Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The components of the hematopoietic system have been traditionally divided into the myeloid tissues, which include the bone marrow and the cells derived from it (e.g., red cells, platelets, granulocytes, and monocytes), and the lymphoid tissues, consisting of the thymus, lymph nodes, and spleen. It is important to recognize, however, that this subdivision is artificial with respect to both the normal physiology of hematopoietic cells and the diseases affecting them. For example, although bone marrow contains relatively few lymphocytes, it is the source of all lymphoid progenitors and the home of long-lived plasma cells and memory lymphocytes. Similarly, neoplastic disorders of myeloid progenitor cells (myeloid leukemias) originate in the bone marrow but secondarily involve the spleen and (to a lesser degree) the lymph nodes. Some red cell disorders (e.g., immunohemolytic anemia, discussed in Chapter 14 ) result from the formation of autoantibodies, indicating a primary disorder of lymphocytes. Thus it is not possible to draw neat lines between diseases involving the myeloid and lymphoid tissues.
Recognizing this difficulty, we somewhat arbitrarily divide diseases of the hematopoietic tissues into two chapters. In this chapter we discuss white cell diseases and disorders affecting the spleen and thymus. In Chapter 14 we consider diseases of red cells and those affecting hemostasis. Before delving into specific diseases, we will briefly discuss the origins of hematopoietic cells, since many disorders of white cells and red cells involve disturbances of their normal development and maturation.
Blood cell progenitors first appear during the third week of embryonic development in the yolk sac. Cells derived from the yolk sac are the source of long-lived tissue macrophages such as microglial cells in the brain and Kupffer cells in the liver ( Chapter 3 ), but the contribution of the yolk sac to blood formation, mainly in the form of embryonic red cells, is only transient. Definitive hematopoietic stem cells (HSCs) arise several weeks later in the mesoderm of the intraembryonic aorta/gonad/mesonephros region. During the third month of embryogenesis, HSCs migrate to the liver, which becomes the chief site of blood cell formation until shortly before birth. HSCs also take up residence in the fetal placenta; this pool of HSCs is of uncertain physiologic relevance but has substantial clinical importance, as HSCs harvested at birth from umbilical cord blood are used in therapeutic HSC transplantation. By the fourth month of development, HSCs shift in location yet again to the bone marrow. By birth, marrow throughout the skeleton is hematopoietically active, and hepatic hematopoiesis dwindles to a trickle, persisting only in scattered foci that become inactive soon after birth. After puberty, hematopoiesis ceases in distal bones and becomes restricted to the axial skeleton. Thus in normal adults, only about half of the marrow space is hematopoietically active.
The formed elements of blood—red cells, granulocytes, monocytes, platelets, and lymphocytes—have a common origin from HSCs, pluripotent cells that sit at the apex of a hierarchy of bone marrow progenitors ( Fig. 13.1 ). Most evidence supporting this scheme comes from studies in mice, but human hematopoiesis is believed to proceed in a similar way. The development of mature blood cells from HSCs involves progressive commitment to increasingly specialized cell populations. HSCs give rise to several kinds of early progenitor cells that have more restricted differentiation potential, such that they ultimately produce mainly myeloid cells or lymphoid cells. The origins of lymphoid cells are revisited when tumors derived from these cells are discussed. These early progenitors in turn give “birth” to progenitors that are further constrained in their differentiation potential. Some of these cells are referred to as colony-forming units (see Fig. 13.1 ) because they produce colonies composed of specific kinds of mature cells when grown in culture. From the various committed progenitors are derived the morphologically recognizable precursors, such as myeloblasts, proerythroblasts, and megakaryoblasts, which are the immediate progenitors of mature granulocytes, red cells, and platelets.
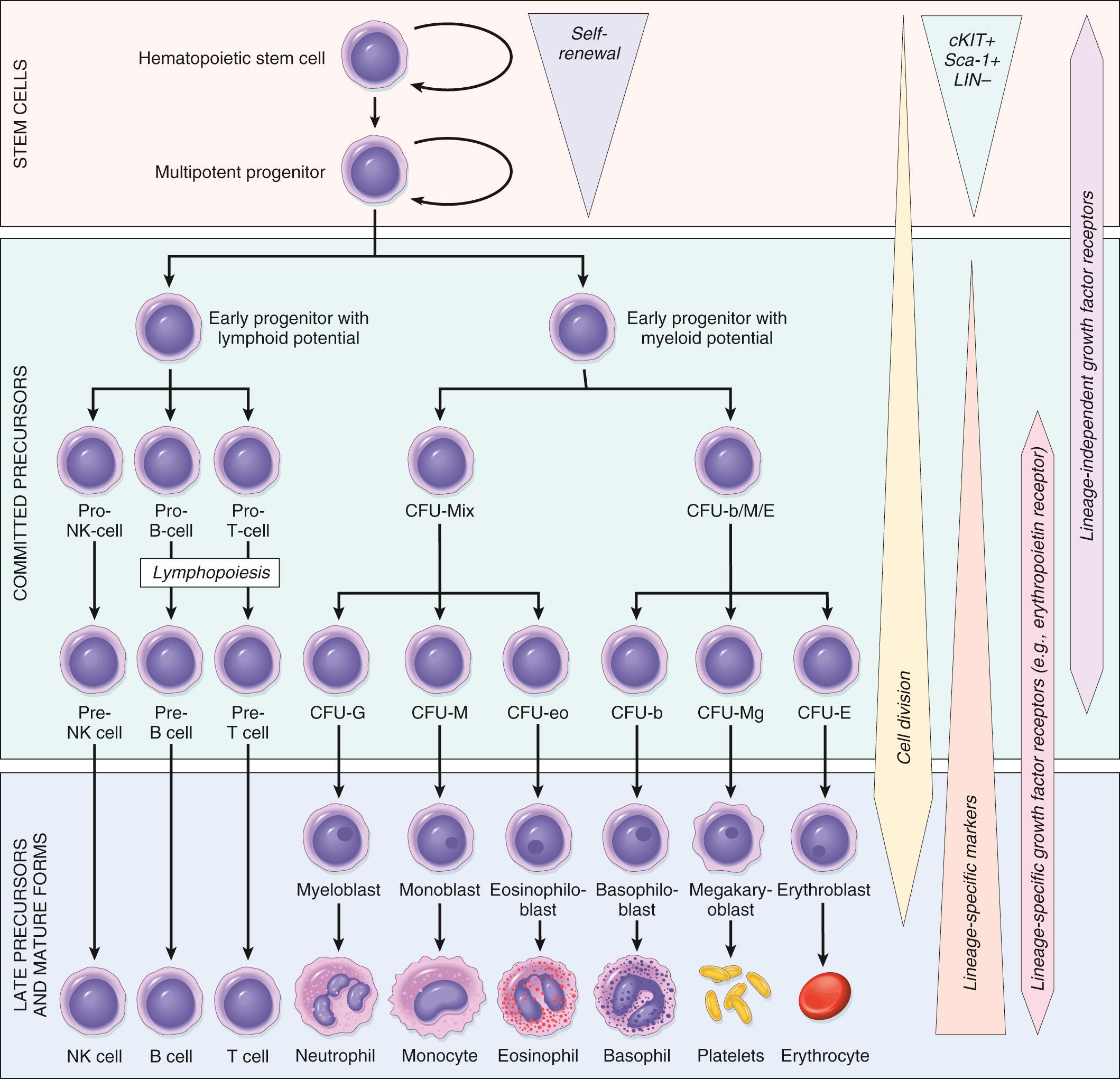
HSCs have two essential properties that are required for the maintenance of hematopoiesis: pluripotency and the capacity for self-renewal. Pluripotency refers to the ability of a single HSC to generate all mature blood cells. When an HSC divides, at least one daughter cell must self-renew to avoid stem cell depletion. Self-renewing divisions occur within a specialized marrow niche, in which stromal cells and secreted factors nurture and protect the HSCs. As one might surmise from their ability to migrate during embryonic development, HSCs are not sessile. Particularly under conditions of stress, such as severe anemia or acute inflammation, HSCs are mobilized from the bone marrow and appear in the peripheral blood. In fact, HSCs used in transplantation are now mainly collected from the peripheral blood of donors treated with granulocyte colony-stimulating factor (G-CSF), one of the factors that can mobilize marrow HSCs from their stem cell niches.
The marrow response to short-term physiologic needs is regulated by hematopoietic growth factors through their effects on the committed progenitors. These growth factors are called colony-stimulating factors (CSFs) because they were discovered by their ability to support the growth of colonies of blood cells in vitro. Because mature blood elements are terminally differentiated cells with finite lifespans, their numbers must be constantly replenished. In current models of hematopoiesis, some divisions of HSCs give rise to cells referred to as multipotent progenitors, which are more proliferative than HSCs but have a lesser capacity for self-renewal (see Fig. 13.1 ). Division of multipotent progenitors gives rise to at least one daughter cell that leaves the stem cell pool and begins to differentiate. Once past this threshold, these newly committed cells lose the capacity for self-renewal and commence an inexorable journey down a road that leads to terminal differentiation and death. However, as these progenitors differentiate, they also proliferate rapidly in response to growth factors, expanding their numbers. Some growth factors, such as stem cell factor (also called KIT ligand) and FLT3 ligand, act through receptors that are expressed on very early committed progenitors. Others, such as erythropoietin, granulocyte-macrophage colony-stimulating factor (GM-CSF), G-CSF, and thrombopoietin, act through receptors that are expressed only on committed progenitors with more restricted differentiation potentials. Feedback loops involving these lineage-specific growth factors tune the marrow output, allowing the numbers of formed blood elements (red cells, white cells, and platelets) to be maintained within appropriate ranges ( Table 13.1 ).
| Cell Type | Range |
|---|---|
| White cells (×10 3 /µL) | 4.8–10.8 |
| Granulocytes (%) | 40–70 |
| Neutrophils (×10 3 /µL) | 1.4–6.5 |
| Lymphocytes (×10 3 /µL) | 1.2–3.4 |
| Monocytes (×10 3 /µL) | 0.1–0.6 |
| Eosinophils (×10 3 /µL) | 0–0.5 |
| Basophils (×10 3 /µL) | 0–0.2 |
| Red cells (×10 6 /µL) | |
| Men | 4.3–5 |
| Women | 3.5–5 |
| Platelets (×10 3 /µL) | 150–450 |
a Reference ranges vary among laboratories. The reference ranges for the laboratory providing the result should always be used.
Many diseases alter the production of blood cells. The marrow is the ultimate source of most cells of the innate and adaptive immune system and responds to infectious or inflammatory challenges by increasing its output of granulocytes under the direction of specific growth factors and cytokines. By contrast, many other disorders are associated with defects in hematopoiesis that lead to deficiencies of one or more types of blood cells. Primary tumors of hematopoietic cells are among the most important diseases that interfere with marrow function, but certain genetic diseases, infections, toxins, and nutritional deficiencies, as well as chronic inflammation from any cause, may also decrease the production of blood cells by the marrow.
Tumors of hematopoietic origin are often associated with mutations that block progenitor cell maturation or abrogate their growth factor dependence. The net effect of such derangements is an unregulated clonal expansion of hematopoietic elements, which replace normal marrow progenitors and often spread to other hematopoietic tissues. In some instances these tumors originate from transformed HSCs that retain the ability to differentiate along multiple lineages, whereas in other instances the origin is a more differentiated progenitor that has acquired an abnormal capacity for self-renewal ( Chapter 7 ).
The bone marrow is a unique microenvironment that supports the orderly proliferation, differentiation, and release of blood cells. It is filled with a network of thin-walled sinusoids lined by a single layer of endothelial cells, which are underlaid by a discontinuous basement membrane and adventitial cells. Within the interstitium lie clusters of hematopoietic cells and fat cells. Differentiated blood cells enter the circulation by transcellular migration through the endothelial cells.
The normal marrow is organized in subtle, but important, ways. For example, normal megakaryocytes lie next to sinusoids and extend cytoplasmic processes that bud off into the bloodstream to produce platelets, while red cell precursors often surround macrophages that dispose of nuclear remnants produced when red cells disgorge their nucleus prior to release into the bloodstream. Processes that distort the marrow architecture, such as deposits of metastatic cancer or granulomatous disorders, can cause the abnormal release of immature precursors into the peripheral blood, a finding that is referred to as leukoerythroblastosis.
Marrow aspirate smears provide the best assessment of the morphology of hematopoietic cells. The most mature marrow precursors can be identified based on their morphology alone. Immature precursors (“blast” forms) of different types are morphologically similar and must be identified definitively using lineage-specific antibodies and histochemical markers (described later under white cell neoplasms). Bone marrow biopsies are a good means for estimating marrow activity. In normal adults the ratio of fat cells to hematopoietic elements is about 1:1. In hypoplastic states (e.g., aplastic anemia) the proportion of fat cells is greatly increased; conversely, fat cells often disappear when the marrow is involved by hematopoietic tumors and in diseases characterized by compensatory hyperplasias (e.g., hemolytic anemias) and neoplastic proliferations such as leukemias. Other disorders (e.g., metastatic cancers and granulomatous diseases) induce local marrow fibrosis. In such cases the marrow usually cannot be aspirated and the lesions are best seen in biopsies.
