Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Methicillin-resistant Staphylococcus aureus (MRSA) is currently the most common isolate in community-acquired deep neck infections in children under 2 years.
Up to 10% of MRSA isolates are clindamycin resistant.
Conservative management without drainage is more likely to succeed with abscess pockets <2.5 cm in diameter that involve a single neck space.
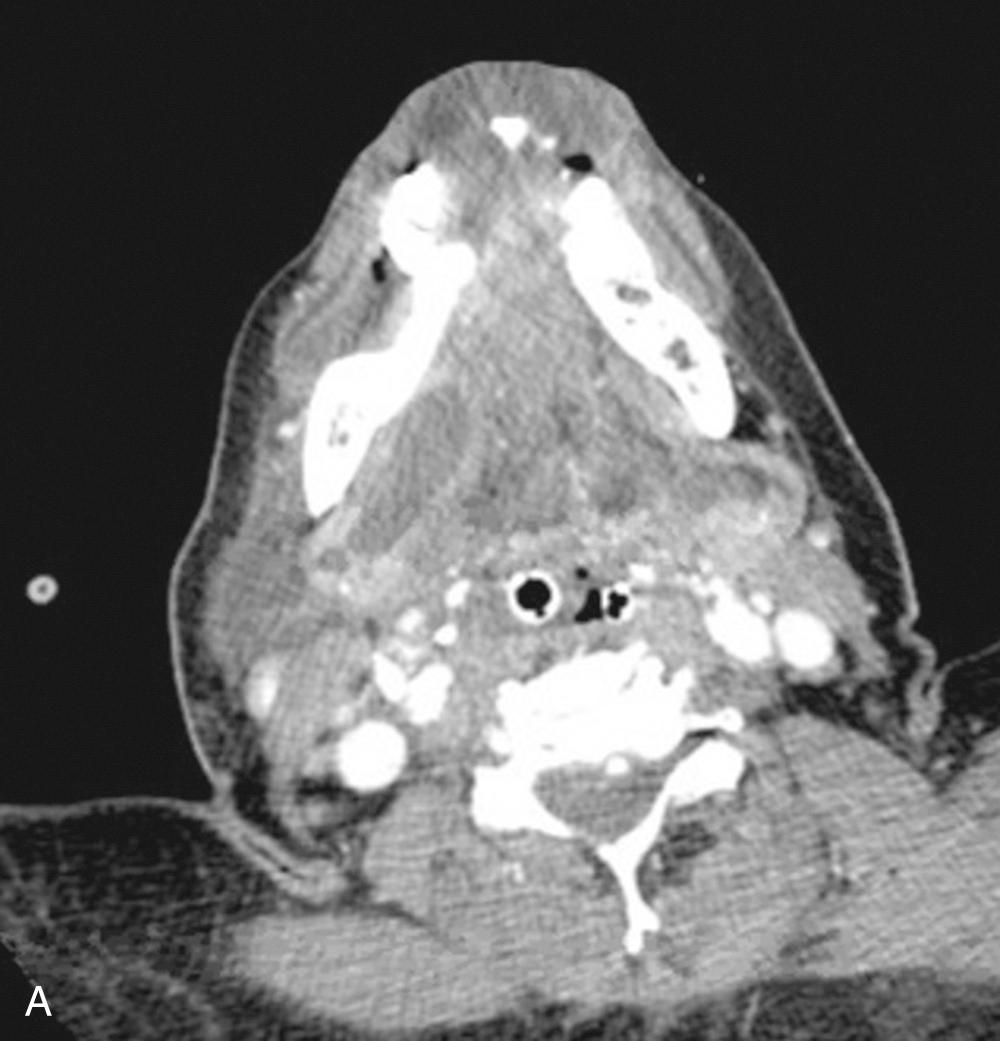
Deep neck infection (DNI) remains a frequent, potentially life-threatening condition in both children and adults. Knowledge of the complex spaces of the neck and how they communicate is necessary for appropriate surgical management. The location of the infection may provide information concerning the origin of the infection and the likely organisms that must be covered when instituting empiric therapy.
Infectious and inflammatory conditions of the upper aerodigestive tract are the primary cause of DNIs. It is estimated that the direct cost of DNI in the United States exceeds $200 million dollars annually. Dental infections are the most common etiology of DNI in adults, whereas Waldeyer ring infections are the most common in children. In pediatric patients alone a recent review of national data estimates the incidence of DNI at 4.6 per 100,000 with >$75 million in charges. In addition, an increased incidence in retropharyngeal abscesses were observed from 2000 to 2009.
Bacterial biofilms are estimated to cause 65% to 80% of human infections and play a key role in the cause of odontogenic infections. Most dental-related infections are localized, minor exacerbations from longstanding decay or periodontal disease. Consequently, there is considerable interest in periodontitis—a chronic inflammatory disease (and by definition a chronic low-grade infection due to a microbial biofilm) of the tooth-supporting structures—as a potential risk factor in the morbidity and mortality of systemic conditions such as cardiovascular disease, diabetes mellitus, and premature birth. Epidemiological studies have revealed an association between periodontal disease and cardiovascular disease, cerebral vascular disease, along with diabetes mellitus, showing that patient's treated for their periodontal disease had decreased hospital costs after undergoing treatment, but research into this association is ongoing.
