typically have an increased dosage of the numerous genes encoded by this chromosome that causes their physical differences (see Chapter 98.2 ).
Neural tube defects (NTDs) are an example of a birth defect that displays multifactorial inheritance in most cases. NTDs and a number of other congenital malformations, such as cleft lip and palate, can recur in families, but inheritance for the majority of affected individuals does not occur in a straightforward, mendelian inheritance pattern, and in this situation, multiple genes and environmental factors together likely contribute to the pathogenesis (see Table 128.2 ). Many of the genes involved in NTDs are unknown, so one cannot predict with certainty the mode of inheritance or a precise recurrence risk in the individual case. Empirical recurrence risks can be provided on the basis of population studies and the presence of single or multiple family members with the same malformation. However, one important gene/environment interaction has been identified for NTDs (see Chapter 609.1 ). Folic acid deficiency is associated with NTDs and can result from a combination of dietary factors and increased utilization during pregnancy. A common variant in the gene for an enzyme in the folate recycling pathway, 5,10-methylene-tetrahydrofolate reductase ( MTHFR ), that makes this enzyme less stable, may also be important in folic acid status. Several teratogenic causes of birth defects have been described (see Tables 128.2 and 128.3 ). Ethanol causes a recognizable malformation syndrome that is variably called fetal alcohol syndrome (FAS), fetal alcohol spectrum disorder (FASD), or fetal alcohol effects (FAE) (see Chapter 126.3 ). Children who were exposed to ethanol during the pregnancy can display microcephaly, developmental delays, hyperactivity, and