Disorders of white blood cells can be classified into two broad categories: proliferative disorders, in which there is an expansion of leukocytes, and leukopenias, which are defined as a deficiency of leukocytes. Proliferations of white cells can be reactive or neoplastic. Reactive proliferations in the setting of infections or inflammatory processes, when large numbers of leukocytes are needed for an effective host response, are fairly common. Neoplastic disorders, though less frequent, are much more important clinically. In the following discussion we will first describe the leukopenic states and summarize the common reactive disorders and then consider in some detail the malignant proliferations of white cells.
The number of circulating white cells may be decreased in a variety of disorders. An abnormally low white cell count (leukopenia) usually results from reduced numbers of neutrophils (neutropenia, granulocytopenia) . Lymphopenia is less common; in addition to congenital immunodeficiency diseases ( Chapter 6 ), it is most commonly observed in advanced human immunodeficiency virus (HIV) infection, following therapy with glucocorticoids or cytotoxic drugs, autoimmune disorders, malnutrition, and certain acute viral infections. In the latter setting, lymphopenia actually stems from lymphocyte redistribution rather than a decrease in the number of lymphocytes in the body. Acute viral infections induce production of type I interferons, which activate T lymphocytes and change the expression of surface proteins that regulate T-cell migration. These changes result in the sequestration of activated T cells in lymph nodes and increased adherence to endothelial cells, both of which contribute to lymphopenia. Granulocytopenia is more common and is often associated with diminished granulocyte function and thus merits further discussion.
Neutropenia, a reduction in the number of neutrophils in the blood, occurs in a wide variety of circumstances. Agranulocytosis, a marked reduction in neutrophils, has the serious consequence of making individuals susceptible to bacterial and fungal infections.
Neutropenia can be caused by (1) inadequate or ineffective granulopoiesis or (2) increased destruction or sequestration of neutrophils in the periphery. Inadequate or ineffective granulopoiesis is observed in the setting of:
Suppression of HSCs, as occurs in aplastic anemia ( Chapter 14 ) and a variety of infiltrative marrow disorders (e.g., tumors, granulomatous disease); in these conditions, granulocytopenia is accompanied by anemia and thrombocytopenia.
Suppression of committed granulocytic precursors by exposure to certain drugs (discussed later).
Disease states associated with ineffective hematopoiesis, such as megaloblastic anemia ( Chapter 14 ) and myelodysplastic syndrome, in which defective precursors die in the marrow.
Rare congenital conditions (e.g., Kostmann syndrome), in which inherited defects in specific genes impair granulocytic differentiation.
Accelerated destruction or sequestration of neutrophils occurs with:
Immunologically mediated injury to neutrophils, which can be idiopathic, associated with a well-defined immunologic disorder (e.g., systemic lupus erythematosus), or caused by exposure to drugs.
Splenomegaly, in which splenic enlargement leads to sequestration and destruction of neutrophils in the spleen and modest neutropenia, sometimes associated with anemia and often with thrombocytopenia.
Increased peripheral utilization, which can occur in overwhelming bacterial, fungal, or rickettsial infections.
The most common cause of agranulocytosis is drug toxicity . Certain drugs, such as alkylating agents and antimetabolites used in cancer treatment, produce agranulocytosis in a predictable, dose-related fashion. Because such drugs cause a generalized suppression of hematopoiesis, production of red cells and platelets is also affected. Agranulocytosis may also occur as an idiosyncratic reaction to a large variety of agents including certain antibiotics, anticonvulsants, antiinflammatory drugs, antipsychotic drugs, and diuretics. The neutropenia induced by antipsychotic agents such as chlorpromazine and related phenothiazines results from a toxic effect on granulocytic precursors in the bone marrow. In contrast, agranulocytosis following administration of other drugs, such as sulfonamides, probably stems from antibody-mediated destruction of neutrophils through mechanisms similar to those involved in drug-induced immunohemolytic anemias ( Chapter 14 ).
In some patients with acquired idiopathic neutropenia, autoantibodies directed against neutrophil-specific antigens are detected. Severe neutropenia may also occur in association with monoclonal proliferations of large granular lymphocytes (so-called LGL leukemia ). The mechanism of this neutropenia is not clear; suppression of granulocytic progenitors by products of the neoplastic cell (usually a CD8+ cytotoxic T cell) is considered most likely.
The alterations in the bone marrow vary with cause. With excessive destruction of neutrophils in the periphery, the marrow is usually hypercellular due to a compensatory increase in granulocytic precursors. Hypercellularity is also the rule with neutropenias caused by ineffective granulopoiesis, as occurs in megaloblastic anemia and myelodysplastic syndrome. Agranulocytosis caused by agents that suppress or destroy granulocyte precursors is understandably associated with marrow hypocellularity.
Infections are a common consequence of agranulocytosis. Ulcerating necrotizing lesions of the gingiva, floor of the mouth, buccal mucosa, pharynx, or elsewhere in the oral cavity (agranulocytic angina) are quite characteristic. These are typically deep, undermined, and covered by gray to green-black necrotic membranes from which numerous bacteria or fungi can be isolated. Less frequently, similar ulcerative lesions occur in the skin, vagina, anus, or gastrointestinal tract. Severe life-threatening invasive bacterial or fungal infections may occur in the lungs, urinary tract, and kidneys. The neutropenic patient is at particularly high risk for deep fungal infections caused by Candida and Aspergillus. Sites of infection often show a massive growth of organisms with little leukocytic response. In the most dramatic instances, bacteria grow in colonies (botryomycosis) resembling those seen on agar plates.
The symptoms and signs of neutropenia are related to infection and include malaise, chills, and fever, often followed by marked weakness and fatigability. With agranulocytosis, infections are often overwhelming and may cause death within hours to days.
Serious infections are most likely when the neutrophil count falls below 500/mm 3 . Because infections are often fulminant, broad-spectrum antibiotics must be given expeditiously whenever signs or symptoms appear. In some instances, such as following myelosuppressive chemotherapy, neutropenia is treated with G-CSF, a growth factor that stimulates the production of granulocytes from marrow precursors.
Leukocytosis refers to an increase in the number of white cells in the blood . It is a common reaction to a variety of inflammatory states.
The peripheral blood leukocyte count is influenced by several factors, including:
The size of the myeloid and lymphoid precursor and storage cell pools in the bone marrow, thymus, circulation, and peripheral tissues.
The rate of release of cells from the storage pools into the circulation.
The proportion of cells that are adherent to blood vessel walls at any time (the marginal pool).
The rate of extravasation of cells from the blood into tissues.
As discussed in Chapter 3 , leukocyte homeostasis is maintained by cytokines, growth factors, and adhesion molecules through their effects on the proliferation, differentiation, and extravasation of leukocytes and their progenitors. Table 13.2 summarizes the major mechanisms of neutrophilic leukocytosis and its causes, the most important of which is infection. In acute infection there is a rapid increase in the egress of mature granulocytes from the bone marrow pool, an alteration that may be mediated through the effects of tumor necrosis factor (TNF) and interleukin-1 (IL-1). If the infection or an inflammatory process is prolonged, IL-1, TNF, and other inflammatory mediators stimulate macrophages, bone marrow stromal cells, and T cells to produce increased amounts of hematopoietic growth factors. These factors enhance the proliferation and differentiation of committed granulocytic progenitors and, over several days, cause a sustained increase in neutrophil production.
| Increased Marrow Production |
|
| Increased Release From Marrow Stores |
|
| Decreased Margination |
|
| Decreased Extravasation Into Tissues |
| Glucocorticoids |
Some growth factors preferentially stimulate the production of a single type of leukocyte. For example, IL-5 mainly stimulates eosinophil production, while G-CSF induces neutrophilia. Such factors are differentially produced in response to various pathogenic stimuli, and, as a result, the five principal types of leukocytosis (neutrophilia, eosinophilia, basophilia, monocytosis, and lymphocytosis) tend to be observed in different clinical settings ( Table 13.3 ).
| Type of Leukocytosis | Causes |
|---|---|
| Neutrophilic leukocytosis | Acute bacterial infections, especially those caused by pyogenic organisms; sterile inflammation caused by, for example, tissue necrosis (myocardial infarction, burns) |
| Eosinophilic leukocytosis (eosinophilia) | Allergic disorders such as asthma, hay fever, parasitic infestations; drug reactions; certain malignancies (e.g., Hodgkin and some non-Hodgkin lymphomas); autoimmune disorders (e.g., pemphigus, dermatitis herpetiformis) and some vasculitides; atheroembolic disease (transient) |
| Basophilic leukocytosis (basophilia) | Rare, often indicative of a myeloproliferative neoplasm (e.g., chronic myeloid leukemia) |
| Monocytosis | Chronic infections (e.g., tuberculosis), bacterial endocarditis, rickettsiosis, and malaria; autoimmune disorders (e.g., systemic lupus erythematosus); inflammatory bowel diseases (e.g., ulcerative colitis) |
| Lymphocytosis | Accompanies monocytosis in many disorders associated with chronic immunologic stimulation (e.g., tuberculosis, brucellosis); viral infections (e.g., hepatitis A, cytomegalovirus, Epstein-Barr virus); Bordetella pertussis infection |
In sepsis or severe inflammatory disorders (e.g., Kawasaki disease), leukocytosis is often accompanied by morphologic changes in neutrophils, such as toxic granulations, Döhle bodies, and cytoplasmic vacuoles ( Fig. 13.2 ). Toxic granules, which are coarser and darker than normal neutrophilic granules, represent abnormal azurophilic (primary) granules. Döhle bodies are patches of dilated endoplasmic reticulum that appear as sky-blue cytoplasmic “puddles.”
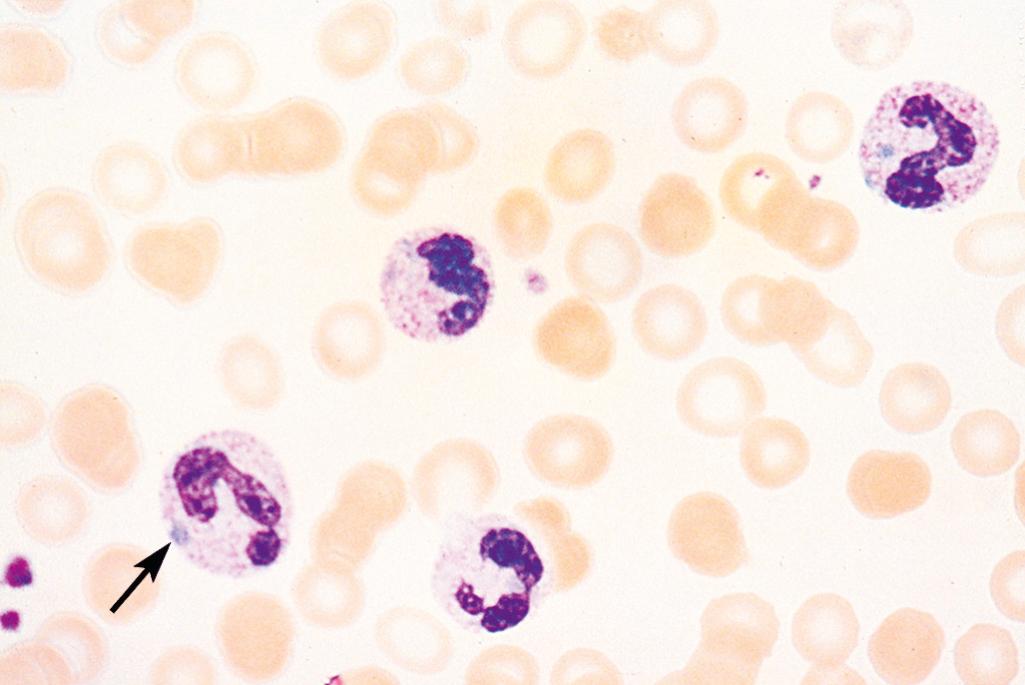
In most instances it is not difficult to distinguish reactive and neoplastic leukocytoses, but uncertainties may arise in two settings. Acute viral infections, particularly in children, can cause the appearance of large numbers of activated lymphocytes that resemble neoplastic lymphoid cells. At other times, particularly in severe infections, many immature granulocytes appear in the blood, mimicking a myeloid leukemia (leukemoid reaction) . Special laboratory studies (discussed later) are helpful in distinguishing reactive and neoplastic leukocytoses.
