Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Kidney malformations account for 20% to 30% of all prenatally diagnosed developmental anomalies and are responsible for 31% of all childhood end-stage kidney disease.
Patients with unilateral kidney agenesis are at risk for hypertension in childhood and chronic kidney disease in adulthood.
Multicystic dysplastic kidney typically presents as a collection of large kidney cysts on ultrasound, and patients generally do well. Autosomal recessive polycystic kidney disease presents as bilaterally enlarged hyperechoic kidneys, often without cysts, and patients may have significant pulmonary and kidney complications.
The outcome for patients with kidney dysplasia depends significantly on the amount of functioning kidney tissue and the associated congenital anomalies.
Infant dialysis has improved rapidly over recent years, and 2-year survival outcomes as high as 80% are now reported by multinational dialysis databases.
Normal kidney development begins with two precursor organs, the pronephros and mesonephros, followed by the definitive metanephros at approximately 5 weeks’ gestation. The metanephros develops from an interaction between a branch of the mesonephric (wolffian) duct, called the ureteric bud, and the surrounding metanephric mesenchyme. The repeatedly branching ureteric bud will become the collecting system, including the collecting ducts, kidney pelvis, ureter, and bladder trigone. The metanephric mesenchyme will form the glomerular podocytes, proximal tubule, loop of Henle, and distal convoluted tubule. Nephrogenesis begins at 9 weeks’ gestation and is complete by 32 to 36 weeks’ gestation. A complex and carefully timed web of signaling “cross-talk” between the mesonephric duct, metanephric mesenchyme, and surrounding tissue is necessary to coordinate proper kidney development. Any interruption of this process can lead to abnormalities in kidney structure and function.
Developmental abnormalities of the kidney and the urinary tract affect 3 to 6 per 1000 births and account for 31% of all children with end-stage renal disease (ESRD) in the United States. Renal malformations represent 20% to 30% of all prenatally diagnosed developmental anomalies; prenatal ultrasound allows malformations that are asymptomatic at birth to be detected. Long-term outcomes for children with development anomalies of the kidney depend on the amount of functional renal tissue at birth, associated urinary tract anomalies, degree of prematurity, urine output, and presence of additional congenital anomalies.
With advances in neonatal critical care, nutritional therapy, and peritoneal dialysis techniques, many children who previously would have died in early infancy are surviving, growing, and receiving a kidney transplant. Reviews of multinational dialysis databases have reported 2-year survival rates as high as 80% in infants who start dialysis. Successful provision of care requires a multidisciplinary team approach that includes the neonatologist or pediatrician, nephrologist, surgeon, urologist, nutritionist, nurse, and social worker. Above all, it requires close communication with and strong support of the child's family, who may bear enormous emotional and financial burdens in caring for their child.
Unilateral kidney agenesis, or a congenital solitary kidney, results from a unilateral early and complete failure in the signaling interaction between the ureteric bud and metanephric mesenchyme. The estimated incidence is 1 in 500 to 1 in 1000 children. Approximately 30% to 40% of children with unilateral kidney agenesis will have other developmental anomalies; approximately 25% will have vesicoureteral reflux, with the remainder having ureter-pelvic junction obstruction and gastrointestinal, cardiac, and musculoskeletal anomalies.
Kidney agenesis was once thought to be a benign condition if the contralateral kidney appeared healthy. However, there is now increasing concern that unilateral kidney agenesis increases the risk for chronic kidney disease in adulthood. The “hyperfiltration” hypothesis suggests that compensatory hyperfiltration in the healthy kidney leads to single-nephron hypertension, glomerulosclerosis, and nephron loss over time. Recent studies of outcomes for children with congenital solitary kidney identified proteinuria and hypertension in 5% to 25% of patients during childhood, while chronic kidney disease affects approximately 13% to 30% of adults with a solitary kidney. The presence of other urinary tract abnormalities or any dysplasia in the remaining kidney worsens the prognosis.
Unilateral kidney agenesis is asymptomatic in infants with a healthy, nonobstructed contralateral kidney. The routine use of voiding cystourethrogram (VCUG) to screen for vesicoureteral reflux in infants without evidence of a urinary tract infection is now controversial. Children with unilateral kidney agenesis should be monitored for appropriate compensatory hypertrophy of the healthy kidney during infancy and should have annual screening for proteinuria and hypertension during childhood ( Table 75.1 ).