In the pediatric population, acute rhinosinusitis, tonsillitis, and pharyngitis are common causes of retropharyngeal lymphadenitis. Oral surgical procedures and endoscopic instrumentation may iatrogenically incite an upper airway infection or traumatize the pharyngoesophageal lumen. Sialadenitis, with or without ductal obstruction, can precipitate infectious spread ( Fig. 9.1 ). Foreign bodies trapped within the upper aerodigestive tract may initiate infections that spread to the deep neck. Superficial infections, such as skin cellulitis, may spread along fascial planes into deeper neck compartments. Penetrating trauma including needle injection associated with intravenous drug use may introduce pathogens into the fascial planes. Congenital or acquired lesions, such as branchial cleft cysts, thyroglossal duct cysts, or laryngoceles may become infected with resulting spread. Congenital cysts account for 10% to 15% of DNI in the pediatric population and should be suspected especially in the setting of recurrent DNI. Acute mastoiditis may progress to a Bezold abscess with spread to the upper neck through bony fissures. Necrotic malignant lymph nodes can form abscesses and demand a high degree of suspicion for cancer if observed in the adult patient over the age of 40 years, in which approximately 5% of cases of DNI are associated with malignant lymph nodes. Immunocompromised patients may present with more virulent or atypical pathogens. Although the etiologic factors are many, a thorough search for the cause often reveals no clear source.
The microbiology of DNI often yields a mixture of aerobic and anaerobic organisms representative of oropharyngeal flora. Studies have isolated approximately 280 species of bacteria in the oral cavity, but less than 50% of the oral flora can be identified through traditional methods. Recent studies have utilized cultivation-independent molecular methods (16rRNA sequencing methods) and identified nearly 600 bacterial species. A list of many of the common “normal” aerobic, facultative, and anaerobic species of bacteria (several fungal species, viruses, and even protozoans can also be found in the “normal oral flora”) is shown in Table 9.1 . In most patients, these normal oral bacteria are the cause of most odontogenic infections. Staphylococcus aureus , in particular, is much more common in the nose and throat, and may participate in mixed odontogenic infections. Multiple recent studies have demonstrated that in pediatric patients there is an increase in S. aureus , likely arising from the Waldeyer ring as the causative organism compared with adults, where it is usually a Streptococcus species of dental origin. Gram-negative anaerobes comprise much of the rest of the mouth's normal flora, and these organisms can increase in numbers, especially in patients with chronic periodontal disease.
| Aerobic Bacteria | Anaerobic Bacteria |
|---|---|
| G ram -P ositive C occi | |
| Streptococcus Staphylococcus aureus (facultative anaerobe) |
Streptococcus Peptococcus Peptostreptococcus |
| G ram -N egative C occi | |
| Neisseria | Veillonella |
| G ram -P ositive B acilli | |
| Diphtheroids | Clostridium Actinomyces Eubacterium Lactobacillus |
| G ram -N egative B acilli | |
| Haemophilus Eikenella |
Prevotella (Bacteroides) Fusobacterium Porphyromonas |
Almost all odontogenic infections are due to mixed flora. Retrospective microbiologic analyses consistently demonstrate common oral pathogens and polymicrobial isolates. Several studies in the past have consistently shown a predominance of certain species associated with odontogenic infections. These include Streptococcus milleri group (a subset of Streptococcus viridans ), Prevotella ( Bacteroides ), Peptostreptococcus , and staphylococci . This finding has been consistent for many years, even as name changes (especially for Bacteroides ) have taken place to better reflect more recent genetic information. Recent studies utilizing molecular methods to identify species from samples taken from odontogenic infections have found a greater prevalence of facultative or obligate anaerobes and a limited number of streptococci. This difference in results may be influenced by the methods for determining speciation where aerobic and anaerobic conventional cultures support the growth of certain microbes over others. Further, as much as 60% of the oral flora is unable to be cultured by routine culture methods.
The proportion of community acquired methicillin-resistant Staphylococcus aureus (MRSA) associated neck space infections is significantly increasing worldwide, especially among the pediatric population. MRSA is more likely to infect younger patients in lateral neck compartments. In pediatrics, three different studies completed in locations across the United States have shown S. aureus as the most common organism in DNIs, with the rate going up in younger infants (<1- to 2-year-olds). Among those studies MRSA rates were 12%, 13.7%, and up to 51% in the most recent report. African American infants were at an increased risk for having an MRSA infection. Clindamycin sensitivity was reported in two of the studies and was 86% to 100%. In adults, a recent study by Kim et al. demonstrated S. viridans was the most common isolate with Staphylococcus the second most common. They compared their results to Rega and colleagues’ prior study and found that clindamycin resistance in Streptococcus and Staphylococcus species rose to 32% and 23% from 13.7% and 10.5%, respectively, although they noted that increased resistance did not appear to change length of stay in the hospital.
Atypical organisms are a relatively common cause of DNI. Actinomyces are endogenous saprophytic organisms of the oral cavity and tonsil. The most common site of cervicofacial actinomycosis is in the vicinity of the angle of the mandible, and this pathogen may cross fascial planes in its route of spread. A granulomatous reaction with central abscess formation and necrosis with “sulfur granules” is characteristic. Tuberculous and nontuberculous infection of the head and neck most commonly presents with cervical lymphadenopathy, often with adherence and ulceration of overlying skin. Histopathologically, caseating necrotizing granulomatous inflammation is present. Cat scratch disease, caused by the pleomorphic gram-negative bacillus Bartonella henselae , manifests with large, tender cervical lymph nodes. Late lesions may form an abscess. Management of atypical neck space infections is often nonsurgical, as incision and drainage procedures may result in a chronic wound or fistulous tract.
