Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Two subsets of pruritoceptive C neurons that respond to histamine versus cowhage and then activate distinct spinothalamic tract neurons
Peripheral itch mediators include histamine, proteases, and interleukin-31, while central itch mediators include opioids, gastrin-releasing peptide, and B-type natriuretic peptide
There is an overlap between chronic itch and chronic pain, including activation of multiple brain areas and associated neuromediators and receptors, e.g. nerve growth factor, neurotrophin 4, transient receptor potential (TRP) channels
Cross-talk between cutaneous nerve fibers and the stratum corneum is a possible mechanism for the pruritus associated with impaired barrier function (e.g. xerosis, atopic dermatitis)
Therapies with topical and systemic drugs that reduce itch sensitization by counteracting the responsible mediators represent promising treatment strategies
The skin is a sensory organ with a dense network of highly specialized afferent sensory nerves that convey sensations such as pain, itch, touch, temperature, vibration, and pressure ( Table 5.1 ); efferent autonomic nerve branches are also present. Neuropeptides such as substance P, calcitonin gene related peptide (CGRP), nerve growth factor (NGF), and other neurotrophins are secreted from nerve fibers, with multiple effects that include immune modulation. Itch (pruritus) is the dominant symptom of many cutaneous diseases. Almost all inflammatory skin disorders can result in pruritus, which patients often perceive as their most unendurable symptom. Pruritus can also occur in association with systemic disease (e.g. renal failure, cholestasis; see Ch. 6 ), psychiatric conditions (see Ch. 7 ), and damage to nerve fibers . Itch is a multidimensional phenomenon with sensory discriminative, cognitive, evaluative, and motivational components. In most instances, itch results from interactions that involve the brain–skin axis.
| PRIMARY AFFERENT NEURONS THAT INNERVATE THE SKIN | ||||
|---|---|---|---|---|
| Fiber | Diameter | Myelination | Conduction velocity | Responds to |
| A-beta (Aβ) | Large | + | >30 m/s | Light touch Moving stimuli |
| A-delta (Aδ) | Small | + | 2–30 m/s | Pain (nociceptors) Itch, cowhage-sensitive ‡ Thermal Mechanical |
| C | Small | − | <2 m/s | Pain (nociceptors) Itch * , histamine-sensitive † Itch * , cowhage-sensitive ‡ Thermal † Mechanical |
* ~5% of total C fibers transmit itch.
‡ Transmit pruritus accompanied by a burning sensation; also sensitive to mechanical stimuli.
† Separate C fibers carry both pruritogenic and thermal stimuli, but not mechanical stimuli.
Pruritus has many similarities to pain. Both are unpleasant sensory experiences that can impair quality of life in affected individuals. However, the behavioral reaction patterns differ – pain elicits a reflex withdrawal, whereas itch leads to a scratching response . Despite being an extremely common complaint and a sensation so rudimentary that almost every two- or four-footed creature experiences it, medical science is still struggling to understand the mechanisms of itch and how it can be inhibited.
The connection between itch and scratching is so close that in some languages the same word refers to both itch and scratch. Itch is restricted to the skin, tracheal mucous membrane, and several mucocutaneous junctions (e.g. conjunctivae). Interestingly, nerves in the deep reticular dermis and subcutaneous fat do not transmit itch.
Itch neurophysiology includes specific itch mediators and nerve fibers that transmit itch peripherally and centrally . While the concept of pruritus as a sensory modality entirely separate from pain was not generally appreciated until the mid-twentieth century, studies have clearly identified individual histaminergic and non-histaminergic C fibers that transmit itch . C fibers that transmit itch have exceptionally slow conduction velocities (0.3–1.0 m/second) and innervate unusually wide territories.
Histamine-sensitive C fibers are sensitive to heat as well as pruritogenic stimuli but not to mechanical stimuli; in contrast, the vast majority of C fibers are sensitive to mechanical and heat stimuli but have little or no response to histamine . The co-responsiveness of itch-transmitting C fibers to temperature explains aggravation of pruritus in a warm environment. However, the ineffectiveness of oral antihistamines for most types of pruritus suggests that other fibers have important roles in itch sensation .