Following their initial development from precursors in the central (also called primary) lymphoid organs—the bone marrow for B cells and the thymus for T cells—lymphocytes circulate through the blood and, under the influence of specific cytokines and chemokines, home to lymph nodes, spleen, tonsils, adenoids, and Peyer patches, which constitute the peripheral (secondary) lymphoid tissues. Lymph nodes, the most widely distributed and easily accessible lymphoid tissue, are frequently examined for diagnostic purposes. They are discrete encapsulated structures that contain separate B-cell and T-cell zones, each richly invested with phagocytes and antigen-presenting cells (see Fig. 6.8 , Chapter 6 ).
The activation of resident immune cells leads to morphologic changes in lymph nodes. Within several days of antigenic stimulation, the primary follicles enlarge and develop pale-staining germinal centers, highly dynamic structures in which B cells acquire the capacity to make high-affinity antibodies against specific antigens. Paracortical T-cell zones may also undergo hyperplasia. The degree and pattern of morphologic change are dependent on the inciting stimulus and the intensity of the response. Trivial injuries and infections induce subtle changes, while more significant infections inevitably produce nodal enlargement and sometimes leave residual scarring. For this reason, lymph nodes in adults are almost never “normal” or “resting,” and it is often necessary to distinguish morphologic changes secondary to past experience from those related to present disease. Infections and inflammatory stimuli often elicit regional or systemic immune reactions within lymph nodes. Some that produce distinctive morphologic patterns are described in other chapters. Most, however, cause stereotypical patterns of lymph node reaction designated acute and chronic nonspecific lymphadenitis.
Acute lymphadenitis in the cervical region is most often due to drainage of microbes or microbial products from infections of the teeth or tonsils, while in the axillary or inguinal regions it is most often caused by infections in the extremities. Acute lymphadenitis also occurs in mesenteric lymph nodes in the setting of acute appendicitis and other inflammatory conditions involving the gut (including self-limiting viral infections), a differential diagnosis that plagues the surgeon. Systemic viral infections (particularly in children) and bacteremia often produce acute generalized lymphadenopathy.
The nodes are swollen, gray-red, and engorged. Microscopically, there is prominence of large reactive germinal centers containing numerous mitotic figures. Macrophages often contain particulate debris derived from dead bacteria or necrotic cells. When pyogenic organisms are the cause, neutrophils are prominent, and the centers of the follicles may undergo necrosis; sometimes the entire node is converted to pus. With less severe reactions, scattered neutrophils infiltrate about the follicles and accumulate within the lymphoid sinuses. The endothelial cells lining the sinuses become activated and enlarge in size.
Nodes involved by acute lymphadenitis are swollen and painful. When abscess formation is extensive the nodes become fluctuant. The overlying skin is red. Sometimes, suppurative infections penetrate through the capsule of the node and track to the skin to produce draining sinuses. Healing of such lesions is associated with scarring.
A wide variety of chronic immunologic stimuli may produce nonspecific lymphadenitis. Several different patterns of morphologic change are seen, often within the same lymph node.
Follicular hyperplasia is caused by stimuli that activate humoral immune responses. It is defined by the presence of large oblong germinal centers (secondary follicles), which are surrounded by a collar of small resting naive B cells (the mantle zone) ( Fig. 13.3 ). Germinal centers are polarized, consisting of two distinct regions: (1) a dark zone with proliferating blast-like B cells (centroblasts) and (2) a light zone composed of B cells with irregular or cleaved nuclear contours (centrocytes). Interspersed among the germinal center B cells is an inconspicuous network of antigen-presenting follicular dendritic cells and macrophages (often referred to as tingible-body macrophages ) containing the nuclear debris of B cells, which undergo apoptosis if they fail to produce an antibody with a high affinity for antigen.

Causes of follicular hyperplasia include rheumatoid arthritis, toxoplasmosis, and early HIV infection. This form of hyperplasia is morphologically similar to follicular lymphoma (discussed later). Features favoring a reactive (nonneoplastic) hyperplasia include (1) preservation of the lymph node architecture, including the interfollicular T-cell zones and the sinusoids, (2) marked variation in the shape and size of the follicles, and (3) the presence of frequent mitotic figures, phagocytic macrophages, and recognizable light and dark zones, all of which tend to be absent from neoplastic follicles.
Paracortical hyperplasia is caused by stimuli that trigger T-cell–mediated immune responses, such as acute viral infections (e.g., infectious mononucleosis). The T-cell regions typically contain immunoblasts, activated T cells three to four times the size of resting lymphocytes that have round nuclei, open chromatin, several prominent nucleoli, and moderate amounts of pale cytoplasm. The expanded T-cell zones encroach on and, in particularly exuberant reactions, may efface the B-cell follicles. In such cases, immunoblasts are so numerous that special studies may be needed to exclude a lymphoid neoplasm. In addition, there is often hypertrophy of sinusoidal and vascular endothelial cells, sometimes accompanied by infiltrating macrophages and eosinophils.
Sinus histiocytosis (also called reticular hyperplasia ) is marked by an increase in the number and size of the endothelial cells that line lymphatic sinusoids and increased numbers of intrasinusoidal macrophages, which expand and distort the sinusoids. This form of hyperplasia may be particularly prominent in lymph nodes draining cancers such as carcinoma of the breast.
Characteristically, lymph nodes in chronic reactions are nontender, as enlargement occurs slowly over time and acute inflammation with associated tissue damage is absent. Chronic lymphadenitis is particularly common in inguinal and axillary nodes, which drain relatively large areas of the body and are frequently stimulated by immune reactions to trivial injuries and infections of the extremities.
Chronic immune reactions also can promote the appearance of organized collections of immune cells in nonlymphoid tissues. These collections are sometimes called tertiary lymphoid organs . A classic example is that of chronic gastritis caused by Helicobacter pylori, in which aggregates of mucosal lymphocytes are seen that simulate the appearance of Peyer patches. A similar phenomenon occurs in rheumatoid arthritis, in which B-cell follicles often appear in the inflamed synovium. Lymphotoxin, a cytokine required for the formation of normal Peyer patches, is probably involved in the establishment of these “extranodal” inflammation-induced collections of lymphoid cells.
Hemophagocytic lymphohistiocytosis (HLH) is a reactive condition marked by cytopenias and signs and symptoms of systemic inflammation related to macrophage activation. For this reason, it is also sometimes referred to as macrophage activation syndrome . Some forms are familial and may appear early in life, even in infants, while other forms are sporadic and may affect people of any age.
The common feature of all forms of HLH is systemic activation of macrophages and CD8+ cytotoxic T cells. The activated macrophages phagocytose blood cell progenitors in the marrow and formed elements in the peripheral tissues, while the “stew” of mediators released from macrophages and lymphocytes suppress hematopoiesis and produce symptoms of systemic inflammation. These effects lead to cytopenias and a shock-like picture, sometimes referred to as “cytokine storm” or the systemic inflammatory response syndrome ( Chapter 4 ).
Familial forms of HLH are associated with several different mutations, all of which impact the ability of cytotoxic T cells (CTLs) and natural killer (NK) cells to properly form or deploy cytotoxic granules. How these defects lead to HLH is not known. One common trigger for HLH is Epstein-Barr virus infection, suggesting that in some instances HLH stems from a defect in the ability of CD8+ CTLs to kill infected cells. As a result of the persistent infection, the CTLs continue to make cytokines, leading to excessive macrophage activation. HLH is also a common complication of peripheral T-cell lymphoma (discussed later), a tumor of mature T cells that is marked by immune dysregulation. Regardless of the trigger, HLH is uniformly associated with extremely high levels of inflammatory mediators such as interferon-γ, TNF-α, IL-6, and IL-12.
Most patients present with an acute febrile illness associated with splenomegaly and hepatomegaly. Hemophagocytosis is usually seen on bone marrow examination, but is neither sufficient nor required to make the diagnosis. Laboratory studies typically reveal anemia, thrombocytopenia, and very high levels of plasma ferritin and soluble IL-2 receptor, both indicative of severe inflammation, as well as elevated liver function tests and triglyceride levels, both related to hepatitis. Coagulation studies may show evidence of disseminated intravascular coagulation. If untreated, this picture can progress rapidly to multiorgan failure, shock, and death.
Treatment involves the use of immunosuppressive drugs, “mild” chemotherapy, and administration of an antibody that neutralizes the activity of interferon-γ. Patients with germline mutations that cause HLH or who have persistent/resistant disease are candidates for HSC transplantation. Without treatment, the prognosis is grim, particularly in those with familial forms of the disease, who typically survive for less than 2 months. With prompt treatment, with or without subsequent HSC transplantation, roughly half of patients survive, though many do so with significant sequelae, such as renal damage in adults and growth retardation and intellectual disability in children.
Malignancies are clinically the most important disorders of white cells. These diseases fall into three broad categories:
Lymphoid neoplasms include a diverse group of tumors of B-cell, T-cell, and NK-cell origin. In many instances the phenotype of the neoplastic cell closely resembles that of a particular lymphocyte class or stage of maturation, a feature that is used in the diagnosis and classification of these disorders.
Myeloid neoplasms arise from early hematopoietic progenitors. Three categories of myeloid neoplasia are recognized: acute myeloid leukemias (AMLs), in which immature progenitor cells accumulate in the bone marrow; myelodysplastic syndromes (MDSs), which are associated with ineffective hematopoiesis and resultant peripheral blood cytopenias; and myeloproliferative neoplasms, in which increased production of one or more terminally differentiated myeloid elements (e.g., granulocytes) usually leads to elevated peripheral blood counts.
The histiocytoses are uncommon proliferative lesions of macrophages and dendritic cells. Although “histiocyte” (literally, “tissue cell”) is an archaic morphologic term, it is still often used. A special type of immature dendritic cell, the Langerhans cell, gives rise to a spectrum of neoplastic disorders referred to as the Langerhans cell histiocytoses.
As in other cancers, the development of white blood cell neoplasms involves genetic alterations, infections, and sometimes a background of chronic inflammation. Different types of tumors show different abnormalities and are, therefore, responsive to different therapies. Before delving into this complexity, we consider themes of general relevance to their etiology and pathogenesis.
Nonrandom chromosomal abnormalities, most commonly translocations, are present in the majority of white cell neoplasms. Many of these alterations are specifically associated with particular neoplasms and have a critical role in their genesis ( Chapter 7 ).
Recurrently affected genes are often those that play crucial roles in the development, growth, or survival of the normal counterpart of the malignant cell. Mutations in certain genes are so strongly associated with specific tumor types that in some instances they are required for particular diagnoses. Some of these mutations produce a “dominant-negative” protein that interferes with a normal function (a loss of function); in others the result is an inappropriate increase in some normal activity (a gain of function).
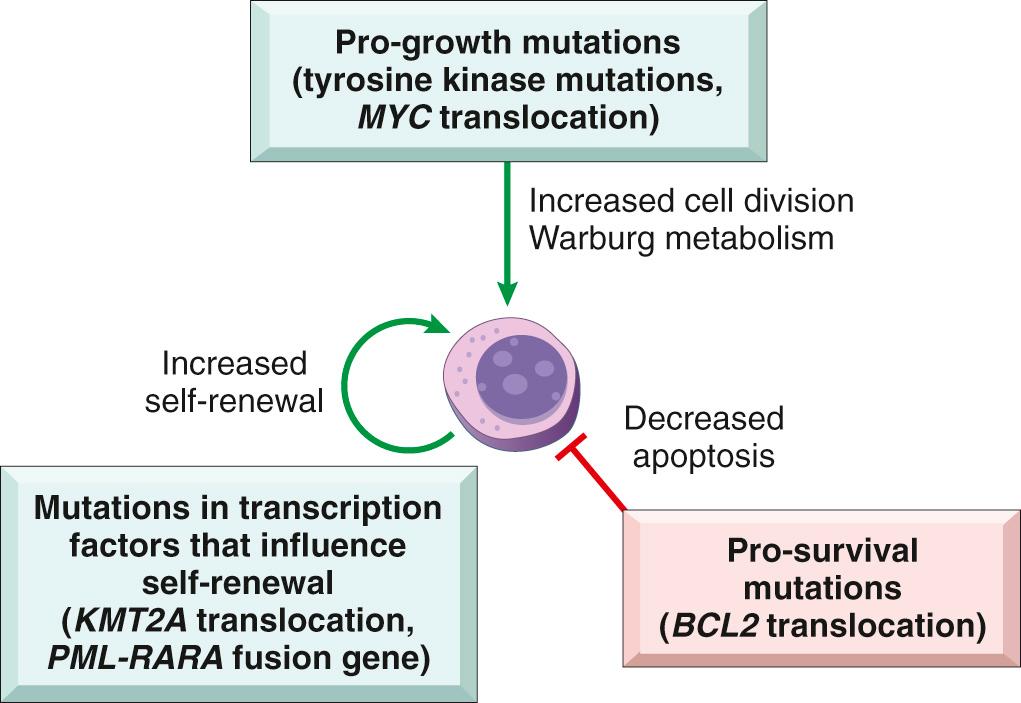
Oncoproteins created by genomic aberrations often block normal maturation, turn on pro-growth signaling pathways, or protect cells from apoptotic cell death. Fig. 13.4 highlights several well-characterized driver mutations and their pathogenic consequences in particular white cell neoplasms.