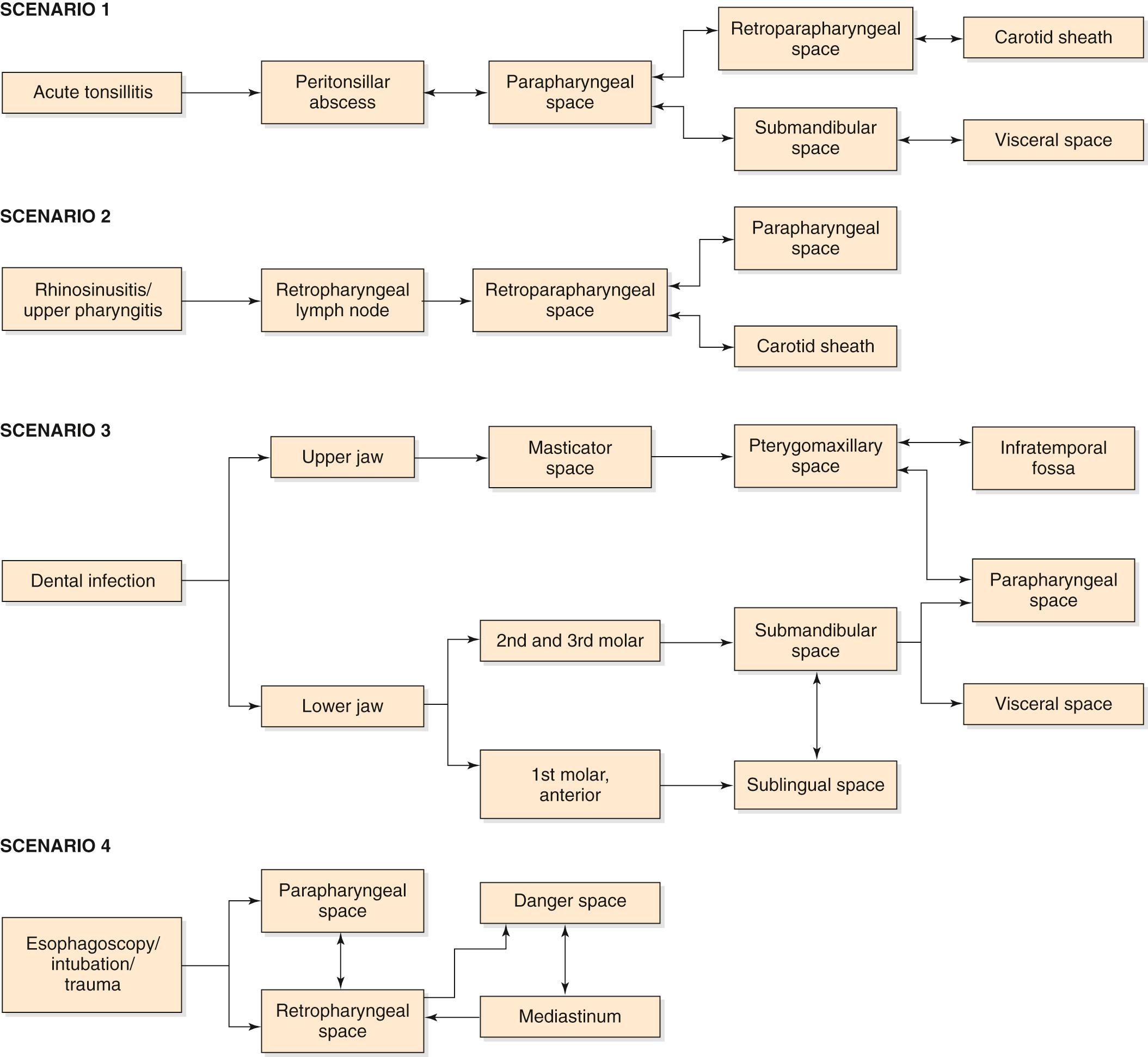
Accurate diagnosis and application of timely treatment requires an understanding of the complex anatomical organization of the neck spaces. Fascial planes divide the neck into true and potential spaces. A description of the neck spaces and contents are summarized in Table 9.2 . Common pathways of spread are outlined in Fig. 9.2 .
| Neck Space | Boundaries | Contents | Communicating Spaces |
|---|---|---|---|
| Peritonsillar |
|
|
|
| Parapharyngeal |
|
|
|
| Infratemporal fossa |
|
|
|
| Pterygomaxillary fossa |
|
|
|
| Temporal fossa |
|
|
|
| Parotid |
|
|
|
| Masticator |
|
|
|
| Submandibular |
|
|
|
| Visceral |
|
|
|
| Carotid sheath |
|
|
|
| Retropharyngeal |
|
|
|
| Danger |
|
|
|
| Prevertebral |
|
|
|
Odontogenic infection from decaying teeth and poor oral hygiene continue to be the most common source of DNI in the adult patient. There are several additional spaces of note with regard to odontogenic infections:
The two primary maxillary spaces that may be involved are the canine space and the buccal space . The canine space becomes infected almost exclusively as a result of apical infection of the root of the maxillary canine tooth. The root must be long enough to ensure that the apex is superior to the insertion of the levator anguli oris muscle. The canine space is located between the anterior surface of the maxilla and the levator labii superioris. When infected, there is usually swelling lateral to the nose and loss of the ipsilateral nasolabial fold. Drainage is most often accomplished with an intraoral stab incision.