Indeed, a distinct parallel pathway of non-histaminergic C fibers that transmit itch has been identified in the peripheral nervous system of humans and the spinothalamic tract of other primates . These fibers are activated by spicules of the tropical legume cowhage ( Mucuna pruriens ), which induces an intense sensation of itch when rubbed, inserted, or injected into the skin, without producing a histaminergic axon reflex . Cowhage spicules induce itch via release of the protease mucunain, which activates proteinase-activated receptor (PAR)-2 and PAR-4 . Cowhage-sensitive fibers transmit a burning sensation together with itch and are also sensitive to mechanical and other stimuli. PAR-2 receptors have a major role in mediating itch in patients with atopic dermatitis (see Ch. 12 ). The non-histaminergic, polymodal C fibers stimulated by mucunain may also have clinical relevance in chronic itch. In addition to C fibers, A-delta fibers contribute to cowhage-evoked itch with a more rapid onset .
Other non-histaminergic C fibers respond to β-alanine and activate distinct populations of primate primary sensory neurons . Intradermal injection of β-alanine elicits itch but not a wheal and flare response. MAS-related G protein-coupled receptor D (MrgprD) is a β-alanine receptor that is exclusively expressed by these C fibers. In mice, toll-like receptor 7 (TLR7) is expressed in C fibers and thought to have a role in itch (especially that elicited by non-histaminergic pruritogens) but not in pain sensations; however, its role in human itch is questionable .
A number of other neuromediators and receptors have been identified in animal models, but their role in itch pathophysiology in humans remains to be determined . Gastrin-releasing peptide receptor (GRPR)-positive neurons in the spinal cord of mice transmit itch, but not pain, via VGLUT2-mediated signaling . Overexpression of gastrin-releasing peptide in cutaneous nerve fibers and GRPR in the spinal cord has also been observed in mice and primates with chronic itch . B-type natriuretic peptide is an itch-selective neuropeptide found in dorsal root ganglia that is expressed at higher levels in mice than in rats and humans . Preprotachykinin A, a substance P precursor, is also expressed by a distinct population of murine dorsal horn neurons that respond to pruritic and noxious stimuli .
The perceived sensation of pruritus can vary greatly in quality . Patients may describe burning, pricking, “insects crawling” on the skin, or even a tickle, but the neurophysiologic and psychologic correlates of these qualitative differences have not yet been elucidated. However, information obtained from itch questionnaires has enabled a better understanding of the different characteristics of itch and its features in various skin diseases .
In the spinal cord, pruriceptive C fibers synapse with secondary sensory neurons in the gray matter of the dorsal horn ( Fig. 5.1 ). These neurons then cross over and ascend in the lateral spinothalamic tract to the thalamus. Studies using in vivo extracellular recordings from cats identified a subclass of lamina I spinothalamic tract neurons that are excited by iontophoretically administered histamine . Neurophysiology experiments in primates then found that cowhage-induced itch stimulates a distinct population of spinothalamic tract neurons that are not involved in the transmission of histamine-mediated itch . Studies in human subjects utilizing functional MRI or positron emission tomography (PET) have shown that histamine and cowhage activate different brain areas, providing insight into the supraspinal processing of itch and the corresponding scratch response .
