Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Uncontrolled hypertension presents a significant health care problem with far-reaching public health and economic consequences.
Novel therapies target neurohumoral modulation and mechanical alteration of vascular compliance to treat hypertension.
Reduction of the sympathetic nervous output from the renal nerves has some supporting clinical data but remains experimental at the time of this publication.
A variety of technologies can be used to modulate renal sympathetic tone through destruction of renal nerves with the most common being radiofrequency ablation.
Hypertension (HTN) is estimated to be present in 77.9 million adults and 60% to 75% of patients older than 60 years. Globally, 51% of stroke and 45% of ischemic heart disease are attributable to HTN. HTN-related stroke, cardiac disease, and renal failure are preventable with adequate lowering of blood pressure (BP), which may be achieved across several modalities.
Surveys suggest that in the United States, nearly 50% of identified and treated hypertensive patients have systolic blood pressure (SBP) >140 mm Hg, and 13% (26% of the uncontrolled) have systolic BPs of >160 mm Hg. Despite the wide availability of effective drugs for BP control, as much as 51% of patients have low adherence, in turn resulting in 38% increased rate of cardiovascular (CV) morbidity relative to those compliant with medications. Even in clinical trial settings, only 57% of patients elect to persist and adhere to medication recommendations after 2 years, signaling the futility of relying on patient compliance for chronic HTN management. Furthermore, a subset of patients who choose to be compliant with complex multidrug formulations, remain above target pressures, and are designated resistant hypertensive patients. Despite compliance with medications, patients with BP above clinical targets continue to have excess CV mortality, stroke, myocardial infarction, and onset of renal insufficiency. Treatment strategies to complement pharmacotherapy to reduce BP would address a large unmet clinical need and potentially reduce the CV morbidity of uncontrolled HTN among patients either nonadherent or resistant to HTN pharmacotherapy.
The pathophysiology of HTN is multifactorial with neurohumoral activation making a significant contribution to its genesis ( Fig. 43.1 ). Secondary pathologies such as arteriosclerosis and loss of vascular compliance further accelerate disease progression. Consequently, a treatment approach addressing the separate components of the pathophysiology resulted in the emergence of several separate and perhaps complimentary device driven strategies: sympathetic nervous modulation to decrease downstream maladaptive events in vascular tone and sodium balance or mechanical alteration of the systemic vascular resistance using an arterial-venous anastomosis to change vascular compliance.
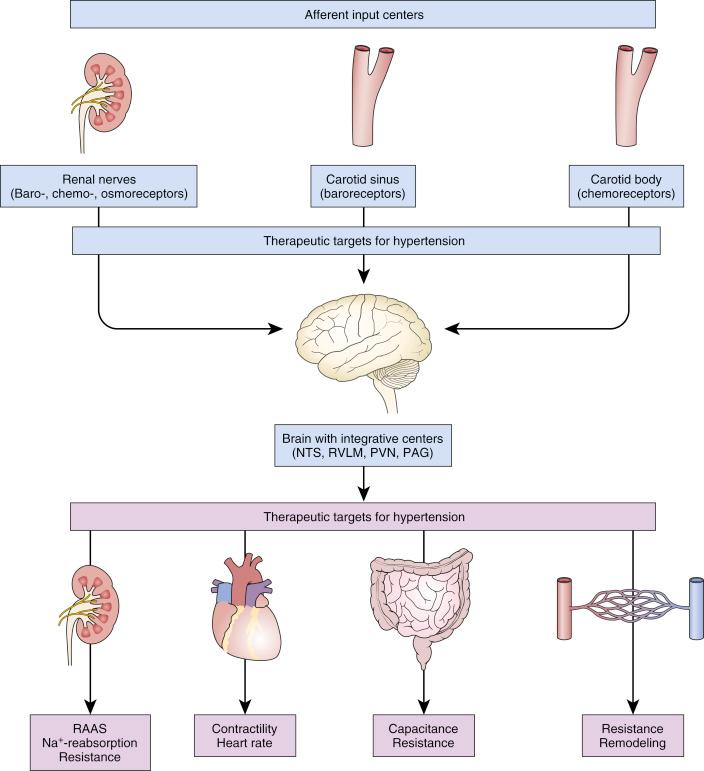
Maintenance of body fluid composition, volume, and vascular tone is a complex integrated process regulated by the autonomic nervous system that is heavily renal driven. The sympathetic nervous system (SNS) acquires signals reflecting changes in renal hydrostatic pressure and chemistry and processes information in the central SNS, and efferent signals from the central nervous system adjust renal contribution to vascular tone and volume homeostasis. Afferents nerve endings from the kidney vasculature and parenchyma relay information to the ipsilateral cell bodies in the dorsal root ganglia to the posterior gray column, and finally the brain stem and mid-brain integrate afferent signals from multiple end-organ sensors and baroreceptors ( Fig. 43.2 ). Efferent fibers from the central nervous system extend to the sympathetic preganglionic neurons in the intermediolateral cell column. At approximately T10 to T12 and L1 to L2, splanchnic nerves connect via prevertebral ganglia (e.g., celiac and mesenteric) to postganglionic neurons and communicate to the kidneys bilaterally. In addition, the renal nervous system may reflexively autoregulate without central nervous system output.