The buccal space becomes involved when the infection of maxillary molar teeth breaks out superior to the attachment of the buccinator muscle. The space itself lies between the buccinator muscle and the skin. All three maxillary molars may cause infection in this space. It is an ovoid space, below the zygomatic arch and above the inferior border of the mandible. Infections in this space are dramatic in appearance and may cause trismus.
The primary mandibular spaces include the submental , sublingual , and submandibular spaces. The primary spaces are those into which infection spreads directly from the teeth through bone. The submental space lies between the anterior bellies of the digastric muscles and between the mylohyoid muscle and the skin. This space is involved with infected mandibular incisors, whose roots are long enough to allow erosion apically to the attachment of the mentalis muscle. Isolated submental space infections are not common.
The sublingual and submandibular spaces exit on the medial aspect of the mandible. They are usually involved because of lingual perforation of infection from the mandibular premolars and molars. The factor that determines whether the sublingual or submandibular space is involved is the location of the perforation relative to the mylohyoid muscle attachment ( Fig. 9.3 ). If the location of the apex of the tooth is superior (premolars, first molar), the sublingual space is involved. Conversely, if the apices are inferior to the mylohyoid (second, third molar), the submandibular space is involved.
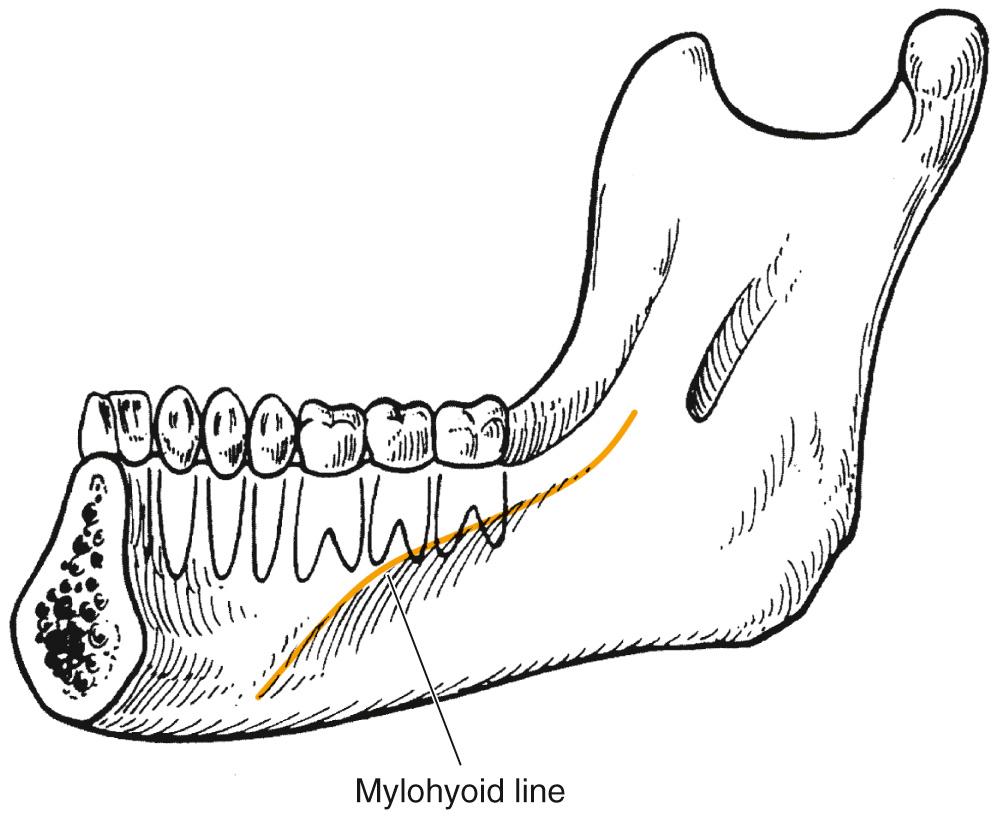
The sublingual space is between the lingual oral mucosa and the mylohyoid muscle. Its posterior boundary is open, so it communicates freely with the submandibular space and the secondary spaces located more posteriorly and superiorly. Clinically there is little extraoral swelling, but there can be marked intraoral lingual swelling in the floor of the mouth, and if bilateral, the tongue will be elevated and swallowing becomes difficult.
The submandibular space lies between the mylohyoid muscle and the skin. Like the sublingual space, it has an open posterior boundary, so it can communicate easily with the secondary spaces as well. When this space becomes infected, the swelling begins at the inferior lateral border of the mandible and extends medially to the digastric area and posteriorly to the hyoid bone.