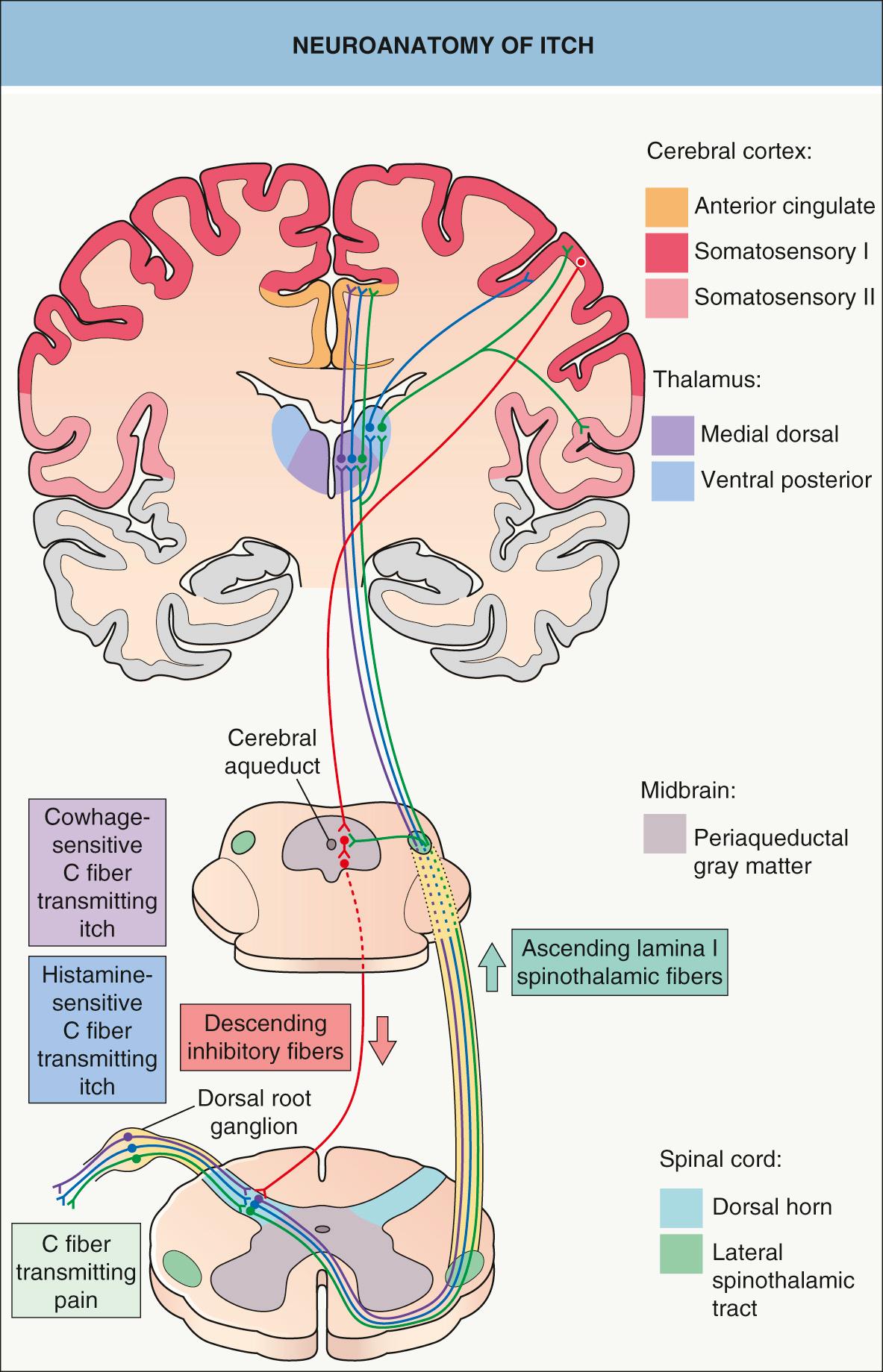
In healthy individuals, induction of itch by histamine or cowhage can elicit activation of the anterior and posterior cingulate cortex, precuneus, somatosensory areas I and II, supramarginal gyrus, inferior parietal lobe, and insula-claustrum complex. In addition to most of the previous areas, cowhage evokes more extensive activation of the insular cortex, claustrum, basal ganglia, putamen, and thalamic nuclei on the contralateral side of stimuli . In one study, atopic dermatitis patients exhibited significantly more activation of the posterior cingulate cortex and precuneus, a cortical area involved in integrative tasks such as visuospatial imagery, episodic memory retrieval and self-awareness, than did healthy controls . This highlights the emotional and affective processing of itch experience in atopic individuals, in whom the degree of brain activation correlated with itch intensity and the severity of the atopic disease .
In healthy subjects, scratching has been found to inhibit the activation of the cingulate cortex and to activate the prefrontal cortex and cerebellum. It is possible that the cerebellum plays a role in coordination of the itch–scratch cycle . Scratching also inhibits histamine-evoked activity of spinothalamic neurons in primates, but not spontaneous activity or that stimulated by pain . Self-scratching also activates areas of the brain involved in reward processing, including the striatum and substantia nigra. While these responses correlate with the pleasure of scratching, activation of other areas is associated with itch relief .
Activation of multiple brain areas argues against a single “itch center”, emphasizing the multidimensionality of the itch sensation. Pain demonstrates a similar pattern of brain activation involving many of the same cortical regions (see Fig. 5.1 ).
