Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The heart is largely derived from mesoderm.
A single heart tube forms with venous and arterial connections. It elongates by addition of cells at either end from the surrounding mesenchyme.
The tube folds to the right and lies in the pericardial cavity.
The atria and ventricles form by ballooning from the tube; septation of the atria and ventricles is largely achieved by this method.
The endocardial cushions at the atrioventricular (AV) junction and outflow tract complete septation of the AV junction and outflow tracts and give rise to the AV and arterial valves.
The epicardium gives rise to the mesenchyme of the heart, the veins and most of wall of the coronary arteries.
The epicardial coronary arteries grow into the aortic root.
The conduction tissue is derived from primitive myocardium of the original heart tube.
A complex series of venous precursors contribute to the abdominal veins.
Aortic arches 3, 4 and 6 give rises to the vessels of the head, neck and thorax.
The heart is an organ largely derived from mesoderm, with a contribution from ectodermally derived neural crest cells. Its development is critically dependent upon a cascade of events tightly regulated in time and space. The first recognisable sign of heart development in the embryo is the formation of the heart tube within the lateral splanchnic mesoderm late in the third week. Invisible to the classical morphologist are a series of preceding genetic steps on which the morphological changes are critically dependent. These include the specification of precursor cells in the bilaminar embryonic disc, their migration to the heart fields and the specification of cell types within the developing heart. By the end of the seventh week, the heart, although tiny, is essentially fully formed. It does not develop in isolation but is intimately related to surrounding structures such as the pharynx, and heart development is both influenced by and influences these surrounding structures. Unsurprisingly, defects in the heart are associated with defects in these associated structures. From very early in its development, the heart is a beating structure and contains a flowing liquid, so mechanical forces are also important in shaping its development.
Heart development is contributed to by multiple genes, many of which have multiple functions in development, with considerable overlap. If a defect develops, it is likely to be modified by growth and subsequent alterations in haemodynamics. Because heart development is a sequential and complex process, no single gene defect leads to a single specific heart defect. There has been a proliferation of information on heart development in recent decades, with genetic experiments in mouse, chick and zebrafish embryos having determined details of cell lineage. Although much remains unknown, the genetic mechanisms regulating heart development are beginning to be understood.
The bulk of the work on early cardiac development comes from studies in fish, chick and mouse embryos.
Within the cardiac crescent (the group of mesodermal cells lying in the splanchnic mesoderm anterior to the buccopharyngeal membrane and that show patterns of gene expression committing them to cardiogenesis), paired endothelial-lined tubes develop with their long axis in the long axis of the embryonic disc ( Fig. 10.1 ).
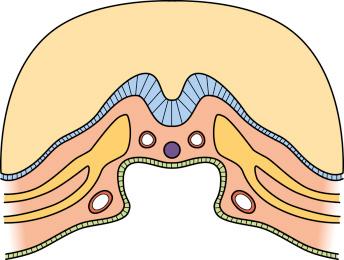
With folding of the embryo, these paired tubes fuse medially over part of their length to form the primitive single heart tube. The surrounding mesoderm of the cardiac crescent differentiates to provide an investing sleeve of myocardium, both layers separated by extracellular matrix termed cardiac jelly . The inflow is caudal, a continuation of the primitive veins, and the outflow cranial and connected to the paired dorsal aortae.
The pericardial cavity develops initially on the dorsal surface of the developing heart as part of the intraembryonic coelom. With folding of the embryo and formation of the single heart tube, the pericardial cavity comes to lie ventral to the heart tube, which is connected dorsally to the mesenchyme by the dorsal mesocardium. The septum transversum grows into the intraembryonic coelom partially to divide the pericardial cavity from the peritoneal cavity. The pericardial cavity is further divided by ingrowth of tissue from the lateral body walls, the pleuropericardial folds, that fuse with each other and the foregut mesenchyme to completely enclose the pericardial cavity and create two pleural cavities still joined to the peritoneal cavity. The dorsal mesenchyme breaks down to leave the heart connected to the body wall only at the arterial and venous poles. The space thus created is the transverse pericardial sinus.