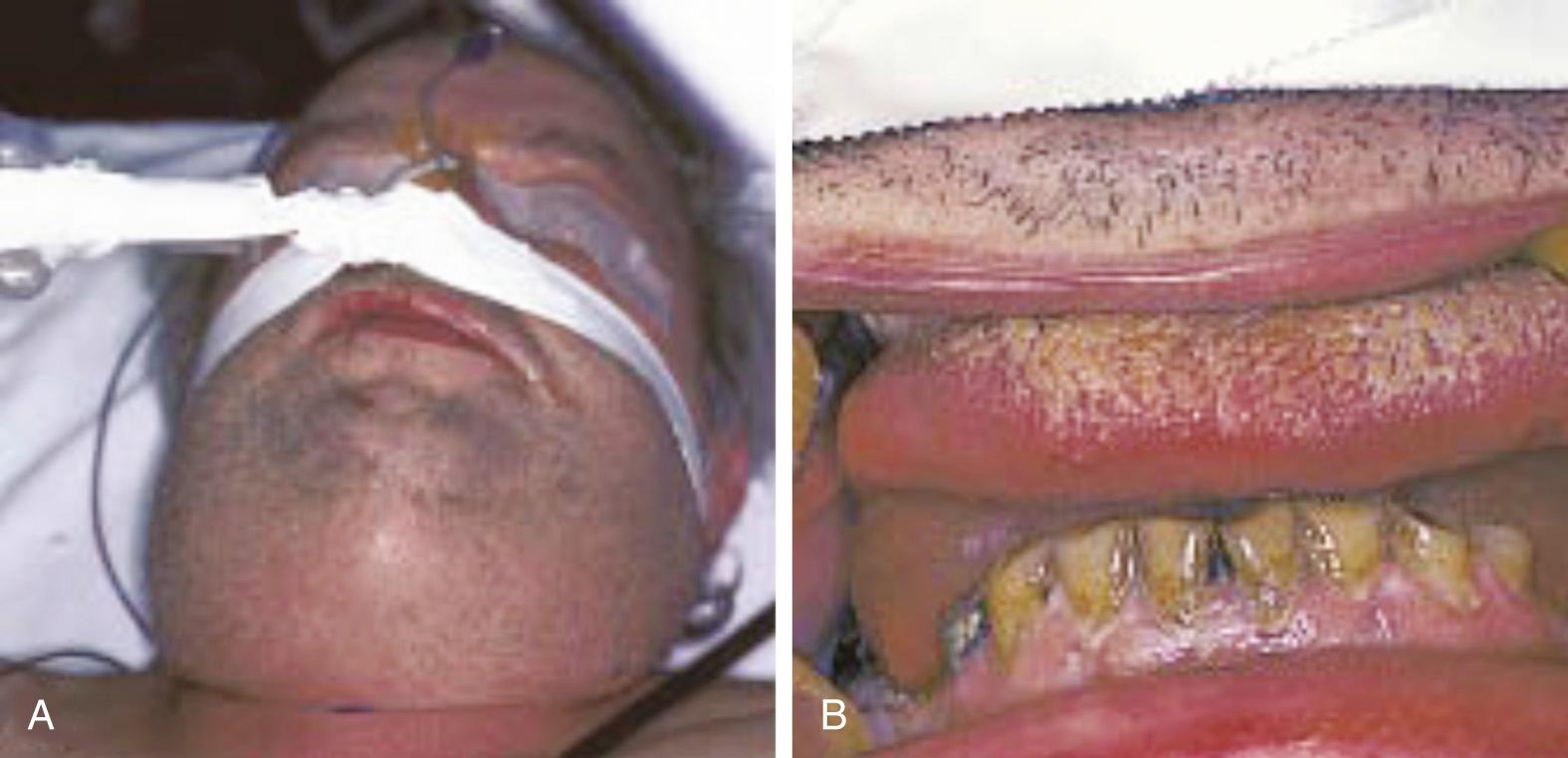
If all three primary mandibular spaces bilaterally become infected, the infection is known as Ludwig angina . Ludwig angina, described by Wilhelm Friedrich von Ludwig in 1836, was not uncommon in the preantibiotic era and was a significant cause of death. It is characterized as a rapid, bilateral gangrenous cellulitis of all three primary spaces. It can extend posteriorly to involve the secondary spaces. It causes gross swelling, elevation and displacement of the tongue, and tense brawny induration of the submandibular region superior to the hyoid bone ( Fig. 9.4 ). Ludwig angina is a clinical diagnosis not requiring advanced imaging, and patients may not tolerate lying supine for a scan due to swelling and oral secretions ( Fig. 9.5 ). There is usually minimal or no fluctuance because of the rapidity of the cellulitic process. The patient experiences severe trismus, drooling, inability to swallow, tachypnea, and dyspnea. Impending compromise of the airway produces marked anxiety. If not treated, the cellulitis can progress with alarming speed, producing airway obstruction and death. The usual cause is an odontogenic infection from a mandibular molar, although other sources such as salivary stones can be a cause. Clearly virulent streptococci are involved, but ultimately, it is a mixed picture as usual.