Seeing other people scratch can induce sensations of itch and an urge to scratch . Psychophysical studies have shown that the perception of itch intensifies in atopic dermatitis patients when they are exposed to visual cues of itch . Recent studies utilizing functional MRI showed that “contagious itch” activates many of the neural regions linked to the physical perception of itch, including anterior insular, primary somatosensory, prefrontal, and premotor cortices as well as the striatum . However, more studies are needed to verify the hypothesis that itch “contagion” involves processing in associative cortical networks such as “mirror neurons” of the prefrontal cortex.
Removal of the epidermis abolishes the perception of itch, suggesting that putative pruritus receptor units are located predominantly within this layer. Light microscopic and ultrastructural studies of human skin have shown the existence of intraepidermal nerve fibers with “free” non-specialized nerve endings extending to the stratum granulosum . A subclass of C fibers in the epidermis that express Mrgprs, in particular MrgprX1, are involved in chloroquine-induced pruritus (see Fig. 5.2F ) . Cathepsin S, an endogenous protease, can cleave and thereby activate both MrgprC11 in mice, which leads to scratching behavior, and MrgprX2 in humans ; this is in addition to its ability to activate PAR-2/4 receptors ( Fig. 5.2 ). In mice, neurons that express MrgprA3 relay itch-specific information to the spinal cord .
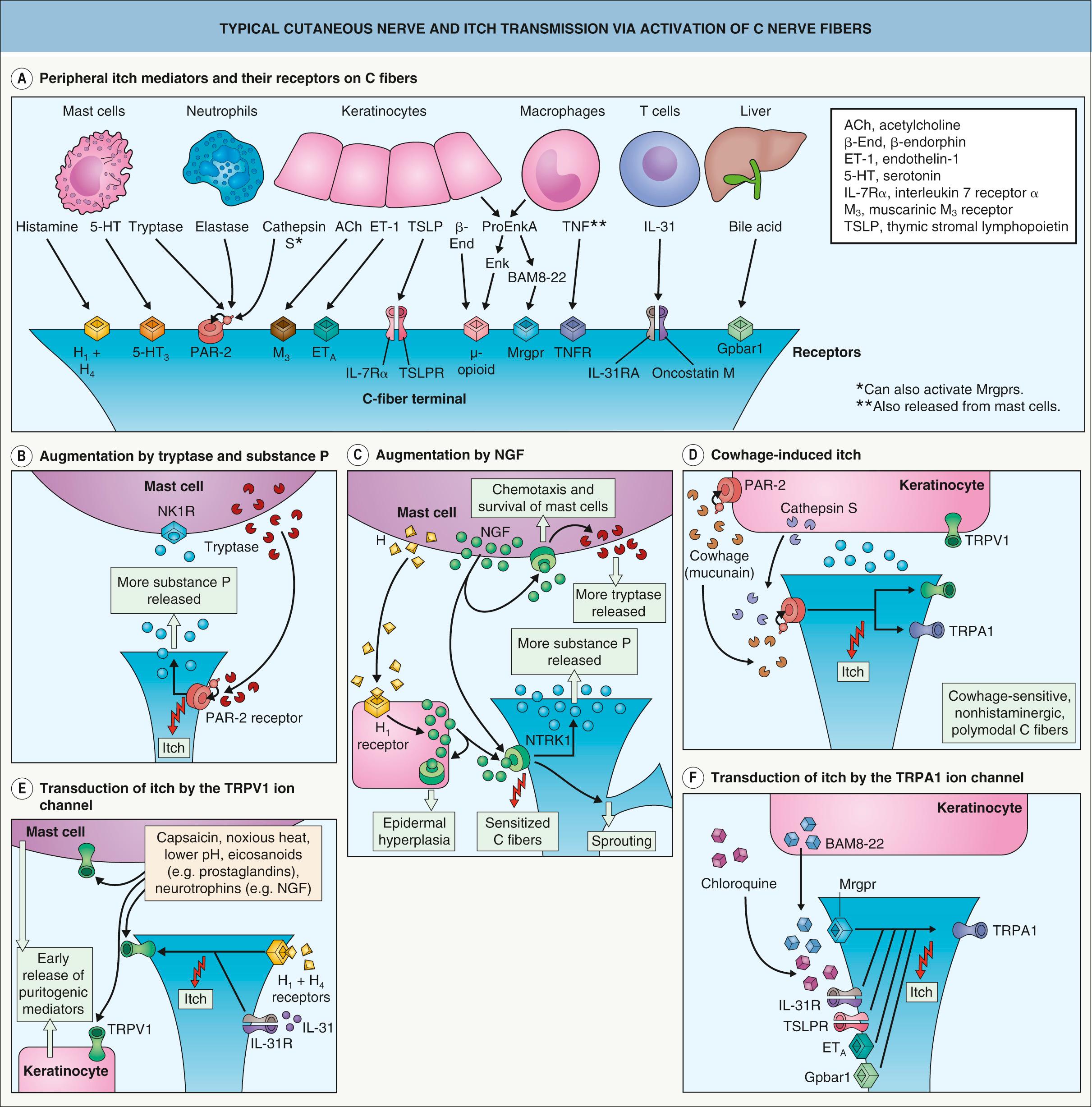
Keratinocytes express a variety of neural mediators and receptors involved in the sensation of itch ( Table 5.2 ). These include opioids, proteases, substance P, NGF, and neurotrophin 4 as well as their respective receptors, including µ- and κ-opioid receptors, PAR-2, neurotrophic tyrosine kinase receptor type 1 (NTRK1 [TRKA]), and transient receptor potential vanilloid ion channels (particularly TRPV1 and TRPV3). Keratinocytes also have ATP-gated ion channels and adenosine receptor ligands similar to those observed in C fibers involved in pain transmission. These structural similarities to nerve fibers suggest that keratinocytes may be involved in the transduction and generation of itch. PAR-2 receptors are thought to be involved in the itch of atopic dermatitis and mediate cowhage-induced itch (see above). Cathepsin S also induces itch via PAR-2/4 receptors and as mentioned previously, Mrgprs .