Many oncoproteins cause an arrest in differentiation, often at a stage when cells are proliferating rapidly. The importance of this mechanism is most evident in the acute leukemias, in which dominant-negative oncogenic mutations involving transcription factors interfere with early stages of lymphoid or myeloid cell differentiation.
Other mutations in transcriptional regulators directly enhance the self-renewal of tumors cells, giving them stem cell–like properties. These types of mutations often collaborate with mutations that constitutively activate tyrosine kinases, which in turn activate RAS and its two downstream signaling arms, the PI3K/AKT and MAPK pathways ( Chapter 7 ), thereby driving cell growth.
Finally, mutations that inhibit apoptosis are prevalent in certain hematologic malignancies.
Proto-oncogenes are often activated in lymphoid cells by errors that occur during attempted antigen receptor gene diversification. Among lymphoid cells, potentially oncogenic mutations occur most frequently in germinal center B cells . After antigen stimulation, B cells enter germinal centers and upregulate the expression of activation-induced cytosine deaminase (AID), a specialized DNA-modifying enzyme that is essential for two types of immunoglobulin (Ig) gene modifications: class switching, an intragenic recombination event in which the IgM heavy-chain constant gene segment is replaced with a different constant segment (e.g., IgG 3 ), leading to a switch in the class (isotype) of antibody produced, and somatic hypermutation, which creates point mutations within Ig genes that may increase antibody affinity for antigen ( Chapter 6 ). Certain proto-oncogenes, such as MYC, are activated in germinal center B-cell lymphomas by translocations to the transcriptionally active Ig locus. Remarkably, AID expression induces MYC/Ig translocations in a small fraction of normal germinal center B cells, apparently because AID creates lesions in DNA that lead to chromosomal breaks. “Mistargeting” of AID also is implicated in point mutations that upregulate the expression and activity of BCL6, an oncogenic transcription factor with an important role in several B-cell malignancies. Another type of regulated genomic instability that is unique to precursor B and T cells is attributable to V(D)J recombinase, which cuts DNA at specific sites within the Ig and T-cell receptor loci, respectively. This process is essential for generating diversity in assembled antigen receptor genes but sometimes goes awry, leading to the joining of proto-oncogenes to antigen receptor gene regulatory elements. The resulting overexpression of the involved proto-oncogene converts it to an oncogene. This mechanism is particularly prevalent in tumors of precursor T cells but is observed in other types of lymphoid neoplasms as well.
As discussed in Chapter 7 , individuals with genetic diseases that promote genomic instability, such as Bloom syndrome, Fanconi anemia, and ataxia telangiectasia, are at increased risk of acute leukemia. In addition, both Down syndrome (trisomy 21) and type I neurofibromatosis are associated with an increased incidence of childhood leukemia.
Three lymphotropic viruses—human T-cell leukemia virus-1 (HTLV-1), EBV, and human herpesvirus-8 (HHV-8; also known as Kaposi sarcoma herpesvirus)—have been implicated as causative agents in particular lymphomas. The possible mechanisms of transformation by viruses are discussed in Chapter 7 . HTLV-1 is associated with adult T-cell leukemia/lymphoma. EBV is found in a subset of Burkitt lymphoma, 30% to 40% of Hodgkin lymphoma (HL), many B-cell lymphomas arising in the setting of T-cell immunodeficiency, and rare NK-cell lymphomas. In addition to Kaposi sarcoma ( Chapter 11 ), HHV-8 is associated with an unusual B-cell lymphoma that presents as a malignant effusion, often in the pleural cavity.
Several agents that cause localized chronic inflammation predispose to lymphoid neoplasia, which almost always arises within the inflamed tissue. Examples include the associations between H. pylori infection and gastric B-cell lymphomas ( Chapter 17 ); gluten-sensitive enteropathy and intestinal T-cell lymphomas; and even breast implants, which are associated with an unusual subtype of T-cell lymphoma. This can be contrasted with HIV infection, which is associated with an increased risk of B-cell lymphomas that may arise within virtually any organ. Early in the course, T-cell dysregulation by HIV infection causes a systemic hyperplasia of germinal center B cells that is associated with an increased incidence of germinal center B-cell lymphomas. In advanced infection (acquired immunodeficiency syndrome [AIDS]), severe T-cell immunodeficiency further elevates the risk for B-cell lymphomas, particularly those associated with EBV and KSHV/HHV-8. These relationships are discussed in more detail in Chapter 6 .
Ironically, radiation therapy and certain forms of chemotherapy used to treat cancer increase the risk of subsequent myeloid and lymphoid neoplasms. This association stems from the mutagenic effects of ionizing radiation and chemotherapeutic drugs on hematolymphoid progenitor cells.
The incidence of AML is increased 1.3- to 2-fold in smokers, presumably because of exposure to carcinogens, such as benzene, in tobacco smoke.
Taken together, the diverse lymphoid neoplasms constitute a complex, clinically important group of cancers, with about 100,000 new cases being diagnosed each year in the United States.
Neoplasms that present with widespread involvement of the bone marrow and (usually, but not always) the peripheral blood are called leukemias. Proliferations of white cells, typically lymphocytes, that usually present as discrete tissue masses are called lymphomas. Originally these terms were attached to what were considered distinct entities, but with time and increased understanding these divisions have blurred. Many entities called “lymphoma” occasionally have leukemic presentations, and evolution to “leukemia” is not unusual during the progression of incurable “lymphomas.” Conversely, tumors identical to “leukemias” sometimes arise as soft tissue masses without detectable bone marrow disease. Hence the terms leukemia and lymphoma merely reflect the usual tissue distribution of each disease at presentation.
Within the large group of lymphomas, Hodgkin lymphoma is segregated from other forms, which constitute the non-Hodgkin lymphomas (NHLs). Hodgkin lymphoma has distinctive pathologic features and is treated in a unique fashion. Another special group of tumors includes plasma cell neoplasms . These most often arise in the bone marrow and only infrequently involve lymph nodes or the peripheral blood.
The clinical presentation of lymphoid neoplasms is most often determined by the anatomic distribution of disease. Two-thirds of NHLs and virtually all Hodgkin lymphomas present as enlarged nontender lymph nodes (often >2 cm). The remaining NHLs present with symptoms related to the involvement of extranodal sites (e.g., skin, stomach, or brain). Lymphocytic leukemias most often come to attention because of signs and symptoms related to the suppression of normal hematopoiesis by tumor cells in the bone marrow, whereas the most common plasma cell neoplasm, multiple myeloma, causes bony destruction of the skeleton and often presents with pain due to pathologic fractures. Other symptoms are frequently caused by proteins secreted from tumor cells or from immune cells responding to the tumor. Specific examples include the plasma cell tumors, in which much of the pathophysiology is related to the secretion of whole antibodies or Ig fragments; Hodgkin lymphoma, which is often associated with fever related to the release of cytokines from nonneoplastic inflammatory cells; and peripheral T-cell lymphomas, tumors of functional T cells that often release pro-inflammatory cytokines and chemokines.
Historically, few areas of pathology evoked as much controversy as the classification of lymphoid neoplasms, but consensus has been reached through use of objective molecular diagnostic tools. The current World Health Organization (WHO) classification scheme ( Table 13.4 ) uses morphologic, immunophenotypic, genotypic, and clinical features to sort the lymphoid neoplasms into five broad categories, separated according to the cell of origin:
Precursor B-cell neoplasms (neoplasms of immature B cells)
Peripheral B-cell neoplasms (neoplasms of mature B cells)
Precursor T-cell neoplasms (neoplasms of immature T cells)
Peripheral T-cell and NK-cell neoplasms (neoplasms of mature T cells and NK cells)
Hodgkin lymphomas (neoplasms of Reed-Sternberg cells and variants)
| I. Precursor B-Cell Neoplasms |
| B-cell acute lymphoblastic leukemia/lymphoma (B-ALL) |
| II. Peripheral B-Cell Neoplasms |
|
| III. Precursor T-Cell Neoplasms |
| T-cell acute lymphoblastic leukemia/lymphoma (T-ALL) |
| IV. Peripheral T-Cell and NK-Cell Neoplasms |
|
| V. Hodgkin Lymphoma |
|
Before discussing the specific entities, some important principles relevant to lymphoid neoplasms should be emphasized.
Lymphoid neoplasia can be suspected based on clinical features, but histologic examination of lymph nodes or other involved tissues is required for diagnosis . Analysis of lineage-specific protein (marker) expression and genetic alterations is an important complement to the morphologic studies. Markers recognized by antibodies that are helpful in the characterization of lymphomas and leukemias are listed in Table 13.5 .
| Antigen Designation | Normal Cellular Distribution |
|---|---|
| Primarily T-Cell Associated | |
| CD1 | Thymocytes and Langerhans cells |
| CD3 | Thymocytes, mature T cells |
| CD4 | Helper T cells, subset of thymocytes |
| CD5 | T cells and small subset of B cells |
| CD8 | Cytotoxic T cells, subset of thymocytes, and some NK cells |
| Primarily B-Cell Associated | |
| CD10 | Pre-B cells and germinal center B cells |
| CD19 | Pre-B cells and mature B cells but not plasma cells |
| CD20 | Pre-B cells after CD19 and mature B cells but not plasma cells |
| CD21 | EBV receptor; mature B cells and follicular dendritic cells |
| CD23 | Activated mature B cells |
| CD79a | Marrow pre-B cells and mature B cells |
| Primarily Monocyte or Macrophage Associated | |
| CD11c | Granulocytes, monocytes, and macrophages; also expressed by hairy cell leukemias |
| CD13 | Immature and mature monocytes and granulocytes |
| CD14 | Monocytes |
| CD15 | Granulocytes; Reed-Sternberg cells and variants |
| CD33 | Myeloid progenitors and monocytes |
| CD64 | Mature myeloid cells |
| Primarily NK-Cell Associated | |
| CD16 | NK cells and granulocytes |
| CD56 | NK cells and a subset of T cells |
| Primarily Stem Cell and Progenitor Cell Associated | |
| CD34 | Pluripotent hematopoietic stem cells and progenitor cells of many lineages |
| Activation Markers | |
| CD30 | Activated B cells, T cells, and monocytes; Reed-Sternberg cells and variants |
| Present on All Leukocytes | |
| CD45 | All leukocytes; also known as leukocyte common antigen (LCA) |
Antigen receptor gene rearrangement generally precedes transformation of lymphoid cells; hence all daughter cells derived from the malignant progenitor share the same antigen receptor gene configuration and sequence and synthesize identical antigen receptor proteins (either Igs or T-cell receptors). In contrast, normal immune responses are comprised of polyclonal populations of lymphocytes that express many different antigen receptors. Thus, analyses of antigen receptor genes and their protein products can be used to distinguish reactive (polyclonal) and malignant (monoclonal) lymphoid proliferations. In addition, each antigen receptor gene rearrangement produces a unique DNA sequence that constitutes a highly specific clonal marker, which can be used to detect small numbers of residual malignant lymphoid cells after therapy.