| Syndrome | Inheritance | Genes | Kidney Disease (%) | Type of Kidney Involvement | Key Features |
|---|---|---|---|---|---|
| Fraser | AR | FRAS1, FREM1, FREM2, GRIP1 | 67 | Bilateral or unilateral kidney agenesis |
|
| Kallman | XR | KAL-1 | 30 |
|
|
| Mayer-Rokitansky-Küster-Hauser | Sporadic or familial | WNT4 | 20–40 | Unilateral kidney agenesis |
|
| OEIS complex | Sporadic | Unknown | 36 |
|
|
| Turner syndrome | Sporadic | XO karyotype | 33 |
|
|
| Thrombocytopenia-absent radius | AR | RBM8A | 23 | Horseshoe kidney |
|
The same embryologic insult that led to failure of kidney development can also cause abnormalities of other mesonephric duct derivatives, including the seminal vesicles, vas deferens, and epididymis and müllerian duct organs. Approximately 32% to 50% of girls with unilateral kidney agenesis will have genital tract abnormalities, including unicornate or didelphic uterus and vaginal obstruction. The syndrome of obstructed hemivagina and ipsilateral kidney agenesis (OHVIRA), presumed to be due to the arrest of müllerian and mesonephric ducts around week 8 of gestation, can lead to unnecessary surgeries for evaluation of misdiagnosed pelvic mass and complications such as abscess formation and endometriosis. Therefore uterine ultrasound screening for genital tract abnormalities in girls with a solitary kidney is recommended but may be technically challenging.
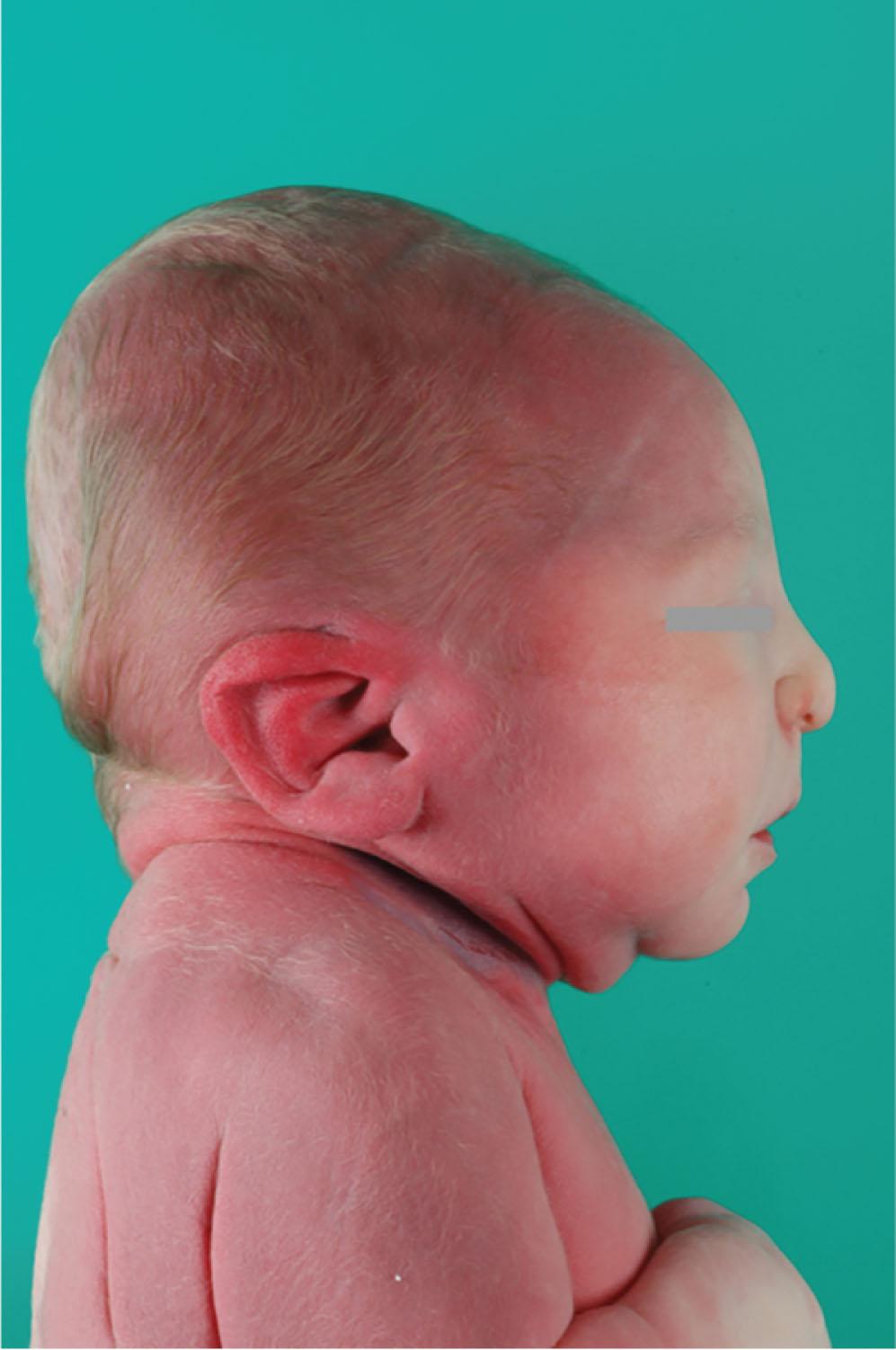
Bilateral kidney agenesis occurs in 1 in 3000 births. It may be an isolated finding or part of a syndrome, such as the brachio-oto-renal dysplasia syndrome or a hereditary kidney adysplasia. It is typically diagnosed prenatally in a pregnancy complicated by severe oligohydramnios or anhydramnios and nonvisualization of the fetal kidneys and bladder. Bilateral kidney agenesis is responsible for approximately 20% of cases of the oligohydramnios sequence (Potter syndrome), in which decreased amniotic fluid causes compression of the fetus. Patients have the classic low-set ears, wide-set eyes with epicanthal folds, flat nose, and receding chin ( Fig. 75.1 ). Historically, infants were typically born alive but died within the first hours to days of life because of pulmonary hypoplasia. There are now multiple case reports of prenatal treatment with serial amniotransfusion that resulted in the birth of infants who survived and received peritoneal dialysis followed by a kidney transplant with minimal respiratory support. As of writing, there is an ongoing clinical trial in the United States to evaluate amnioinfusions with normal saline or lactated ringers solution for management of bilateral kidney agenesis ( https://clinicaltrials.govNCT03101891 ).