The primitive myocardium of this primary heart tube shows regular contractions by the third week (embryonic age). This heart tube elongates by addition of mesodermal cells at its two poles (and from the dorsal mesocardium until that structure involutes) and undergoes rightward looping ( Fig. 10.2 ). The primitive heart tube is the scaffold to which the atrial and ventricular chambers are added by ballooning from its myocardium. Septa are formed, dividing the right and left sides of the atria, ventricles and great arteries.
Heart formation is completed with the development of valves, the conduction system and the formation of the coronary arteries by the ingrowth of extracardiac tissues derived from the neural crest and from the proepicardium situated in the septum transversum.
Heart progenitor cells are located in two small patches, one on either side of the midline in the epiblast of the bilaminar embryonic disc, close to the cranial part of the primitive streak. The earliest known committed cardiac precursors express the T-box transcription factor Eomesodermin\Tbr2, which activates another transcription factor MESP1 . These progenitor cells migrate together through the primitive streak and form two plates of lateral mesoderm cells positioned anteriorly. Specification of cardiogenic mesoderm has already started during this cellular migration. The progenitor cells migrate such that the medial-lateral arrangement of these cells will become the cranial-caudal axis of a linear heart tube.
With the formation of the embryonic coelom, they occupy the splanchnic mesoderm and fuse in the midline cranially to form the cardiac crescent. The cells of this cardiac mesoderm express the cardiac specific transcription regulator genes NKX2.5 and GATA-4 .
The cells of the splanchnic mesoderm, one on each side of the body, interact with adjacent tissues ( Fig. 10.3 ). The splanchnic mesoderm is positioned adjacent to the foregut endoderm (see Fig. 10.1 ), which is thought to provide inductive signals to begin myocardial differentiation. Endoderm also has a mechanical role in assembly of the heart tube. By its active contraction, it pulls the bilateral fields of cardiogenic mesoderm towards the midline, permitting them to fuse and form a single heart tube.
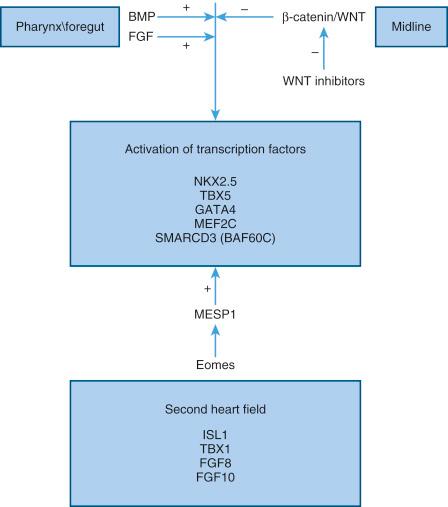
The primitive, single heart tube initially functions not so much to support the embryonic circulation as to provide a scaffold into which the cells from the second heart field migrate to effect chamber formation. The second heart field cells are first located medial to the cardiac crescent and subsequently lie in mesoderm underlying the pharynx before they are added to the heart. The cranial part of the second heart field, the anterior heart field, which is identified by FGF10 expression, contributes to the formation of right ventricular and outflow tract myocardium. Cells in the posterior second heart field express Isl1, but not anterior heart field markers, contribute to atrial myocardium.
The nomenclature of the heart fields may be confusing because not all authors use exactly the same terms, and the heart fields are dynamic, changing in position and with time.
The first and second heart fields develop sequentially, and the anterior and posterior heart fields describe anatomical positions.
It appears that the overall cellular environment is critical for this process because cells taken from a different location or at a different developmental time point can contribute to the heart when placed in the appropriate location.