| MAJOR MEDIATORS OF PRURITUS: RELATIVE POTENCIES WITH REGARD TO PRURITUS AND PAIN | ||
|---|---|---|
| Mediator | Pruritus | Pain |
| Primary mediators | ||
| Histamine | +++ * | + * |
| Tryptase (protease) | +++ | + |
| Cathepsin S (protease) | +++ | ? |
| Interleukin-31 | +++ | ? |
| Secondary mediators | ||
| Prostaglandin E 1,2 | + † | + |
| Substance P ‡ | + | + |
| µ-Opioid receptor agonists | ++ | − |
| Nerve growth factor (NGF) | + | ++ |
| Interleukin-2 | +++ | +/− |
* Superficial (intraepidermal) injection of histamine causes pruritus; deep dermal injection causes pain.
Various mediators that act centrally and/or peripherally are involved in pruritus, including histamine, proteases, substance P, opiates, NGF, and prostaglandins. In inflammatory skin diseases, proinflammatory mediators produce pruritus and other signs of inflammation, in particular erythema due to vasodilation and edema from increased vascular permeability. The relative potencies of the major mediators with regard to these responses (including pruritus) are listed in Table 5.2 . Some of these mediators cause pruritus indirectly by evoking release of histamine and tryptase from mast cells or by potentiating the actions of mediators such as prostaglandins E 1 and E 2 .
Histamine is the archetypal mediator of signs and symptoms of inflammation, including pruritus. In the skin, histamine is contained primarily within the granules of dermal mast cells. Histamine can be released from mast cells upon activation of a range of receptors, including the high-affinity IgE receptor (FcεRI), the KIT receptor for stem cell factor, and receptors for neuropeptides (e.g. substance P, NGF) and complement C5a. In IgE-mediated acute urticaria, histamine is released when a specific antigen/allergen cross-links adjacent receptor-bound specific IgE antibodies. In autoimmune chronic urticaria, similar cross-linking occurs via functional circulating IgG that react with epitopes expressed on the α-chain of adjacent FcεRIs or less commonly anti-IgE autoantibodies (see Fig. 18.3 ). Histologically, dermal mast cells and unmyelinated neurons are closely juxtaposed (see Fig. 5.2B, C, E ), raising the possibility of a close (“synapsis-like”) functional relationship between the immune and nervous systems.