Kidney ectopia occurs when the kidney fails to ascend from its embryologic position in the fetal pelvis to its final position in the kidney fossa. Ectopia can be simple, with the kidney located ipsilateral to its ureteral insertion, or crossed, with the kidney located contralaterally. Crossed ectopic kidneys typically fuse to the orthotopic kidney. It is found in 1 in 1000 individuals on autopsy and is typically asymptomatic; approximately 90% of patients are never diagnosed, and ectopic kidneys with ectopic ureters can become obstructed or associated with incontinence. Approximately 20% to 30% of patients will have vesicoureteral reflux usually into the orthotopic kidney, which may be associated with infections. Ectopia is not associated with hypertension, proteinuria, or chronic kidney disease. Patients are typically not followed closely if there is no evidence of reflux or obstruction.
A horseshoe kidney occurs when the two kidneys are fused, typically at the lower poles, by a parenchymal or fibrous isthmus. This fusion impedes the embryologic ascent of the horseshoe kidney past the origin of the inferior mesenteric artery. It occurs in 1 in 400 children. Twenty-two percent of cases are associated with other systemic abnormalities, including vertebral anomalies, anorectal malformations, and Turner syndrome, and 20% to 30% of children will have vesicoureteral reflux. Urinary tract infections and kidney stones are common. The risk of progression to chronic kidney disease is primarily dependent on the amount of kidney damage suggested by proteinuria, hypertension, and kidney scarring.
Kidney ectopia occurs when the kidney fails to ascend from its embryologic position in the fetal pelvis to its final position in the kidney fossa. Ectopia can be simple, with the kidney located ipsilateral to its ureteral insertion, or crossed, with the kidney located contralaterally. Crossed ectopic kidneys typically fuse to the orthotopic kidney. It is found in 1 in 1000 individuals on autopsy and is typically asymptomatic; approximately 90% of patients are never diagnosed, and ectopic kidneys with ectopic ureters can become obstructed or associated with incontinence. Approximately 20% to 30% of patients will have vesicoureteral reflux usually into the orthotopic kidney, which may be associated with infections. Ectopia is not associated with hypertension, proteinuria, or chronic kidney disease. Patients are typically not followed closely if there is no evidence of reflux or obstruction.