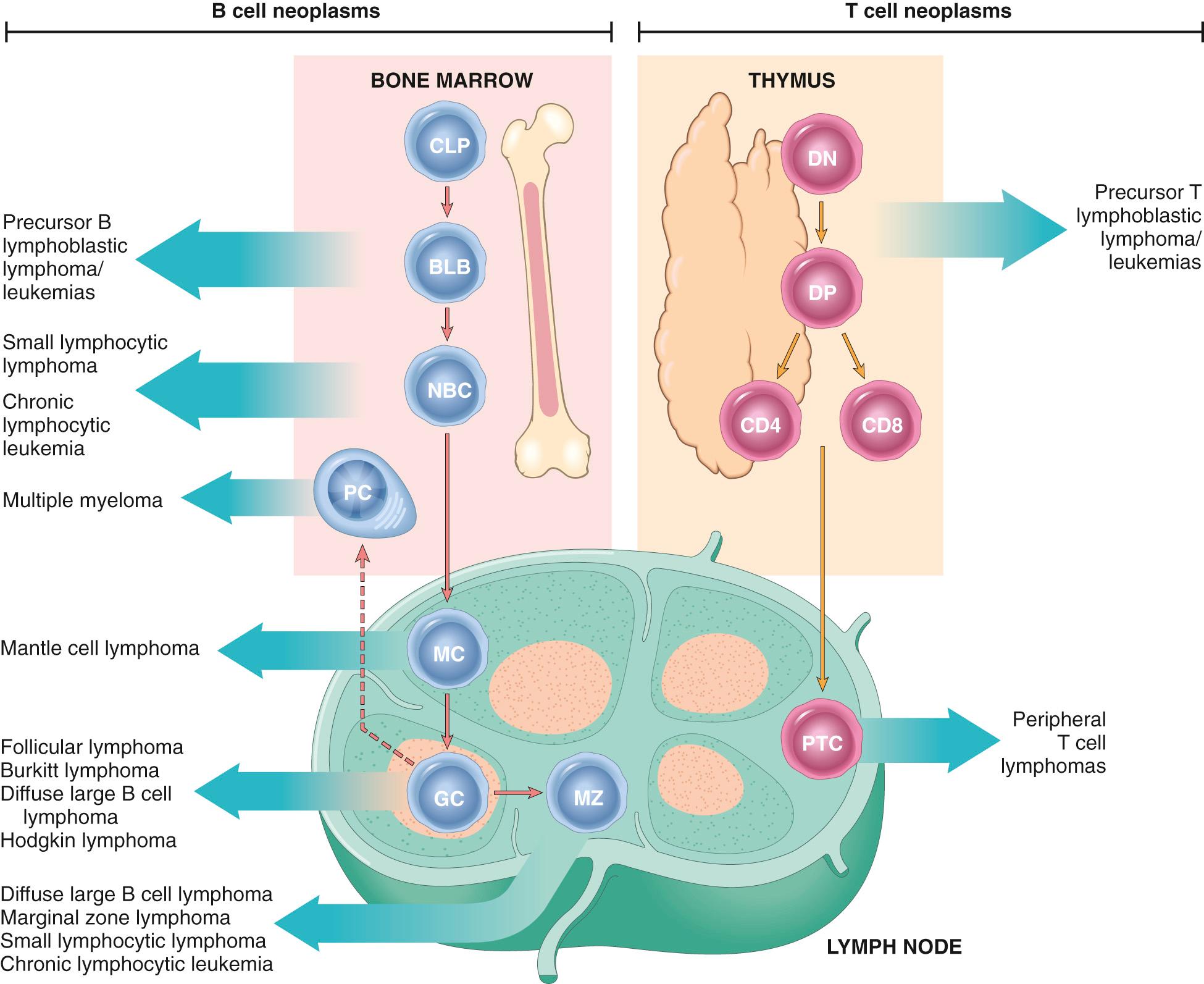
Most lymphoid neoplasms resemble some recognizable stage of B- or T-cell differentiation ( Fig. 13.5 ), a feature that is used in their classification. The vast majority (85% to 90%) of lymphoid neoplasms are of B-cell origin, with most of the remainder being T-cell tumors; tumors of NK-cell origin are rare.
Lymphoid neoplasms are often associated with immune abnormalities. Both a loss of protective immunity (susceptibility to infection) and a breakdown of tolerance (autoimmunity) may be seen, sometimes in the same patient. In a further ironic twist, individuals with inherited or acquired immunodeficiency are themselves at high risk of developing certain lymphoid neoplasms, particularly those caused by oncogenic viruses (e.g., EBV).
Neoplastic B and T cells tend to recapitulate the behavior of their normal counterparts. Like normal lymphocytes, neoplastic B and T cells express adhesion molecules and chemokine receptors that govern their homing to certain tissue sites, leading to characteristic patterns of involvement. For example, follicular lymphomas home to germinal centers in lymph nodes, whereas cutaneous T-cell lymphomas home to the skin. Variable numbers of neoplastic B and T lymphoid cells also recirculate through the lymphatics and peripheral blood to distant sites; as a result, most lymphoid tumors are widely disseminated at the time of diagnosis. Notable exceptions to this rule include Hodgkin lymphomas, which are sometimes restricted to one group of lymph nodes, and marginal zone B-cell lymphomas, which are often restricted to sites of chronic inflammation.
Hodgkin lymphoma spreads in an orderly stepwise fashion, whereas most forms of NHL disseminate widely and somewhat unpredictably early in their course. Hence, while lymphoma staging provides useful prognostic information, it is of most utility in guiding therapy in Hodgkin lymphoma.
We begin our discussion of specific entities with neoplasms of immature lymphoid cells and then move to mature B-cell tumors, plasma cell neoplasms, and tumors of T-cells and NK-cells. Some of the salient molecular and clinical features of these neoplasms are summarized in Table 13.6 . We will finish by discussing Hodgkin lymphoma.
| Diagnosis | Cell of Origin | Genotype | Salient Clinical Features |
|---|---|---|---|
| Neoplasms of Immature B and T Cells | |||
| B-cell acute lymphoblastic leukemia/lymphoma a | Bone marrow precursor B cell | Diverse chromosomal translocations; t(12;21) involving RUNX1 and ETV 6 present in 25% | Predominantly children; symptoms relating to marrow replacement and pancytopenia; aggressive |
| T-cell acute lymphoblastic leukemia/lymphoma | Precursor T cell (often of thymic origin) | Diverse chromosomal translocations; NOTCH1 mutations (50%–70%) | Predominantly adolescent males; thymic masses and variable bone marrow involvement; aggressive |
| Neoplasms of Mature B Cells | |||
| Burkitt lymphoma a | Germinal center B cell | Translocations involving MYC and Ig loci, usually t(8;14); subset EBV-associated | Adolescents or young adults with extranodal masses; uncommonly presents as “leukemia”; aggressive |
| Diffuse large B-cell lymphoma b | Germinal center or post–germinal center B cell | Diverse chromosomal rearrangements, most often of BCL6 (30%), BCL2 (10%), or MYC (5%) | All ages, but most common in older adults; often appears as a rapidly growing mass; 30% extranodal; aggressive |
| Extranodal marginal zone lymphoma | Memory B cell | t(11;18), t(1;14), and t(14;18) creating MALT1-IAP2, BCL10-IGH, and MALT1-IGH fusion genes, respectively | Arises at extranodal sites in adults with chronic inflammatory diseases; may remain localized; indolent |
| Follicular lymphoma b | Germinal center B cell | t(14;18) creating BCL2-IGH fusion gene | Older adults with generalized lymphadenopathy and marrow involvement; indolent |
| Hairy cell leukemia | Memory B cell | Activating BRAF mutations | Older men with pancytopenia and splenomegaly; indolent |
| Mantle cell lymphoma | Naive B cell | t(11;14) creating cyclin D1– IGH fusion gene | Older men with disseminated disease; moderately aggressive |
| Multiple myeloma/solitary plasmacytoma b | Post–germinal center bone marrow homing plasma cell | Diverse rearrangements involving IGH ; 13q deletions | Myeloma: older adults with lytic bone lesions, pathologic fractures, hypercalcemia, and renal failure; moderately aggressive |
| Plasmacytoma: isolated plasma cell masses in bone or soft tissue; indolent | |||
| Small lymphocytic lymphoma/chronic lymphocytic leukemia | Naive B cell or memory B cell | Trisomy 12, deletions of 11q, 13q, and 17p; NOTCH1 mutations; splicing factor mutations | Older adults with bone marrow, lymph node, spleen, and liver disease; autoimmune hemolysis and thrombocytopenia in a minority; indolent |
| Neoplasms of Mature T Cells or NK Cells | |||
| Adult T-cell leukemia/lymphoma | Helper T cell | HTLV-1 provirus present in tumor cells | Adults with cutaneous lesions, marrow involvement, and hypercalcemia; occurs mainly in Japan, West Africa, and the Caribbean; aggressive |
| Peripheral T-cell lymphoma, unspecified | Helper or cytotoxic T cell | No specific chromosomal abnormality | Mainly older adults; usually presents with lymphadenopathy; aggressive |
| Anaplastic large-cell lymphoma | Cytotoxic T cell | Rearrangements of ALK (anaplastic large cell lymphoma kinase) in a subset | Children and young adults, usually with lymph node and soft tissue disease; aggressive |
| Extranodal NK/T-cell lymphoma | NK-cell (common) or cytotoxic T cell (rare) | EBV-associated; no specific chromosomal abnormality | Adults with destructive extranodal masses, most commonly sinonasal; aggressive |
| Mycosis fungoides/Sézary syndrome | Helper T cell | No specific chromosomal abnormality | Adult patients with cutaneous patches, plaques, nodules, or generalized erythema; indolent |
| Large granular lymphocytic leukemia | Two types: cytotoxic T cell and NK cell | Point mutations in STAT3 | Adult patients with splenomegaly, neutropenia, and anemia, sometimes accompanied by autoimmune disease |
Acute lymphoblastic leukemia/lymphomas (ALLs) are neoplasms composed of immature B (pre-B) or T (pre-T) cells, which are referred to as lymphoblasts. About 85% are B-ALLs, which typically manifest as childhood acute leukemias. The less common T-ALLs tend to present in adolescent males as thymic lymphomas. There is, however, considerable overlap in the clinical behavior of B-ALL and T-ALL; for example, B-ALL uncommonly presents as a mass in the skin or a bone, and many T-ALLs present with or evolve to a leukemic picture. Because of their morphologic and clinical similarities, the various forms of ALL are discussed here together.
ALL is the most common cancer of children. Approximately 2500 new cases are diagnosed each year in the United States, most occurring in individuals younger than 15 years of age. ALL is almost three times more common in Caucasians than in African-Americans and is slightly more frequent in boys than in girls. Hispanics have the highest incidence of any ethnic group. B-ALL peaks in incidence at about the age of 3, perhaps because the number of normal bone marrow pre-B cells (the cell of origin) is greatest very early in life. Similarly the peak incidence of T-ALL is in adolescence, the age when the thymus reaches maximum size. B-ALL and T-ALL also occur less frequently in adults of all ages.
Many of the chromosomal aberrations seen in ALL dysregulate the expression and function of transcription factors required for normal B- and T-cell development. Most T-ALLs have mutations in NOTCH1, a gene that is essential for T-cell development, while a high fraction of B-ALLs have mutations affecting genes such as PAX5, TCF3, ETV6, and RUNX1, all of which are required for the proper differentiation of early hematopoietic precursors. By disturbing the expression and function of “master” regulatory factors, these mutations promote maturation arrest and increased self-renewal, a stem cell–like phenotype. Similar themes are relevant in the genesis of AML (discussed later).
In keeping with the multistep origin of cancer ( Chapter 7 ), mutations in transcription factor genes are not sufficient to produce ALL. The identity of other driver mutations is incomplete, but aberrations that promote cell growth, such as mutations that increase tyrosine kinase activity and RAS signaling, are commonly present. Emerging data from deep sequencing of ALL genomes suggest that fewer than 10 mutations are sufficient to produce full-blown ALL; hence compared to solid tumors, ALL is genetically simple.
Approximately 90% of ALLs have numerical or structural chromosomal changes. Most common is hyperploidy (>50 chromosomes), but hypoploidy and a variety of balanced chromosomal translocations also are seen. Changes in chromosome numbers are of uncertain pathogenic significance but are important because they frequently correlate with immunophenotype and sometimes prognosis. For example, hyperdiploidy and hypodiploidy are seen only in B-ALL and are associated with better and worse prognoses, respectively. In addition, B-ALL and T-ALL are associated with completely different sets of translocations; thus, while morphologically identical, they are genetically quite distinct.
In leukemic presentations, the marrow is hypercellular and packed with lymphoblasts, which replace normal marrow elements. Mediastinal thymic masses occur in 50% to 70% of T-ALLs, which are also more likely than B-ALL to be associated with lymphadenopathy and splenomegaly. In both B-ALL and T-ALL, the tumor cells have scant basophilic cytoplasm and nuclei somewhat larger than those of small lymphocytes ( Fig. 13.6A ). The nuclear chromatin is delicate and finely stippled, and nucleoli are usually small and often demarcated by a rim of condensed chromatin. The nuclear membrane is often deeply subdivided, imparting a convoluted appearance. In keeping with the aggressive clinical behavior, the mitotic rate is high. As with other rapidly growing lymphoid tumors, interspersed macrophages ingesting apoptotic tumor cells may impart a “starry sky” appearance (shown in Fig. 13.15 ).