Within the cardiac crescent, the primitive heart tube forms. The horseshoe-shaped cardiac crescent forms a tube with two caudolateral inlets, or venous pole, and one craniomedial outlet, or arterial pole. The process starts with the formation of endothelial-lined channels in the mesenchyme that form a plexus that eventually coalesces to form paired endothelial tubes. Folding of the embryo then brings these paired tubes together in the midline where they fuse to form a single endothelial-lined tube with a surrounding cuff of mesenchyme, separated from it by extracellular matrix termed cardiac jelly. The endocardium develops simultaneously with the myocardium and is a specialised endothelial cell type derived from the splanchnic mesoderm that, via an unknown mechanism, differs from the myocardial precursors.
The linear heart tube subsequently grows by addition of cells from a proliferating pool of precursors located external to it in the heart fields and not by division of myocytes in situ. Being added to the heart, their fate is not fixed, and their identity depends on their eventual location.
The cardiac precursors are therefore initially situated cranial and lateral to the future mouth but caudal to the mesoderm that forms the transverse septum and contributes to the formation of the diaphragm. During the process of folding, the cardiac precursors end up between the mouth on the cranial aspect, the diaphragm at the caudal aspect, and ventral to the foregut, as in the adult situation.
By folding of the embryo, the lateral parts of the cardiac mesoderm are brought together, forming the ventral part of the heart tube. The inner curvature of the cardiac crescent forms the dorsal side of the tube and is contiguous with the dorsal mesocardium, the attachment of the heart to the body wall.
The peripheral part of the cardiac crescent will eventually face the transverse septum and form the venous pole of the heart, and the central part of the crescent, which forms the outflow tract, is contiguous with the pharyngeal mesenchyme. This intimate association of the cardiac and facial region during development explains the high incidence of combined cardiac and facial malformations in syndromic conditions.
Retinoic acid appears critical in specifying these posterior precursor cells to become the inflow, or venous parts of the heart, the sinus venosus and atria,
The myocardium shows regular contractions by the third week, meaning that the structural requirements for contraction, namely contractile proteins, sarcoplasmic reticulum and gap junctions, are already present in the myocardial cells of the primitive heart tube. It would appear that the initial contractions, at least in the chick embryo, are not essential for tissue oxygen and nutrient supply but likely have an important function providing mechanical stimuli for further development of the heart.
The myocardium of the primitive heart tube demonstrates TBX2 and TBX3 expression. Traditionally, five consecutive segments of the tube are recognised: venous, atrial, left ventricular, right ventricular and arterial. Between each of the segments is a transition zone – the sinoatrial ring, the atrioventricular (AV) canal, the primary heart fold and the ventricular outflow tract. It is important to recognise that the cardiac chambers develop as outgrowths from these segments and that the original segments form the connections of the cardiac chambers rather than the chambers themselves.
Looping of the straight heart tube follows activation of a gene cascade that determines right–left symmetry. Left–right differentiation has already started at the time of gastrulation with the formation of the Hensen node. Nodal cilia beat in one direction and cause a gradient of molecules across the bilaminar embryo. These genes are already expressed in the cardiac mesoderm before formation of the heart tube and include NODAL , LEFTY and PITX2, of which PITX2 is the effector gene. Furthermore, the direction of looping is not random but is controlled by genes not yet understood and is not under the control of PITX2 .
The tube loops to the right (see Fig. 10.2 ), which causes the transition zones to be brought into close proximity on the inner curvature of the loop. This is absolutely essential for establishing the correct connections of the chambers. Actin polymerisation-driven myocardial cell shape changes have been found to contribute to the bending of the heart tube, but the torsional component of looping is largely caused by forces from its encapsulating membrane, the splanchnopleure.
The looped heart tube has unidirectional blood flow, which was originally thought to be peristaltic but is now regarded as functioning as a Liebau pump (unidirectional and pulsatile flow resulting when an elastic tube containing fluid is periodically squeezed ). At this stage, the AV cushions function as primitive valves to permit unidirectional flow of blood in the looped heart tube.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here