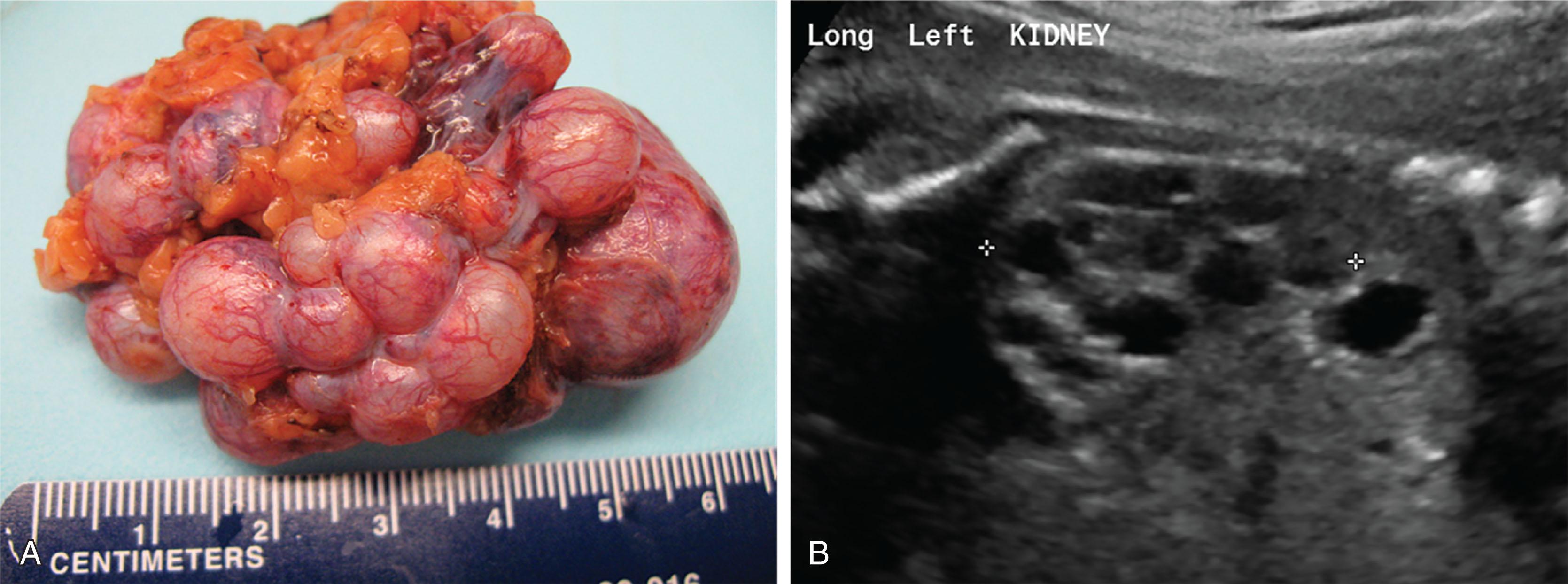
A multicystic dysplastic kidney (MCDK) develops when there is an impairment of nephrogenesis, resulting in branched ducts surrounded by connective tissue and undifferentiated cells. Occasionally there will be areas of recognizable kidney tissue, but the ureter is not patent and the kidney is nonfunctional. MCDK is typically a sporadic and unilateral finding that occurs in 1 in 4300 births. It is the second most common cause of a flank mass in newborns. MCDK can be accurately diagnosed on ultrasound as a collection of large cysts that do not communicate with the kidney pelvis ; a nuclear medicine scan is not necessary for diagnosis ( Fig. 75.2 ). Bilateral MCDK is functionally similar to bilateral kidney agenesis, with similar management and outcomes.
Approximately 20% of patients with MCDK have vesicoureteral reflux in the contralateral kidney; 40% of these children will have severe (grade III to V) reflux. However, given the low rate of clinical intervention in some recent cohorts, the need for routine screening VCUG is controversial. If VCUG is not performed, the parents should be given instructions on monitoring for urinary tract infections. Five percent of MCDKs are associated with a contralateral ureteropelvic or ureterovesical junction obstruction; approximately 15% will have other developmental abnormalities.
MCDKs undergo spontaneous involution over time, with 5% of prenatally detected MCDKs no longer visible at birth and 40% to 55% involuted by age 5 years. If the contralateral kidney is normal, MCDK is usually asymptomatic, with a prognosis similar to unilateral kidney agenesis (see earlier). In rare cases MCDK may be a bilateral finding; these infants generally die soon after birth because of pulmonary hypoplasia. Patients should have routine ultrasound monitoring in infancy to ensure involution of the MCDK and appropriate compensatory hypertrophy of the contralateral kidney, with annual screening throughout childhood for hypertension and proteinuria. Lack of compensatory hypertrophy of the contralateral kidney is a risk factor for progressive kidney disease. Nephrectomy is generally only performed if there is a clear indication. Older studies associated MCDK with an increased incidence of Wilms tumor, but more recent cohorts have not found an association. Currently, routine tumor monitoring is not recommended for MCDK.