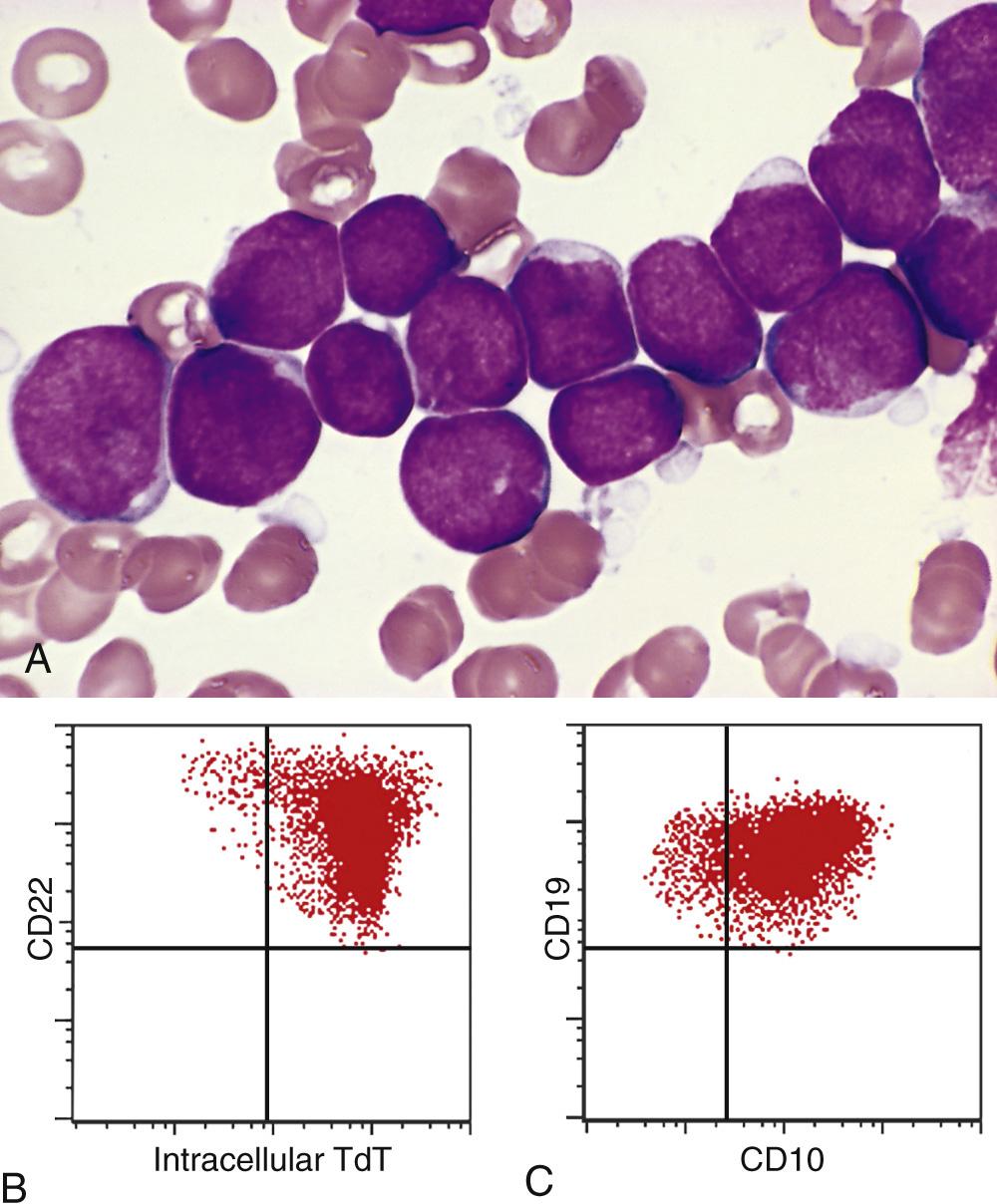
Because of their different responses to chemotherapy, ALL must be distinguished from AML, a neoplasm of immature myeloid cells that can cause identical signs and symptoms. Compared with myeloblasts, lymphoblasts have more condensed chromatin, less conspicuous nucleoli, and smaller amounts of cytoplasm that usually lacks granules. However, these morphologic distinctions are not absolute, and definitive diagnosis relies on stains performed with antibodies specific for B- and T-cell antigens ( Fig. 13.6B and C ). Histochemical stains are also helpful, in that (in contrast to myeloblasts) lymphoblasts are myeloperoxidase-negative and often contain periodic acid–Schiff–positive cytoplasmic material.
Immunostaining for terminal deoxynucleotidyl transferase (TdT), a specialized DNA polymerase that is expressed only in pre-B and pre-T lymphoblasts, is positive in more than 95% of cases ( Fig. 13.6B ). B-ALLs are arrested at various stages of pre–B-cell development, which correlate with the expression of certain proteins. The lymphoblasts usually express the pan B-cell marker CD19 and the transcription factor PAX5 as well as CD10. In very immature B-ALLs, CD10 is negative. Alternatively, more mature “late pre-B” ALLs express CD10, CD19, CD20, and cytoplasmic IgM heavy chain (µ chain). Similarly, T-ALLs are arrested at various stages of pre–T-cell development. In most cases the cells are positive for CD1, CD2, CD5, and CD7. The more immature tumors are usually negative for surface CD3, CD4, and CD8, whereas “late” pre–T-cell tumors are positive for these markers.
Although ALLs and AMLs are genetically and immunophenotypically distinct, they are clinically very similar. In both, the accumulation of neoplastic “blasts” in the bone marrow suppresses normal hematopoiesis by physical crowding, competition for growth factors, and other poorly understood mechanisms. The common features and those more characteristic of ALL are the following:
Abrupt stormy onset within days to a few weeks of the first symptoms.
Symptoms related to depression of marrow function, including fatigue due to anemia; fever, reflecting infections secondary to neutropenia; and bleeding due to thrombocytopenia.
Mass effects caused by neoplastic infiltration (which are more common in ALL), including bone pain resulting from marrow expansion and infiltration of the subperiosteum; generalized lymphadenopathy, splenomegaly, and hepatomegaly; testicular enlargement; and, in T-ALL, complications related to compression of large vessels and airways in the mediastinum.
Central nervous system manifestations such as headache, vomiting, and nerve palsies resulting from meningeal spread, all of which are also more common in ALL.
Treatment of pediatric ALL is one of the great success stories of oncology. With aggressive chemotherapy about 95% of children with ALL obtain a complete remission, and 75% to 85% are cured. Despite these achievements, however, ALL remains a leading cause of cancer deaths in children, and only 35% to 40% of adults are cured. Several factors are associated with a worse prognosis: (1) age younger than 2 years, largely because of the strong association of infantile ALL with translocations involving the MLL gene; (2) presentation in adolescence or adulthood; and (3) peripheral blood blast counts greater than 100,000, which probably reflects a high tumor burden. Favorable prognostic markers include (1) age between 2 and 10 years; (2) a low white cell count; (3) hyperdiploidy; (4) trisomy of chromosomes 4, 7, and 10; and (5) the presence of a t(12;21). Notably, the molecular detection of residual disease after therapy is predictive of a worse outcome in both B-ALL and T-ALL and is being used to guide clinical trials.
Although most chromosomal aberrations in ALL alter the function of transcription factors, the t(9;22) instead creates a fusion gene that encodes a constitutively active BCR-ABL tyrosine kinase (described in more detail under Chronic Myeloid Leukemia ). In B-ALL, the BCR-ABL protein is usually 190 kDa in size and has stronger tyrosine kinase activity than the form of BCR-ABL that is found in chronic myeloid leukemia, in which a BCR-ABL protein of 210 kDa in size is usually seen. Treatment of t(9;22)-positive ALLs with BCR-ABL kinase inhibitors in combination with conventional chemotherapy is highly effective and has greatly improved the outcome for this molecular subtype of B-ALL in children and adults. Of interest, cryptic rearrangements involving genes encoding tyrosine kinases other than ABL also have been described in B-ALL, particularly in adults, and these too may be therapeutic targets. The outlook for adults with ALL lacking “targetable” molecular lesions remains more guarded, in part because of differences in the molecular pathogenesis of adult and childhood ALL, and also because older adults cannot tolerate the intensive chemotherapy regimens that are curative in children. Dramatic responses in B-ALL have been achieved with chimeric antigen receptor T cells directed against the B-cell antigen CD19 ( Chapter 7 ), but at high cost and with associated toxicities that have sometimes proved fatal.
Most common type of cancer in children; may be derived from either precursor B or T cells.
Highly aggressive tumors manifest with signs and symptoms of bone marrow failure or as rapidly growing masses.
Tumor cells contain genetic lesions that block differentiation, leading to the accumulation of immature, nonfunctional blasts.
A subset of tumors contains activating mutations in tyrosine kinases (e.g., BCR-ABL) that are important targets of therapy.
Chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL) differ only in the degree of peripheral blood lymphocytosis. Most affected patients have sufficient lymphocytosis to fulfill the diagnostic requirement for CLL (absolute lymphocyte count >5000/mm 3 ). CLL is the most common leukemia of adults in the Western world. There are about 15,000 new cases of CLL each year in the United States. The median age at diagnosis is 60 years, and there is a 2:1 male predominance. In contrast, SLL constitutes only 4% of NHLs. CLL/SLL is much less common in Japan and other Asian countries than in the West.
Unlike most other lymphoid malignancies, chromosomal translocations are rare in CLL/SLL. The most common genetic anomalies are deletions of 13q14.3, 11q, and 17p and trisomy 12q. Molecular characterization of the region deleted on chromosome 13 has implicated two microRNAs, miR-15a and miR-16-1, tumor suppressor genes. Loss of these mIRs is believed to result in overexpression of the anti-apoptotic protein BCL2, which is uniformly observed in CLL/SLL. DNA sequencing has revealed that the Ig genes of some CLL/SLL are somatically hypermutated, whereas others are not, suggesting that the cell of origin may be either a post–germinal center memory B cell or a naive B cell. For unclear reasons, tumors with unmutated Ig segments (those putatively of naive B-cell origin) pursue a more aggressive course. Deep sequencing of CLL genomes has also revealed gain-of-function mutations involving the NOTCH1 receptor in 10% to 18% of tumors, as well as frequent mutations in genes that regulate RNA splicing.
The growth of CLL/SLL cells is largely confined to proliferation centers (described later), where tumor cells receive critical cues from the microenvironment. Stromal cells in proliferation centers express a variety of factors that stimulate the activity of the transcription factors nuclear factor kappa B (NF-κB), which promotes cell survival, and MYC, which promotes cell growth. Other critical signals are generated by the B-cell receptor (membrane bound Ig), which triggers a signaling pathway that includes Bruton tyrosine kinase (BTK). The importance of BTK in B cells is emphasized by congenital X-linked agammaglobulinemia ( Chapter 6 ), a failure of B-cell development that is caused by loss-of-function mutations in the BTK gene. Notably, BTK inhibitors produce sustained therapeutic responses in a high fraction of CLL patients, indicating that CLL cells depend on B-cell receptor signaling and BTK activity for their growth and survival.
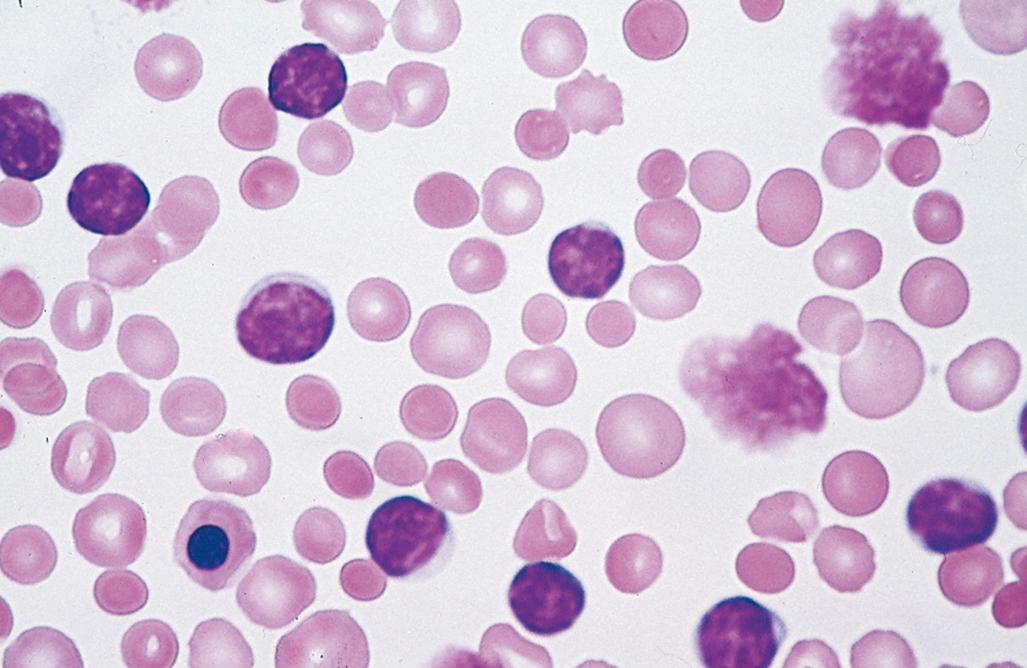
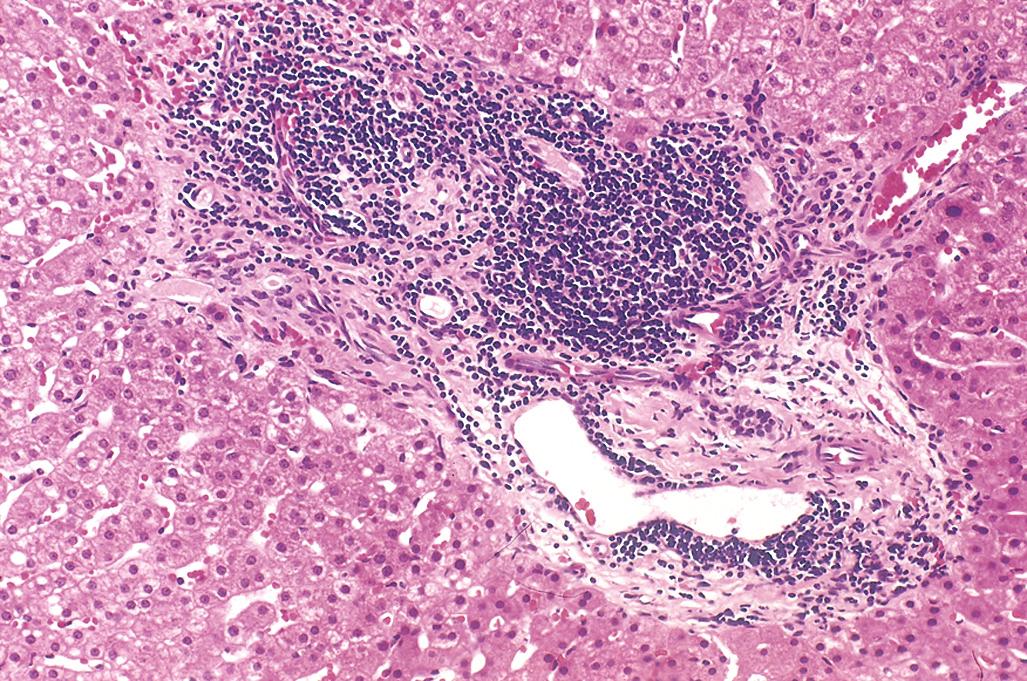
Lymph nodes are diffusely effaced by predominantly small lymphocytes 6 to 12 µm in diameter with round to slightly irregular nuclei, condensed chromatin, and scant cytoplasm ( Fig. 13.7 ). Admixed are variable numbers of larger activated lymphocytes that often gather in loose aggregates referred to as proliferation centers that contain mitotically active cells. When present, proliferation centers are pathognomonic for CLL/SLL. The blood contains variable numbers of small round lymphocytes with scant cytoplasm ( Fig. 13.8 ). Some of these cells are usually disrupted in the process of making smears, producing so-called smudge cells. In almost all cases, the bone marrow, spleen, and liver ( Fig. 13.9 ) also are involved, albeit to widely varying degrees.