facial dysmorphic features. Ethanol, which is toxic to the developing central nervous system (CNS), causes cell death in developing neurons.
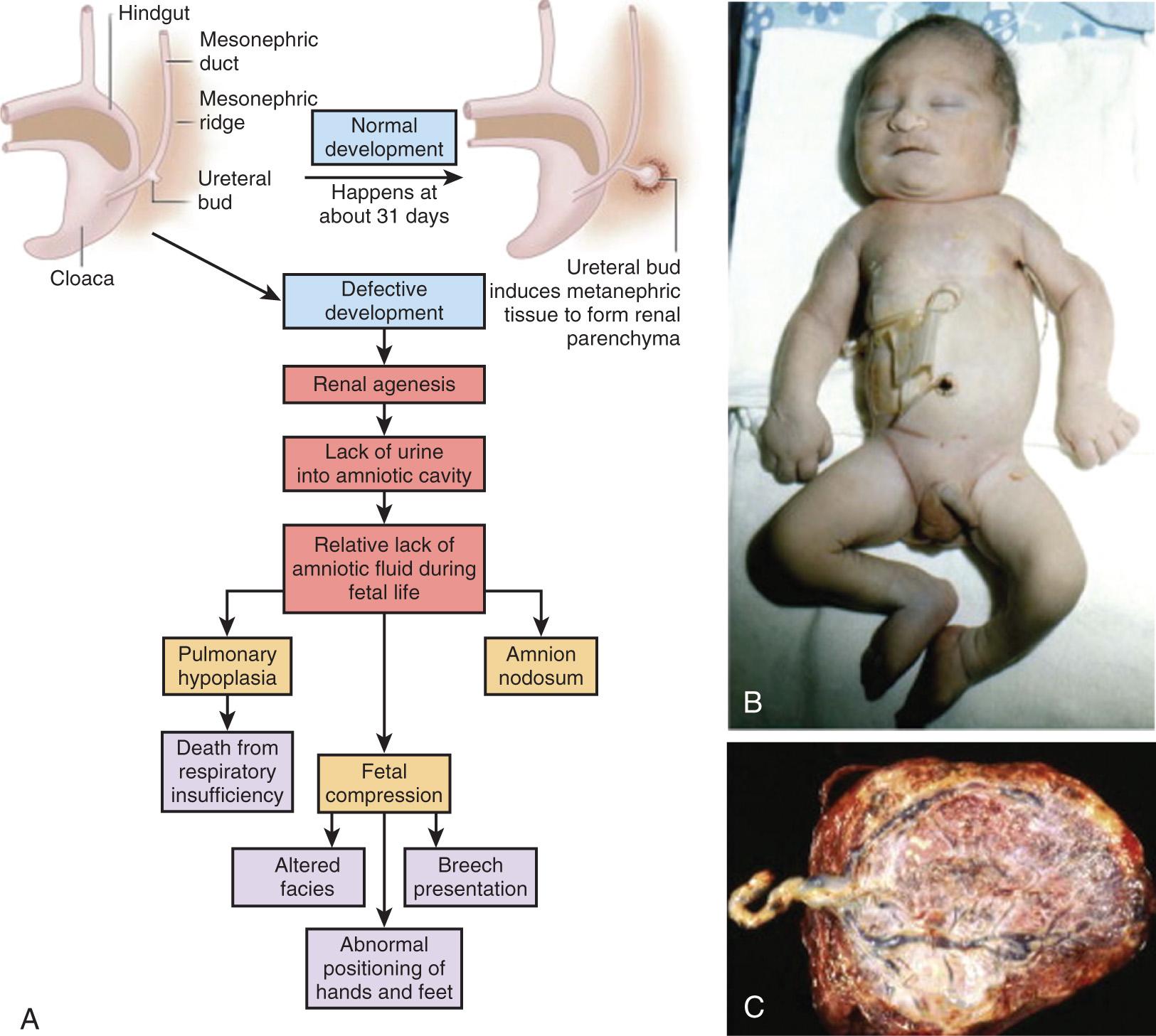
Many deformations involve the musculoskeletal system ( Fig. 128.4 ). Fetal movement is required for the proper development of the musculoskeletal system, and restriction of fetal movement can cause musculoskeletal deformations such as clubfoot (talipes). Two major intrinsic causes of deformations are primary neuromuscular disorders and oligohydramnios, or decreased amniotic fluid, which can be caused by fetal renal defects. The major extrinsic causes of deformation are those that result in fetal crowding and restriction of fetal movement. Examples of extrinsic causes include oligohydramnios resulting from chronic leakage of amniotic fluid and abnormal shape of the amniotic cavity. When a fetus is in the breech position ( Fig. 128.5 ), the incidence of deformations is increased 10-fold. The shape of the amniotic cavity also has a profound effect on the shape of the fetus and is influenced by many factors, including uterine shape, volume of amniotic fluid, and the size and shape of the fetus ( Fig. 128.6 ).
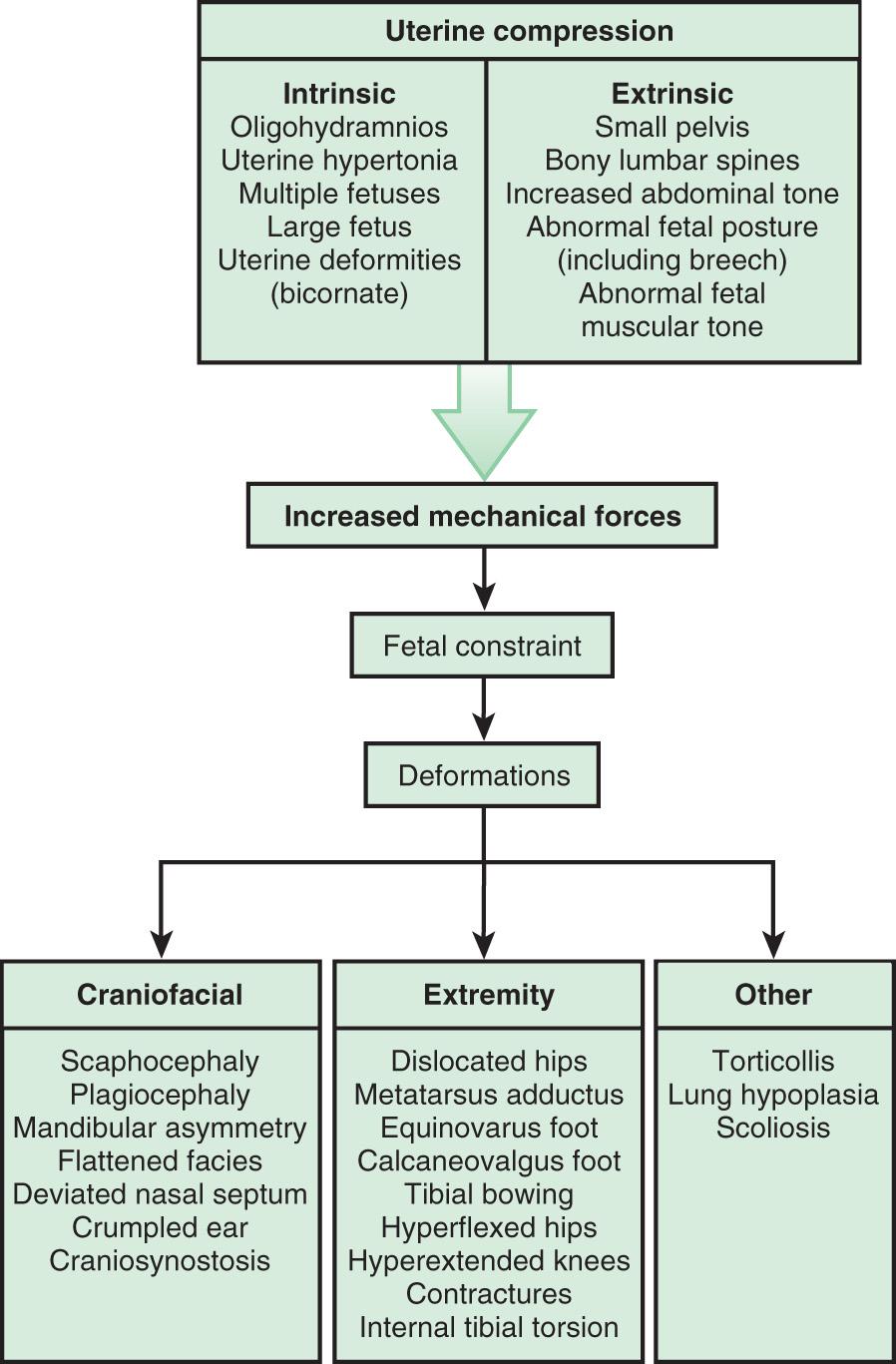
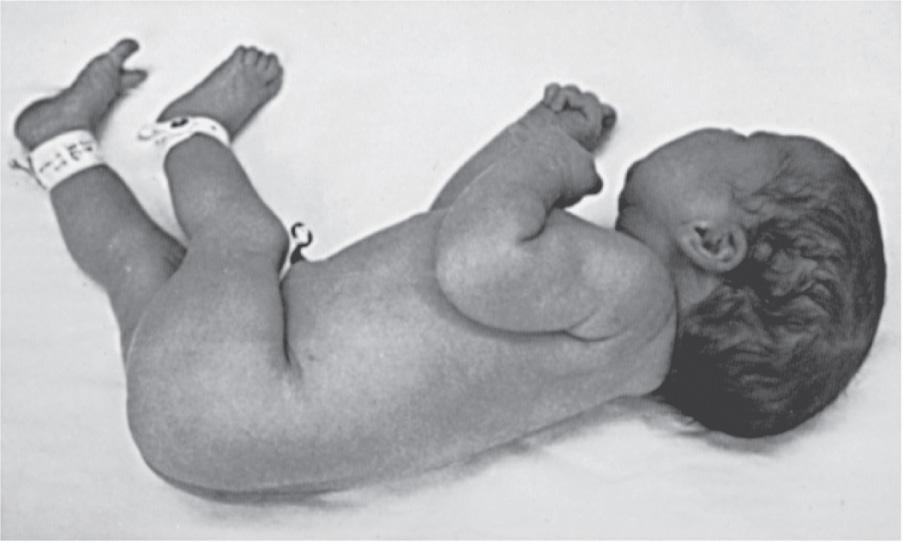
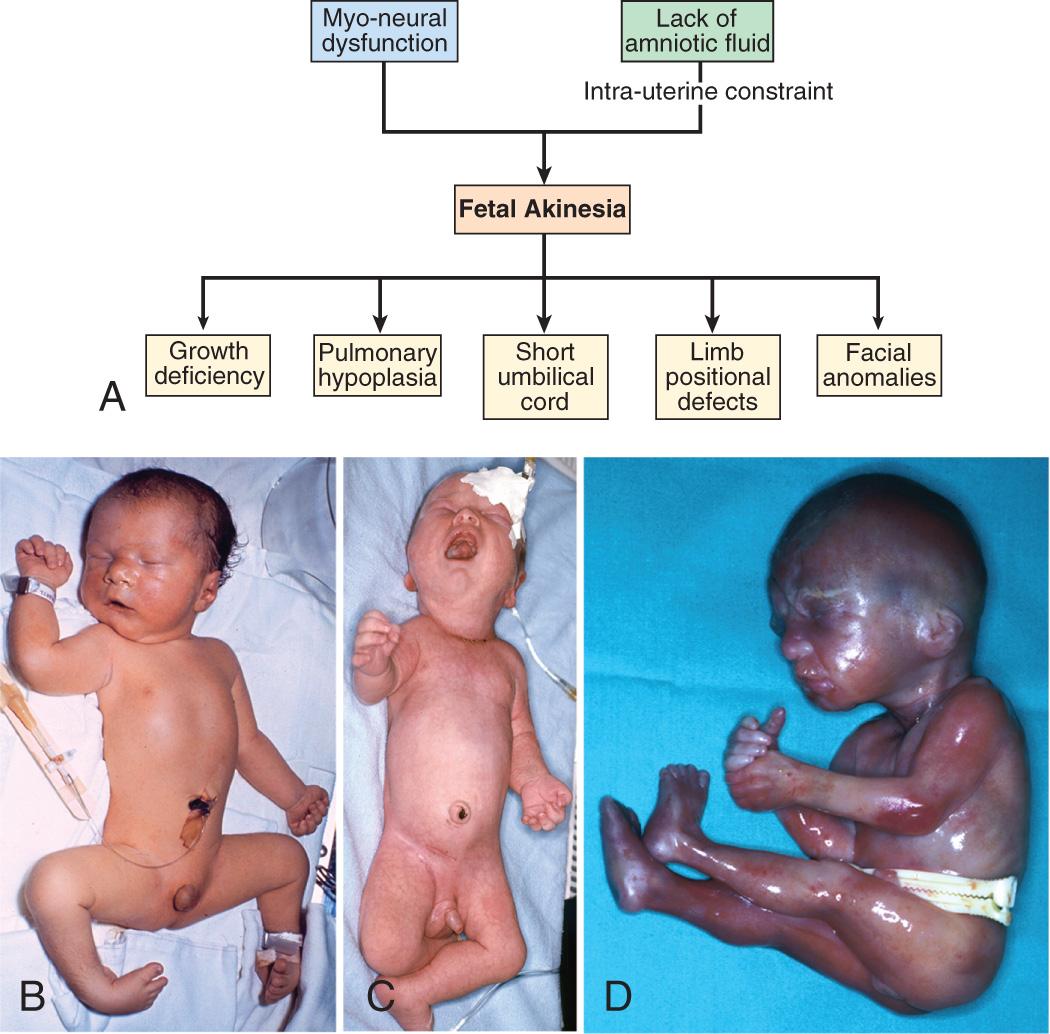
It is important to determine whether deformations result from intrinsic or extrinsic causes. Most children with deformations from extrinsic causes are otherwise completely normal, and their prognosis is usually excellent. Correction typically occurs spontaneously. Deformations caused by intrinsic factors, such as multiple joint contractures resulting from CNS or peripheral nervous system defects, have a different prognosis and may be much more significant for the child ( Fig. 128.7 ).