Kidney dysplasia occurs when either failure of ureteric bud-metanephric mesenchyme signaling or early urinary flow obstruction disrupts the normal development and differentiation of the fetal kidney. The tissue is made up of primitive ducts, branches of the ureteric bud, surrounded by a ring of fibromuscular tissue and disorganized lobar development. Kidney dysplasia may be unilateral or bilateral, isolated or syndromic, and sporadic or genetic. Mutations in ITGA8 , an integrin important to cell structure and signaling, and FGF20 , a fibroblast growth factor with a variety of functions in growth and development, have been associated with dysplasia and/or agenesis. Kidney dysplasia occurs in 0.1% to 3% of births and is the most common cause of childhood ESRD.
Kidney dysplasia is typically diagnosed prenatally or postnatally with the appearance of large, bright kidneys on ultrasound. Cysts may or may not be present. Treatment and prognosis depend on the degree of dysplasia and associated findings. Children with unilateral kidney dysplasia and a normal contralateral kidney may have outcomes similar to children with unilateral kidney agenesis. Children with bilateral kidney dysplasia have variable outcomes depending on the degree of residual kidney function. Mild bilateral dysplasia may result in adequate amniotic fluid production for lung development; however, in general, there is a progressive decline in kidney function in infancy or childhood. Severe bilateral dysplasia has significantly worse postnatal outcomes, particularly if children develop the oligohydramnios sequence (Potter syndrome) with pulmonary hypoplasia. Treatment may include dialysis, but the appropriateness of dialysis is typically determined on a case-by-case basis after discussion of the multidisciplinary care team and parents.
Prognosis for children with kidney dysplasia associated with genetic syndromes often depends on the patient's other developmental abnormalities. Some of the more common syndromes are presented as follows and in Table 75.2 .
| Syndrome | Inheritance | Genes | Kidney Disease (%) | Type of Kidney Involvement | Key Features |
|---|---|---|---|---|---|
| Alagille | AD | JAG1, NOTCH2 | 40 with JAG1 |
|
|
| Brachio-oto-renal | AD | EYA-1, SIX1, SIX5 | 67 |
|
|
| Cornelia de Lange | AD, XD | NIPBL, SMC1A, SMC3, RAD21, HDAC8 | 36 |
|
|
| DiGeorge | AD | 22q11.2 deletion | 30 |
|
|
| Ecterodactyly, ectodermal dysplasia, and cleft lip/palate (EEC1) | AD | 7q11.2–q21.3 | 20 |
|
|
| Fanconi anemia | XR | Heterogeneous | 5 |
|
|
| Fryns | AR | Unknown | 35 |
|
|
| Hypoparathyroidism-deafness-renal dysplasia | AD | GATA3 | >60 |
|
|
| Pallister-Hall | AD | GLI3 | 21–36 |
|
|
| Prune-belly | AR | Unknown | >97 |
|
|
| Renal coloboma | AD | PAX2 | >90 |
|
|
| Townes-Brocks |
|
SALL1 | 27–42 |
|
|
| VACTERL | Sporadic | Unknown | 65–80 |
|
|
| Wolf-Hirschhorn | Sporadic | 4p16 deletion | 40 |
|
|
Renal coloboma syndrome (a.k.a. papillorenal syndrome) is an autosomal dominant disorder caused by mutations in PAX2 , a transcription factor involved in development. Affected children have optic nerve coloboma (dysplasia) and small dysplastic kidneys. Vesicoureteral reflux, high-frequency hearing loss, and central nervous system anomalies may also be present. Most patients have progressive kidney dysfunction, although the timing is highly variable, even in families with the same PAX2 mutation. PAX2 mutations have also been associated with congenital anomalies of the urinary tract and autosomal dominant childhood-onset focal segmental glomerulosclerosis (FSGS).
Branchio-oto-renal syndrome is an autosomal dominant condition affecting 1 in 40,000 newborns. It is caused by mutations in EYA-1, SIX1 , or SIX5 , which interact in the development of the branchial arches, inner ear, and kidney. Clinical manifestations include branchial arch anomalies (clefts, fistula, cysts), preauricular pits, hearing impairment (conductive or sensorineural), and kidney anomalies ranging from unilateral dysplasia to bilateral agenesis.
Hypoparathyroidism-deafness-renal dysplasia syndrome (a.k.a. Barakat syndrome) is an autosomal dominant disorder with variable penetrance caused by mutations in GATA3 , a transcription factor involved in embryologic development. Patients can present at any age with symptomatic hypocalcemia secondary to hypoparathyroidism or early-onset bilateral sensorineural hearing loss that worsens with age. The associated kidney abnormalities include unilateral or bilateral kidney dysplasia or agenesis, although vesicoureteral reflux, proteinuria, and progressive chronic kidney disease have been described.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here