CLL/SLL has a distinctive immunophenotype. The tumor cells express the pan B-cell markers CD19 and CD20, as well as CD23 and CD5, the latter a marker that is found on a small subset of normal B cells. Low-level expression of surface Ig (usually IgM or IgM and IgD) is also typical, as is high-level expression of BCL2.
Patients are often asymptomatic at diagnosis. When symptoms appear, they are nonspecific and include easy fatigability, weight loss, and anorexia. Generalized lymphadenopathy and hepatosplenomegaly are present in 50% to 60% of symptomatic patients. The leukocyte count is highly variable; leukopenia can be seen in individuals with SLL and marrow involvement, while counts in excess of 200,000/mm 3 are sometimes seen in CLL patients with heavy tumor burdens. A small monoclonal Ig “spike” is present in the blood of some patients. At the other end of the spectrum are asymptomatic patients with monoclonal B cells in their peripheral blood that number too few to merit the diagnosis of CLL. This condition, referred to a monoclonal lymphocytosis of uncertain significance, is considered to be a precursor lesion and progresses to symptomatic CLL at a rate of 1% per year.
CLL/SLL disrupts normal immune function through uncertain mechanisms. Hypogammaglobulinemia is common and contributes to an increased susceptibility to infection, particularly those caused by bacteria. Conversely, 10% to 15% of patients develop hemolytic anemia or thrombocytopenia due to autoantibodies made by nonneoplastic B cells.
The course and prognosis are extremely variable and depend primarily on the clinical stage. Overall median survival is 4 to 6 years but is more than 10 years in individuals with minimal tumor burden at diagnosis. Other variables that correlate with a worse outcome include (1) the presence of deletions of 11q and 17p (the latter involving TP53 ), (2) a lack of somatic hypermutation, (3) the expression of ZAP-70, a protein that augments signals produced by the Ig receptor, and (4) the presence of NOTCH1 mutations. Symptomatic patients are generally treated with “gentle” chemotherapy and immunotherapy with antibodies against proteins found on the surface of CLL/SLL cells, particularly CD20. Newly available, highly active targeted therapies include BTK inhibitors and BCL2 inhibitors; their impact on the disease course is still being determined.
Another factor that impacts patient survival is the tendency of CLL/SLL to transform to a more aggressive tumor. Most commonly this takes the form of a transformation to diffuse large B-cell lymphoma (DLBCL), so-called Richter syndrome (approximately 5% to 10% of patients). Richter syndrome is often heralded by the development of a rapidly enlarging mass within a lymph node or the spleen. It is often associated with acquisition of new abnormalities involving TP53 or MYC and is an ominous event, with most patients surviving less than 1 year.
Follicular lymphoma is the most common form of indolent NHL in the United States, trailing only diffuse large B-cell lymphoma (discussed later) in frequency among lymphomas. It affects 15,000 to 20,000 individuals per year. It usually presents in middle age and afflicts males and females equally. It is less common in Europe and rare in Asian populations.
Follicular lymphoma is strongly associated with chromosomal translocations involving BCL2. Its hallmark is a (14;18) translocation that juxtaposes the IGH locus on chromosome 14 and the BCL2 locus on chromosome 18. The t(14;18) is seen in up to 90% of follicular lymphomas and leads to overexpression of BCL2 (see Fig. 13.12 ). BCL2 antagonizes apoptosis ( Chapters 2 and 7 ) and promotes the survival of follicular lymphoma cells. Notably, while normal germinal centers contain numerous B cells undergoing apoptosis, follicular lymphoma is characteristically devoid of apoptotic cells. Deep sequencing of follicular lymphoma genomes have identified mutations in the KMT2D gene in about 90% of cases as well. KMT2D encodes a histone methyltransferase, suggesting that epigenetic abnormalities such as changes in the patterns of histone marks have an important role in this neoplasm.
Particularly early in the disease, follicular lymphoma cells growing in lymph nodes are found within a network of reactive follicular dendritic cells admixed with macrophages and T cells. Expression profiling studies have shown that differences in the genes expressed by these stromal cells are predictive of outcome, implying that the response of follicular lymphoma cells to therapy is influenced by the surrounding microenvironment.
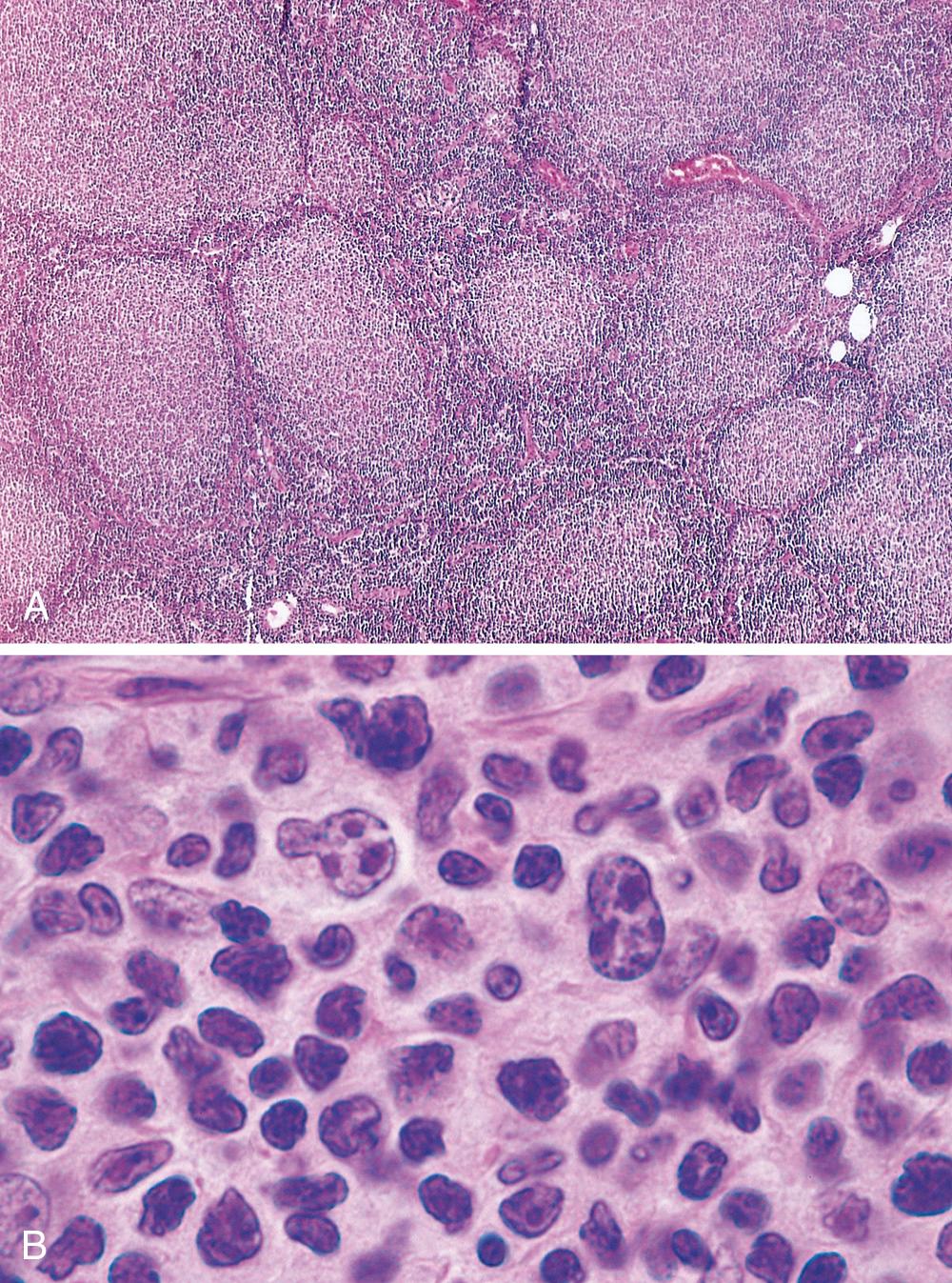
In most cases, a nodular or nodular and diffuse growth pattern is observed in involved lymph nodes ( Fig. 13.10A ). Two principal cell types are present in varying proportions: (1) small cells with irregular or cleaved nuclear contours and scant cytoplasm, referred to as centrocytes (small cleaved cells), and (2) larger cells with open nuclear chromatin, several nucleoli, and modest amounts of cytoplasm, referred to as centroblasts ( Fig. 13.10B ). In most follicular lymphomas, centrocytes are in the majority. Peripheral blood involvement sufficient to produce lymphocytosis (usually <20,000 cells/mm 3 ) is seen in about 10% of cases. Bone marrow involvement occurs in 85% of cases and characteristically takes the form of paratrabecular lymphoid aggregates. The splenic white pulp ( Fig. 13.11 ) and hepatic portal triads are also frequently involved.
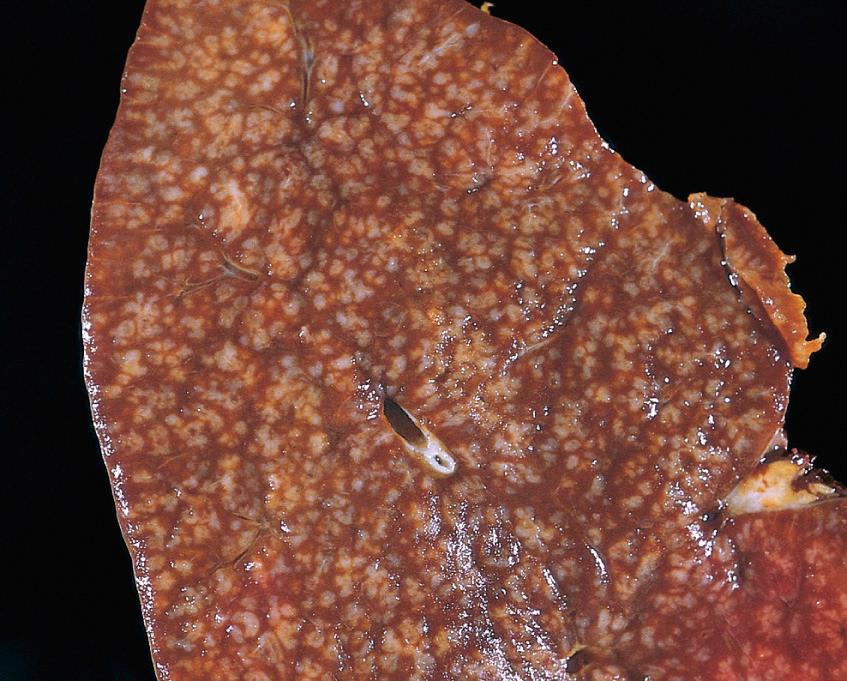
The neoplastic cells closely resemble normal germinal center B cells, expressing CD19, CD20, CD10, surface Ig, and BCL6. Unlike CLL/SLL and mantle cell lymphoma, CD5 is not expressed. BCL2 is expressed in more than 90% of cases, in distinction to normal follicular center B cells, which are BCL2-negative ( Fig. 13.12 ).