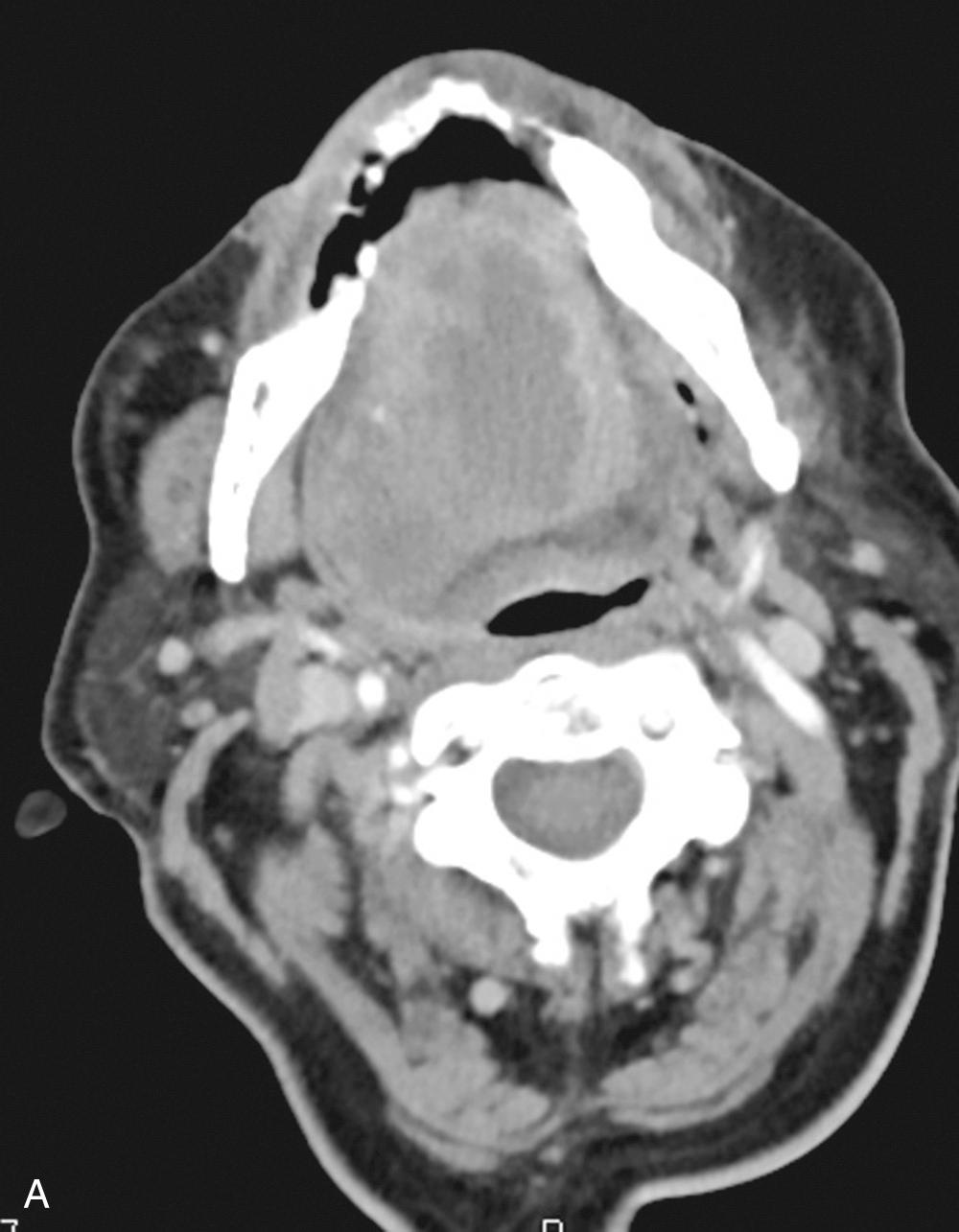
A full description of the anatomy of the neck is beyond the scope of this chapter, but it is imperative that the treating head and neck surgeon have a comprehensive understanding of the relationships within the neck. There are multiple fascial planes from superficial to deep, investing all of the muscles, visceral organs, and coming together around the vasculature. Due to the nature of these relationships and the loose areolar tissue connecting them, it creates potential spaces that allow for bacteria to spread to other locations. Streptococcus anginosus ( S. milleri group) are known to produce tissue-destroying enzymes that can help them to spread and infect along these planes, similar with anerobic bacteria that produce hyaluronidase, collagenase, and fibrinolysin.
The parapharyngeal space is an inverted pyramid in shape extending superiorly from the skull base inferiorly to the hyoid. It communicates with many of the other spaces in the head and neck, which allows infection to spread here easily, making it a common site for a DNI, and subsequently to spread to the other connected spaces. It is separated by the styloid and its muscular attachments into prestyloid and poststyloid compartments. The poststyloid compartment contains the vascular and nervous structures (sympathetic chain, cranial nerves IX, X, XI, XII), which allows an infection here to lead to an infected thrombus in the internal jugular vein (Lemierre syndrome), carotid artery aneurysm, and Horner syndrome or other cranial nerve palsies. Sichel et al. noted in their study of parapharyngeal abscesses that children often had poststyloid infections that were contained within a lymph node and more responsive to IV antibiotics, whereas infections of the prestyloid space were more likely to liquefy the fat and had a high potential to spread to other neck spaces.
One of the main sites for spread is to the retropharyngeal space , which is located in the midline posterior to the pharynx and esophagus, extending superiorly from the skull base to the level of the second thoracic vertebrae. Lymph nodes in this location drain the sinuses, nasopharynx, and Waldeyer ring. Infections here are primarily seen in children secondary to regression of lymphoid tissue in adults. The alar fascia is posterior to the retropharyngeal space and the only barrier from spread to the danger space . Communication into the danger space allows spread to the diaphragm and access to the superior mediastinum, which can lead to intrathoracic extension of an infection and mediastinitis.
A separate access site to the mediastinum is via the carotid sheath . Spread along the vasculature can go to the superior mediastinum at the aortic arch, dubbed the “Lincoln highway” by H. P. Mosher when originally described in 1929 ( Fig. 9.6 ). Intimate knowledge of the neck fascial planes and the spaces they create allows the surgeon to know where the infection can spread and where to look on imaging as well as intraoperatively.