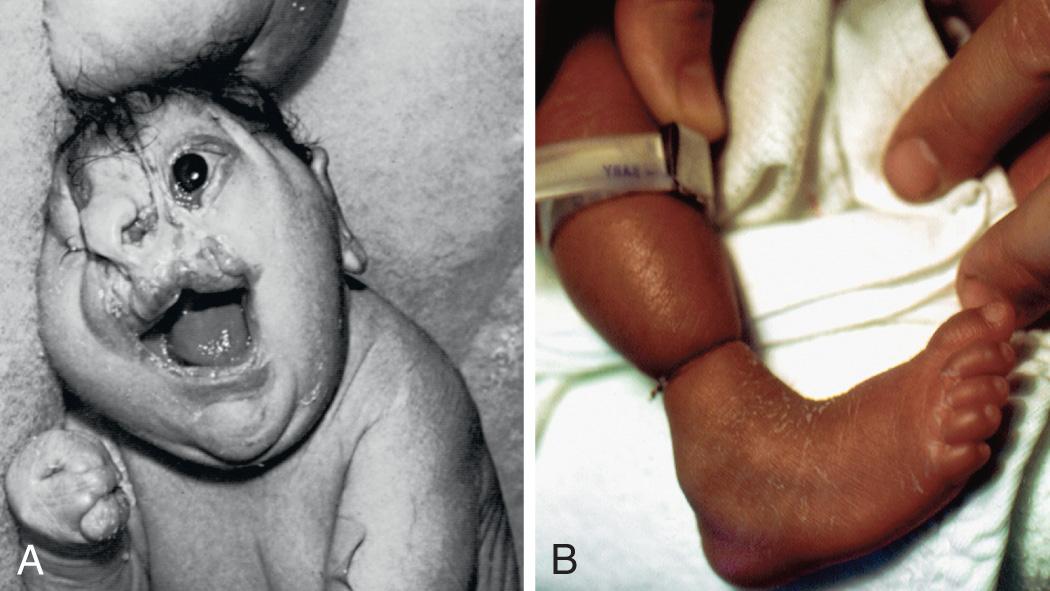
Disruptions are caused by destruction of a previously normally formed organ or body part. At least 2 mechanisms are known to produce disruptions. One involves entanglement, followed by tearing apart or amputation, of a normally developed structure, usually a digit or limb, by strands of amnion floating within amniotic fluid (amniotic bands) ( Fig. 128.8 ). The other mechanism involves interruption to the blood supply to a developing part, which can lead to infarction, necrosis, and resorption of structures distal to the insult. If interruption to the blood supply occurs early in gestation, the disruptive defect typically involves atresia, or absence of a body part. Genetic factors were previously considered to play a minor role in the pathogenesis of disruptions; most occur as sporadic events in otherwise healthy individuals. The prognosis for a disruptive defect is determined entirely by the extent and location of the tissue loss.
The pattern of multiple anomalies that occurs when a single primary defect in early development produces multiple abnormalities because of a cascade of secondary and tertiary developmental anomalies is called a sequence (see Fig. 128.9 ). When evaluating a child with multiple congenital anomalies, the physician must differentiate between multiple anomalies that are caused by a single localized error in morphogenesis (a sequence) from syndromes with multiple malformations. In the former, recurrence risk counseling for the multiple anomalies depends entirely on the risk of recurrence for the single, localized malformation. Pierre-Robin sequence is a pattern of multiple anomalies produced by mandibular hypoplasia. Because the tongue is relatively large for the oral cavity, it drops back (glossoptosis), blocking closure of the posterior palatal shelves and causing a U -shaped cleft palate. There are numerous causes of mandibular hypoplasia, all of which can result in characteristic features of Pierre-Robin sequence.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here