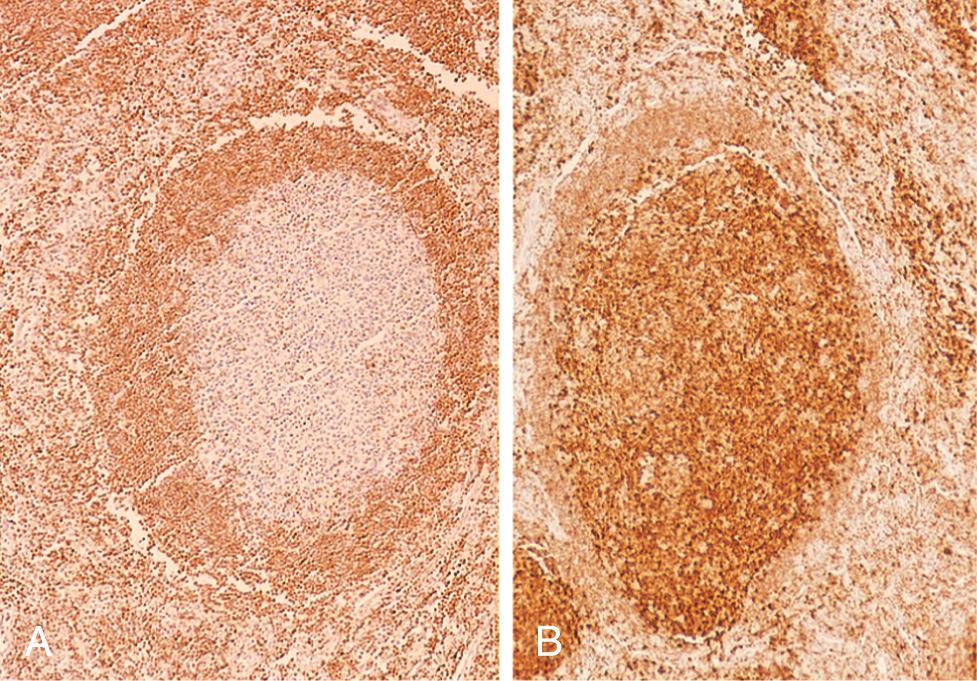
Follicular lymphoma tends to present with painless, generalized lymphadenopathy. Involvement of extranodal sites, such as the gastrointestinal tract, central nervous system, or testis, is relatively uncommon. Although incurable, it usually follows an indolent waxing and waning course. Survival (median, 7 to 9 years) is not improved by aggressive therapy; hence the usual approach is to palliate patients with low-dose chemotherapy or immunotherapy (e.g., anti-CD20 antibody) when they become symptomatic. Like CLL, it also is responsive to inhibitors of B-cell receptor signaling (e.g., BTK inhibitors) and inhibitors of BCL2.
Histologic transformation occurs in 30% to 50% of follicular lymphomas, most commonly to DLBCL. These transformation events are frequently associated with aberrations that increase the expression of MYC, which you will recall drives Warburg metabolism and rapid cell growth. Follicular lymphomas show evidence of ongoing somatic hypermutation, which may promote transformation by causing point mutations or chromosomal aberrations. The median survival is less than 1 year after transformation.
Diffuse large B-cell lymphoma (DLBCL) is the most common form of NHL. Each year in the United States there are about 25,000 new cases. There is a slight male predominance. The median patient age is about 60 years, but DLBCL also occurs in young adults and children.
Genetic, gene expression profiling, and immunohistochemical studies indicate that DLBCL is molecularly heterogeneous. One frequent pathogenic event is dysregulation of BCL6, a DNA-binding zinc-finger transcriptional repressor that is required for the formation of normal germinal centers. About 30% of DLBCLs contain various translocations that have in common a breakpoint in BCL6 at chromosome 3q27. Acquired mutations in BCL6 promoter sequences that abrogate BCL6 autoregulation (an important negative-regulatory mechanism) are seen even more frequently. It is hypothesized that both types of lesions are inadvertent by-products of somatic hypermutation that result in overexpression of BCL6, which has several important consequences. BCL6 represses the expression of factors that normally serve to promote germinal center B-cell differentiation, growth arrest, and apoptosis, and each of these effects is believed to contribute to the development of DLBCL. Mutations similar to those found in BCL6 are also seen in multiple other oncogenes, including MYC, suggesting that somatic hypermutation in DLBCL cells is “mistargeted” to a wide variety of loci.
Another 10% to 20% of tumors are associated with the t(14;18) (discussed earlier under Follicular Lymphoma), which leads to the overexpression of the antiapoptotic protein BCL2. Tumors with BCL2 rearrangements usually lack BCL6 rearrangements, suggesting that these rearrangements define two distinct molecular classes of DLBCL. Some tumors with BCL2 rearrangements may arise from unrecognized underlying follicular lymphomas, which frequently transform to DLBCL. Roughly 5% of DLBCLs are associated with translocations involving MYC; these tumors may have a distinctive biology (discussed later under Burkitt lymphoma). Finally, sequencing of DLBCL genomes has identified frequent mutations in genes encoding histone acetyltransferases such as p300 and CREBP, proteins that regulate gene expression by modifying histones and altering chromatin structure.
The common features are a relatively large cell size (usually four to five times the diameter of a small lymphocyte) and a diffuse pattern of growth ( Fig. 13.13 ). Other morphologic features show substantial variation. Most commonly, the tumor cells have a round or oval nucleus that appears vesicular due to margination of chromatin to the nuclear membrane, but large multilobated or irregular nuclei are prominent in some cases. Nucleoli may be two to three in number and located adjacent to the nuclear membrane or single and centrally placed. The cytoplasm is usually moderately abundant and may be pale or basophilic. More anaplastic tumors may contain multinucleated cells with large inclusion-like nucleoli that resemble Reed-Sternberg cells (the malignant cell of Hodgkin lymphoma).
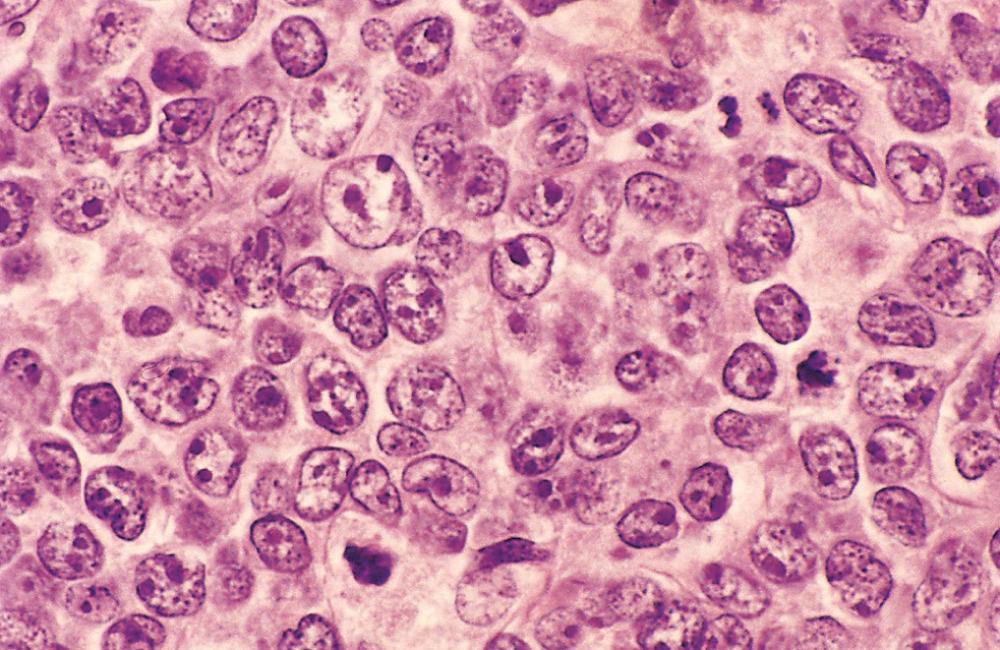
These mature B-cell tumors express CD19 and CD20 and show variable expression of germinal center B-cell markers such as CD10 and BCL6. Most have surface Ig. High-level expression of both MYC and BCL2 proteins is seen in some cases and may predict more aggressive behavior.
Several subtypes of DLBCL are sufficiently distinctive to merit brief discussion.
Immunodeficiency-associated large B-cell lymphoma occurs in the setting of T-cell immunodeficiency (e.g., in advanced HIV infection and in recipients of organ or HSC transplants). The neoplastic B cells are usually infected with EBV, which plays a critical pathogenic role. Restoration of T-cell immunity may lead to regression of these proliferations.
Primary effusion lymphoma presents as a malignant pleural or ascitic effusion, mostly in patients with advanced HIV infection or in older adults. The tumor cells are often anaplastic in appearance and typically fail to express surface B- or T-cell markers, but have clonal IGH gene rearrangements. In all cases the tumor cells are infected with HHV-8, which appears to have a causal role.
DLBCL typically presents as a rapidly enlarging mass at a nodal or extranodal site. It can arise virtually anywhere in the body. Waldeyer ring, the oropharyngeal lymphoid tissue that includes the tonsils and adenoids, is involved commonly. Primary or secondary involvement of the liver and spleen may take the form of large destructive masses ( Fig. 13.14 ). Extranodal sites include the gastrointestinal tract, skin, bone, brain, and other tissues. Bone marrow involvement is relatively uncommon and usually occurs late in the course. Rarely, a leukemic picture emerges.
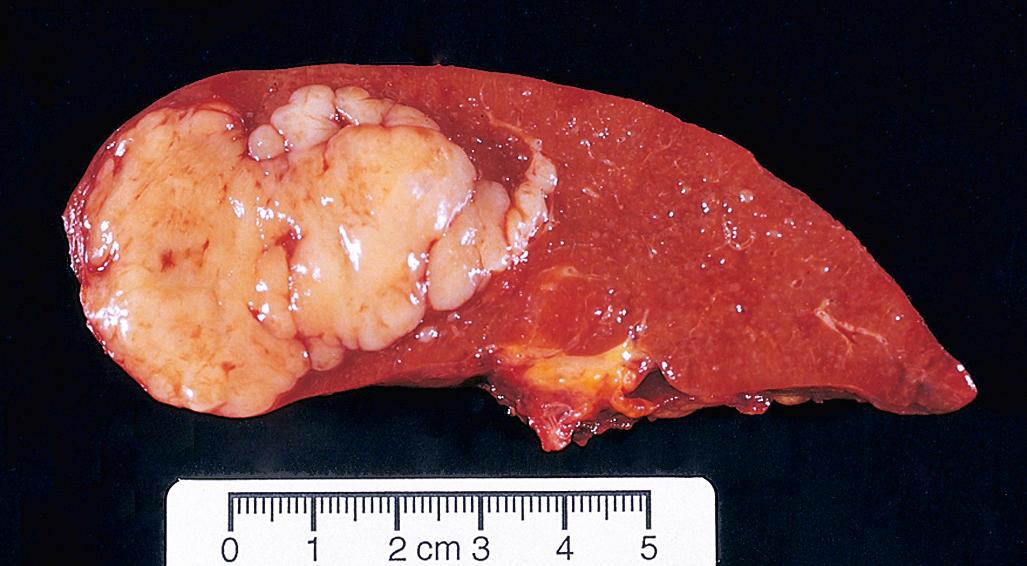
DLBCLs are aggressive tumors that are rapidly fatal without treatment. With intensive combination chemotherapy, 60% to 80% of patients achieve a complete remission, and 40% to 50% are cured. Adjuvant therapy with anti-CD20 antibody improves both the initial response and the overall outcome. Individuals with limited disease fare better than those with widespread disease or bulky tumor masses. Expression profiling has identified several distinct molecular subtypes, including one resembling germinal center B cells and a second resembling activated post–germinal center B cells, each with differing clinical outcomes. DLBCLs with MYC translocations have a worse prognosis than those without and may be better treated with chemotherapy regimens that are now standard for Burkitt lymphoma. CAR T cells directed against the B-cell antigen CD19 are now available for the treatment of patients with relapsed refractory DLBCL.
Within the category of Burkitt lymphoma fall (1) African (endemic) Burkitt lymphoma, (2) sporadic (nonendemic) Burkitt lymphoma, and (3) a subset of aggressive lymphomas occurring in individuals infected with HIV. Burkitt lymphomas occurring in these three settings are histologically identical but have distinct clinical, genotypic, and virologic characteristics.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here