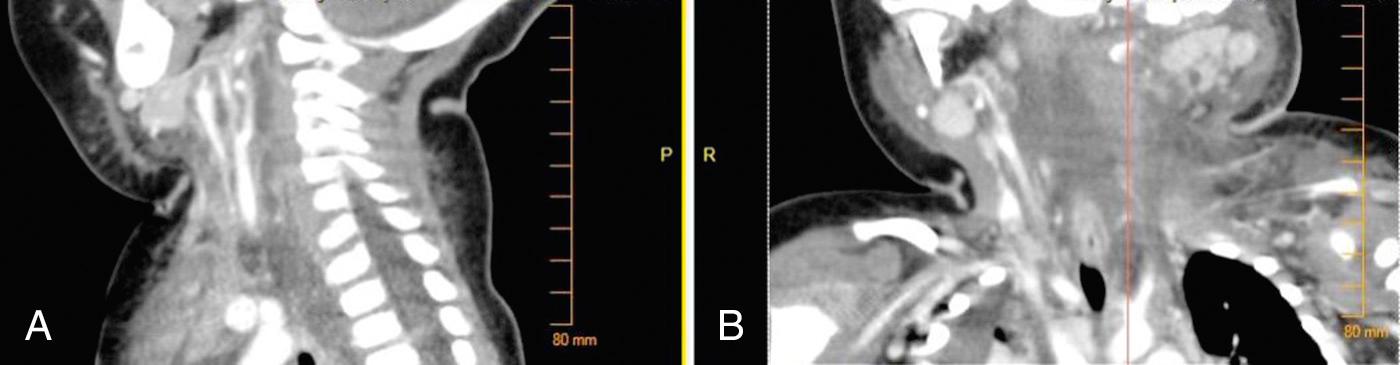
The symptoms of DNI are determined by both the generalized inflammatory process and localizing symptoms at the site of infection. Inflammatory symptoms such as pain, fever, swelling, and redness are common. Symptoms such as dysphagia, odynophagia, drooling, “hot potato” voice, hoarseness, dyspnea, trismus, and ear pain offer further clues about the location of the inflammatory process as well as its potential severity. The onset and duration of symptoms should be elicited. Recent events such as dental work, upper airway surgery or intubation, intravenous drug use, sinusitis, pharyngitis, otitis, or blunt or penetrating soft tissue trauma that preceded worsening symptoms should be identified in order to formulate a differential of likely microorganisms and common pathways of spread.
The past medical history should be reviewed to account for antibiotic allergies and immunodeficiency status. Patients with a history of human immunodeficiency virus, hepatitis, diabetes, collagen vascular diseases, hematologic malignancy, and recent chemotherapy or steroid use are at increased risk of atypical pathogens and rapidly progressive disease that may not display an acute inflammatory response. Even if on appropriate therapy, immunocompromised patients are at increased risk of DNI. In a large retrospective study, 10,000 HIV-infected individuals on standard retroviral therapy had two times risk of DNI over 6 years of observation compared with 50,000 controls.
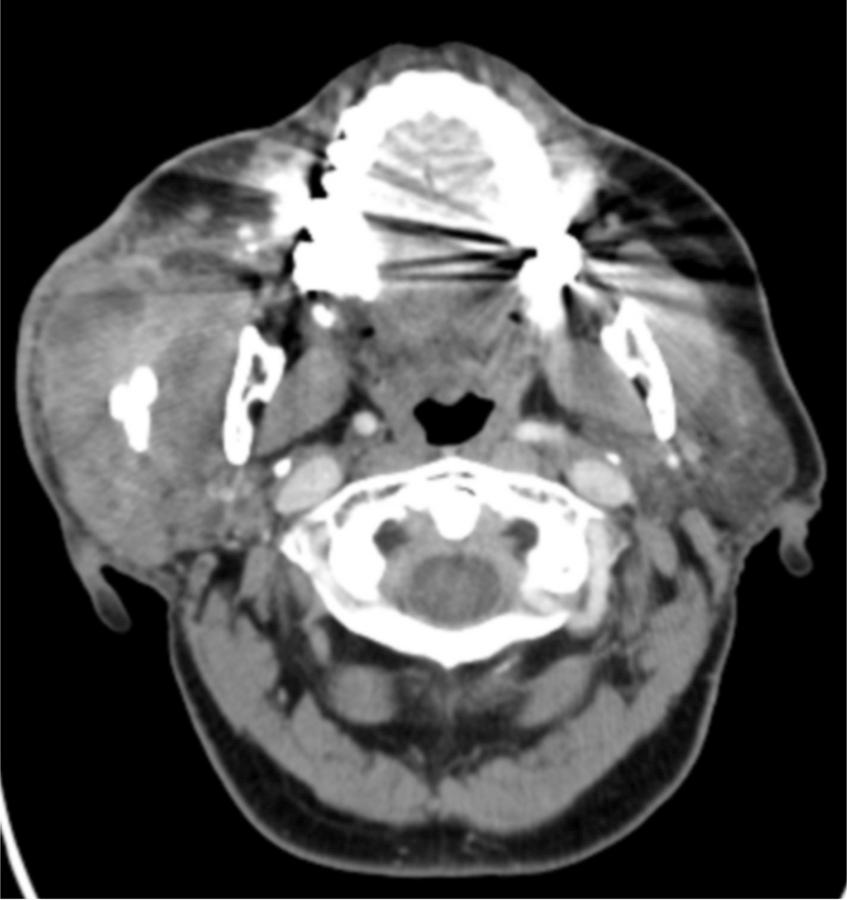
A complete head and neck physical examination is required in all patients with potential DNI. Palpation of the neck and face may identify localizing tenderness or fluctuance as well as crepitus caused by airway trauma or gas-producing organisms. Otoscopic examination of the ear and nasal passages can rapidly reveal edema, purulence, drainage, and tenderness, as well as rule out obstructing foreign bodies. Examination of the oral cavity and oropharynx is facilitated by the use of a headlight, which frees up the hands for bimanual examination. Difficulty with mouth opening indicates that inflammation has already spread to the parapharyngeal, pterygoid, or masseteric spaces. An odontogenic source of infection should be considered in the presence of alveolar swelling, and decayed, loose, tender, or broken teeth. The floor of mouth should be assessed for visible edema which may cause posterior deflection of the oral tongue. The Stensen and Wharton ducts should be assessed for purulent discharge and palpated for obstructing stones ( Fig. 9.7 ).