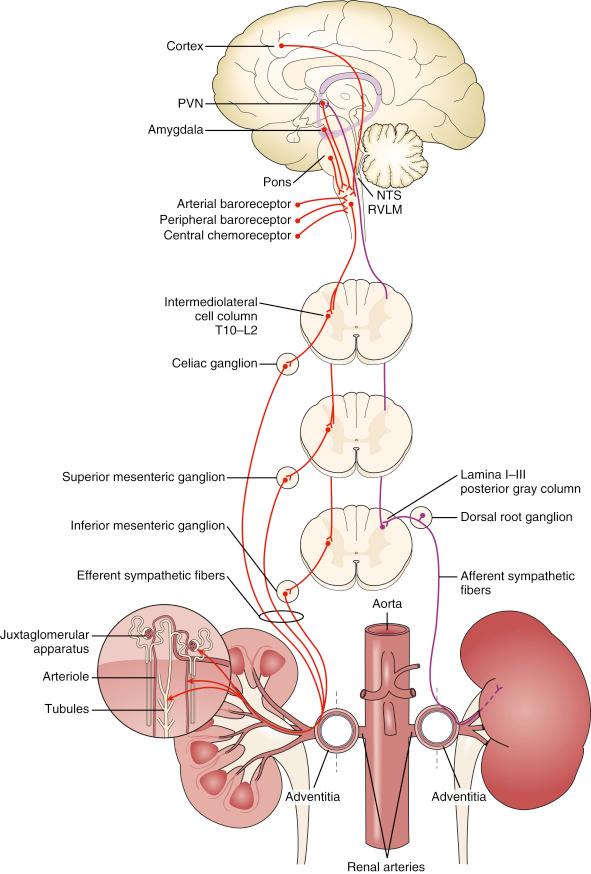
Efferent and afferent nerves function in a reflex loop where afferent signals from the kidney to the central nervous system regulate the efferent sympathetic nerve output back to the kidney. Renal afferent nerves are complex and diverse because they represent a heterogeneous population of fibers including myelinated and nonmyelinated fibers. Two main physiologic types of renal afferents are responsible for signaling information to the central nervous system, mechanosensitive and chemosensitive receptors. Mechanosensitive receptors transmit hydrostatic data from the renal arteries, veins, and pelvis. Renal ischemia, ionic composition, adenosine, osmolar concentration, oxidative stress, and chemicals (i.e., bradykinin) activate renal chemoreceptors. Functionally, the renal afferent fibers can be divided in (1) pressor, (2) renorenal, and (3) depressor types. This means that some fibers contribute to the elevation of the sympathetic tone (sympathostimulant), whereas others reduce the sympathetic tone (sympatholytic). This is supported by increasing preclinical and clinical evidence suggesting that endovascular electrical mapping with stimulation of renal arteries can result in a differential response including an elevation in BP, no effect, and in some instances a reduction in BP. Several investigators have suggested a specific anatomic distribution of the functional fibers, with preferentially sympathostimulatory fibers in the proximal renal artery and sympathoinhibitory fibers more distal in the renal hilum.
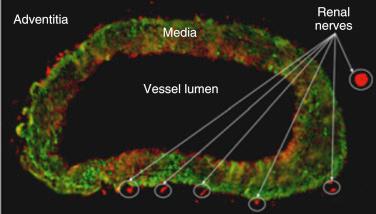
The efferent arm of the reflex loop innervates renal tubules, the juxtaglomerular apparatus, and renal vasculature. Increased renal sympathetic efferent activity stimulates the β1 receptor of the juxtaglomerular apparatus to release renin. Alpha-1B subtype receptor stimulation increases sodium resorption. Alpha-1A stimulation vasoconstricts renal arterioles, decreasing renal blood flow. Graded response of low- or high-frequency signals from the central nervous system modify renal activity. Coincidentally, the efferents are colocated with the afferents in the renal adventitia, providing the anatomic feasibility for percutaneous ablation technology ( Fig. 43.3 ).
Of the vast preclinical research on renal sympathetic innervation in HTN, a few studies merit mention. Electrical stimulation of feline and canine renal afferents increases vasoconstriction-mediated BPs rises. Interruption of afferent fibers via dorsal rhizotomy in a rodent nephrectomy model of chronic renal failure model causes a decrease in norepinephrine (NE) turnover, attenuates HTN, and improves renal function confirming the renal contribution to BP regulation. Surgical renal denervation (RDN) across several HTN models consistently lowers BP and improves natriuresis.
Prior to the development of antihypertensive pharmacotherapy, surgical thoracolumbar splanichnectomy served as a therapeutic option for reducing BP. A large, prospective, observational study where 1266 and 467 patients undergoing splanchnectomy and medical therapy, respectively, showed durable BP lowering in conjunction with improved 5-year survival (surgical 81% vs. medical 46%). In addition, symptomatic relief for headaches and precordial pain, reduction in cardiac size, and renal function improvement were observed at the cost of significant operative mortality (5%). Subsequent studies in nephrectomized patients observed postsurgical reductions in resting sympathetic tone corresponding to substantial lowering of BP, reinforcing the pathophysiological evidence for modulating renal sympathetic output as a treatment for HTN.
Sympathetic modulation for treatment of HTN was rediscovered, and various methodologies have been explored to ablate renal nerves. Given a lack of techniques to screen for different functional afferent renal fiber types, the initial development targeted an undifferentiated global renal denervation. The most developed technology is endovascular radiofrequency (RF) ablation. Initial devices use unipolar or bipolar RF ablation in repeated spiral patterns in energy levels, attempting to achieve sufficient depth to reach the adventitia and disrupt nerve traffic. Delivery of pharmacoablative agents or transmission of focused ultrasonic energy to ablate renal nerves are investigational at the time this chapter is authored.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here