Histamine's pruritic action can be potentiated by prostaglandin E 1 and E 2 . Evidence for histamine as the main mediator of pruritus is limited to a few skin diseases, including acute and chronic urticaria and mastocytosis (e.g. urticaria pigmentosa). H 1 antihistamines are usually effective in these disorders.
Recognition of the histamine H 4 receptor has expanded our understanding of the physiologic actions of histamine. The H 4 receptor is expressed by neurons and bone marrow-derived cells such as eosinophils, mast cells, dendritic cells, monocytes, and CD8 + T cells. It mediates chemotaxis in the latter group and is thought to have a role in the inflammation and pruritus of atopic dermatitis. H 4 antagonists are under development and have been shown to alleviate experimental pruritus. In animal models of itch, the effects of H 4 antagonists are synergistic with those of centrally acting H 1 antihistamines (e.g. diphenhydramine) .
As noted above, itch-specific GRPR-positive neurons in the dorsal horn of the spinal cord have been identified in mice . Although named for its role in regulating gastrointestinal functions, the GRP ligand of GRPRs is widely expressed in the CNS. Whether peripheral sensory neurons express GRP is still debated .
B-type natriuretic peptide (BNP; natriuretic polypeptide B) is a 32-amino acid polypeptide that is secreted by the cardiac ventricles to regulate blood pressure and fluid balance. BNP is also expressed by a subset of C fibers and may function in itch transmission , likely driving an itch circuit that includes GRPR-expressing neurons in the spinal cord.
Human dermal mast cells produce two proteases, tryptase and chymase . Tryptase released by activated mast cells cleaves PAR-2, a G protein-coupled receptor present on C-fiber terminals (see Fig. 5.2B, D ); this exposes a tethered ligand domain and thereby “self-activates” PAR-2, leading to itch transmission. PAR-2 activation results in local release of neuropeptides, including substance P and calcitonin gene-related peptide, which induce neurogenic inflammation . Kallikrein and cathepsins in the skin can also activate PAR-2 in a similar manner. In addition, cleavage of murine MrgprC11 or human MrgprX2 by cathepsin S activates these receptors and evokes itch (see above).
Tryptase levels are elevated fourfold in non-lesional forearm skin of atopic dermatitis patients , while expression of PAR-2 is significantly increased in the epidermis and cutaneous nerve fibers of eczematous lesions and to a lesser degree in non-lesional skin. Proteases such as cathepsin B can also be found in common allergens (e.g. grass pollen, house dust mites) , and Staphylococcus aureus can induce secretion of proteases; both of these exogenous factors are known to aggravate atopic dermatitis and itch.
Transgenic mice overexpressing a serine protease exhibit severe itch and scratching ( Table 5.3 ). In Netherton syndrome, serine protease inhibitor deficiency leads to excess epidermal protease activity, resulting in pruritus and atopic manifestations (see Ch. 57 ). These observations suggest that interactions between proteases and receptors on C fibers play important roles in itch and cutaneous inflammation.
| MURINE MODELS OF ITCH | ||
|---|---|---|
| Model | Clinical and laboratory features | Pathogenesis/comments |
| Transgenic mice overexpressing PAR-2 | Epidermal hyperplasia and scaling as well as pruritus and scratching | |
| Transgenic mice overexpressing a serine protease | Pruritus and scratching | |
| TRPV1-deficient mice and Pirt-deficient mice | Lack of response to pruritogens | Pirt normally plays an important role in sensing itch (histaminergic and non-histaminergic) as well as pain via TRPV1 channels (and also TRPV1-independent itch) |
| Mrgpr-deficient mice and TRPA1-deficient mice | Lack of scratching response to chloroquine and BAM8-22, but preserved response to histamine | Mrgprs are normally activated by chloroquine and BAM8-22, so the lack of response in TRPA1-deficient mice implicates this receptor in downstream signaling |
| GRPR-deficient mice | Lack scratching response to pruritogens, but preserved response to painful stimuli | GRPR is expressed in the dorsal spinal cord |
| Bhlhb5-deficient mice | Enhanced scratching response to pruritogens | Selective loss of inhibitory interneurons in the dorsal horn that regulate pruritus |
| µ-Opioid receptor-deficient mice | Thinner epidermis, higher density of epidermal nerve endings, and less scratching after induction of dry skin | |
| Transgenic mice overexpressing interleukin-31 | Pruritus, scratching, excoriations and alopecia | |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here