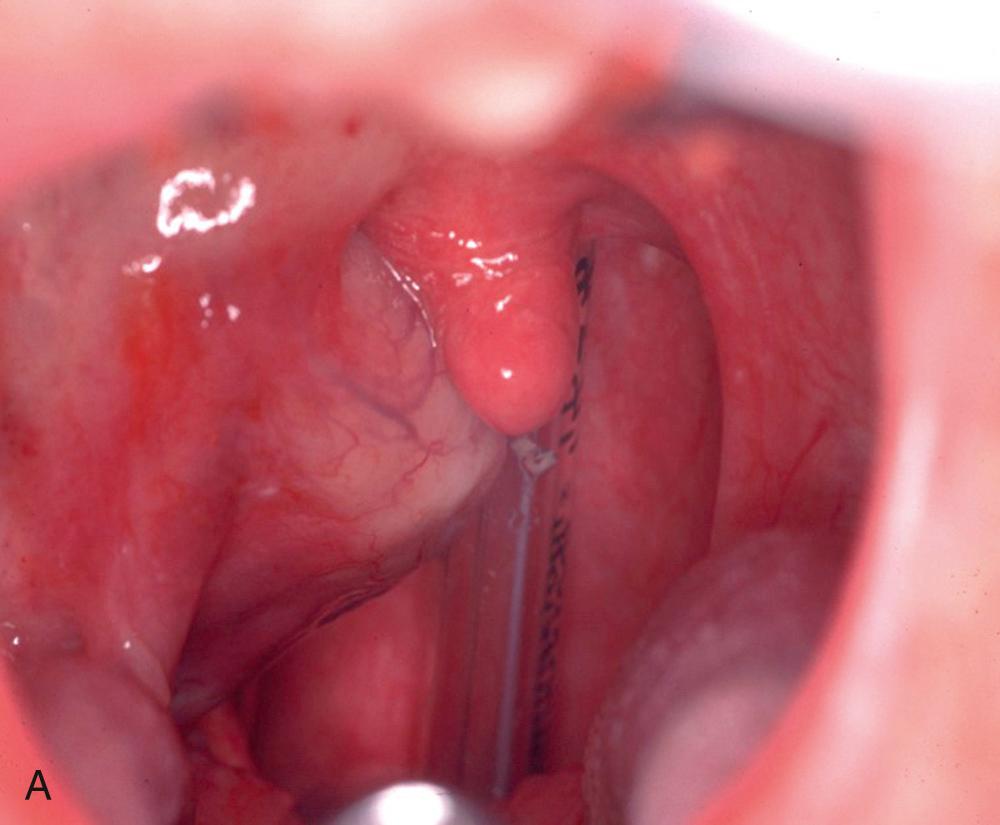
Visualization of the oropharynx is necessary to assess for asymmetric lateral or posterior wall swelling, and/or deviation of the uvula. Unilateral pharyngeal wall swelling in the absence of associated inflammatory symptoms such as fever or mucosal erythema should raise the possibility of parapharyngeal tumors, which should not be biopsied or incised without further evaluation ( Fig. 9.8 ). A unilaterally enlarged, irregular, or ulcerated tonsil, especially in the setting of prolonged exposure to tobacco and alcohol, may indicate the presence of tonsillar malignancy.
A complete cranial nerve examination is recommended. Infections of the upper dentition, paranasal sinuses, facial soft tissues, and parotid place the orbits at increased risk due to retrograde flow through the facial and valveless ophthalmic veins ( Fig. 9.9 ). Edematous eyelids must be manually separated in order to assess the underlying globe. Reduced mobility of the globe and/or an absent papillary light reflex indicates orbital inflammation and or abscesses, which require urgent attention in order to save the eye.
Awake flexible fiberoptic evaluation of the upper airway is indicated in most cases of suspected DNI, especially if the patient has hoarseness, dyspnea, stridor, and/or dysphagia, or odynophagia without obvious cause on oropharyngeal examination. A normal pulse oximetry does not eliminate the need for direct airway evaluation, since the oxygen saturation is a poor proxy for airway status and typically does not fall until the airway is completely occluded. The presence of a patent, midline, nonedematous airway should be documented prior to transport for radiographic evaluation, in order to prevent an airway emergency while the patient is supine in the radiology suite. Direct evaluation of the airway will identify patients who may be difficult to intubate by standard technique, should surgery be required.
An initial complete blood count (CBC) typically demonstrates leukocytosis in cases of DNI. A lack of a leukocytic response may indicate viral illness, immunodeficiency, or a condition such as tumor, which can be confused with DNI. Daily measures of the white blood count may be helpful in monitoring a patient's response to treatment such as intravenous antibiotics and/or surgical drainage. Intravenous steroids are often necessary to reduce upper airway inflammation in DNI and should not be withheld out of concern that steroid-related leukocytosis will make it difficult to monitor treatment response. C-reactive protein can also help indicate severity of disease on admission and trended to observe course of disease. A basic electrolyte panel should be obtained to assess glucose level, bodily hydration, and renal function should general anesthesia become necessary during treatment.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here